Abstract
The bone marrow (BM) microenvironment consists of extracellular-matrix and the cellular compartment including immune cells. Multiple myeloma (MM) cell and BM accessory cell interaction promotes MM survival via both cell-cell contact and cytokines. Immunomodulatory agents (IMiDs) target not only MM cells, but also MM cell-immune cell interactions and cytokine signaling. Here we examined the in vitro effects of IMiDs on cytokine signaling triggered by interaction of effector cells with MM cells and BM stroma cells. IMiDs diminished interleukin-2, interferonγ, and IL-6 regulator suppressor of cytokine signaling (SOCS)1 expression in immune (CD4T, CD8T, natural-killer T, natural-killer) cells from both BM and PB of MM patients. In addition, coculture of MM cells with healthy PBMCs induced SOCS1 expression in effector cells; conversely, treatment with IMiDs down-regulated the SOCS1 expression. SOCS1 negatively regulates IL-6 signaling and is silenced by hypermethylation in MM cells. To define the mechanism of inhibitory-cytokine signaling in effector cells and MM cells, we next analyzed the interaction of immune cells with MM cells that were epigenetically modified to re-express SOCS1; IMiDs induced more potent CTL responses against SOCS1 re-expressing–MM cells than unmodified MM cells. These data therefore demonstrate that modulation of SOCS1 may enhance immune response and efficacy of IMiDs in MM.
Introduction
Multiple myeloma (MM) is characterized by accumulation of malignant plasma cells in the bone marrow (BM), bone lesions, and immunodeficiency. The interaction between myeloma cells with BM accessory cells and the extracellular matrix induces autocrine and paracrine tumor growth, as well as immune suppressor response mediated by suppressors of cytokine/growth factor signaling. The growth and antiapoptotic factors interleukin-6 (IL-6), insulin-like growth factor, vascular endothelial growth factor, and tumor necrosis factor-α, as well as chemokines and other secreted molecules in the BM milieu, play crucial roles in MM disease progression and pathophysiology.1,2 As in other cancers, the immune system can modulate MM cell growth, tumor development can promote immunosuppression, and conversely, immunosuppression may support tumor development.3-6 Novel biologic agents targeting not only tumor cells but also tumor cell–host interactions, cytokines, and the BM microenvironment, may affect mechanisms of both tumor cell growth and immunosuppression. For example, immunomodulatory drugs (IMiDs) have been used to overcome conventional drug resistance and improve patient outcome in MM. In addition to their direct anti-MM effect, IMiDs also stimulate T-cell proliferation, IL-2 and interferonγ (IFNγ) production,7 and enhance cytotoxic T lymphocyte (CTL) and natural killer (NK) effector cell activity against MM cells.8 Lenalidomide is more potent than thalidomide in both stimulating T-cell proliferation via the T-cell receptor (TCR) and in enhancing IL-2 and IFNγ production. In addition, lenalidomide decreases secretion of IL-6, tumor necrosis factor-α, and IL-10.9 Another IMiD, pomalidomide, also triggers significantly increased serum IL-2 receptor and IL-12 levels, with associated activation of T cells, monocytes, and macrophages.1
Regulation of the immune response is mainly mediated by cytokines. Signaling through the cytokine receptor family initiates formation of a functional cytokine receptor and appropriate cellular response such as differentiation, proliferation, and further cytokine production.10,11 However, tight control of cytokine signaling is required to modulate cytokine level for proper cell response; conversely, lack of control can support tumor development and growth.6,10-12 In the cytokine signaling cascade, binding of cytokine to its receptor initiates intracellular signaling through activating cytoplasmic kinases (JAKs). The JAK proteins phosphorylate tyrosine residues within the receptor chains, creating docking sites for the signal transducers and activators of transcription protein (STATs). The homo- or heterodimer forms of STATs translocate to the nucleus, where they regulate target gene expression for appropriate cell response. These target genes also include negative regulators of cytokine signaling and production, the suppressor of cytokine signaling (SOCS) genes. The SOCS family includes intracellular cytokine-inducible proteins SOCS1-7 and CIS, composed of a central SH2 domain, an amino-terminal domain, and a carboxy-terminal 40-amino acid module (SOCS box) involved in proteasomal targeting.5,10,11,13,14 Each SOCS can be induced and then, in turn, regulates its corresponding cytokine signaling. SOCS proteins can inhibit or attenuate JAK/STAT cytokine signaling pathway by directly inhibiting JAK tyrosine kinase activity via kinase inhibitory region (KIR), competing with JAK/STAT binding sites or by acting as E3-ubiquitin ligases to mediate degradation of proteins.10,11,13-15 Within the SOCS family, SOCS1 is known as a common negative regulator of IL-2, IFNγ, and IL-6 signaling.13,15-20 IL-6 is one of the essential growth factors for survival and growth of MM cells, while suppressing immune cell proliferation and response against MM. IL-6 induces MM cell growth via IL-6R/JAK/STAT pathway, whereas negative feedback of IL-6-induced JAK/STAT pathway is mediated by SOCS3; moreover, the inhibitory effect of SOCS1 on JAK/STAT pathway is even stronger than SOCS3.21-24 Importantly, SOCS1 gene is silenced in 75% of MM patients by DNA hypermethylation of CpG dinucleo-tides in the promoter region,25-27 thereby resulting in uncontrolled IL-6 signaling, MM cell growth, and immune suppression.
We have previously demonstrated that lenalidomide triggers tyrosine phosphorylation of CD28 and activation of nuclear factor κB (NFκB) in T cells, as well as promotes the nuclear translocation of nuclear factor of activated T cells-2 and activator protein-1 via activation in PI3-K/Akt pathway, thereby stimulating IL-2 secretion.7,28 However, the molecular mechanisms regulating IMiDs-induced IL-2 and IFNγ production in immune effector cells, as well as immune responses in the MM BM and peripheral blood (PB) milieu, are undefined. Here we examined effects of IMiDs on molecular mechanisms regulating cytokine signaling in immune effector cells (CD4T cells, CD8T cells, NKT cells, and NK cells) in the BM and PB milieu. We showed that IMiDs down-regulate SOCS1 in immune effector cells, thereby inducing IL-2 and IFNγ production. Moreover, IMiDs induce more potent CTL response against MM cells epigenetically modified to re-express SOCS1 gene than MM cells lacking SOCS1. Taken together, our data therefore demonstrate that SOCS1 may be playing an important role in the pathogenesis of MM.
Methods
Cell lines and cell isolation
Human MM cell lines MM1.S (kindly provided by Dr Steven Rosen, Northwestern University, Chicago, IL), RPMI 8226, U266 (ATCC), OPM1, and plasma cell leukemia cell line INA-6 (kindly provided by Dr Edward Thompson, University of Texas Medical Branch, Houston, TX) were maintained in RPMI 1640 (Bio Whittaker) containing 10% fetal calf serum and 100 U/mL penicillin and 100 μg/mL streptomycin (Life Technologies). Heparinized venous blood samples and/or aspirates of BM from patients with untreated or relapsed MM and healthy donors were obtained after written informed consent per the Declaration of Helsinki and approval by the Institutional Review Board of the Dana-Farber Cancer Institute. Mononuclear cells were separated by ficoll-hypaque density gradient centrifugation. CD3T cells from patients with MM and healthy donors were negatively selected by depleting B cells, monocytes, granulocytes, platelets, early erythroid precursor cells, and NK cells using a magnetically labeled cocktail of hapten-modified anti-CD14, -CD16, -CD36, -CD56, -CD123, TCRγδ, glycophorin-A MoAbs (Miltenyi Biotec). The purity of the isolated T cells was detected using anti-CD3 Ab.
Reagents and compounds
IMiDs CC-5013 (Revlimid/Lenalidomide) and CC-4047 (Pomalidomide/Actimid; Celgene) were dissolved in dimethyl sulfoxide at 10mM and stored at −20°C. 5-Azacytidine (5-AzaC; 10μM) and Trichostatin A (TsA; 5μM) were reconstituted in culture medium and stored at −20°C (Sigma-Aldrich). Anti-CD3 MoAb (10 μg/mL; Coulter) was used to stimulate cells.
Cell culture and treatment
BM stroma cells (BMSCs) were generated from BM aspirates of MM patients or healthy donors by culturing 4-6 weeks in DMEM1α medium complemented with 20% fetal bovine serum, 100 U/mL penicillin, and 100 μg/mL streptomycin (Life Technologies). Mononuclear cells from PB (PBMCs) or BM (BMMCs) of MM patients or healthy donors were cocultured with MM cell lines MM1.S, RPMI8226, U266, OPM1, INA-6, or BMSCs in the absence or presence of lenalidomide or pomalidomide at 1μM for 1 hour to 7 days.
DNA sodium bisulfite modification and MSP
To quantify DNA methylation at the SOCS1 gene promoter region, genomic DNA was isolated from MM cell lines (MM1.S, RPMI8226, U266, OPM1, INA-6) incubated for 4 days either alone or in combination with IMiDs (lenalidomide and pomalidomide, 1μM; Celgene), AzaC (0.5μM) followed by TsA (0.1μM) for additional 16 hours and sodium bisulfite-modified (CpGenome DNA Modification Kit; Millipore). MSP was then performed using methylation-specific polymerase chain reaction (MSP) primers that specifically recognize unmethylated or methylated SOCS1 gene sequences (CpG-WIZ SOCS1 Amplification Kit).
Results
IMiDs activate effector immune cells and reduce inhibitory immune cells in MM in vitro
To assess effects of IMiDs on immune effector cells, PBMCs and BMMCs from healthy donors and MM patients were analyzed for effector cell activation status by measuring positive costimulatory molecule profile, cell proliferation, and cytokine profile using flow cytometry. Lenalidomide and pomalidomide induced expression of costimulatory molecules CD28 (lenalidomide, 63%; pomalidomide, 48% average positive cells; P < .05), inducible costimulator (ICOS; lenalidomide, 40%; pomalidomide, 48% average positive cells; P < .05), and inducible costimulator ligand (ICOSL; lenalidomide, 55%; pomalidomide, 66% average positive cells; P < .05) in CD3+ gated effector cells within MM-PBMCs (Figure 1A).
IMiDs activate effector immune cells and reduce inhibitory immune cells in MM in vitro. PBMCs from patients with MM (MM-PBMC) were stimulated with anti-CD3 Ab and incubated for 6 days in the absence or presence of lenalidomide and pomalidomide. IMiDs effect on effector cell activation was analyzed using flow cytometry and cytokine protein array. (A) Histogram plots by flow cytometric analysis show membrane-expression of positive-costimulatory signaling molecules CD28, ICOS, and ICOSL on gated MM-PBMC CD3+ cells incubated for 3 days in the absence or presence of lenalidomide and pomalidomide. Shadowed profiles indicate isotype-matched control immunoglobulin (Ig) staining. One representative of 3 independent experiments is shown. Statistical significance indicated (t test, 1-tailed distribution, P < .05). (B) IMiDs induced proliferation of effector cells in MM-PBMCs is shown by CFSE-staining flow cytometric analysis. Proliferating effector cells were identified by CFSE costaining in gated CD4 PECy5 (CD4T cells), CD8 PECy7 (CD8 T cells), or CD56 PE/CD8 PECy7 (NKT cells) positive subpopulations. Proliferation is represented by division index of each cell population. One representative of 3 independent experiments is shown. Statistical significance indicated (t test, 1-tailed distribution, P < .05). (C) IMiDs effect on cytokine production was determined in MM-PBMC effector cells by intracytoplasmic cytokine staining flow cytometric analysis. MM-PBMCs were stimulated with anti-CD3 Ab and cultured for 16 hours with or without lenalidomide and pomalidomide, and intracytoplasmic expression (percent positive stained cells) of IL-2 (PE) was shown in gated effector cell subpopulations, as shown in the figure by side scatter dot plots. y-axis represents IL-2 PE staining and x-axis represents CD4T cells (top panel), CD8 T cells (middle panel), and NKT cells (bottom panel). One representative of 3 independent experiments is shown. Statistical significance indicated (t test, 1-tailed distribution, P < .05). (D) Secreted IFNγ, IL-2, and IL-6 were measured by cytokine protein array in the supernatants of anti-CD3 Ab-stimulated MM-BMMCs in the absence or presence of lenalidomide and pomalidomide for 24 hours. Expression of cytokines was determined using ImageJ 1.37v (http://rsb.info.nih.gov/ij/) densitometric analysis. Average and SD of triplicate from 1 representative of 3 independent experiments is shown. Statistical significance indicated (t test, 1-tailed distribution, P < .05). (E) Inhibitory CD4T cells were identified in the MM-BMMCs incubated in the absence or presence of lenalidomide and pomalidomide for 48 hours to 6 days. CD4T cells were gated and further analyzed for membrane CD25 and intracellular FOXP3 coexpression by flow cytometry. Top panel demonstrates CD4+CD25+FOXP3+ regulatory CD4T cells at the end of 6 days culture, and bottom panel represents IL-17 producing CD4 T cells (Th17) at the end of a 48-hour culture. One representative of 3 independent experiments is shown. Statistical significance indicated (t test, 1-tailed distribution, P < .05).
IMiDs activate effector immune cells and reduce inhibitory immune cells in MM in vitro. PBMCs from patients with MM (MM-PBMC) were stimulated with anti-CD3 Ab and incubated for 6 days in the absence or presence of lenalidomide and pomalidomide. IMiDs effect on effector cell activation was analyzed using flow cytometry and cytokine protein array. (A) Histogram plots by flow cytometric analysis show membrane-expression of positive-costimulatory signaling molecules CD28, ICOS, and ICOSL on gated MM-PBMC CD3+ cells incubated for 3 days in the absence or presence of lenalidomide and pomalidomide. Shadowed profiles indicate isotype-matched control immunoglobulin (Ig) staining. One representative of 3 independent experiments is shown. Statistical significance indicated (t test, 1-tailed distribution, P < .05). (B) IMiDs induced proliferation of effector cells in MM-PBMCs is shown by CFSE-staining flow cytometric analysis. Proliferating effector cells were identified by CFSE costaining in gated CD4 PECy5 (CD4T cells), CD8 PECy7 (CD8 T cells), or CD56 PE/CD8 PECy7 (NKT cells) positive subpopulations. Proliferation is represented by division index of each cell population. One representative of 3 independent experiments is shown. Statistical significance indicated (t test, 1-tailed distribution, P < .05). (C) IMiDs effect on cytokine production was determined in MM-PBMC effector cells by intracytoplasmic cytokine staining flow cytometric analysis. MM-PBMCs were stimulated with anti-CD3 Ab and cultured for 16 hours with or without lenalidomide and pomalidomide, and intracytoplasmic expression (percent positive stained cells) of IL-2 (PE) was shown in gated effector cell subpopulations, as shown in the figure by side scatter dot plots. y-axis represents IL-2 PE staining and x-axis represents CD4T cells (top panel), CD8 T cells (middle panel), and NKT cells (bottom panel). One representative of 3 independent experiments is shown. Statistical significance indicated (t test, 1-tailed distribution, P < .05). (D) Secreted IFNγ, IL-2, and IL-6 were measured by cytokine protein array in the supernatants of anti-CD3 Ab-stimulated MM-BMMCs in the absence or presence of lenalidomide and pomalidomide for 24 hours. Expression of cytokines was determined using ImageJ 1.37v (http://rsb.info.nih.gov/ij/) densitometric analysis. Average and SD of triplicate from 1 representative of 3 independent experiments is shown. Statistical significance indicated (t test, 1-tailed distribution, P < .05). (E) Inhibitory CD4T cells were identified in the MM-BMMCs incubated in the absence or presence of lenalidomide and pomalidomide for 48 hours to 6 days. CD4T cells were gated and further analyzed for membrane CD25 and intracellular FOXP3 coexpression by flow cytometry. Top panel demonstrates CD4+CD25+FOXP3+ regulatory CD4T cells at the end of 6 days culture, and bottom panel represents IL-17 producing CD4 T cells (Th17) at the end of a 48-hour culture. One representative of 3 independent experiments is shown. Statistical significance indicated (t test, 1-tailed distribution, P < .05).
We next verified effects of IMiDs on effector cell proliferation by carboxyfluorescein succinimidyl ester (CFSE)–flow cytometric analysis. Consistent with previous studies,8,28 IMiDs induced in vitro proliferation of T cells from patients with MM (Figure 1B). Flow cytometric analysis of CFSE-stained MM-PBMCs showed a significant increase in dividing CD4T cells (control division index,1.36; lenalidomide division index,1.52; pomalidomide division index,1.69; P < .05), CD8T cells (control division index, 0.19; lenalidomide division index, 0.3; pomalidomide division index, 0.48; P < .05), and CD3+CD8+CD56+NKT cells (control division index, 0.11; lenalidomide division index, 0.2; pomalidomide division index, 0.27; P < .05) in the presence of lenalidomide and pomalidomide (Figure 1B). Similarly, there was also increased proliferation of effector cells in MM-BMMCs cultured with lenalidomide and pomalidomide (data not shown).
Direct stimulatory effects of IMiDs on type I cytokine profile in effector cells of PBMCs and BMMCs from MM patients was next confirmed by intracytoplasmic cytokine staining and fluorescence-activated cell sorting analysis. Both lenalidomide and pomalidomide stimulated intracytoplasmic IFNγ and IL-2 (Figure 1C) expression in CD4T cells (average 2-fold, P < .05), CD8T cells (average 1.9-fold, P < .05), and NKT cells (average 0.8-fold, P < .05) from MM patients (Figure 1C). There was also significantly increased secretion of IFNγ (average 2.5-fold, P < .05) and IL-2 (average 4.5-fold, P < .05), as well as decreased secretion of IL-6 (average 0.7-fold, P < .05) in MM-BMMCs cultured with lenalidomide and pomalidomide, respectively (Figure 1D). In addition, IMiDs reduced the number of IL-17 producing CD4T cells (Th17) at 2 days (Figure 1E) as well as FOXP3 expressing CD4+CD25+ T cells (Treg) in MM-BM at 6 days.
IMiDs regulate SOCS1-mediated cytokine-negative feedback mechanism in effector cells in MM
To delineate the mechanism of modulatory effect of IMiDs on cytokine signaling pathway mediating IL-2, IFNγ, and IL-6 in effector cells, expression of SOCS1 protein was next evaluated in cultured PBMCs and BMMCs from healthy donors and MM patients. Both lenalidomide and pomalidomide reduced SOCS1 expression in CD4T cells, CD8T cells, NK cells, and NKT cells from healthy donors (data not shown) and MM patients, including both PB (supplemental Figure 1A, available on the Blood Web site; see the Supplemental Materials link at the top of the online article) and BM (supplemental Figure 1B).
In MM, several immune defects have been identified, particularly involving T cells.29-32 To eliminate intrinsic abnormalities, characteristics of MM and to confirm the effect of IMiDs on SOCS1 expression in immune cells, we next assessed the direct impact of tumor cells and microenvironment on the SOCS1 expression in normal donor effector cells. Mimicking the MM-BM environment, we performed direct cell-cell contact coculture experiments using healthy PBMCs, MM cell lines, and MM BMSCs (MM-BMSCs). Healthy PBMCs were cocultured for 16 hours with either MM cell lines (MM1.S and U266) or MM cell lines and MM-BMSCs together, with or without lenalidomide and pomalidomide. To limit allogeneic immune response effect in control cultures, healthy PBMCs were cocultured with allogeneic healthy PBMCs. Intracytoplasmic expression of SOCS1 protein was then evaluated by flow cytometry in CD4T cells, CD8T cells, NKT cells, and NK cells. Interestingly, SOCS1 protein expression was markedly induced in all effector cell subpopulations in the presence of both MM cell lines and MM-BMSC: CD4T cells (Figure 2A), CD8T cells (Figure 2B), NKT cells (Figure 2C), and NK cells (Figure 2D). Conversely, lenalidomide and pomalidomide significantly reduced the expression of SOCS1 protein in all effector cells cocultured either with MM cell lines alone or with MM cell lines and MM-BMSC (Figure 2). IFNγ-dependent JAK/STAT pathway was significantly activated in all effector cells in MM by lenalidomide and pomalidomide
IMiDs regulate SOCS1-dependent cytokine-negative feedback mechanism in effector cells in MM. Flow cytometric analysis of intracellular expression of SOCS1 protein in effector cells from cocultures are shown as representative dot plots. To provide direct tumor cell-effector cell contact and -BM contact, healthy donor's PBMCs were cocultured for 16 hours with either MM cell lines (MM1.S and U266) or with MM cell lines and MM-BMSCs, in the absence or presence of lenalidomide and pomalidomide. Intracytoplasmic expression of SOCS1 (PE) protein was then evaluated by flow cytometry in the gated CD4 PECy5 (CD4T cells; A), CD8 PEcy7 (CD8T cells; B), CD56 PE+CD8 PECy7+ (NKT cells; C), and CD56PE+CD8PECy7− (NK cells). (D) Top panel demonstrates intracellular SOCS1 protein expression in effector cells of healthy PBMC cocultured with allogeneic healthy PBMCs. Middle panel demonstrates intracellular SOCS1 protein expression in effector cells of healthy PBMC cocultured with MM cell line MM1.S, and bottom panel demonstrates intracellular SOCS1 protein expression in effector cells of healthy PBMCs cocultured with MM cell line MM1.S and MM-BMSC. Numbers indicate percentage of positive cells. One representative of 3 independent experiments is shown. Statistical significance indicated (t test, 1-tailed distribution, P < .05).
IMiDs regulate SOCS1-dependent cytokine-negative feedback mechanism in effector cells in MM. Flow cytometric analysis of intracellular expression of SOCS1 protein in effector cells from cocultures are shown as representative dot plots. To provide direct tumor cell-effector cell contact and -BM contact, healthy donor's PBMCs were cocultured for 16 hours with either MM cell lines (MM1.S and U266) or with MM cell lines and MM-BMSCs, in the absence or presence of lenalidomide and pomalidomide. Intracytoplasmic expression of SOCS1 (PE) protein was then evaluated by flow cytometry in the gated CD4 PECy5 (CD4T cells; A), CD8 PEcy7 (CD8T cells; B), CD56 PE+CD8 PECy7+ (NKT cells; C), and CD56PE+CD8PECy7− (NK cells). (D) Top panel demonstrates intracellular SOCS1 protein expression in effector cells of healthy PBMC cocultured with allogeneic healthy PBMCs. Middle panel demonstrates intracellular SOCS1 protein expression in effector cells of healthy PBMC cocultured with MM cell line MM1.S, and bottom panel demonstrates intracellular SOCS1 protein expression in effector cells of healthy PBMCs cocultured with MM cell line MM1.S and MM-BMSC. Numbers indicate percentage of positive cells. One representative of 3 independent experiments is shown. Statistical significance indicated (t test, 1-tailed distribution, P < .05).
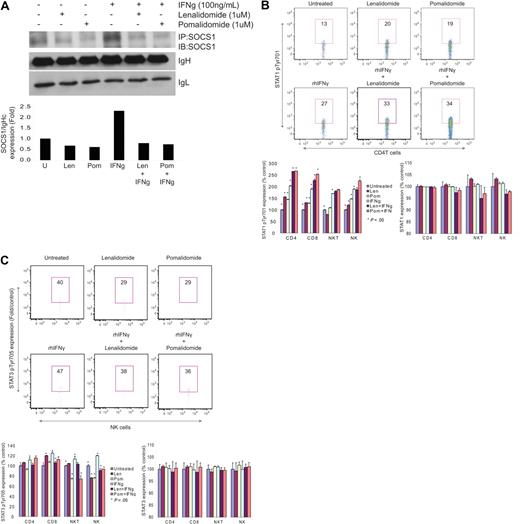
Specificity of IMiDs effect on SOCS1 expression in effector cells was also confirmed by coimmunoprecipitation–Western blot analysis. Highly purified CD3T cells from healthy donors were cultured with or without lenalidomide or pomalidomide in the presence of anti-CD3 stimulation. T cells were then exogenously stimulated with SOCS1 inducing cytokine IFNγ for 15 minutes to 4 hours to induce SOCS1 expression. Exogenous IFNγ significantly induced SOCS1 expression (2.3-fold increase) compared with unstimulated cells (Figure 3A); conversely, lenalidomide and pomalidomide significantly reduced SOCS1 protein expression in both unstimulated and IFNγ stimulated CD3T cells (Figure 3A). Moreover, lenalidomide and pomalidomide also induced SOCS1 downstream target molecule STAT1 phosphorylation in all effector cells (Figure 3B). Intracytoplasmic expression of phosphorylated STAT1 detection by flow cytometry showed that lenalidomide and pomalidomide alone induced STAT1 phosphorylation in CD4T cells, which was further augmented in combination with IFNγ (average 2.7-fold, P < .05), CD8T cells (average 2.3-fold, P < .05), NKT cells (average 1.8-fold, P < .05), and NK cells (average 2.1-fold, P < .05). In contrast, STAT3 phosphorylation was reduced in all effector cells by lenalidomide and pomalidomide in combination with IFNγ (Figure 3C), particularly in CD8T (average 0.8-fold, P < .05), NKT (average 0.8-fold, P < .05), and NK cells (average 0.8-fold, P < .05). Lenalidomide and pomalidomide induced no significant change on the expression of total STAT1 and STAT3 protein (Figure 3B-C) These data suggest that less activation of STAT3 phosphorylation might be because IFNγ specifically stimulates STAT1, rather than STAT3, phosphorylation. In addition, STAT5 phosphorylation was highly induced by lenalidomide alone or in combination with IFNγ (data not shown), indicating that IL-2/IFNγ–dependent JAK-STAT pathway was significantly activated in the presence of lenalidomide and pomalidomide in all effector cells in MM.
IMiDs regulate ex vivo induced SOCS1 expression and STAT activation. (A) SOCS1 protein expression was shown by immunoprecipitation-Western blot in effector cells, incubated in the presence of aCD3 Ab and lenalidomide (1μM) or pomalidomide (1μM) for 16 hours, and then stimulated with SOCS1 inducing cytokine IFNγ (100 ng/mL) for 15 minutes to 4 hours (top panel). Expression of SOCS1 protein relative to IgH chain was demonstrated (fold) using ImageJ 1.37v (http://rsb.info.nih.gov/ij/) densitometric analysis (bottom panel). One representative of 3 independent experiments is shown. Intracellular expression of STAT1 pTyr701 and STAT1 (B) and STAT3 pTyr705 and STAT3 (C) are shown in PBMCs from healthy donors (n = 3) by flow cytometric analysis. aCD3 Ab stimulated PBMCs were incu-bated in the absence or presence of lenalidomide and pomalidomide for 16 hours, and then stimulated with IFNγ for 15 minutes. Expression of phosphorylated STATs is shown as a percent expression relative to control baseline levels in aCD3 Ab–stimulated cells. Top panels demonstrate phosphorylated STATs expression in CD4 T cells as representative plots, and bottom panels demonstrate phosphorylated STATs expression in all effector cells as percent expression relative to control baseline levels in aCD3 Ab stimulated cells. Numbers indicate percentage of positive cells. One representative of 3 independent experiments is shown. Statistical significance indicated (t test, 1-tailed distribution, P < .05).
IMiDs regulate ex vivo induced SOCS1 expression and STAT activation. (A) SOCS1 protein expression was shown by immunoprecipitation-Western blot in effector cells, incubated in the presence of aCD3 Ab and lenalidomide (1μM) or pomalidomide (1μM) for 16 hours, and then stimulated with SOCS1 inducing cytokine IFNγ (100 ng/mL) for 15 minutes to 4 hours (top panel). Expression of SOCS1 protein relative to IgH chain was demonstrated (fold) using ImageJ 1.37v (http://rsb.info.nih.gov/ij/) densitometric analysis (bottom panel). One representative of 3 independent experiments is shown. Intracellular expression of STAT1 pTyr701 and STAT1 (B) and STAT3 pTyr705 and STAT3 (C) are shown in PBMCs from healthy donors (n = 3) by flow cytometric analysis. aCD3 Ab stimulated PBMCs were incu-bated in the absence or presence of lenalidomide and pomalidomide for 16 hours, and then stimulated with IFNγ for 15 minutes. Expression of phosphorylated STATs is shown as a percent expression relative to control baseline levels in aCD3 Ab–stimulated cells. Top panels demonstrate phosphorylated STATs expression in CD4 T cells as representative plots, and bottom panels demonstrate phosphorylated STATs expression in all effector cells as percent expression relative to control baseline levels in aCD3 Ab stimulated cells. Numbers indicate percentage of positive cells. One representative of 3 independent experiments is shown. Statistical significance indicated (t test, 1-tailed distribution, P < .05).
IMiDs epigenetically regulate SOCS1 methylation in MM cells in vitro
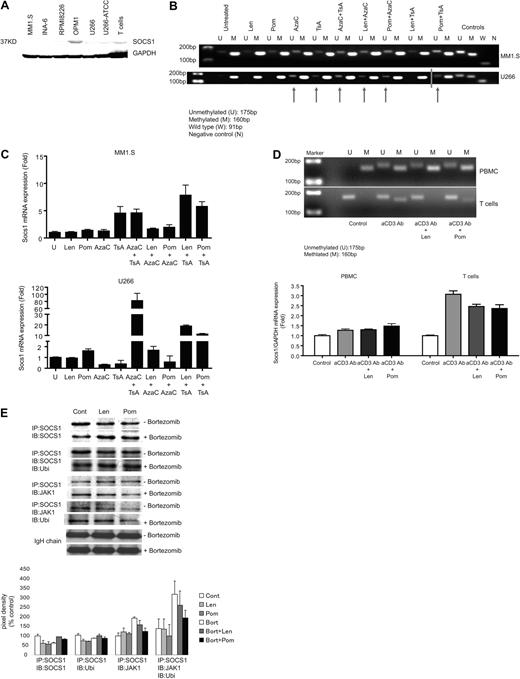
To further delineate the role of SOCS1 in regulating cytokines triggered by interaction between MM cells, effector cells, and the microenvironment, we next assessed immune response against MM cells in which SOCS1 gene expression was restored. Because SOCS1 gene can be silenced by hypermethylation at the promoter region, we first confirmed SOCS1 protein expression in MM cell lines including MM1.S-, RPMI8226-, OPM1-, U266-, and IL-6–dependent INA-6 plasma cell leukemia cell line, as well as in unstimulated CD3T cells from healthy donors by Western blot. SOCS1 protein expression was low in all MM cell lines tested except OPM1 cells, which show higher expression of SOCS1 protein (Figure 4A).
IMiDs epigenetically regulate SOCS1 methylation in MM cells in vitro. (A) Baseline SOCS1 protein expression in MM cell lines and unstimulated CD3+ T cells is shown by Western blot. Glyceraldehyde 3-phosphate dehydrogenase (GAPDH) expression was determined as an internal control for loading. Data are demonstrated as a representative of 3 separate experiments. (B) Methylation analysis of SOCS1 in MM cell lines MM1.S and U266 is assessed by MSP. MM1.S and U266 cells were incubated either alone or in combination with IMiDs (lenalidomide and pomalidomide; 1μM), 5-Azacytidine (5-AzaC; 0.5μM), and Trichostatin A (TsA; 0.1μM); sodium bisulfite–modified genomic DNA was then subjected to MSP using MSP primers that specifically recognize unmethylated or methylated SOCS1 gene sequences. U indicates the presence of unmethylated SOCS1 gene (175 bp); and M indicates the presence of methylated SOCS1 gene (160 bp). In vitro unmethylated and methylated DNA were used as controls for methylation. W indicates wild-type SOCS1 and N indicates water as control for PCR (blue line indicates the merged photomicrograps run on the same gel but in 2 lanes). (C) SOCS1 gene transcriptional expression is shown by quantitative real-time PCR in MM1.S and U266 cells. Cells were incubated either alone or in combination with IMiDs (1μM), 5-AzaC (0.5μM), and TsA (0.1μM); SOCS1 gene expression was then analyzed by real time, quantitative PCR using SYBR-Green labeling. Data are demonstrated as the mean fold increase relative to baseline levels (untreated cells). All quantitative RT-PCR data are normalized to expression level of GAPDH mRNA. (D) Methylation analysis of SOCS1 in PBMCs and T cells from healthy donors by MSP. After bisulfite modification, genomic DNA of PBMCs and T cells purified from the same PBMCs was subjected to SOCS1-specific MSP (top panel). U indicates unmethylated (175 bp) and M indicates methylated (160 bp) SOCS1 gene. Bottom panel shows expression of SOCS1 mRNA measured by quantitative RT-PCR in PBMCs and T cells from the same healthy donor. Data are demonstrated as the mean fold increase relative to baseline levels (untreated cells). All quantitative RT-PCR data are normalized to expression level of GAPDH mRNA. One representative of 3 independent experiments is shown. (E) Coimmunoprecipitation analysis of SOCS1 mediated JAK1 ubiquitination is shown in CD3T cells incubated in the absence or presence of lenalidomide and pomalidomide after preincubation with or without bortezomib. Cells were stimulated with IFNγ for 15 minutes to induce SOCS1 expression. SOCS1 protein was immunoprecipitated and blotted with anti-SOCS1 moAb, anti-JAK1 Ab, and ubiquitin moAb. Top panel demonstrates IP:IB bands. Bottom panel demonstrates pixel density values of each band expression relative to IgH chain expression. Fold expression of proteins are determined relative to control. One representative of 3 independent experiments is shown.
IMiDs epigenetically regulate SOCS1 methylation in MM cells in vitro. (A) Baseline SOCS1 protein expression in MM cell lines and unstimulated CD3+ T cells is shown by Western blot. Glyceraldehyde 3-phosphate dehydrogenase (GAPDH) expression was determined as an internal control for loading. Data are demonstrated as a representative of 3 separate experiments. (B) Methylation analysis of SOCS1 in MM cell lines MM1.S and U266 is assessed by MSP. MM1.S and U266 cells were incubated either alone or in combination with IMiDs (lenalidomide and pomalidomide; 1μM), 5-Azacytidine (5-AzaC; 0.5μM), and Trichostatin A (TsA; 0.1μM); sodium bisulfite–modified genomic DNA was then subjected to MSP using MSP primers that specifically recognize unmethylated or methylated SOCS1 gene sequences. U indicates the presence of unmethylated SOCS1 gene (175 bp); and M indicates the presence of methylated SOCS1 gene (160 bp). In vitro unmethylated and methylated DNA were used as controls for methylation. W indicates wild-type SOCS1 and N indicates water as control for PCR (blue line indicates the merged photomicrograps run on the same gel but in 2 lanes). (C) SOCS1 gene transcriptional expression is shown by quantitative real-time PCR in MM1.S and U266 cells. Cells were incubated either alone or in combination with IMiDs (1μM), 5-AzaC (0.5μM), and TsA (0.1μM); SOCS1 gene expression was then analyzed by real time, quantitative PCR using SYBR-Green labeling. Data are demonstrated as the mean fold increase relative to baseline levels (untreated cells). All quantitative RT-PCR data are normalized to expression level of GAPDH mRNA. (D) Methylation analysis of SOCS1 in PBMCs and T cells from healthy donors by MSP. After bisulfite modification, genomic DNA of PBMCs and T cells purified from the same PBMCs was subjected to SOCS1-specific MSP (top panel). U indicates unmethylated (175 bp) and M indicates methylated (160 bp) SOCS1 gene. Bottom panel shows expression of SOCS1 mRNA measured by quantitative RT-PCR in PBMCs and T cells from the same healthy donor. Data are demonstrated as the mean fold increase relative to baseline levels (untreated cells). All quantitative RT-PCR data are normalized to expression level of GAPDH mRNA. One representative of 3 independent experiments is shown. (E) Coimmunoprecipitation analysis of SOCS1 mediated JAK1 ubiquitination is shown in CD3T cells incubated in the absence or presence of lenalidomide and pomalidomide after preincubation with or without bortezomib. Cells were stimulated with IFNγ for 15 minutes to induce SOCS1 expression. SOCS1 protein was immunoprecipitated and blotted with anti-SOCS1 moAb, anti-JAK1 Ab, and ubiquitin moAb. Top panel demonstrates IP:IB bands. Bottom panel demonstrates pixel density values of each band expression relative to IgH chain expression. Fold expression of proteins are determined relative to control. One representative of 3 independent experiments is shown.
The methylation status of SOCS1 gene promoter in these MM cells lines was next analyzed in bisulfite modified genomic DNA by MSP. SOCS1 gene promoter was hypermethylated in MM1.S, OPM1, U266, and INA6 cells, but both methylated and unmethylated alleles of SOCS1 gene were present in RPMI8226 cells (data not shown). To evaluate the mechanism whereby IMiDs regulate SOCS1 expression, we next explored whether IMiDs induce epigenetic changes in the SOCS1 gene promoter thereby regulating SOCS1 expression. Methylation status of SOCS1 gene promoter was assessed by MSP in MM1.S and U266 cells cultured with lenalidomide, pomalidomide, and demethylating agents AzaC and TsA, alone or in combinations (Figure 4B). SOCS1 gene expression was restored in both MM cell lines treated with AzaC and TsA, alone or in combination. Importantly, we show that pomalidomide also demethylates SOCS1 gene promoter, either alone or in combination with demethylating agents, whereby lenalidomide demethylates SOCS1 gene only in combination with demethylating agents in MM cells (Figure 4B).
The effects of IMiDs on SOCS1 gene expression was next confirmed by measuring SOCS1 mRNA expression using quantitative reverse transcription polymerase chain reaction (RT-PCR) in MM1.S and U266 cell lines treated with IMiDs and demethylating agents, alone or in combinations (Figure 4C). Analysis of SOCS1 mRNA expression demonstrated that SOCS1 was partially demethylated and re-expressed in MM cell lines incubated with lenalidomide and pomalidomide or demethylating agents AzaC or TsA. Interestingly, SOCS1 demethylation and re-expression was considerably increased in MM cell lines cultured with both AzaC and TsA. Likewise, SOCS1 gene was highly demethylated and re-expressed in MM cell lines cultured with the lenalidomide or pomalidomide in combination with TsA or AzaC (Figure 4C).
We next analyzed the effect of IMiDs on the methylation status and the transcriptional expression of SOCS1 gene in PBMCs and T cells isolated from the same healthy donors (Figure 4D). Anti-CD3 Ab-stimulated PBMCs were incubated with or without IMiDs for 16 hours. CD3T cells were then purified from these PBMCs; genomic DNA was isolated from both PBMCs and T cells and then subjected to MSP. As shown in Figure 4D (top gel photomicrograph), SOCS1 gene promoter was methylated in untreated control PBMCs, but it was unmethylated in T cells (bottom gel photomicrograph) purified from same PBMCs. Importantly, stimulation of cells with anti-CD3 Ab led to partial demethylation of SOCS1 gene in PBMCs and its partial methylation in T cells, indicating active cytokine signaling in these stimulated cells. Importantly, when SOCS1 gene is unmethylated, IMiDs showed no significant effect on the methylation status of SOCS1 gene, as seen in T cells. In addition, there was no significant change in the methylation status of SOCS1 gene in PBMCs after IMiDs exposure (Figure 4D top panel). These data suggest that IMiDs may be regulating methylation of SOCS1 gene specifically in MM cells, as shown in Figure 4B. Consistent with the methylation status of SOCS1 gene promoter in these normal cells, quantitative RT-PCR revealed no significant change in the expression of SOCS1 gene transcripts in PBMCs incubated with IMiDs compared with untreated as well as anti-CD3 Ab-stimulated cells (Figure 4D bottom panel). In contrast, anti-CD3 Ab stimulation induced significant expression of SOCS1 mRNA in T cells. Whereas, both lenalidomide and pomalidomide reduced SOCS1 mRNA expression in these anti-CD3 Ab-stimulated T cells (Figure 4D bottom panels).
We next attempted to determine whether IMiDs modulate SOCS1 expression in effector cells at the transcriptional and/or translational mechanisms. After blockade of proteasome activity by Bortezomib preincubation, effector cells were stimulated with anti-CD3 Ab and lenalidomide or pomalidomide. SOCS1 expression was then induced by rhIFNγ (15 minutes to 4 hours). To determine whether IMiDs reduce SOCS1 protein expression through proteasomal degradation, we assessed changes induced by IMiDs on SOCS1 ubiquitination. In addition, we analyzed SOCS1/JAK1 binding and ubiquitination in these effector cells. Both lenalidomide and pomalidomide increased SOCS1 ubiquitination in IFNγ-stimulated (as early as 15 minutes) and proteasome activity–inhibited effector cells. However, there was decreased ubiquitination of SOCS1/JAK1 immune complex induced by lenalidomide and pomalidomide (Figure 4E). These data indicate that lenalidomide and pomalidomide may modulate SOCS1 expression at the transcriptional, rather than translational, level.
IMiDs enhance CTL-mediated cytotoxicity against SOCS1 re-expressing MM cells
We hypothesized that loss of SOCS1 expression by promoter hypermethylation leading to increased IL-6 signaling in MM cells may be contributing to the decreased susceptibility of tumor cells to CTL-mediated lysis. To address our hypothesis, we evaluated MM cell–specific cytotoxic T cell activity against SOCS1 gene-silenced and –re-expressing target MM cells by 51Cr-release assay (Figure 5A). MM cell–specific CTLs were pretreated with lenalidomide or pomalidomide. To induce re-expression of SOCS1 gene in MM target cells, they were preincubated in the absence or presence of lenalidomide, pomalidomide, AzaC, or TsA, alone or in com-bination. 51Cr-release cytotoxicity assay demonstrated that CTL-mediated lysis was significantly increased against SOCS1 re-expressing MM cells compared with SOCS1 gene silenced MM cells. Moreover, there was significantly increased CTL-mediated lysis of target MM cells treated with lenalidomide and AzaC (77% killing) or TSA (70% killing) combination compared with lenalidomide alone (58% killing; Figure 5A top panel). Similarly, pomalidomide pretreatment induced more potent CTL cytotoxic activity against MM cells in the presence of pomalidomide and AzaC (83% killing) or TSA (71% killing) than pomalidomide alone (68% killing; Figure 5A bottom panel). In addition, CFSE flow cytometric analysis demonstrated that incubation with lenalidomide and pomalidomide in combination with AzaC or TsA markedly increased CD8T cell proliferation compared with treatment with lenalidomide or pomalidomide alone (Figure 5B). In addition to enhancing CTL proliferation and CTL-mediated tumor cell lysis, lenalidomide and pomalidomide in combination with AzaC directly inhibited MM cell proliferation (Figure 5C).
IMiDs enhance CTL-mediated cytotoxicity against SOCS1-expressing MM cells. (A) Specific cytotoxic T cell activity against SOCS1 gene re-expressing target MM cell lines MM.1S and U266 were evaluated by 51Cr-release assay. To re-express SOCS1 gene, target MM cell lines were incubated either alone or in combination with AzaC, TsA, lenalidomide, or pomalidomide. MM cell–specific CTLs were generated from healthy donors by restimulation with γ-irradiated MM1.S or U266 apoptotic bodies using dendritic cells. CTLs were pretreated with lenalidomide or pomalidomide for 24 hours before use as effector cells against SOCS1 gene re-expressing MM cells in 51Cr-release assay. x-axis represents ratio of effector (IMiDs-pretreated MM cell–specific CTLs) to target SOCS1 gene re-expressing MM cells. y-axis represents specific lysis (percent) of target cells by effector cells. Top panel shows lenalidomide pretreated effector CTL lytic activity against SOCS1 gene re-expressing MM1.S cells; bottom panel shows pomalidomide-pretreated effector CTL lytic activity against SOCS1 gene re-expressing MM1.S cells. One representative of 3 independent experiments is shown. (B) Proliferation of CD8T cells cultured with the combination of lenalidomide and pomalidomide with AzaC or Tsa compared with lenalidomide or pomalidomide alone was evaluated by CFSE-flow cytometric analysis. x-axis represents CFSE stained proliferating cells, y-axis represents gated CD8+ T cells. Data are shown as histo-gram proliferation plots using Flowjo analysis software. One representative of 3 independent experiments is shown. (C) Direct antitumor activity of lenalidomide and pomalidomide, either alone or in combination with AzaC, was shown by 3[H]thymidine incorporation assay in MM1.S and U266 cells at 96 hours of culture. Data represent mean ± standard deviation of triplicate cultures. (*) indicates statistical significance determined by Student t test, 1-tailed distribution P < .01. One representative of 3 separate experiments is shown.
IMiDs enhance CTL-mediated cytotoxicity against SOCS1-expressing MM cells. (A) Specific cytotoxic T cell activity against SOCS1 gene re-expressing target MM cell lines MM.1S and U266 were evaluated by 51Cr-release assay. To re-express SOCS1 gene, target MM cell lines were incubated either alone or in combination with AzaC, TsA, lenalidomide, or pomalidomide. MM cell–specific CTLs were generated from healthy donors by restimulation with γ-irradiated MM1.S or U266 apoptotic bodies using dendritic cells. CTLs were pretreated with lenalidomide or pomalidomide for 24 hours before use as effector cells against SOCS1 gene re-expressing MM cells in 51Cr-release assay. x-axis represents ratio of effector (IMiDs-pretreated MM cell–specific CTLs) to target SOCS1 gene re-expressing MM cells. y-axis represents specific lysis (percent) of target cells by effector cells. Top panel shows lenalidomide pretreated effector CTL lytic activity against SOCS1 gene re-expressing MM1.S cells; bottom panel shows pomalidomide-pretreated effector CTL lytic activity against SOCS1 gene re-expressing MM1.S cells. One representative of 3 independent experiments is shown. (B) Proliferation of CD8T cells cultured with the combination of lenalidomide and pomalidomide with AzaC or Tsa compared with lenalidomide or pomalidomide alone was evaluated by CFSE-flow cytometric analysis. x-axis represents CFSE stained proliferating cells, y-axis represents gated CD8+ T cells. Data are shown as histo-gram proliferation plots using Flowjo analysis software. One representative of 3 independent experiments is shown. (C) Direct antitumor activity of lenalidomide and pomalidomide, either alone or in combination with AzaC, was shown by 3[H]thymidine incorporation assay in MM1.S and U266 cells at 96 hours of culture. Data represent mean ± standard deviation of triplicate cultures. (*) indicates statistical significance determined by Student t test, 1-tailed distribution P < .01. One representative of 3 separate experiments is shown.
Discussion
MM is characterized by immune defects that may predispose to infections causing morbidity and mortality, as well as tumor growth and resistance to chemotherapy. Pathogenesis of immune defects in MM is not well defined, but direct contact between MM cells and immune cells or BMSCs, as well as cytokines in the microenvironment, play a role. Impaired differentiation and function of B cells, T cells, NKT cells, NK cells, and dendritic cells have been identified in MM.30,33,34 In addition, decreased numbers of circulating CD19B cells, abnormal T cell subsets with an inversion of CD4:CD8 T cells and Th1:Th2 ratio, as well as increased inhibitory T cell subsets have also been demonstrated in MM.31-33,35-37 Interaction between the tumor cell and BM cellular or microenvironment is crucial for the development and progression of MM.38,39 Even though MM cell-BMSCs interaction has been extensively studied, tumor cell interaction with other BM accessory cells (particularly immune cells) and associated sequelae due to cytokines in the MM-BM milieu are not fully defined. BMSCs, via adhesion molecules and cytokines, provide a protective and supportive environment for tumor cell survival, growth, angiogenesis, drug resistance, and abnormal bone metabolism in MM.1,38-40 For example, immunosuppressive cytokines including IL-6, IL-10, transforming growth factor-β, vascular endothelial growth factor, and metalloproteinases are important regulatory factors within the BM microenvironment.2,40 As in other cancers, cytokine receptor/JAK/STAT pathway is constitutively activated in MM, leading to tumor cell proliferation, mobility, drug resistance, and protection against antitumor immunity.1,6,11 Disruption of BM cellular and cytokine support for tumor cells within the BM milieu is therefore essential to provide more effective novel therapeutic approaches in MM. In this context, immunomodulatory drugs have been exploited to repair immune defects and thereby enhance direct as well as immunomodulatory antitumor activity in MM. In this study, we assessed the effects of IMiDs lenalidomide and pomalidomide on cytokine signaling pathway in immune effector cells within the MM-BM or -PB microenvironment.
T cell activation and proliferation require a secondary costimulation in addition to activation via TCR. In MM, decreased expression of positive costimulatory signaling molecule CD28, as well as down-regulation of its signaling molecules p56Lck, p59Fyn, PI3K, and ZAP70, have been shown.29,41,42 We have previously demonstrated that CD3 stimulation is necessary for lenalidomide to induce T cell proliferation and secretion of IFNγ. In addition, we have shown that lenalidomide triggers tyrosine phosphorylation of CD28 and its downstream signaling molecule nuclear factor κB in anti-CD3 Ab prestimulated purified healthy donor T cells.28 Here we show that both lenalidomide and pomalidomide induce cell surface expression not only of CD28, but also of other positive costimulatory molecules ICOS and ICOSL, in T cells. ICOS and its ligand ICOSL, members of the CD28 family of costimulatory molecules, are expressed primarily on activated T cells. ICOS regulates T-cell–mediated immune responses by controlling T:B cell interactions, as well as both Th1 and Th2 cell activation and effector function.43 In addition to T cell activation and proliferation, up-regulation of CD28, ICOS, and ICOSL by IMiDs suggests that lenalidomide and pomalidomide may also regulate humoral immune responses in MM. We and others have also shown that thalidomide and lenalidomide induce cell proliferation of purified T cells.2,28 Here we demonstrate that IMiDs not only induce proliferation of purified T cells, but also induce proliferation of other effector cells including NKT cells and NK cells. In addition, lenalidomide and pomalidomide induce both IL-2 and IFNγ, but abrogate IL-6 expression in MM-PB or -BM milieu. While inducing activation and proliferation of effector cells as in other studies,44 IMiDs here also reduced inhibitory T cell populations in the MM-BM milieu, including regulatory Th cells (Treg) and IL-17 producing Th cells (Th17), indicating a positive regulatory function in enhancing anti-MM immune response.
Immune cell derived cytokines IL-2 and IFNγ mediate T-cell activation, proliferation, and antitumor immune response via JAK/STAT cytokine signaling pathway. IFNγ signaling is mediated through receptor bound JAK1, JAK2, and STAT1, whereas IL-2 signaling is mediated through JAK1, JAK3, and STAT1, and STAT5 transducers in immune effector cells. However, IL-6 signaling is mediated by JAK1, JAK2, and STAT1, STAT3 transducers.45 Even though each cytokine is regulated by its own specific negative feedback mechanism, SOCSs, to prevent excessive cytokine production and signaling, SOCS1 is the common suppressor of cytokine signaling molecule for IL-2, IFNγ, and IL-6.6,10,13-18,20-23,45-47 Because immunomodulatory drugs mainly regulate IL-2, IFNγ, and IL-6 secretion in MM, we determined whether lenalidomide and pomalidomide might be regulating these cytokines in effector cells via SOCS1. We observed that lenalidomide and pomalidomide markedly reduced SOCS1 expression in all effector cells in normal and MM-BM, as well as in PB. Because functional and developmental immune defects are characteristic of MM, we performed coculture experiments using normal donor effector cells to elucidate the direct impact of lenalidomide and pomalidomide on SOCS1 expression in effector cells. In these cocultures, we showed that MM cells, more prominently MM cells in the presence of MM-BMSCs, induced high levels of SOCS1 expression in healthy effector cells; interestingly, both lenalidomide and pomalidomide reduced SOCS1 expression in normal donor effector cells in these cocultures. Moreover, lenalidomide and pomalidomide also reduced SOCS1 expression in effector cells that were stimulated with IFNγ to exogenously overexpress SOCS1, indicating the specificity of the effect. Consequently, lenalidomide and pomalidomide enhanced phosphorylation of STAT1, a direct target molecule of SOCS1, to inhibit JAK/STAT signaling in all effector cells.
To elucidate the role of SOCS1 regulated-cytokine pathway in effector cell-MM cell interaction in the MM-BM environment, we assessed effector cell function against MM cells. SOCS1 regulated immune suppressor cytokine IL-6 is essential for survival and growth of MM cells.21-24 It has been shown that SOCS1 gene is silenced in 75% of MM patients by DNA hypermethylation of CpG dinucleotides in the promoter region.25-27 Silencing of SOCS1 genes by hypermethylation may result in uncontrolled IL-6 signaling, leading to MM cell growth and immune suppression. Even though it has been shown that re-expression of SOCS1 gene using SOCS1 expressing viral-vectors has suppressed IL-6 downstream signaling leading to anti-MM response,24 the role of SOCS1 regulating anti-MM immune response remained unknown. However, ectopic expression of SOCS1 in MM as well as other tumors indicated that SOCS1 has a tumor suppressor activity.48 In this study, using myeloma-specific CTLs, we determined that SOCS1 gene was re-expressed in target MM cell lines by lenalidomide or pomalidomide, either alone or in combination with AzaC, TsA, and CTLs, particularly lenalidomide- or pomalidomide-pretreated CTLs, had more potent anti-MM cytotoxic activity against SOCS1 re-expressing MM cell lines compared with SOCS1 gene–silenced MM cell lines. It has been shown that demethylating agent AzaC and selective inhibitor of class I and II histone deacetylases TsA, alone and more potently in combination, induce re-expression of epigenetically silenced SOCS1 in MM.49 Here, we determined that lenalidomide and pomalidomide alone were able to demethylate and induce SOCS1 gene transcription in MM cells. Our data suggest that, re-expression of SOCS1 in addition to suppression of IL-6 signaling in MM cells, also enhanced susceptibility of MM cell lines to CTL-mediated cytotoxicity. The observation that both lenalidomide and pomalidomide effects were more potent in inducing SOCS1 gene demethylation and expression in the presence of TsA indicates that promoter methylation and histone deacetylation work together to shut down the gene. Moreover, as in previous studies,24 re-expression of SOCS1 may be involved in direct anti-MM activity of AzaC by suppressing IL-6 signaling in MM. Interestingly, lenalidomide and pomalidomide in combination with AzaC-enhanced T-cell proliferation compared with lenalidomide or pomalidomide alone, suggesting that combination of IMiDs with AzaC might be balancing SOCS1-mediated negative feedback mechanism of action in T cells, resulting in T-cell proliferation and activation. SOCS inhibits JAK/STAT cytokine pathway by competing with JAK/STAT binding sites, acting as an ubiquitin E3 ligase, or by inducing JAK degradation via proteasomes.14,20,46,48 Finally, even though lenalidomide and pomalidomide induced ubiquitination of SOCS1, there was decreased ubiquitination of SOCS1/JAK1 protein complex in the proteasome activity inhibited effector cells. In addition, we observed that lenalidomide and pomalidomide, alone or in combination with AzaC and TsA, induced SOCS1 mRNA expression in MM cells, but reduced SOCS1 mRNA expression in effector cells. Therefore, these data show that lenalidomide and pomalidomide modulate SOCS1 expression at the transcriptional level rather than translational level through proteasomal degradation. Lenalidomide and pomalidomide show differential SOCS1-mediated regulatory functions in MM cells and effector cells. While there was a decreased SOCS1 expression in SOCS1 gene unmethylated effector cells, lenalidomide and pomalidomide induced demethylation of SOCS1 gene promoter in tumor cells thereby resulting in increased SOCS1 gene expression. These differential effects on MM cells and effector cells may be caused by epigenetic changes in tumor cells but not in effector cells. Demethylation of hypermeth-ylated genes may become a target to restore gene expression, whereas unmethylated or hypomethylated genes may not be targeted thereby resulting in decreased IL-6 in SOCS1 gene hypermethylated tumor cells, as well as increased IL-2 and IFNγ in SOCS1 gene unmethylated effector cells.
Taken together, our data demonstrate that tumor suppressor molecule SOCS1 plays an important role in the tumor cell-immune cell-BM microenvironment interaction in MM. Importantly, lenalidomide and pomalidomide induce epigenetic modifications of SOCS1 gene in MM cells, as well as modulate cytokine signaling via SOCS1-mediated cytokine signaling in effector cells. Furthermore, characterization of the molecular mechanisms of immunomodulatory effects of lenalidomide or pomalidomide on immune cells in the BM environment will both define their role in disease pathogenesis and suggest further novel immune-based targeted therapies. For example, further studies assessing the combination of IMiDs with epigenetic modulating drugs, such as histone deacetylase inhibitors and AzaC, may provide more potent immunomodulatory therapy in MM. Finally, given the new promising clinical activity of pomalidomide even in lenalidomide refractory myeloma,50 ongoing efforts are attempting to delineate direct and epigenetic mechanisms to account for this important difference.
The online version of this article contains a data supplement.
The publication costs of this article were defrayed in part by page charge payment. Therefore, and solely to indicate this fact, this article is hereby marked “advertisement” in accordance with 18 USC section 1734.
Acknowledgments
We thank Dr Robert Knight, Dr Peter Schafer, and Dr Mindy Yang (Celgene) for their invaluable support and for providing IMiDs.
This study was supported by National Institutes of Health/National Cancer Institute (NIH/NCI) Specialized Program of Research Excellence in Myeloma P50 CA100707 (K.C.A.); NIH/NCI Host-Tumor Cell Interactions in Myeloma: Therapeutic Applications P01 CA78378 (K.C.A.), and NIH/NCI Molecular Sequelae of Myeloma-Bone Marrow Interactions: Therapeutic Applications R01 CA50947 (K.C.A.) grants. Lenalidomide and pomalidomide were provided by Celgene.
National Institutes of Health
Authorship
Contribution: G.G. designed the research; G.G., E.C., E.S., T.H., G.P., M.B., Y.H., D.C., L.S., N. Mimura, and C.F. performed experiments; N.R., N. Munshi, P.R., and K.C.A. provided the clinical samples; Y.T.-T. and S.N. distributed the clinical samples; G.G. analyzed results and made figures; and G.G. and K.C.A. wrote the paper.
Conflict-of-interest disclosure: K.C.A. is on the Board of Directors or advisory committees for Celgene, Millennium, and Novartis. N.M. is on the Board of Directors or advisory committees for Celgene, Millennium, and Novartis. P.R. is on the Board of Directors or advisory committees of Celgene, Millennium, and Johnson & Johnson. N.R. has received research grants from Amgen, Celgene, and Novartis and has consulted for Astrazeneca. The remaining authors declare no competing financial interests.
Correspondence: Güllü Görgün, Dana-Farber Cancer Institute, Department of Medical Oncology, 44 Binney St, Rm M557, Boston, MA 02115; e-mail: gullu_gorgun@dfci.harvard.edu.



![Figure 5. IMiDs enhance CTL-mediated cytotoxicity against SOCS1-expressing MM cells. (A) Specific cytotoxic T cell activity against SOCS1 gene re-expressing target MM cell lines MM.1S and U266 were evaluated by 51Cr-release assay. To re-express SOCS1 gene, target MM cell lines were incubated either alone or in combination with AzaC, TsA, lenalidomide, or pomalidomide. MM cell–specific CTLs were generated from healthy donors by restimulation with γ-irradiated MM1.S or U266 apoptotic bodies using dendritic cells. CTLs were pretreated with lenalidomide or pomalidomide for 24 hours before use as effector cells against SOCS1 gene re-expressing MM cells in 51Cr-release assay. x-axis represents ratio of effector (IMiDs-pretreated MM cell–specific CTLs) to target SOCS1 gene re-expressing MM cells. y-axis represents specific lysis (percent) of target cells by effector cells. Top panel shows lenalidomide pretreated effector CTL lytic activity against SOCS1 gene re-expressing MM1.S cells; bottom panel shows pomalidomide-pretreated effector CTL lytic activity against SOCS1 gene re-expressing MM1.S cells. One representative of 3 independent experiments is shown. (B) Proliferation of CD8T cells cultured with the combination of lenalidomide and pomalidomide with AzaC or Tsa compared with lenalidomide or pomalidomide alone was evaluated by CFSE-flow cytometric analysis. x-axis represents CFSE stained proliferating cells, y-axis represents gated CD8+ T cells. Data are shown as histo-gram proliferation plots using Flowjo analysis software. One representative of 3 independent experiments is shown. (C) Direct antitumor activity of lenalidomide and pomalidomide, either alone or in combination with AzaC, was shown by 3[H]thymidine incorporation assay in MM1.S and U266 cells at 96 hours of culture. Data represent mean ± standard deviation of triplicate cultures. (*) indicates statistical significance determined by Student t test, 1-tailed distribution P < .01. One representative of 3 separate experiments is shown.](https://ash.silverchair-cdn.com/ash/content_public/journal/blood/116/17/10.1182_blood-2010-04-279893/4/m_zh89991059360005.jpeg?Expires=1769135858&Signature=icLHM~xklhoawxdspEGupnwGQKgVufXg-gAkQEbKevQSQppMj~eBVgCJvbk5PPOuyJwoZP-Yr5DGIDP-9o0Q37NTnp~6x~xoP-GKYc~tSJ9Rxz1DksjGHanbBWzPNJTaqmuASoyaq1e2N6cS~JbMUBCTgxF~lsRdlrejIrcdcPh5ZFsmf6cuoHaDpqOaxatSGCLdBwCBzy1DDzJUl7mkC5C7m9~6T872IuqtUnIdyYU6OOEmffCkvei0WV0R2eDmf-ZLG2mkmoLkFespyBqPH80FDNqQYtXY6uloUXy6YMC~B0b6vsUy4igOGeOws0xcL7MxVFm7PqZd6h7OIeXU2Q__&Key-Pair-Id=APKAIE5G5CRDK6RD3PGA)
This feature is available to Subscribers Only
Sign In or Create an Account Close Modal