To the editor:
Chronic neutrophilic leukemia (CNL) is a rare myeloproliferative neoplasm characterized by sustained elevation of neutrophil count, splenomegaly, and poor prognosis. Activating mutations in the colony-stimulating factor 3 receptor (CSF3R), also known as the granulocyte colony-stimulating factor (G-CSF) receptor, have recently been identified in 80% of patients studied with CNL.1,2 The most common mutation is T618I, which renders the receptor ligand independent through constitutive JAK/STAT activation.1,3 The strong association of activating CSF3R mutations with CNL has led to the addition of a CSF3R T618I mutation or other activating CSF3R mutation to the diagnostic criteria for CNL in the 2016 revision to the World Health Organization (WHO) classification of myeloid neoplasms and acute leukemia.4
CNL is generally a disease of older adults with a median survival of <2 years (range, 1-106 months).2 The natural history remains poorly understood due to its rarity, insidious onset, and delay in diagnosis.5 Approximately 200 cases of CNL have been reported, largely as single case reports and small case series.6
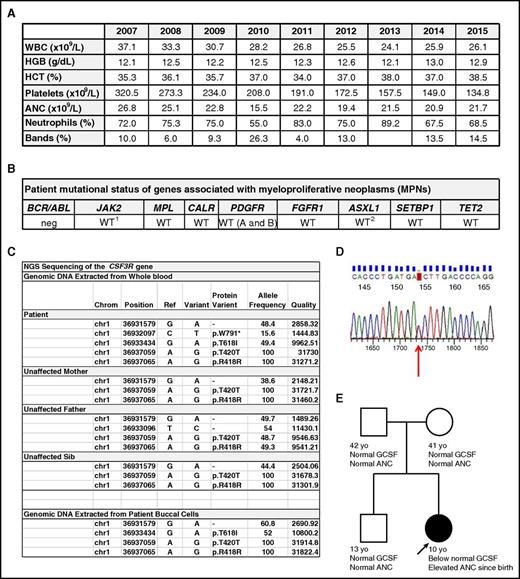
We report a child with CNL and a germ line CSF3R T618I mutation with stable leukocytosis for 11 years. The patient was a full-term infant girl with leukocytosis at birth (leukocyte count, 35 000 per cubic millimeter; absolute neutrophil count, 29 000). Bone marrow studies at 5 months revealed hypercellular marrow with granulocytic hyperplasia, <1% myeloblasts, no dysplastic features, and normal karyotype/fluorescent in situ hybridization. At 4 years of age, splenomegaly was demonstrated by ultrasound. The patient remained healthy without evidence of leukocyte adhesion defect or chronic infections or inflammation. Serial blood counts showed persistent leukocytosis (range, 24 000-37 000) with normal hemoglobin and platelet count (Figure 1A). Both parents and the only sibling had normal blood counts. At 10 years of age, due to increasing splenomegaly and a mildly decreased platelet count of 134 × 109/L, a bone marrow examination was repeated, and was unchanged. Cytogenetics and fluorescent in situ hybridization were normal, and no mutations were detected in JAK2, MPL, CALR, PDGFR A/B, or FGFR1. Polymerase chain reaction for BCR-ABL was negative (Figure 1B). Serum G-CSF was undetectable. Deep sequencing revealed a CSF3R T618I mutation that was confirmed by Sanger sequencing of patient DNA from both buccal and blood cells with an ∼50% mutant allele frequency (Figure 1C-E). No mutations were identified in ASXL1, SETBP1, or TET2 (Figure 1B), which are frequently reported in association with a T618I mutation in patients with CNL.5 A CSF3R W791X mutation was detected at a low frequency (∼16%) in the patient’s blood cells but not buccal cells (Figure 1C). Repeat molecular studies 14 months later were unchanged with no additional mutations identified or changes in the mutant CSF3R allele frequencies detected.
Heterozygous germ line CSF3R T618I mutation in a child with CNL. (A) Serial blood counts by year showing white blood count (WBC), hemoglobin (HGB), hematocrit (HCT), platelet count, absolute neutrophil count (ANC), percentage of neutrophils, and percentage of bands. (B) Mutational analysis of genes frequently implicated in myeloproliferative neoplasms (MPNs) using next-generation and/or Sanger sequencing (WT, wild type; neg, negative). 1V617F and exon 12 mutations were not detected. A single nucleotide polymorphism (SNP) in exon 9 (L393V) was detected. 2Mutations in ASXL1 were not detected. M1249V, a known SNP, was identified. (C) Using next-generation sequencing (NGS), the entire CSF3R gene was sequenced from genomic DNA (gDNA) from whole blood from the patient, the unaffected parents, and the unaffected sibling, as well as from gDNA isolated from a buccal swab from the patient. A heterozygous T618I mutation was identified in the gDNA isolated from both blood and buccal cells from the patient. CSF3R W791* was detected in blood cells only from the patient at low frequency (16%). (D) Confirmation of the CSF3R T618I mutation in patient cells in panel C by Sanger sequencing. (E) Patient pedigree showing the CSF3R T618I mutation present in the patient but undetectable in either parent or the unaffected sibling. Serum G-CSF levels were below normal in the patient but within the normal range in the parents and unaffected sibling.
