Key Points
Normal induced pluripotent stem cells exhibit donor-specific gene expression signatures and the capacity for hematopoietic development.
CNVs acquired during reprogramming or selection of rare CNVs present in the starting cell population may alter iPSC developmental potential.
Abstract
Induced pluripotent stem cells (iPSCs) hold great promise for modeling human hematopoietic diseases. However, intrinsic variability in the capacities of different iPSC lines for hematopoietic development complicates comparative studies and is currently unexplained. We created and analyzed 3 separate iPSC clones from fibroblasts of 3 different normal individuals using a standardized approach that included excision of integrated reprogramming genes by Cre-Lox mediated recombination. Gene expression profiling and hematopoietic differentiation assays showed that independent lines from the same individual were generally more similar to one another than those from different individuals. However, one iPSC line (WT2.1) exhibited a distinctly different gene expression, proliferation rate, and hematopoietic developmental potential relative to all other iPSC lines. This “outlier” clone also acquired extensive copy number variations (CNVs) during reprogramming, which may be responsible for its divergent properties. Our data indicate how inherent and acquired genetic differences can influence iPSC properties, including hematopoietic potential.
Introduction
Patient-derived induced pluripotent stem cells (iPSCs) offer valuable approaches to model human blood disorders.1-4 However, different wild-type iPSC lines vary greatly in hematopoietic potential, complicating the ability to ascertain phenotypic abnormalities in mutant lines by comparative studies.5,6 Reasons for this observed variability between normal iPSC lines are unknown but may involve genetic and/or epigenetic differences. These differences may derive from donor-specific properties related to the cell type of origin or baseline genotype. In addition, dissimilar reprogramming strategies7 and the persistence8 or reactivation of reprogramming transgenes5 may alter the properties of iPSCs. Finally, acquired chromosomal aberrations, including point mutations or copy number variations (CNVs) in iPSCs with apparently normal karyotypes, could cause phenotype variation.9,10 The utility of iPSCs for modeling hematopoietic disorders hinges on demonstrating their consistent and reproducible features, including in vitro developmental capacity. In this report, we show that iPSC lines generated from the same individual exhibit gene expression signatures and hematopoietic development that are more similar to one another than those of iPSCs generated from different individuals. Our findings provide a new set of standardized iPSC reference lines for future studies, begin to unravel the mechanisms for variation among different iPSC clones, and offer important lessons regarding the use of iPSCs for modeling human disease.
Methods
Normal fibroblasts from Coriell Institute Cell Repository (GM00969 and GM05565) and American Type Culture Collection (ATCC-CRL-2097) were transduced using STEMCCA lentiviral-based vectors.11 iPSC-like colonies were expanded and Southern blots were performed to identify single integrations. Further characterization included quantitative polymerase chain reaction (PCR) and flow cytometry for pluripotency marker expression and teratoma analysis. SNP karyotyping (Illumina610) was performed on iPSCs12 and compared with the parental fibroblasts. Global gene arrays (ST1.0 array) were performed on iPSCs enriched by fluorescence-activated cell sorting (FACS) sorted for pluripotency markers SSEA3 and TRA-1-81. In vitro differentiations were performed to generate hematopoietic lineages. Detailed methods are described in the supplementary information available online. The worked presented in this manuscript was approved by the Children’s Hospital of Philadelphia IACUC and Boston University Intitutional Review Board committees, and this study was conducted in accordance with the Declaration of Helsinki.
Results and discussion
We sought to create panels of “isogenic” reference iPSCs consisting of multiple clones from the same sources and to begin to explore the mechanisms associated with clonal variation in hematopoietic development. We used the STEMCCA lentiviral vector to reprogram human fibroblast lines from 3 unrelated normal subjects.11 Three different iPSC lines from each individual were characterized (supplemental Figure 1). Southern blot analysis identified lines containing a single lentiviral integration site, and the reprogramming gene cassettes were removed subsequently by CRE recombinase–mediated excision. All lines analyzed further displayed normal embryonic stem cell (ESC)-like morphology, normal karyotype, expression of endogenous pluripotency markers, and the capacity to form cells representing 3 germ layers in teratomas. All of these standard criteria for pluripotency were maintained after more than 20 cell passages.
We performed transcriptome analysis from cells grown under identical conditions. FACS was used to isolate cells expressing similar levels of the pluripotency markers SSEA3 and TRA-1-81 to reduce variability in gene expression caused by low levels of spontaneous differentiation (Figure 1A). Using principal component analysis (PCA), iPSC lines generated from the same individual tended to cluster, suggesting a donor-related genetic component to the expression signature (Figure 1Bi). However, clone CHOPWT2.1 mapped independently of any group. We used Significance Analysis of Microarrays (SAM) (GEO accession number GSE49231) to compare transcripts across WT2 lines and found that the 2.2 and 2.3 clones were more similar to each other than to 2.1, in agreement with PCA. We identified 50 genes that were differentially regulated (>1.5-fold) between these groups (Figure 1Bii and supplemental Table 1). Differences in 2 of these transcripts, DPPA3 and ID3, were validated by quantitative real-time PCR (Figure 1Bii and supplemental Table 1). We observed little difference in the transcriptome profiles between human ESCs and iPSCs, in agreement with published studies.12,13
Variable mRNA expression, CNV, and growth of iPSC lines from the same and different fibroblast source. (A) Representative FACS purification of human iPSCs (or ESCs) expressing high levels of pluripotency markers SSEA3 and Tra-1-81. Presorted culture (left) gated at a high level of SSEA-3/TRA-1-81 expression and purity analysis of postsorted cells. (B) mRNA expression analysis of sorted iPSCs and ESCs. (i) PCA representation of gene expression variations across individuals and within the same individual: CHOPWT1 (black), CHOPWT2 (red), BMC (green), and ESCs (blue) and (ii) comparison of gene expression from CHOPWT2 cell lines. The heat map shows the expression of 25 upregulated and 25 downregulated genes between the WT2 cell lines, criteria Student t test (P < .05), and at least a 1.5-fold change. Genes were ordered according to decreasing average expression ratio. Quantitative real-time PCR plots of BCLX and 2 examples of genes that were either upregulated or downregulated in WT2.1 compared with WT2.2 and WT2.3 iPSC lines (*P < .05 and **P < .01). Data are presented as relative expression compared with the CHOPWT2.1 expression level. (C) Total number of genes acquired or deleted during the reprogramming process for each iPSC line established. (D) Seven-day cell expansion of undifferentiated iPSC lines: CHOPWT1 (green), CHOPWT2 (blue), BMC (red). iPSC expansion expressed as fold change above starting cell numbers. Data are representative of 3 experiments (mean ± SEM, *P < .05).
Variable mRNA expression, CNV, and growth of iPSC lines from the same and different fibroblast source. (A) Representative FACS purification of human iPSCs (or ESCs) expressing high levels of pluripotency markers SSEA3 and Tra-1-81. Presorted culture (left) gated at a high level of SSEA-3/TRA-1-81 expression and purity analysis of postsorted cells. (B) mRNA expression analysis of sorted iPSCs and ESCs. (i) PCA representation of gene expression variations across individuals and within the same individual: CHOPWT1 (black), CHOPWT2 (red), BMC (green), and ESCs (blue) and (ii) comparison of gene expression from CHOPWT2 cell lines. The heat map shows the expression of 25 upregulated and 25 downregulated genes between the WT2 cell lines, criteria Student t test (P < .05), and at least a 1.5-fold change. Genes were ordered according to decreasing average expression ratio. Quantitative real-time PCR plots of BCLX and 2 examples of genes that were either upregulated or downregulated in WT2.1 compared with WT2.2 and WT2.3 iPSC lines (*P < .05 and **P < .01). Data are presented as relative expression compared with the CHOPWT2.1 expression level. (C) Total number of genes acquired or deleted during the reprogramming process for each iPSC line established. (D) Seven-day cell expansion of undifferentiated iPSC lines: CHOPWT1 (green), CHOPWT2 (blue), BMC (red). iPSC expansion expressed as fold change above starting cell numbers. Data are representative of 3 experiments (mean ± SEM, *P < .05).
CNV, reflected by gain or loss of chromosomal DNA segments, can occur in iPSCs and contribute to their phenotypic variation.9,10,14 The CNVs that differ between the original fibroblast lines and their iPSC progeny are described in Figure 1C and supplemental Table 2. Three lines (BMC1, BMC2, and CHOPWT2.2) had no detectable reprogramming-associated CNVs The BMC3 line and the 3 CHOPWT1 lines had CNVs affecting 1 or 2 genes. CHOPWT2.3 iPSCs had 2 larger duplications affecting 21 genes, whereas CHOPWT2.1 had a large deletion and 3 duplications (>200 KB) affecting 48 genes. Considering that growth-promoting mutations can be selected for in culture, we examined the expansion rate of the iPSC lines. All iPSC lines expanded at similar rates, except for CHOPWT2.1, which grew approximately twofold faster (Figure 1D). This could reflect increased expression of genes associated with multiple CNVs present including ID1 or BCL2L1 (BCLX),9,10 which were upregulated in the CHOPWT2.1 line (Figure 1Bii). These data are consistent with prior work suggesting that a chromosome 20 minimal amplicon involving region q11.1-q11.21, which is acquired in CHOPWT2.1, can confer a growth advantage to ESCs and iPSCs.
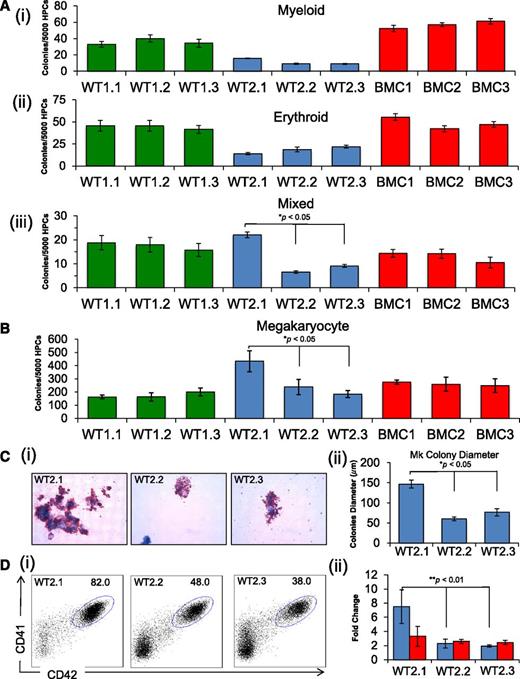
We performed in vitro differentiation of the 9 iPSC clones and 3 ESC lines to compare their hematopoietic capacities. We used an adherent monolayer differentiation culture protocol in serum-free medium with sequential addition of growth factors that stimulate the formation of hematopoietic progenitors (supplemental Methods). At days 7 to 9 of differentiation, the cultures shed multipotent CD43+CD41+CD235+ hematopoietic progenitor cells (HPCs) (supplemental Figure 2A) into the medium with erythroid, myeloid, megakaryocytic, and multilineage (mixed) capacity.15 HPC populations from different iPSC lines from the same fibroblast source generated similar numbers of myeloid and erythroid progenitors in methylcellulose colony assays (Figure 2Ai-ii). In contrast, frequencies of myeloid and erythroid colonies varied between sets of iPSCs derived from different fibroblast lines and between different human ESC lines (Figure 2A and supplemental Figure 2B). For example, progenitors from the 3 BMC iPSC clones produced more myeloid colonies than WT1 and WT2 iPSC clones and ESC lines H9 and CHB8, but had similar numbers to the H1 ESC line. The iPSC lines BMC and WT1 and ESC lines H1, H9, and CHB8 all produced similar numbers of erythroid colonies that were greater than those produced by WT2 clones. Thus, intrinsic genetic or epigenetic variation from donor-derived sources likely influences hematopoietic development of iPSCs and ESCs. However, clone CHOPWT2.1 iPSC, which exhibited the most extreme number of CNVs and increased growth rate, also displayed distinct hematopoietic features, including increased frequencies of multilineage and megakaryocytic colonies within the HPC population compared with other clones from the same source (CHOPWT2.2 and 2.3) (Figures 2Aiii,B and supplemental Figure 2C). The CHOPWT2.1 line also produced larger Mk colonies, reflecting increased proliferative capacity (Figure 2C). In liquid cultures, CHOPWT2.1 generated increased absolute numbers (∼threefold) and proportion (∼twofold) of CD41+CD42+ Mks compared with the other 2 iPSC clones from the same fibroblast source (Figure 2Di-ii). Thus, the CHOPWT2.1 iPSC line exhibits distinct differences from the 8 other lines, including an altered transcriptome, increased expansion rate, and enhanced production of multilineage and megakaryocytic hematopoietic progenitors. It is possible that these differences result from altered gene dosages associated with CNVs For example, the antiapoptotic gene BCL2L1/BCLX, which is amplified in CHOPWT2.1 and previously implicated in human ESC expansion, has also been shown to promote megakaryopoiesis.16-18 However, selected genes within the CNV regions (FYN, HSBP1, TBL1XR1, CDH12, ID1, and BCL2L1/BCLX) were expressed at similar levels in Mks generated from all WT2 clones (data not shown). Thus, we cannot directly implicate these genes in the enhanced expansion of Mks produced by clone CHOPWT2.1. However, dysregulation of these genes in CHOPWT2.1-derived multipotent hematopoietic progenitors could influence their developmental capacity, including their commitment to the Mk lineage.
In vitro differentiation of iPSC lines showing selective skewing toward megakaryopoiesis. (A) Methylcellulose progenitor assay of iPSC-derived HPCs from day 7 of differentiation. The panels show myeloid (i), erythroid (ii), and mixed (iii) colonies (mean ± SEM for 3 independent experiments, *P < .05). (B) Collagen-based colony assay to assess megakaryocyte (Mk) potential of iPSC-derived HPCs. (C) Images (i) of Mk colonies were captured by the Zeiss Axioskop2 microscope (Munich, Germany) (original magnification ×20), and (ii) size as a measure of Mk colony diameter (μm) (mean ± SEM for 3 independent experiments, *P < .05). (D) Assessment of megakaryocytic development from HPCs grown in liquid culture with thrombopoietin, interleukin 3, and SCF. The percentage of CD41a (αIIb, x-axis) and CD42a (GPIX, y-axis) Mk (i) are compared for CHOPWT2 lines, and (ii) total Mk number (blue) and non-Mk (red) cells are represented as fold change above the starting number of cells (mean ± SEM for 3 independent experiments, **P < .01).
In vitro differentiation of iPSC lines showing selective skewing toward megakaryopoiesis. (A) Methylcellulose progenitor assay of iPSC-derived HPCs from day 7 of differentiation. The panels show myeloid (i), erythroid (ii), and mixed (iii) colonies (mean ± SEM for 3 independent experiments, *P < .05). (B) Collagen-based colony assay to assess megakaryocyte (Mk) potential of iPSC-derived HPCs. (C) Images (i) of Mk colonies were captured by the Zeiss Axioskop2 microscope (Munich, Germany) (original magnification ×20), and (ii) size as a measure of Mk colony diameter (μm) (mean ± SEM for 3 independent experiments, *P < .05). (D) Assessment of megakaryocytic development from HPCs grown in liquid culture with thrombopoietin, interleukin 3, and SCF. The percentage of CD41a (αIIb, x-axis) and CD42a (GPIX, y-axis) Mk (i) are compared for CHOPWT2 lines, and (ii) total Mk number (blue) and non-Mk (red) cells are represented as fold change above the starting number of cells (mean ± SEM for 3 independent experiments, **P < .01).
Several important conclusions and caveats regarding the use of iPSCs for examining normal and pathologic hematopoietic development derive from our studies. First, individual iPSC lines from same-patient fibroblasts are more similar than those derived from different-patient samples, suggesting that intrinsic heritable genetic and/or epigenetic features of donor somatic cells contribute to the properties of progeny iPSCs. Thus, in studies comparing normal and disease-associated iPSCs, it is important to examine multiple lines from different affected subjects and controls to distinguish between effects caused by pathologic mutations vs normal variation. Second, altered developmental potential of iPSCs may correlate with their acquisition of CNVs, which could occur either during reprogramming or from clonal expansion of a rare CNV-containing cell within the parental somatic cells.19 Either way, SNP array analysis and/or genome sequencing should complement standard karyotypic analysis of newly created iPSC lines. Our study highlights the inherent reproducibility in the hematopoietic development potential of carefully selected iPSC lines and reinforces the importance of carefully characterizing iPSC lines to avoid artifactual effects on iPSC differentiation caused by mutations acquired during cell culture.
The online version of this article contains a data supplement.
The publication costs of this article were defrayed in part by page charge payment. Therefore, and solely to indicate this fact, this article is hereby marked “advertisement” in accordance with 18 USC section 1734.
Acknowledgments
M.J.W. is a former Scholar of the Leukemia Lymphoma Society and The Jane Fishman Grinberg Professor of Pediatrics.
This work was supported by National Institutes of Health grants RC2 HL101606 (M.J.W.), P30DK090969 (M.J.W.), U01 HL099656 (M.J.W., D.L.F., P.G.), and RC2HL101535 (D.N.K, G.M., P.G.).
Authorship
Contribution: J.A.M., P.P., L.L., A.M.G., D.X., Y.Y., S.K.S., H.M., and L.Y. designed and performed experiments; L.Y., K.W., S.S., A.G., L.S., and A.S. performed data analysis; A.O., J-C.J., G.M., and D.N.K. provided reagents and materials (STEMCCA reprograming vectors) and edited manuscript; and J.A.M., D.L.F., M.J.W., and P.G. designed experiments and wrote the manuscript.
Conflict-of-interest disclosure: The authors declare no competing financial interests.
Correspondence: Paul Gadue, The Children’s Hospital of Philadelphia, Department of Pathology and Laboratory Medicine, Colket Translational Research Building, 3501 Civic Center Blvd, Room 5012, Philadelphia, PA 19104; e-mail: gaduep@email.chop.edu; or Mitchell Weiss, The Children’s Hospital of Philadelphia, Division of Hematology, The Abramson Research Center 316B, 3615 Civic Center Blvd, Philadelphia, PA 19104; e-mail: weissmi@email.chop.edu


This feature is available to Subscribers Only
Sign In or Create an Account Close Modal