To the editor:
Primary myelofibrosis (PMF) is characterized by the presence of bone marrow fibrosis associated with increased abundance of megakaryocytes and atypia in bone marrow histology.1-3 The main driver mutations found in PMF are JAK2 (50% to 60% of patients), MPL (3% to 5% of patients), and CALR (25% to 30% of patients).4 Various other nondriver mutations have also been identified.4 PMF is associated with high risk of thrombotic events5 and leukemic transformation.6 Currently, the only curative treatment is allogeneic stem-cell transplantation, with significant treatment-related toxicity.7
The mechanisms of thrombotic events associated with PMF have not been fully elucidated.5,8,9 A multifactorial mechanism is suspected, with concerted contribution from platelets, leukocytes, endothelial cells, and soluble factors.8 We found lysyl oxidase (LOX), a secreted enzyme that oxidizes lysine residues in proteins, to be upregulated in megakaryocytes of a mouse model of PMF (GATA-1low myelofibrotic mice).10 Elevated LOX in PMF megakaryocytes might be a result of hyperproduction of factors known to upregulate Lox gene expression, such as transforming growth factor β.11,12 We also reported that transgenic upregulation of platelet LOX in normal mice enhances platelet binding to monomeric collagen via α2β1 receptors, but not binding to fibrillar collagen (preferred by glycoprotein VI [GPVI] receptors). As discussed in that report,13 highly conserved extracellular lysine residues found in α2β1 receptors could be a target of secreted LOX. Importantly, parallel to its effect on cell adhesion to collagen, LOX also augments propensity for thrombosis in mice.13
Although mouse GATA-1low megakaryocytes have significantly upregulated LOX,10 it is important to confirm this finding in cells with the more prevalent JAK2V617F mutation. In this regard, we first analyzed megakaryocytes from transgenic mice carrying the human JAK2V617F mutation, resulting in hallmarks of PMF.14 Upregulated LOX was evident in these cells, as was enhanced adhesion to collagen, compared with matching controls (supplemental Figure 1A-B, available on the Blood Web site). Furthermore, after treatment with the LOX-specific inhibitor β-aminopropionitrile (BAPN), megakaryocyte ability to adhere to collagen was significantly reduced (supplemental Figure 1C).
To test the relevance of these findings to human PMF, we next examined whether LOX level is dysregulated in cells of patients with PMF compared with controls. Data were derived from analyses of 10 patient samples (5 women and 5 men; age range, 30-55 years) with fibrosis grades from MF-1 to MF-3 (scaled as per World Health Organization); 60% carried the JAK2V617F mutation, and 40% carried the CALR mutation. Peripheral blood platelet values were approximately 300 to 700 × 109/L. None of the patients had received previous aspirin, ruxolitinib, or interferon treatment. Healthy subjects (HSs), used as controls, comprised 3 men and 2 women, ranging in age from 30 to 40 years and without previous treatment history. No mutation analysis or bone marrow fibrosis grading was performed in HSs. Their peripheral blood platelet values were approximately 200 to 300 × 109/L. Experiments were performed under institutional review board protocol 2011-0004143 (IRCCS Policlinico San Matteo Foundation, 26 September 2011).
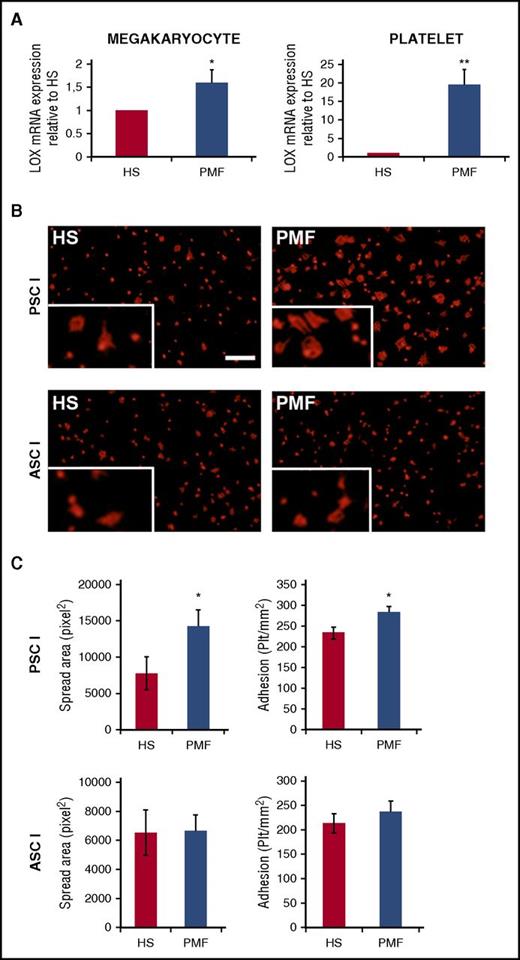
As shown in Figure 1A, LOX expression was mildly but significantly upregulated in CD34+-derived PMF megakaryocytes compared with controls (cells were processed as described in supplemental Methods). Interestingly, LOX expression was found to be quite higher in platelets of patients with PMF compared with controls (Figure 1B). It is possible that among the pool of megakaryocytes analyzed, the large ones (typically platelet yielding) expressed much higher LOX. Indeed, after our earlier mouse studies,10 a report of LOX immunohistochemistry in human bone marrow aspirates showed its significant upregulation in megakaryocytes of patients with myeloproliferative neoplasms compared with controls.15
Quantitative reverse-transcriptase polymerase chain reaction analysis of LOX in peripheral blood platelets and megakaryocytes differentiated from control HSs and patients with PMF. (A) Equal numbers of cells were evaluated in the experimental groups, and values were normalized to GAPDH messenger RNA (mRNA) and expressed relative to HSs. Data are expressed as mean ± standard deviation (SD) of 10 samples from patients with PMF and 5 from HSs. *P < .05, **P < .01. (B) An equal number of washed platelets from peripheral blood of HSs or patients with PMF were plated on pepsinated monomeric type I collagen (PSCI) or triple helical native acid soluble type I collagen (ASCI). Adherent platelets were fixed and stained with anti β-actin antibody (red). Scale bar, 50 µm. (C) Platelets (plt) were counted and expressed as platelets per mm2. Spread area was measured using Olympus LCmicro software and expressed as pixels. Data of adhesion to monomeric collagen (top bars) or fibrillar collagen (lower bars) are expressed as mean ± SD. *P < .05. Supplemental Methods provides additional details on procedures and statistics.
Quantitative reverse-transcriptase polymerase chain reaction analysis of LOX in peripheral blood platelets and megakaryocytes differentiated from control HSs and patients with PMF. (A) Equal numbers of cells were evaluated in the experimental groups, and values were normalized to GAPDH messenger RNA (mRNA) and expressed relative to HSs. Data are expressed as mean ± standard deviation (SD) of 10 samples from patients with PMF and 5 from HSs. *P < .05, **P < .01. (B) An equal number of washed platelets from peripheral blood of HSs or patients with PMF were plated on pepsinated monomeric type I collagen (PSCI) or triple helical native acid soluble type I collagen (ASCI). Adherent platelets were fixed and stained with anti β-actin antibody (red). Scale bar, 50 µm. (C) Platelets (plt) were counted and expressed as platelets per mm2. Spread area was measured using Olympus LCmicro software and expressed as pixels. Data of adhesion to monomeric collagen (top bars) or fibrillar collagen (lower bars) are expressed as mean ± SD. *P < .05. Supplemental Methods provides additional details on procedures and statistics.
Considering our current finding of elevated LOX in human PMF platelets and our earlier reported effect of transgenic mouse platelet LOX on adhesion to monomeric collagen,13 we tested this property in human PMF cells. A greater number of PMF-derived platelets adhered on monomeric collagen compared with controls (Figure 1B-C). In addition, the spread area of adherent platelets was significantly higher compared with that of control platelets (Figure 1C). In accordance with the transgenic mouse studies,13 enhanced adhesion of PMF-derived platelets was observed only on monomeric collagen (PSCI, preferred by α2β1) and not on fibrillar collagen (triple helical native acid soluble type I collagen, preferred by GPVI; Figure 1C). As was the case in the LOX transgenic mice,13 no differences were detected between the experimental groups when tested on matrices coated with bovine serum albumin (control) or fibrinogen (mediated by αIIbβ3 receptors; data not shown). Importantly, the observed phenotype was not mediated by changes in collagen receptor expression in human PMF samples compared with controls (supplemental Figure 2).
Next, we tested whether increased LOX expression in PMF-derived megakaryocytes affects collagen adhesion. As shown in Figure 2, PMF megakaryocytes showed a greater tendency to adhere and spread to monomeric collagen (PSCI; mean ± standard deviation, 25 ± 6 and 39 ± 5, respectively, for control and PMF-derived megakaryocytes; P < .05; Figure 2A), although this difference was not observed on fibrillar collagen (data not shown). The influence of LOX was confirmed by treating megakaryocytes with the LOX-specific inhibitor BAPN, which restored PMF-derived megakaryocyte adhesion to levels comparable to that in control cells (Figure 2A). The effect of BAPN on control cells (which express LOX, albeit at lower levels) seemed variable.
Megakaryocyte adhesion assay. Megakaryocytes were differentiated from CD34+ hematopoietic progenitor cells derived from control HSs or patients with PMF. Because possible oxidation of extracellular residues by secreted LOX would take place during cellular development, it was important to add the LOX inhibitor during this process. At day 2 of differentiation, cells were treated or not with 100 µm of LOX inhibitor BAPN until the end of culturing. BAPN concentration was selected based on consideration of the incubation time, so as to inhibit LOX but avoid effects on cell number (confirmed). At day 13, megakaryocytes were collected and counted, and equal numbers of HS and PMF megakaryocytes were plated on PSCI-coated glass coverslips for 3 hours. Adherent megakaryocytes were fixed and stained with anti-β1 tubulin antibody (green) and counterstained with Hoechst (blue). (A) Adherent HS- and PMF-derived megakaryocytes (Mks) were counted and expressed as number (n) of β1 tubulin+ megakaryocytes per field. Data are expressed as mean ± standard deviation of 5 samples from patients with PMF and 3 HS samples. *P < .05. Ctrl, control. (B) Images of adhering cells; scale bar, 50 µm. See supplemental Methods for additional details on cell culturing, cell isolation, and other procedures and statistics.
Megakaryocyte adhesion assay. Megakaryocytes were differentiated from CD34+ hematopoietic progenitor cells derived from control HSs or patients with PMF. Because possible oxidation of extracellular residues by secreted LOX would take place during cellular development, it was important to add the LOX inhibitor during this process. At day 2 of differentiation, cells were treated or not with 100 µm of LOX inhibitor BAPN until the end of culturing. BAPN concentration was selected based on consideration of the incubation time, so as to inhibit LOX but avoid effects on cell number (confirmed). At day 13, megakaryocytes were collected and counted, and equal numbers of HS and PMF megakaryocytes were plated on PSCI-coated glass coverslips for 3 hours. Adherent megakaryocytes were fixed and stained with anti-β1 tubulin antibody (green) and counterstained with Hoechst (blue). (A) Adherent HS- and PMF-derived megakaryocytes (Mks) were counted and expressed as number (n) of β1 tubulin+ megakaryocytes per field. Data are expressed as mean ± standard deviation of 5 samples from patients with PMF and 3 HS samples. *P < .05. Ctrl, control. (B) Images of adhering cells; scale bar, 50 µm. See supplemental Methods for additional details on cell culturing, cell isolation, and other procedures and statistics.
In the past, we identified LOX as an amplifier of bone marrow fibrosis in a mouse model of PMF, resulting from lysine oxidation of collagen and its consequent crosslinking.10 The present report provides evidence that platelets and megakaryocytes from patients with PMF overexpress LOX and show higher adhesion to collagen that is dependent on LOX activity. Future proteomic studies will identify residues in collagen receptors that are potentially targeted by secreted LOX. Increased platelet adhesion to collagen is central to augmented risk for thrombus formation and cardiovascular disease (as reviewed by Kunicki et al16 ). Moreover, higher adhesion of megakaryocytes to this matrix suggests an interesting prospect, because adhesion to extracellular matrix has been suggested to affect a plethora of biological functions, including clonal expansion.17 Currently, no specific therapy apart from aspirin is recommended for prevention of thrombosis in PMF, despite the fact that thrombotic events are one of the major life-threatening events associated with PMF.18 Taken together, our findings suggest that LOX, in addition to controlling bone marrow fibrotic phenotype, could modulate changes in platelet properties associated with PMF. Future studies could focus on exploring the relevance of these findings to various forms of human myeloproliferative neoplasms.
The online version of this article contains a data supplement.
Authorship
Acknowledgments: The authors thank Kirin Brewery for providing PEG-MGDF and Joe Z. Zhao for providing JAK2V617 mice.
This work was supported in part by the Associazione Italiana per la Ricerca sul Cancro (AIRC; Milano, Italy) “Special Program Molecular Clinical Oncology 5 × 1000” to AIRC–Gruppo Italiano Malattie Mieloproliferative (A.B., V.R.), grants from the Myeloproliferative Neoplasms Foundation and from the National Heart, Lung, and Blood Institute, National Institutes of Health (HL080442) (K.R., an American Heart Association Established Investigator), and by a fellowship funded by Collegio Ghislieri, Pavia progetto “Progressi in Biologia e Medicina” (V.A.).
Contribution: V.A. performed experiments with human samples and analyzed and interpreted data; V.C. contributed to conceptualization of the project and scientific discussion; V.R. performed megakaryocyte cultures and cell adhesion and contributed to the final version of the paper; O.L. performed LOX immunostaining and participated in discussions; S.M. performed experiments with mouse samples and analyzed and interpreted data and cowrote the manuscript; A.B. coincepted the project, analyzed and interpreted data, and cowrote the manuscript; and K.R. coincepted the project, analyzed and interpreted data, and cowrote the manuscript.
Conflict-of-interest disclosure: The authors declare no competing financial interests.
Correspondence: Alessandra Balduini, Department of Molecular Medicine, University of Pavia, via Forlanini 6, 27100 Pavia, Italy; e-mail: alebal04@unipv.it; and Katya Ravid, Boston University School of Medicine, 700 Albany St, W-6, Boston, MA 02118; e-mail: kravid@bu.edu.


This feature is available to Subscribers Only
Sign In or Create an Account Close Modal