Heterozygous germ line CSF3R T618I mutation in a child with CNL. (A) Serial blood counts by year showing white blood count (WBC), hemoglobin (HGB), hematocrit (HCT), platelet count, absolute neutrophil count (ANC), percentage of neutrophils, and percentage of bands. (B) Mutational analysis of genes frequently implicated in myeloproliferative neoplasms (MPNs) using next-generation and/or Sanger sequencing (WT, wild type; neg, negative). 1V617F and exon 12 mutations were not detected. A single nucleotide polymorphism (SNP) in exon 9 (L393V) was detected. 2Mutations in ASXL1 were not detected. M1249V, a known SNP, was identified. (C) Using next-generation sequencing (NGS), the entire CSF3R gene was sequenced from genomic DNA (gDNA) from whole blood from the patient, the unaffected parents, and the unaffected sibling, as well as from gDNA isolated from a buccal swab from the patient. A heterozygous T618I mutation was identified in the gDNA isolated from both blood and buccal cells from the patient. CSF3R W791* was detected in blood cells only from the patient at low frequency (16%). (D) Confirmation of the CSF3R T618I mutation in patient cells in panel C by Sanger sequencing. (E) Patient pedigree showing the CSF3R T618I mutation present in the patient but undetectable in either parent or the unaffected sibling. Serum G-CSF levels were below normal in the patient but within the normal range in the parents and unaffected sibling.
This is the first report of a germ line CSF3R T618I mutation and the youngest reported patient to date with CNL. Our patient is also the longest living patient reported with CNL. The patient’s sustained neutrophilia, splenomegaly, and morphological and molecular studies fulfill the 2016 WHO diagnostic criteria for CNL. A previous report by Plo et al identified a different activating germ line CSF3R mutation as the basis for hereditary chronic neutrophilia.7 These investigators identified an autosomal-dominant CSF3R T640N point mutation (reported as T617N using the traditional CSF3R numbering system that does not include the signal peptide) with complete penetrance in 12 of 16 members of a 3-generation pedigree. The T640N mutation affects the transmembrane domain of CSF3R and not the membrane proximal region to which the T618I mutation localizes. Like the T618I mutation, constitutive phosphorylation of the JAK/STAT signaling pathway by the T640N mutation was demonstrated, which could be abrogated by the JAK2 inhibitor AZD1480. Transplantation of hematopoietic stem cells expressing the CSF3R T640N mutant in mice also induced a myeloproliferative-like disorder.
More recently, Maxson et al identified a T640N mutation in 2 patients meeting WHO criteria for true CNL/atypical chronic myeloid leukemia (CML).8 The features of many of the affected family members in the pedigree reported by Plo et al with the T640N mutation also satisfy the 2016 WHO diagnostic criteria for CNL. Notably, 6 of the 12 affected family members were 20 years of age or younger. These data and our own demonstrate that CNL is a disease that is not restricted to the elderly. We recommend that CNL be included in the differential diagnosis of unexplained neutrophilia in children and that mutational analysis of CSF3R be performed.
Germ line analyses were not performed in the initial studies linking presumed somatic CSF3R mutations to CNL, so it is possible that a congenital origin for CNL may have been missed in these cases.1,2 The presence of a germ line CSF3R T618I mutation in our patient that was not detected in either parent suggests that the mutation arose as a result of genetic mosaicism or as a spontaneous mutation in utero. Our patient and the pedigree reported by Plo et al emphasize the importance of performing germ line studies in patients with CNL. Such studies should help to better define the causality and natural history of CNL.
A clinical course for CNL similar to CML that is composed of a chronic phase, an accelerated phase, and blast transformation has been suggested by several investigators.5 Blast transformation has been reported to occur in 20% of patients with CNL at a median of 21 months from diagnosis. To date, all reported cases have been myeloid, and most of these patients have been found to have mutations in other genes such as ASXL1 and SETBP1 in addition to an activating CSF3R mutation.5 These observations, together with our own demonstrating stable leukocytosis over a 11-year time period in a patient with CNL harboring a CSF3R T618I mutation but lacking mutations in other genes, provide strong evidence that additional mutations are required for progression of CNL, analogous to the secondary molecular alterations observed in transformation of CML.9 The pathophysiologic relevance of the acquired low-frequency CSF3R W791X mutation in our patient that has remained stable for 14 months and was previously described in 1 other patient with CNL and a T618I mutation remains unknown.1 Serial molecular studies in patients with CNL should help define genetic abnormalities associated with different phases of CNL and their impact on prognosis and therapy.
Authorship
Contribution: L.J.D. designed experiments, reviewed data, and helped write the manuscript; D.P.M. provided clinical data and helped write the manuscript; N.S. performed research and helped write the manuscript; A.E.P. and A.L. performed research; J.M.G. reviewed the data and helped write the manuscript; and B.R.A. designed experiments, reviewed the data, and wrote the manuscript.
Conflict-of-interest disclosure: The authors declare no competing financial interests.
Correspondence: Belinda R. Avalos, The Levine Cancer Institute, 1021 Morehead Medical Dr, Suite 5002, Charlotte, NC 28204; e-mail: belinda.avalos@carolinashealthcare.org.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal