Abstract
Survivin and XIAP, members of the protein family known as the inhibitors of apoptosis, interfere with the activation of caspases, called the “cell death executioners.” We examined Survivin (n = 116) and XIAP (n = 172) expression in primary acute myeloid leukemia (AML) blasts and assessed the impact of their expression on prognosis. They were detected in all samples analyzed. However, no correlation was observed with cytogenetics, remission attainment, or overall survival of patients with AML. To investigate the importance of caspases in chemotherapy-induced apoptosis in AML, we treated OCI-AML3 cells with Ara-C, doxorubicin, vincristine, and paclitaxel, which induced caspase cleavage and apoptosis. Blocking of caspase activation by pan-caspase inhibitor abolished poly(adenosine diphosphate [ADP]-ribose) polymerase cleavage and DNA fragmentation but did not prevent chemotherapy-induced cell death and did not inhibit, or only partially inhibited, mitochondrial release of cytochrome c, Smac, apoptosis-inducing factor (AIF), or loss of mitochondrial membrane potential. Caspase inhibition also did not protect AML blasts from chemotherapy-induced cell death in vitro. These results suggest that expression levels of Survivin or XIAP have no prognostic impact in AML patients. Although anticancer drugs induced caspase cleavage and apoptosis, cell killing was caspase independent. This may partially explain the lack of prognostic impact of XIAP and Survivin and may suggest caspase-independent mechanisms of cell death in AML. (Blood. 2003;102:4179-4186)
Introduction
Apoptosis is genetically programmed cell death regulated by multiple proteins. Inhibitors of apoptosis proteins (IAPs) are a family of related proteins that suppress cell death by inhibiting upstream and terminal caspases.1-8 These proteins suppress apoptosis induced by a variety of stimuli, including tumor necrosis factor, Fas, menadione, staurosporine, etoposide, paclitaxel, and growth factor withdrawal.1,4,5,9 The expression of at least some IAPs is up-regulated by various growth factors10-12 supporting their roles in survival. Among them, Survivin is considered important because it links cell death and cell proliferation.13,14 Survivin is not expressed in adult terminal differentiated tissues, but it is expressed in most human cancers and cancer cell lines.1,13 Disruption of Survivin expression causes cell death and cell division defects, which result in polyploidy and multinucleated cells.15-17 Studies from different groups have demonstrated that the elevated expression of Survivin is associated with poor prognosis and increased tumor recurrence in many cancers18-21 and in acute myelogenous leukemia (AML).22 Our previous studies showed that Survivin is overexpressed in the blasts of AML patient samples analyzed.10,23
XIAP is the most widely expressed IAP. It is also the most potent inhibitor of caspases, with an inhibition constant (Ki) in the low nanomolar range.2,3,24 Elevated expression of XIAP has been reported in certain cancers and cancer cell lines.25,26 Down-regulation of XIAP has been shown to induce apoptosis in chemoresistant human ovarian cancer cells27 and to sensitize non-small cell lung carcinoma to low-dose γ-irradiation.28 We recently reported variable expression levels of XIAP in primary AML samples and found its expression inversely correlated with the survival of patients with AML.29
Most current anticancer therapies, including chemotherapy, trigger tumor cell death by inducing apoptosis. Caspases are believed to function as the central mediators of apoptosis. On activation, they selectively cleave a variety of key structural proteins of the cytoskeleton and nucleus and numerous proteins involved in DNA repair and signaling pathways, resulting in DNA fragmentation.30 Inhibiting caspase activation has been shown to prevent apoptosis.30,31 This is supported by the fact that the overexpression of IAPs inhibits apoptotic cell death.1-5 However, recent studies suggest that caspase activation is not the only pathway by which anticancer drugs induce cell death. Anticancer drugs can also induce caspase-independent cell death32 and necrosis.33 Recently, a number of caspase-independent cell death factors, which are released from mitochondria along with cytochrome c, have been described, among them apoptosis-inducing factor (AIF),34-36 endonuclease, EndoG,37 and HtrA2 (omi), which may be able to trigger caspase-independent cell death through its intrinsic serine protease activity.38-41 Caspase-independent cell death has not been carefully studied in leukemia.
To assess the clinical significance of caspase regulators and their role in AML, we examined Survivin and XIAP expression in AML and correlated their expression with clinical outcome. XIAP expression was also correlated with other proteins whose expressions affect cell survival in the same samples. We then investigated the importance of caspase-dependent apoptosis in chemotherapy-induced cell death by inhibiting caspase activation. We here report that the expression levels of neither Survivin nor XIAP are associated with prognosis in AML and that cell death induced by chemotherapy in AML can be triggered independently of caspase activation.
Materials and methods
Cells and cell cultures
Human leukemic cells OCI-AML3 were cultured in RPMI 1640 medium containing 10% heat-inactivated fetal calf serum (FCS), 1 mM l-glutamine, and 50 μg/mL penicillin/streptomycin. Bone marrow specimens and peripheral blood from patients with AML with high (more than 70%) blast counts were obtained after signed, informed consent, according to institutional guidelines. Mononuclear cells were purified by Ficoll-Hypaque (Sigma Chemical, St Louis, MO) density-gradient centrifugation and were recultured in AIM-V medium (Gibco-BRL, Gaithersburg, MD) supplemented with 10% FCS, 100 U/mL granulocyte-macrophage colony-stimulating factor (GM-CSF) (Immunex, Seattle, WA), and 100 ng/mL stem cell factor (Amgen, Thousand Oaks, CA).
Treatment of cells
OCI-AML3 cells at a density of 1 × 106/mL were treated with various anticancer drugs, including Ara-C (2 μM), doxorubicin (Dox) (500 ng/mL), vincristine (10 ng/mL), and paclitaxel (Taxol; Bristol-Myers Squibb, Princeton, NJ) (50 ng/mL) for up to 48 hours. Drug concentrations were based on their IC50 and were adjusted experimentally. IDN-1529 and IDN-1965 are 2 irreversible, cell-permeable, broad-spectrum caspase inhibitors developed by Idun Pharmaceuticals (La Jolla, CA).42 For caspase inhibition, 20 μM IDN-1529, 20 μM IDN-1965, or 50 μM Z-VAD-FMK (Trevigen, Gaithersburg, MD) was added to the cells 1 hour before drug administration. Experiments were conducted at least in triplicate.
Cell death assays
Cell viability was determined by trypan blue exclusion. Early apoptotic events were detected by the flow cytometric measurement of externalized phosphatidylserine43 with the Annexin-V-FLUOS Staining Kit (Roche Diagnostics, Indianapolis, IN). To measure mitochondrial membrane potential (Δψm), cells were loaded with CMXRos (300 nM) and MitoTracker Green (100 μM; both from Molecular Probes, Eugene, OR) for 1 hour at 37°C. The Δψm was then assessed by measuring CMXRos retention (red fluorescence) while simultaneously adjusting for mitochondrial mass (green fluorescence).44 For the DNA fragmentation assay, cells were washed twice with phosphate-buffered saline (PBS) and resuspended in lysis buffer (10 mM Tris-HCl, 1 mM EDTA [ethylenediaminetetraacetic acid], 0.025% Triton X-100, pH 8.0) for 10 to 20 minutes on ice. Lysates were digested with RNase A (50 μg/mL) and then proteinase K (2 mg/mL) for 1 hour each at 37°C. DNA samples were separated on 1.8% agarose gel containing ethidium bromide and visualized by UV light.
Determination of intracellular localization of AIF
Cells were placed on slides by cytospin and fixed with 4% paraformaldehyde in PBS for 10 minutes at room temperature. Cells on slides were permeabilized with TBST buffer (10 mM Tris, 15 mM NaCl, 0.1% Triton X-100) for 15 minutes (3 times TBST, 5 minutes each) and rinsed with TBS buffer for 5 minutes. Next cells were blocked with 5% goat serum in PBS for 30 minutes and incubated with anti-AIF antibody (1:100; Santa Cruz Biotechnology, Santa Cruz, CA) overnight at 4°C in a humidified chamber. Excess antibody was removed by washing the slides with PBS. Then cells were incubated with secondary antibody, fluorescein isothiocyanate (FITC)-labeled swine antigoat IgG (H+L, Calgat, Burlingame, CA; 1:250), for 30 minutes at 37°C. After washing with PBS, cells were analyzed using a fluorescence microscope and a confocal laser scanning biologic microscope (Fluoview/FV500; Olympus America, Melville, NY). To-Pro 3 (Molecular Probes) was used for nuclei staining.
Immunoblotting
Cells were washed twice with PBS buffer and lysed at a density of 2 × 104 cells/μL in 2 × protein lysis buffer (0.25 M Tris-HCl, 2% sodium dodecyl sulfate [SDS], 4% β-mercaptoethanol, 10% glycerol, 0.02% bromophenol blue). An equal amount of cell lysate (equivalent to 2-3 × 105 cells) was loaded onto a 12% sodium dodecyl sulfate-polyacrylamide gel electrophoresis (SDS-PAGE) gel (Bio-Rad, Hercules, CA). To analyze Smac and cytochrome c release from mitochondria, cells were suspended in an ice-cold buffer (25 mM Tris and 5 mM MgCl2, pH 7.4) and centrifuged for 5 minutes at 16 000g to isolate cytosolic lysates. Supernatants were mixed with 2 × protein lysis buffer and subjected to 15% SDS-PAGE. After electrophoresis, the proteins were transferred to Hybond-P membranes (Amersham Pharmacia Biotech, Buckinghamshire, England). Membranes were blotted overnight at 4°C with PBS buffer containing 7% nonfat dry milk (Bio-Rad) and 0.3% Tween-20, followed by incubation with the monoclonal poly(adenosine diphosphate [ADP]-ribose) polymerase (PARP) antibody (1:2000; PharMingen, San Diego, CA); polyclonal caspase-3 antibody (CPP32, 1:1000; PharMingen); caspases-7, -8, and -9 antibodies (1:1000; Cell Signaling Technology, Beverly, MA); caspase-2 antibody (1:2000; kindly provided by Dr Y. Lazebnik, Cold Spring Harbor Laboratory, Cold Spring Harbor, NY); cytochrome c antibody (1:250; PharMingen); Smac antibody (1:2000; kindly provided by Dr Xiaodong Wang, University of Texas Medical Center, Dallas); omi antibody (1:2000; gift of Dr E.S. Alnemri, Kimmel Cancer Institute, Thomas Jefferson University, Philadelphia, PA); or anti-β-actin (Sigma Chemical) for 2 hours at room temperature. Membranes were washed 3 times with PBS buffer containing 0.3% Tween-20 and were probed with a second antibody. After washing, the membranes were reacted with ECLTH chemiluminescent Western Blotting Detection Reagents (Amersham Pharmacia Biotech), and the signals were detected by PhosphoImager Storm 860 (Version 4.0; Molecular Dynamics, Sunnyvale, CA) and quantitated by Scion Image software (Scion, Frederick, MD). Beta-actin was used as loading control.
Survivin expression in 116 AML samples was analyzed as described previously.10 For analyses of XIAP (antibody from Transduction Laboratories [Lexington, KY] and PharMingen), total ERK2, (antibody from Santa Cruz Biotechnology) and phosphorylated ERK2 (pERK2; antibody from Cell Signaling Technology) from the 172 AML patient samples, immunoblotting was performed on cell lysates derived from the Ficoll-Hypaque-generated mononuclear fraction of peripheral blood with high blast counts, as described previously.29 After applying secondary antibodies and subsequent washing, membranes were exposed to SuperSignal West Dura extended-duration substrate (Pierce, Rockford, IL) for 5 minutes in accordance with manufacturer's recommendations, and the blots were then directly imaged and quantitated in a ChemiImager 4400 low-light imaging system (Alpha Innotech, San Leandro, CA). Survivin, XIAP, total ERK2, and pERK2 expression levels were determined by PhosphoImager Storm 860 and were quantitated by Scion Image software using β-actin as a loading control or determined by densitometry and were normalized against the respective signals from HeLa cells for possible variations caused by differences in antibody concentration or length of blot exposure to x-ray film.
Results
Survivin expression and prediction of clinical outcome in primary AML
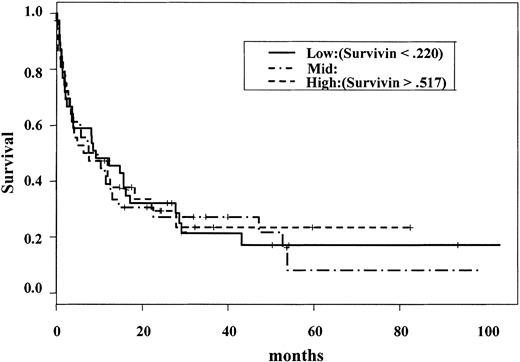
We previously compared Survivin expression in normal CD34+ cells (n = 7) and blasts from AML patients (n = 18) and found that Survivin is overexpressed in AML.10 Here we analyzed Survivin expression in 116 AML samples to determine whether there was any significant effect of Survivin level on clinical outcome of AML patients. Immunoblotting analyses showed that Survivin was expressed in the blast cells of all 116 AML patients. However, there was no correlation of Survivin level with age, white blood cell count (WBC), blood or bone marrow blast count, cytogenetics, complete remission (CR), CR duration, or survival of AML patients. In this group, age (P = .02), cytogenetics, and WBC count were of major significance (P = .00 by log rank test, P = .007 by Cox analysis, respectively) on survival, as expected. The risk for death increased as Survivin level increased, but this pattern was not statistically significant (log rank test, P = .944) (Figure 1). However, Martingale residual analysis identified a small group of patients with very high Survivin levels (n = 6) (exceeding the Survivin levels of HL-60 cells) at high risk for death, but this group was too small to be significant. Cox regression analysis also failed to identify prognostic significance for Survivin, whereas age, WBC count, and cytogenetics were significant. Among various French-American-British (FAB) groups, the difference in Survivin expression has borderline significance (P = .05). Patients with promyelocytic leukemia (FABM3) expressed lower levels of Survivin. Among 13 paired samples from peripheral blood and bone marrow, no significant difference in Survivin level was observed for samples from different sources.
Overall survival of primary AML compared with Survivin levels. Survivin levels were analyzed by Western blot in 116 primary AML samples and plotted against survival. The risk for death increased, but was not statistically significant, as Survivin level increased (log-rank test; P = .944).
Overall survival of primary AML compared with Survivin levels. Survivin levels were analyzed by Western blot in 116 primary AML samples and plotted against survival. The risk for death increased, but was not statistically significant, as Survivin level increased (log-rank test; P = .944).
Expression of XIAP protein in primary AML samples: assessment of prognostic impact
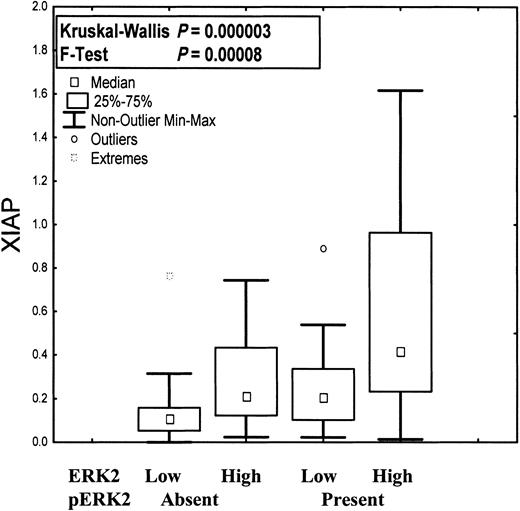
Our initial expression study on 76 patient samples suggested that XIAP protein levels were inversely correlated with survival in AML.29 To extend this study, we analyzed XIAP expression in additional AML patient blasts using immunoblotting analysis. Our results demonstrate that of 172 primary AML patient samples analyzed, all expressed XIAP protein but within a broad range. The range of XIAP expression was not correlated with sex, age, WBC count, percentage of blasts and promyelocytes, cytogenetics, performance status, history of antecedent hematologic disorder, remission, or survival. The variability of expression was wider in patients with FABM2 and significantly narrower among patients with promyelocytic leukemia (FABM3) or t(15;17). XIAP expression was strongly correlated with total ERK2 expression (r = 0.61; r2 = 0.4; P < .00001), and this correlation held regardless of whether pERK was present (r = 0.64; r2 = 0.41; P < .000001) or absent (r = 0.53; r2 = 0.29; P < .0000001) (Figure 2). XIAP protein levels, however, did not significantly correlate with pERK (r = 0.2; r2 = 0.04; P = .02). We generated r and r2 using a least-squares fit between 2 independent variables with Statistica version 6.1 (StatSoft, Tulsa, OK) and P using Pearson correlation. XIAP protein levels also did not correlate with levels of Rb, Bcl-2, Bax, caspase-2, caspase-3, or protein kinase C α (PKC-α), which had previously been measured in the same AML samples.45-48
Expression of XIAP, total ERK, and pERK in primary AML. XIAP expression as a function of ERK2 expression and ERK2 phosphorylation. XIAP was significantly more highly expressed in samples (n = 172) with high rather than low ERK2 levels. This correlation was independent of ERK2 phosphorylation status.
Expression of XIAP, total ERK, and pERK in primary AML. XIAP expression as a function of ERK2 expression and ERK2 phosphorylation. XIAP was significantly more highly expressed in samples (n = 172) with high rather than low ERK2 levels. This correlation was independent of ERK2 phosphorylation status.
Induction of caspase-independent cell death by anticancer agents
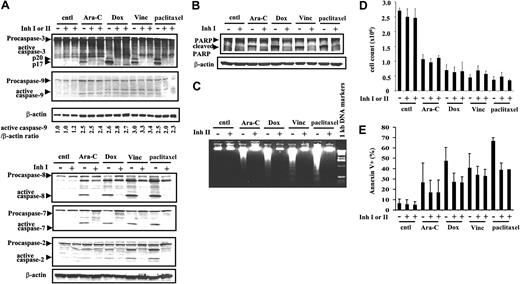
Because IAP blocks apoptosis by inhibiting caspases and because the overexpression of Survivin and XIAP has been shown to inhibit apoptosis induced by various stimuli,2,3,9 we questioned whether cells escape death if caspases, the executioners of cell death, are completely inhibited. To answer this, we treated OCI-AML3 cells with Ara-C, Dox, vincristine, and paclitaxel with or without exogenous caspase inhibitors. We then examined the activation of caspases, the cleavage of PARP (a caspase substrate), the fragmentation of DNA, which is catalyzed by caspase-activated DNase,49 and cell death in response to anticancer drugs. The treatment of OCI-AML3 cells with Ara-C (2 μM), Dox (500 ng/mL), vincristine (10 ng/mL), and paclitaxel (50 ng/mL) resulted in the activation of caspases-3, -9, -8, -7, and -2 (Figure 3A), PARP cleavage (Figure 3B), and DNA fragmentation (Figure 3C) at 48 hours. In the presence of caspase inhibitor IDN-1529 or IDN-1965, the activation of caspases-3, -8, -7, and -2 were inhibited (Figure 3A). Although caspase-9 was not inactivated under our experimental conditions (Figure 3A), the inhibition of caspases-3 and -7 was sufficient to block PARP cleavage and DNA fragmentation (Figure 3B-C), indicating blockade of the caspase-mediated apoptosis pathway. As expected, these anticancer drugs induced cell killing, as shown by cell counts (Figure 3D) and Annexin-V-FLUOS staining (Figure 3E) at 48 hours. Interestingly, cell death was not prevented by caspase inhibition when the number of viable cells was determined (Figure 3D), whereas Annexin-V positivity was suppressed partially (Figure 3E). Similar results were obtained at 24 hours, showing no effect of caspase inhibition. At 72 and 96 hours, no differences were found in the number of viable cells after drug treatment with or without the caspase inhibitor. By 96 hours, 18.3% or 19.1% of cells were viable when treated with Ara-C, and 11.3% or 11.7% were viable after Dox treatment, with or without the inhibitor, respectively. In vincristine- or paclitaxel-treated OCI-AML3 cells, less than 1% of viable cells were found with or without caspase inhibitor (results not shown). With a commercially available general caspase inhibitor, Z-VAD-FMK at 50 μM, caspase-3 activation was inhibited, but cell death was not prevented in OCI-AML3 cells treated with 500 ng/mL Dox (not shown). Our results suggest that a caspase-independent pathway is involved in drug-induced death of these leukemic cells. Externalization of phosphatidylserine (PS) as measured by annexin V binding was shown to be caspase dependent50 and is therefore not a good measure of cell death under these experimental conditions.
Effects of various drugs on caspase activation and cell death with or without caspase inhibitors. OCI-AML3 cells were treated with 2 μM Ara-C, 500 ng/mL Dox, 10 ng/mL vincristine (Vinc), or 50 ng/mL paclitaxel for up to 48 hours, with or without the caspase inhibitor IDN-1529 (Inh I) or IDN-1965 (Inh II) (20 μM). (A) Caspases-3, -9, -8, -7, and -2 processing at 48 hours, measured by immunoblotting. (B) Immunoblotting analysis of caspase-dependent PARP cleavage at 48 hours. (C) DNA fragmentation assay at 48 hours. (D) Cell counts at 48 hours. (E) Annexin V analysis at 48 hours. Results are expressed as mean ± SE. cntl indicates control.
Effects of various drugs on caspase activation and cell death with or without caspase inhibitors. OCI-AML3 cells were treated with 2 μM Ara-C, 500 ng/mL Dox, 10 ng/mL vincristine (Vinc), or 50 ng/mL paclitaxel for up to 48 hours, with or without the caspase inhibitor IDN-1529 (Inh I) or IDN-1965 (Inh II) (20 μM). (A) Caspases-3, -9, -8, -7, and -2 processing at 48 hours, measured by immunoblotting. (B) Immunoblotting analysis of caspase-dependent PARP cleavage at 48 hours. (C) DNA fragmentation assay at 48 hours. (D) Cell counts at 48 hours. (E) Annexin V analysis at 48 hours. Results are expressed as mean ± SE. cntl indicates control.
Participation of mitochondria in caspase-independent cell death induced by chemotherapeutic drugs
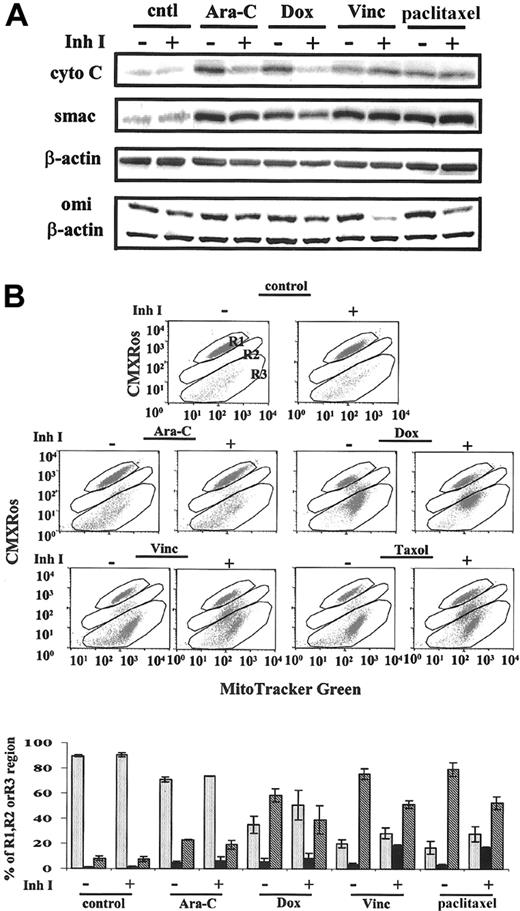
To examine the involvement of mitochondria in chemotherapy-induced cell death, we determined cytochrome c, Smac, and omi (HtrA2) release from mitochondria and changes in mitochondrial membrane potential (MMP) in OCI-AML3 cells on drug treatment with or without caspase inhibitors. As shown in Figure 4, these drugs induced the release of cytochrome c, Smac, and omi from mitochondria (Figure 4A) and the loss of MMP (Figure 4B) measured at 48 hours. In Ara-C- and Dox-treated OCI-AML3 cells, the release of cytochrome c and Smac was partially inhibited, whereas the decrease of MMP was not affected by the caspase inhibitor IDN-1529. In vincristine- and paclitaxel-treated cells, the release of cytochrome c and Smac was not blocked, whereas the loss of MMP was partially inhibited by the caspase inhibitor. Vincristine and paclitaxel, under the conditions used, affected a more complete depolarization of mitochondria (R3 = 75.6% ± 4.3% and 79.3% ± 5.4%, respectively; Figure 4B), and caspase inhibited, in part, the complete depolarization (R3 = 51.4% ± 3%; P = .025; and R3 = 52.7% ± 5.1%; P = .006, respectively). Caspase inhibitors partially prevented cytochrome c and Smac release when the MMP loss was less severe and did not block cytochrome c or Smac release when MMP loss was significant. The overall partial inhibition of MMP loss by caspase inhibition was not sufficient to prevent cytochrome c and Smac release when vincristine or paclitaxel induced significant damage to mitochondria. There was a basal level of omi in the cytosol without drug exposure. With drug treatment, omi release was increased (Figure 4A). Omi release was inhibited by the caspase inhibitor to basal levels in the Ara-C- and Dox-treated cells and to less than basal levels in vincristine- and paclitaxel-treated cells. Under the same conditions, the activation of caspase-8 and caspase-2, and the effector caspases-3 and -7 induced by these drugs, was inhibited, suggesting that though IAPs function by inhibiting caspases and increasing chemoresistance, when caspase-dependent apoptosis pathways are blocked, changes in mitochondria upstream of the caspase cascade, induced by chemotherapeutic agents, are sufficient to cause cell death. Omi release is probably caspase dependent in OCI-AML3.
Effects of various drugs on mitochondria with or without caspase inhibitor IDN-1529. OCI-AML3 cells were treated with 2 μM Ara-C, 500 ng/mL Dox, 10 ng/mL vincristine (Vinc), or 50 ng/mL paclitaxel for up to 48 hours, with or without the caspase inhibitor IDN-1529 (Inh I) (20 μM). (A) Immunoblotting analysis of cytochrome c, Smac, and Omi releases from mitochondria. (B) Changes in mitochondrial membrane potential determined by CMXRos and MitoTracker green. cntl indicates control; R1, cells maintaining Δψm; R2, cells partially losing Δψm; R3, cells losing Δψm. Results are expressed as mean ± SE.
Effects of various drugs on mitochondria with or without caspase inhibitor IDN-1529. OCI-AML3 cells were treated with 2 μM Ara-C, 500 ng/mL Dox, 10 ng/mL vincristine (Vinc), or 50 ng/mL paclitaxel for up to 48 hours, with or without the caspase inhibitor IDN-1529 (Inh I) (20 μM). (A) Immunoblotting analysis of cytochrome c, Smac, and Omi releases from mitochondria. (B) Changes in mitochondrial membrane potential determined by CMXRos and MitoTracker green. cntl indicates control; R1, cells maintaining Δψm; R2, cells partially losing Δψm; R3, cells losing Δψm. Results are expressed as mean ± SE.
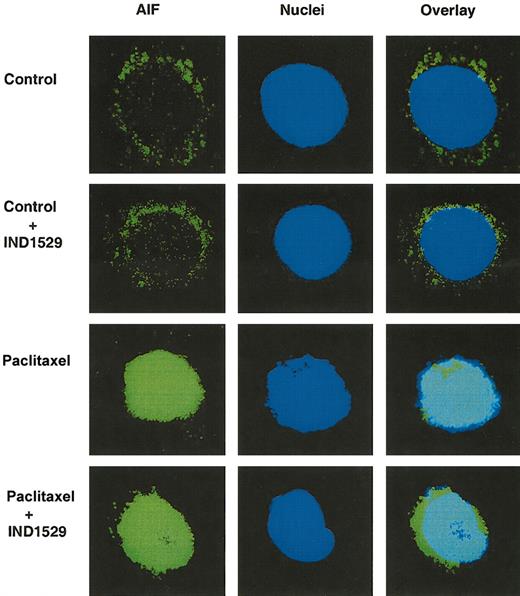
Chemotherapy-induced nuclear translocation of AIF is partially caspase independent
To understand the mechanism of mitochondrial participation of caspase-independent cell death, we analyzed the nuclear translocation of AIF after drug treatment with or without caspase inhibitor IDN-1529. In untreated OCI-AML3 cells with or without caspase inhibitor IDN-1529, AIF positivity was found in the perinuclear area. Strong, bright AIF immunostaining was observed in the nuclei of cells treated with Ara-C, Dox, vincristine, or paclitaxel. This nuclear translocation of AIF was observed in spite of caspase inhibition. Figure 5 shows a representative graph demonstrating nuclear translocation of AIF in paclitaxel-treated OCI-AML3 cells with or without IDN-1529. The percentages of AIF-positive nuclei, measured at 48 hours, in vincristine- (37.5% ± 3.8%) and paclitaxel- (47% ± 4.8%) treated cells were higher than those in Ara-C- (11.3% ± 4.4%) and Dox- (12.9% ± 4.1%) treated cells. This is in agreement with the increased loss of MMP in vincristine- and paclitaxel-treated cells compared with cells exposed to Ara-C and Dox. In the presence of IDN-1529, AIF nuclear translocation was partially inhibited in vincristine- (15.1% ± 2.3%; P = .0005), paclitaxel- (16.8% ± 5%; P = .0034), and Ara-C- (8.1% ± 4.1%; P = .005) treated cells. IDN-1529 also inhibited AIF translocation in Dox- (4.3% ± 1.2%) treated cells, but the inhibition was not significant (P = .097). These data indicate that AIF translocation is caspase dependent and independent. Caspase-independent AIF translocation could contribute to caspase-independent cell death induced by chemotherapeutic drugs in leukemic cells.
Effects of paclitaxel on nuclear translocation of AIF with or without caspase inhibitor IDN-1529. OCI-AML3 cells were treated with 50 ng/mL paclitaxel for up to 48 hours, with or without the caspase inhibitor IDN-1529 (Inh I) (20 μM). Cells were fixed, immunofluorescence stained, and analyzed using a fluorescence microscope and a confocal laser scanning biologic microscope. Images on the left show AIF immunofluorescence staining (FITC-green). Images in the center show nuclei stained with To-Pro 3. Images on the right demonstrate the overlay of AIF and nuclear staining. Green signals surrounding the nuclei represent the cytoplasmic AIF (control and control + IDN-1529 cells). The overlay of green and blue in the nuclei represents the nuclear AIF (Taxol and Taxol+IDN-1529-treated cells). Original magnification is × 90 for all images.
Effects of paclitaxel on nuclear translocation of AIF with or without caspase inhibitor IDN-1529. OCI-AML3 cells were treated with 50 ng/mL paclitaxel for up to 48 hours, with or without the caspase inhibitor IDN-1529 (Inh I) (20 μM). Cells were fixed, immunofluorescence stained, and analyzed using a fluorescence microscope and a confocal laser scanning biologic microscope. Images on the left show AIF immunofluorescence staining (FITC-green). Images in the center show nuclei stained with To-Pro 3. Images on the right demonstrate the overlay of AIF and nuclear staining. Green signals surrounding the nuclei represent the cytoplasmic AIF (control and control + IDN-1529 cells). The overlay of green and blue in the nuclei represents the nuclear AIF (Taxol and Taxol+IDN-1529-treated cells). Original magnification is × 90 for all images.
Caspase inhibition does not protect primary AML blasts cultured in vitro from chemotherapy-induced cell death
Purified primary bone marrow AML blasts were treated with various chemotherapeutic agents under the same conditions used in OCI-AML3 cells. Because of the limitation in cell numbers, not all samples could be treated with all 4 agents. Cell count, annexin V positivity, and MMP were measured at 48 hours. As shown in Table 1, each patient responded differently to various drugs. Patients 2, 3, and 4 were resistant to Ara-C and paclitaxel. Patients 1 and 5 responded to all drugs, and patient 4 responded to Dox treatment (Table 1, asterisk). Among those samples that showed responses to chemotherapy, AML blasts were not protected from death by caspase inhibition, as determined by cell count. Although the annexin positivity was partially inhibited, the loss of MMP was not or was partially prevented by caspase inhibition (Table 1). Results confirm those observed in OCI-AML3 cells.
Cell viability of bone marrow samples from primary AML blasts
. | % Blasts . | Control IDN-1529 . | . | Ara-C IDN-1529 . | . | Dox IDN-1529 . | . | Vincristine IDN-1529 . | . | Paclitaxel IDN-1529 . | . | |||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Bone marrow . | . | − . | + . | − . | + . | − . | + . | − . | + . | − . | + . | |||||
| Patient 1 | 91 | — | — | — | — | |||||||||||
| Cell count | 100 | 82.5 | 35.9 * | 32.0 * | 69.9 * | 66.0 * | ||||||||||
| Annexin V+ | 13.5 | 7.3 | 63.5 * | 50.0 * | 24.2 * | 13.3 * | ||||||||||
| CMXRos-MTG | ||||||||||||||||
| R1 | 73.5 | 77.7 | 16.9 * | 24.3 * | 59.0 * | 67.7 * | ||||||||||
| R2 | 3.7 | 4.8 | 4.3 * | 11.5 * | 5.5 * | 7.0 * | ||||||||||
| R3 | 21.1 | 15.0 | 77.1 * | 59.8 * | 33.4 * | 23.3 * | ||||||||||
| Patient 2 | 82 | — | — | — | — | |||||||||||
| Cell count | 100 | 92.4 | 82.3 | 96.8 | 83.1 | 85.8 | ||||||||||
| Annexin V+ | 13.4 | 7.1 | 14.6 | 14.7 | 10.3 | 10.3 | ||||||||||
| CMXRos-MTG | ||||||||||||||||
| R1 | 19.1 | 22.5 | 27.5 | 18.7 | 19.7 | 24.7 | ||||||||||
| R2 | 56.6 | 59.1 | 37.4 | 48.7 | 61.4 | 56.4 | ||||||||||
| R3 | 21.7 | 15.4 | 30.6 | 28.4 | 15.8 | 17.5 | ||||||||||
| Patient 3 | 78 | — | — | — | — | |||||||||||
| Cell count | 100 | 103.4 | 106.7 | 101.1 | 112.4 | 101.1 | ||||||||||
| Annexin V+ | 4.7 | 2.3 | 7.7 | 5.5 | 6.3 | 3.4 | ||||||||||
| CMXRos-MTG | ||||||||||||||||
| R1 | 86.8 | 82.9 | 85.9 | 86.3 | 89.0 | 84.2 | ||||||||||
| R2 | 6.7 | 13.2 | 8.2 | 8.4 | 6.5 | 11.9 | ||||||||||
| R3 | 2.6 | 2.3 | 4.0 | 3.4 | 3.3 | 2.4 | ||||||||||
| Patient 4 | 41 | — | — | |||||||||||||
| Cell count | 100 | 101.0 | 88.5 | 89.6 | 46.9 * | 57.3 * | 96.9 | 99.0 | ||||||||
| Annexin V+ | 15.4 | 14.6 | 15.8 | 16.4 | 36.6 * | 28.2 * | 14.5 | 11.3 | ||||||||
| CMXRos-MTG | ||||||||||||||||
| R1 | 95.8 | 95.3 | 93.3 | 94.0 | 89.0 * | 76.0 * | 96.0 | 94.8 | ||||||||
| R2 | 3.7 | 4.3 | 6.02 | 5.0 | 10.2 * | 23.4 * | 3.5 | 4.6 | ||||||||
| R3 | 0.01 | 0.03 | 0.07 | 0.08 | 0.01 * | 0.01 * | 0.11 | 0.11 | ||||||||
| Patient 5 | 82 | |||||||||||||||
| Cell count | 100 | 110.0 | 40.0 * | 39.0 * | 49.0 * | 47.0 * | 70.0 * | 65.0 * | 69.0 * | 68.0 * | ||||||
| Annexin V+ | 15.2 | 4.3 | 78.9 * | 30.6 * | 58.7 * | 30.5 * | 19.0 * | 13.2 * | 17.6 * | 13.1 * | ||||||
| CMXRos-MTG | ||||||||||||||||
| R1 | 93.6 | 93.6 | 8.7 * | 48.8 * | 41.3 * | 53.7 * | 78.2 * | 73.0 * | 78.8 * | 70.6 * | ||||||
| R2 | 1.4 | 2.0 | 13.2 * | 9.3 * | 6.9 * | 9.1 * | 7.0 * | 5.3 * | 5.1 * | 5.4 * | ||||||
| R3 | 3.1 | 2.1 | 75.7 * | 38.6 * | 41.3 * | 32.1 * | 11.5 * | 18.8 * | 12.5 * | 20.4 * | ||||||
. | % Blasts . | Control IDN-1529 . | . | Ara-C IDN-1529 . | . | Dox IDN-1529 . | . | Vincristine IDN-1529 . | . | Paclitaxel IDN-1529 . | . | |||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Bone marrow . | . | − . | + . | − . | + . | − . | + . | − . | + . | − . | + . | |||||
| Patient 1 | 91 | — | — | — | — | |||||||||||
| Cell count | 100 | 82.5 | 35.9 * | 32.0 * | 69.9 * | 66.0 * | ||||||||||
| Annexin V+ | 13.5 | 7.3 | 63.5 * | 50.0 * | 24.2 * | 13.3 * | ||||||||||
| CMXRos-MTG | ||||||||||||||||
| R1 | 73.5 | 77.7 | 16.9 * | 24.3 * | 59.0 * | 67.7 * | ||||||||||
| R2 | 3.7 | 4.8 | 4.3 * | 11.5 * | 5.5 * | 7.0 * | ||||||||||
| R3 | 21.1 | 15.0 | 77.1 * | 59.8 * | 33.4 * | 23.3 * | ||||||||||
| Patient 2 | 82 | — | — | — | — | |||||||||||
| Cell count | 100 | 92.4 | 82.3 | 96.8 | 83.1 | 85.8 | ||||||||||
| Annexin V+ | 13.4 | 7.1 | 14.6 | 14.7 | 10.3 | 10.3 | ||||||||||
| CMXRos-MTG | ||||||||||||||||
| R1 | 19.1 | 22.5 | 27.5 | 18.7 | 19.7 | 24.7 | ||||||||||
| R2 | 56.6 | 59.1 | 37.4 | 48.7 | 61.4 | 56.4 | ||||||||||
| R3 | 21.7 | 15.4 | 30.6 | 28.4 | 15.8 | 17.5 | ||||||||||
| Patient 3 | 78 | — | — | — | — | |||||||||||
| Cell count | 100 | 103.4 | 106.7 | 101.1 | 112.4 | 101.1 | ||||||||||
| Annexin V+ | 4.7 | 2.3 | 7.7 | 5.5 | 6.3 | 3.4 | ||||||||||
| CMXRos-MTG | ||||||||||||||||
| R1 | 86.8 | 82.9 | 85.9 | 86.3 | 89.0 | 84.2 | ||||||||||
| R2 | 6.7 | 13.2 | 8.2 | 8.4 | 6.5 | 11.9 | ||||||||||
| R3 | 2.6 | 2.3 | 4.0 | 3.4 | 3.3 | 2.4 | ||||||||||
| Patient 4 | 41 | — | — | |||||||||||||
| Cell count | 100 | 101.0 | 88.5 | 89.6 | 46.9 * | 57.3 * | 96.9 | 99.0 | ||||||||
| Annexin V+ | 15.4 | 14.6 | 15.8 | 16.4 | 36.6 * | 28.2 * | 14.5 | 11.3 | ||||||||
| CMXRos-MTG | ||||||||||||||||
| R1 | 95.8 | 95.3 | 93.3 | 94.0 | 89.0 * | 76.0 * | 96.0 | 94.8 | ||||||||
| R2 | 3.7 | 4.3 | 6.02 | 5.0 | 10.2 * | 23.4 * | 3.5 | 4.6 | ||||||||
| R3 | 0.01 | 0.03 | 0.07 | 0.08 | 0.01 * | 0.01 * | 0.11 | 0.11 | ||||||||
| Patient 5 | 82 | |||||||||||||||
| Cell count | 100 | 110.0 | 40.0 * | 39.0 * | 49.0 * | 47.0 * | 70.0 * | 65.0 * | 69.0 * | 68.0 * | ||||||
| Annexin V+ | 15.2 | 4.3 | 78.9 * | 30.6 * | 58.7 * | 30.5 * | 19.0 * | 13.2 * | 17.6 * | 13.1 * | ||||||
| CMXRos-MTG | ||||||||||||||||
| R1 | 93.6 | 93.6 | 8.7 * | 48.8 * | 41.3 * | 53.7 * | 78.2 * | 73.0 * | 78.8 * | 70.6 * | ||||||
| R2 | 1.4 | 2.0 | 13.2 * | 9.3 * | 6.9 * | 9.1 * | 7.0 * | 5.3 * | 5.1 * | 5.4 * | ||||||
| R3 | 3.1 | 2.1 | 75.7 * | 38.6 * | 41.3 * | 32.1 * | 11.5 * | 18.8 * | 12.5 * | 20.4 * | ||||||
AML blasts were treated with Ara-C (2 μM), Dox (500 ng/mL), vincristine (10 ng/mL), or paclitaxel (50 ng/mL) with or without caspase inhibitor IDN-1529 (20 μM) at 48 hours. Results are expressed as percentages. — indicates not done.
Samples responded to the drugs.
Discussion
The IAP family of antiapoptotic genes is widely expressed in tumors of hematopoietic and epithelial origin.1,10,19,21,22,29,51 We have previously reported the expression of the IAP family members Survivin, XIAP, cIAP1, and cIAP2 in leukemia cells.10,29,52 In this study, we examined the expression of Survivin and XIAP in additional primary AML samples (n = 116 and n = 172, respectively) and assessed the clinical impact of IAP expression, extending our previous studies. To examine the importance of caspases, the targets of IAPs, in chemotherapeutic drug-induced cell death in AML, we studied the activation of caspases and cell death induced by various anticancer drugs in the absence and presence of caspase inhibitors in OCI-AML3 cells. We found no correlation between Survivin and XIAP expression, cytogenetics, remission attainment, or overall survival of patients with AML. Several commonly used anticancer agents such as Ara-C, Dox, vincristine, and paclitaxel induced caspase activation, PARP cleavage, DNA fragmentation, releases of cytochrome c and Smac, nuclear translocation of AIF, loss of MMP, and cell death. Caspase inhibition blocked caspase-dependent apoptosis but had no effect on the number of surviving cells, although it diminished mitochondrial changes associated with apoptosis. Spindle blockers had a partial effect on MMP by allowing only partial depolarization in a subpopulation of cells.
Elevated Survivin protein expression levels associated with poor prognosis and increased cancer reoccurrence have been reported by several groups.18-20 One study was also reported in AML22 ; Survivin expression was analyzed in 125 AML patients by immunofluorescence and confocal microscopy. Sixty percent of samples were found to express Survivin. By Western blot analysis, we found Survivin expression in all our 116 AML samples. Our assay method may be more sensitive and quantitative than immunofluorescence staining followed by confocal microscopy, which was scored as a percentage of positive-staining cells. Adida et al did not see significant differences in complete remission rates or overall survival rates between Survivin-positive and Survivin-negative AML patients using the log-rank test. Only when Survivin expression was adjusted with the Cox model for established prognostic factors in AML (cytogenetics, age, and WBC), it became an independent negative prognostic factor. Our results did not identify a correlation between Survivin expression, cytogenetics, age, or WBC, and we therefore conclude that this IAP has no prognostic importance.
XIAP expression in relation to clinical outcome has not been well studied in leukemia. Our initial observations on 76 primary AML samples suggested an inverse correlation between XIAP protein levels and survival in patients with AML.29 However, when a larger sample size (172 more samples) was analyzed, this correlation did not hold. No differences in age, cytogenetics, antecedent hematologic disorder, performance status, WBC, or complete remission rate were found between the 2 groups of patients in the XIAP study. The only difference was that the relapse rate was much higher in the original group than in the present group (70% vs 52%). This likely reflected differences in therapy used during the different time periods, but it cannot explain the different outcome of our study. A study from another group in patients with advanced non-small cell lung cancer (n = 55) also showed a lack of correlation between the expression of XIAP and the response to chemotherapy, progression, or overall survival.53 In addition, the same group showed that in patients with radically resected non-small cell lung cancer (n = 144), those expressing high levels of XIAP had longer overall survival times than those expressing lower levels of XIAP.54 Hence, the role of XIAP levels in predicting outcomes in patients with cancer is still controversial.
The lack of prognostic impact of Survivin or XIAP expression in AML does not imply biologic irrelevance. Given the poor overall outcome in AML, these IAPs could make a major contribution to drug resistance. Recently, cIAP1 was shown to predict outcome in patients with cancer treated with radiotherapy.55 Furthermore, the IAPs are a family of proteins. It may not be accurate to predict clinical outcome determining only one of them. XIAP-/- mice had a normal phenotype because of the compensatory up-regulation of other IAPs.56 To further complicate analysis, IAPs are negatively regulated by mitochondria-released Smac/Diablo.57,58 In addition, XIAP-associated factor I (XAF-I)25,59 and HtrA2 (Omi),38-41 once released from mitochondria, bind and eliminate XIAP function. The function of IAPs depends not only on their protein expression levels and localization but also on the levels and localization of their negative regulators. It would be useful to analyze the profile of all known IAPs and their regulators in future studies and to correlate this profile with clinical outcomes in AML. Although the expression of Survivin or XIAP itself did not predict the outcome in AML patients, considering the prosurvival properties of these proteins, their overexpression may contribute to tumorigenesis by giving cells a survival advantage and resistance to therapy. IAPs could therefore be excellent cancer therapy targets. Indeed, our recent studies demonstrated that the down-regulation of Survivin and XIAP expression in HL-60 cells by antisense oligonucleotides resulted in caspase activation and cell death and sensitized HL-60 cells to chemotherapy-induced cell death.60,61 Others also showed that inhibiting Survivin and XIAP expression overcomes resistance and sensitizes tumor cells to therapy.27,28,62,63
Our present study showed that XIAP expression strongly correlated with total ERK but not with pERK. The meaning of this is not understood at this time. Our recent study, however, showed that XIAP expression is regulated through the mitogen-activated protein kinase (MAPK) pathway60 and that ERK could be another downstream target of this signaling pathway. Others also demonstrated that the expression of XIAP is translationally regulated by a cap-independent mechanism mediated by an internal ribosome entry site64 and is transcriptionally regulated by the nuclear factor κB (NFκB) signaling pathway.65
Increasing evidence supports the notion that caspases are not always critical for cell death and that caspase activation is not the only pathway by which anticancer drugs induce cell death. The release of cytochrome c from mitochondria plays an essential role in regulating cell death. Studies have shown that cytochrome c release from mitochondria promotes cell death in 2 ways: promoting apoptosis by activating caspases through Apaf-1 and promoting necrosis by interrupting electron-chain transport, thereby preventing oxidative phosphorylation, promoting free-radical production, and eventually depleting cellular adenosine triphosphate (ATP) pools.66 Other caspase-independent cell death factors that have been described include AIF,34-36 endonuclease, EndoG,35,37 and HtrA2/Omi. Omi is released from mitochondria along with cytochrome c and is able to trigger caspase-independent cell death through its intrinsic serine protease activity.38-41 In addition, our studies demonstrate that caspase inhibitors have partial effects on mitochondria (cytochrome c and Smac release, loss of MMP, and nuclear translocation of AIF) and that cell death occurs in AML cells in response to various anticancer drugs despite caspase inhibition. Changes in mitochondria induced by drugs might be sufficient for the induction of cell death when the caspase cascade is blocked. Involvement of a caspase-dependent apoptotic pathway may just make cell death more efficient, more physiologic, and less dramatic. Support for the concept of caspase-independent cell death also came from our recent in vivo study of chemotherapy-induced cell death in patients with AML undergoing chemotherapy in whom no caspase activation was found.67 Therefore, caspases are not the only executioners of cell death relevant to mechanisms of chemotherapy action. Of course, we cannot exclude the possibility that other uncharacterized caspases not inhibited by IDN-1529 and IDN-1965 are responsible for mediating drug-induced cell death in these leukemic cells, given the fact that caspase-9 was not inhibited.
We noticed in our experiments that the release of cytochrome c and Smac from mitochondria and that changes in MMP did not always coincide. For example, the release of cytochrome c and Smac was partially inhibited by caspase inhibition, whereas changes in MMP were not when OCI-AML3 cells were treated by Ara-C or Dox. Changes in MMP were partially inhibited, but the release of cytochrome c and Smac was not inhibited when these cells were treated using vincristine or paclitaxel. This observation is in agreement with the studies from Boise,68 who demonstrated that cytochrome c release and mitochondrial depolarization are separate events. A more complete understanding of the diverse mechanisms of cell death will help to design more powerful strategies that target the caspase-dependent death pathway in apoptosis-sensitive forms of cancers and the caspase-independent death pathway in apoptosis-resistant forms of cancers.
Prepublished online as Blood First Edition Paper, August 14, 2003; DOI 10.1182/blood-2003-03-0960.
Supported in part by National Institutes of Health (NIH) grants PO1 CA55164, CA16672, and PO1 CA49639.
The publication costs of this article were defrayed in part by page charge payment. Therefore, and solely to indicate this fact, this article is hereby marked “advertisement” in accordance with 18 U.S.C. section 1734.
We thank Patty Contreras (Idun Pharmaceuticals, La Jolla, CA) for supplying us with caspase inhibitors. We thank Rosemarie Lauzon for her assistance in preparing the manuscript and C. Ellen Jackson and Kimberly Edwards for procuring the clinical samples.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal