Key Points
The binding of administered FVIIa to endogenous EPCR enhances its ability to bypass FVIII or FIX deficiency in vivo.
EPCR modulation of function of pharmacologic FVIIa administration may be exploited in protein or gene-based FVIIa therapeutics.
Abstract
Recombinant activated human factor VII (rhFVIIa) is an established hemostatic agent in hemophilia, but its mechanism of action remains unclear. Although tissue factor (TF) is its natural receptor, rhFVIIa also interacts with the endothelial protein C receptor (EPCR) through its γ-carboxyglutamic acid (Gla) domain, with unknown hemostatic consequences in vivo. Here, we study whether EPCR facilitates rhFVIIa hemostasis in hemophilia using a mouse model system. Mouse activated FVII (mFVIIa) is functionally homologous to rhFVIIa, but binds poorly to mouse EPCR (mEPCR). We modified mFVIIa to gain mEPCR binding using 3 amino acid changes in its Gla domain (L4F/L8M/W9R). The resulting molecule mFVIIa-FMR specifically bound mEPCR in vitro and in vivo and was identical to mFVIIa with respect to TF affinity and procoagulant functions. In macrovascular injury models, hemophilic mice administered mFVIIa-FMR exhibited superior hemostatic activity compared with mFVIIa. This was abolished by blocking mEPCR and was absent in ex vivo whole blood coagulation assays, implicating a specific mFVIIa-FMR and endothelial mEPCR interaction. Because mFVIIa-FMR models the TF-dependent and EPCR binding properties of rhFVIIa, our data unmask a novel contribution of EPCR on the action of rhFVIIa administration in hemophilia, prompting the rational design of improved and safer rhFVIIa therapeutics.
Introduction
High-dose recombinant human activated factor VII (rhFVIIa) is an effective hemostatic regimen for hemophilia patients with inhibitors to factor VIII (FVIII) or factor IX (FIX).1 In addition to hemophilia, use of rhFVIIa is widespread as an emergency hemostatic agent in both pediatric and adult populations.2,3 However, off-label use carries potentially life-threatening complications, as increased thrombotic risk has been reported.3-5
Despite its widespread use, the mechanism of action of rhFVIIa in hemophilia remains controversial. It has been postulated that rhFVIIa generates hemostatic amounts of factor Xa (FXa) via a tissue factor (TF)-rhFVIIa complex on the surface of TF-expressing cells at the site of injury. As a result, the therapeutic rhFVIIa doses are high because rhFVIIa must compete with endogenous factor VII (FVII) for binding to TF.6,7 Alternatively, rhFVIIa administration is hemostatic through an activated platelet-dependent but TF-independent mode of action. Specifically, the poor affinity of FVII for anionic phospholipid membranes8 necessitates large doses of rhFVIIa for treatment to generate hemostatic amounts of FXa.9,10 This mode of action is supported by the greater clinical efficacy of an analog of rhFVIIa with enhanced TF-independent activity and platelet-binding sites.11-13 Additional evidence was recently provided by the hemostatic capacity in a hemophilia mouse injury model of a chimeric mouse FVIIa (mFVIIa) molecule harboring the γ-carboxyglutamic acid (Gla) domain of mouse FIX that does not bind mouse TF (mTF).14 However, it is unclear if those effects resulted through novel binding partner(s) for the chimeric mFVIIa molecule and/or its (potentially) altered pharmacokinetics.14
In part, procoagulant reactions are regulated by the anticoagulant function of activated protein C (PC). The activation of PC is enhanced ∼20-fold by its binding through its Gla domain to the endothelial PC receptor (EPCR).15 Intriguingly, FVIIa also interacts with EPCR through its Gla domain,16 with affinity similar to that of PC.17 However, little is known on the EPCR-FVIIa interaction in vivo or its subsequent functional consequences. Mouse models of EPCR overexpression or deficiency have suggested that EPCR is involved in the biodistribution and tissue persistence of administered rhFVIIa in vivo.18 Previous studies on the role of EPCR in rhFVIIa-dependent FXa and thrombin generation17,19-21 have not clarified whether EPCR contributes to the mechanism of action of high-dose rhFVIIa administration in hemophilia. An appropriate model system to address this in vivo would require normal interactions between rhFVIIa and TF, as well as EPCR. Unfortunately, standard mouse models are unsuitable because of species-specific restrictions in its factor VII/factor VIIa interaction with TF and EPCR. Specifically, mouse FVIIa (mFVIIa) binds mTF22,23 but has negligible affinity for mouse EPCR (mEPCR).21,24 Conversely, rhFVIIa can interact with mEPCR but has limited hemostatic capacity in mice due to poor interaction with mTF.23
A better understanding of rhFVIIa’s mechanism of action could suggest ways to improve its therapeutic and, possibly, its safety profile. Here, by manipulating the mouse system, we studied the contribution of an EPCR-rhFVIIa interaction in rhFVIIa-mediated hemostasis in vivo. We modified the Gla domain of mFVIIa with 3 amino acid changes (L4F/L8M/W9R) to create a gain in function in its ability to bind mEPCR without altering affinity for TF or the resulting function of the TF-bound protease. Thus, the resulting molecule (mFVIIa-FMR) suitably models the TF-dependent and EPCR binding properties of rhFVIIa. This allowed us to use it in hemophilic mice to evaluate the hemostatic effects of the EPCR-rhFVIIa interaction for on-demand rhFVIIa administration in hemophilia.
Methods
Plasmid construction
Expression plasmids for all constructs were based on pcDNA3.1 and are described in detail in the supplemental Materials available on the Blood Web site.
Expression and purification of recombinant proteins
Mouse FVIIa, mFVIIa chimeras, and mouse soluble EPCR (msEPCR) were expressed in human embryonic kidney (HEK 293) cells, following stable DNA transfection, and purified using the included C-terminal HPC4 tag, as previously described.25 Mouse FVIIa and chimeras were further purified on a CHT5-I hydroxyapatite column (BioRad Laboratories, Hercules, CA), to separate the fully γ-carboxylated material.26 Protein concentration was determined using the following molecular weight (in Da) and extinction coefficients (E2800.1%): mFVIIa and chimeras, 50 000 and 1.39; mouse factor X (mFX), 59 000 and 1.16; msEPCR, and 39 700 and 0.66 (see supplemental Materials for msEPCR molecular weight and E2800.1% determination). γ-Carboxyglutamic acid analysis on all recombinant proteins was performed as previously described.27
Generation of CHO-K1 stable cell lines expressing full-length mEPCR and TF
Stable CHO-K1 cells (ATCC CCL-61) expressing full-length mEPCR or mTF on their surface were screened by flow cytometry using an allophycocyanin (APC)-labeled anti-mEPCR antibody (anti-mouse CD201-APC at 0.5 µg/mL; eBioscience, San Diego, CA) or anti-mTF antibody (goat anti-mTF at 4 µg/mL, Cat. AF3178; R&D Systems, Minneapolis, MN) followed by an Alexa-Fluor 488-labeled donkey anti-goat IgG (2 µg/mL, Cat. A-11055; Life Technologies, Green Island, NY). Geneticin- or zeocin-resistant CHO-K1 cells (negative controls for mEPCR- and mTF-expressing CHO-K1 cells, respectively) were generated by stable transfection of an empty pcDNA3 vector harboring only the resistance marker.
Binding assays
The methodology used in isothermal titration calorimetry experiments is described in the supplemental Materials. Determination of ligand binding to the cell surface exposed receptor (mEPCR or mTF) was performed as previously described17 with some modifications. Specifically, confluent monolayers in a 24-well plate of mEPCR-expressing (CHO-K1-mEPCR) or G418-resistant CHO-K1 cells were incubated with increasing concentration of ligand in buffer B (10 mM N-2-hydroxyethylpiperazine-N′-2-ethanesulfonic acid, 150 mM NaCl, 4 mM KCl, 11 mM glucose, 1 mg/mL bovine serum albumin, 1.6 mM Ca2+, and 0.6 mM Mg2+, pH 7.45) at 4°C on ice for 1 hours. After 4 washes with ice-cold buffer B, the surface-bound ligand was eluted with buffer C (10 mM N-2-hydroxyethylpiperazine-N′-2-ethanesulfonic acid, 150 mM NaCl, 4 mM KCl, 11 mM glucose, 1 mg/mL bovine serum albumin, and 5 mM EDTA, pH 7.45) and subjected to polyacrylamide gel electrophoresis and detection by western blotting. Protein presence in the eluates was detected with a mouse monoclonal anti-HPC4 antibody (5 µg/mL) and a secondary goat anti-mouse IgG antibody (IR-Dye CW800, 1:10 000 dilution; Li-Cor Biosciences, Lincoln, NE). Blots were imaged at 169 µm with an Odyssey imager (Li-Cor Biosciences). After image background subtraction, band intensities were quantified and corrected by nonspecific binding using the ImageJ software.28
Thrombin generation assay, coagulation assays, mFVIIa antigen determination, rotational thromboelastometry, and factor Xa generation assay
Thrombin generation was determined as previously described.29,30 Prothrombin time (PT) assays were determined as previously described22 using human factor VII-deficient plasma (George King Bio-Medical, Overland Park, KS) and Innovin (Siemens Healthcare, Malvern, PA). When necessary, quantification was performed using standard curves of increasing concentration of each recombinant protein. Determination of uninhibited (free) mFVIIa or mFVIIa-FMR in mouse plasma was performed by an enzyme-linked immunosorbent assay, as previously described,22 and/or a PT-based assay with standard curves of increasing concentration of each recombinant protein. Whole blood coagulation by rotational thromboelastometry (ROTEM) and the factor Xa generation assay are described in the supplemental Methods.
Animal experiments and procedures
All experimental procedures were reviewed and approved by the Institutional Animal Care and Use Committee at The Children's Hospital of Philadelphia. Weight- and age (8-12 weeks)- matched male hemophilia B31 or hemophilia A32 mice backcrossed on a C57BL/6 background for ≥6 generations and hemostatically normal male mice (C57BL/6 background; The Jackson Laboratory, Bar Harbor, ME) were used in all experiments. Blood was collected via the retro-orbital plexus into 3.8% citrate. The ferric chloride-induced injury model and tail clip assay were performed essentially as previously described.30
Statistical analysis
Two-tailed Mann-Whitney U and Kruskal-Wallis tests were performed using GraphPad Prism v5.0b. Statistical differences were considered significant when P < .05.
Results
Generation and expression of mFVIIa chimeras with affinity to mEPCR
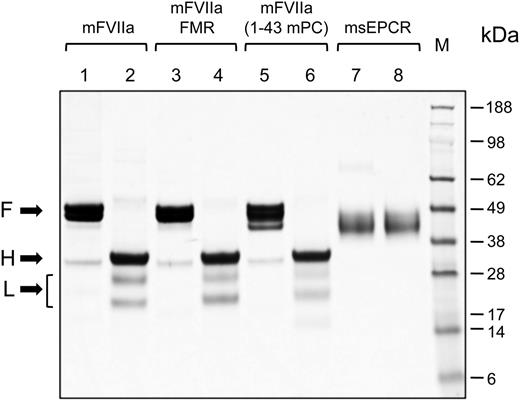
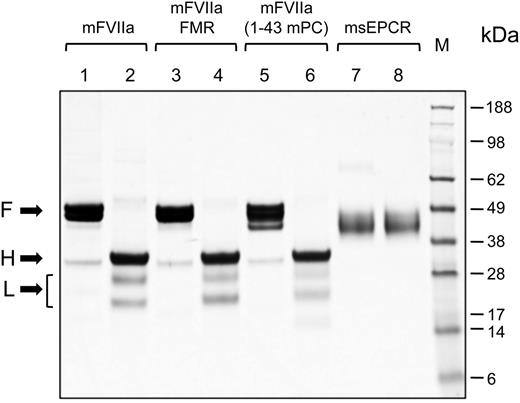
We wanted to engineer the mEPCR binding capacity to mFVIIa by using parts of the mouse PC Gla domain, because PC interacts with EPCR through the Gla domain.33 We generated 2 chimeric mFVIIa molecules with modified Gla domains using either part (mFVIIa-FMR) or the entire Gla domain of mouse PC (mFVIIa-[1-43 mPC]) (Figure 1). Recombinant mFVIIa, mFVIIa-FMR, mFVIIa-(1-43 mPC), or msEPCR were expressed in HEK293 and purified. Electrophoresis under reducing or nonreducing conditions revealed that mFVIIa and its chimeras were secreted and purified in the activated 2-chain form (Figure 2, lanes 2, 4, and 6).
Structure of the recombinant proteins used in this study. All proteins have a Gla domain, 2 epidermal growth factor (EGF) domains (EGF-1 and EGF-2), and a catalytic domain. A recognition site (Arg-Lys-Arg-Arg-Lys-Arg [RKRRKR]) for a paired amino acid cleaving enzyme (PACE)/furin type protease is inserted after the Arg at position 152. The sequence of the Gla domain is shown and highlighted in blue for each protein. Changes in the Gla domain compared with mFVIIa taken from the mouse PC Gla domain are highlighted in orange. Numbering refers to the mature form of each protein.
Structure of the recombinant proteins used in this study. All proteins have a Gla domain, 2 epidermal growth factor (EGF) domains (EGF-1 and EGF-2), and a catalytic domain. A recognition site (Arg-Lys-Arg-Arg-Lys-Arg [RKRRKR]) for a paired amino acid cleaving enzyme (PACE)/furin type protease is inserted after the Arg at position 152. The sequence of the Gla domain is shown and highlighted in blue for each protein. Changes in the Gla domain compared with mFVIIa taken from the mouse PC Gla domain are highlighted in orange. Numbering refers to the mature form of each protein.
Coomassie brilliant blue staining of sodium dodecyl sulfate-polyacrylamide gel electrophoresis gel of purified proteins. Five micrograms of mFVIIa, mFVIIa chimeras, or msEPCR were electrophoresed under reducing or nonreducing conditions. The apparent molecular weights of the marker standard (M) are indicated in kilodaltons. Lanes 1, 3, 5, and 7, nonreducing conditions; lanes 2, 4, 6, and 8, reducing conditions. Arrows point to the full-length (F) (under nonreducing conditions) and the heavy (H) and light (L) chains (under reducing conditions) of each protease. The bracket indicates 2 N-glycosylation variants of the light chain of mFVIIa (or its chimeras), as confirmed by treatment with peptide-N-glycosidase F treatment (data not shown).
Coomassie brilliant blue staining of sodium dodecyl sulfate-polyacrylamide gel electrophoresis gel of purified proteins. Five micrograms of mFVIIa, mFVIIa chimeras, or msEPCR were electrophoresed under reducing or nonreducing conditions. The apparent molecular weights of the marker standard (M) are indicated in kilodaltons. Lanes 1, 3, 5, and 7, nonreducing conditions; lanes 2, 4, 6, and 8, reducing conditions. Arrows point to the full-length (F) (under nonreducing conditions) and the heavy (H) and light (L) chains (under reducing conditions) of each protease. The bracket indicates 2 N-glycosylation variants of the light chain of mFVIIa (or its chimeras), as confirmed by treatment with peptide-N-glycosidase F treatment (data not shown).
mFVIIa chimeras bind to mEPCR on the cell surface or in solution
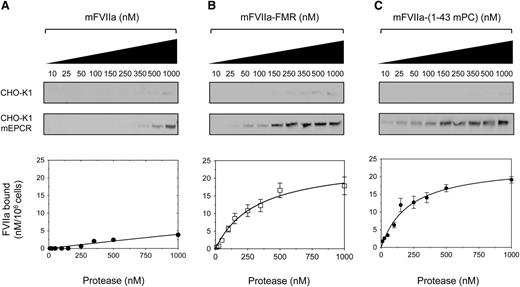
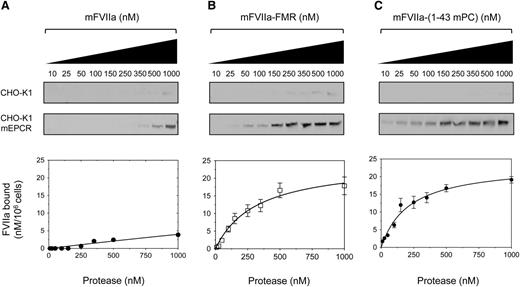
To assess the ability of the mFVIIa chimeras to bind to mEPCR as a gain of function, we generated a stable cell line expressing full-length mEPCR (CHO-K1-mEPCR). To control for nonspecific binding, CHO-K1 cells stably transfected with empty expression vector were also generated. Both cell types were incubated with increasing concentration of ligand (mFVIIa or its chimeras) at 4°C in the presence of physiological concentrations of Ca2+ and Mg2+ ions to ensure optimal binding.34 Analysis of bound ligand showed that mFVIIa lacked appreciable affinity to mEPCR (Figure 3A) even at a high concentration (1 µM), as expected.21,24 In contrast, both mFVIIa-FMR and mFVIIa-(1-43 mPC) showed specific and dose-dependent binding to mEPCR (Figure 3B-C, respectively). Fluorescent densitometric analysis allowed us to estimate a Kd of interaction of 0.34 ± 0.07 and 0.19 ± 0.06 µM for mFVIIa-FMR and mFVIIa-(1-43 mPC), respectively.
Recombinant mFVIIa chimeras bind to mEPCR on CHO-K1 cells, in contrast to mFVIIa. Increasing concentration of ligand (10-1000 nM) (A) mFVIIa, (B) mFVIIa-FMR, or (C) mFVIIa-(1-43 mPC) was incubated on CHO-K1 or CHO-K1-mEPCR cells in the presence of 1.6 mM Ca2+ and 0.6 mM Mg2+ for 1 hour at 4°C. Bound fraction was eluted and visualized by fluorescence-based western blotting (top, images shown in greyscale) and band intensities were quantified and corrected for nonspecific binding (on CHO-K1 cells). Integrated densities (bottom) were plotted, and equilibrium dissociation constants (Kd) were calculated using a 1-site–specific binding curve fitting with GraphPad Prism v5.0b (GraphPad Software). Conversion to nanomolar was done using the integrated densities of known amounts of ligand in buffer C, electrophoresed, and blotted as described in the Methods, and values were normalized for cell numbers. Depicted curves for each protein are fitted from 3 independent experiments. Data points are shown as mean ± standard deviation (SD). The y-axis of all graphs depicts FVIIa bound material.
Recombinant mFVIIa chimeras bind to mEPCR on CHO-K1 cells, in contrast to mFVIIa. Increasing concentration of ligand (10-1000 nM) (A) mFVIIa, (B) mFVIIa-FMR, or (C) mFVIIa-(1-43 mPC) was incubated on CHO-K1 or CHO-K1-mEPCR cells in the presence of 1.6 mM Ca2+ and 0.6 mM Mg2+ for 1 hour at 4°C. Bound fraction was eluted and visualized by fluorescence-based western blotting (top, images shown in greyscale) and band intensities were quantified and corrected for nonspecific binding (on CHO-K1 cells). Integrated densities (bottom) were plotted, and equilibrium dissociation constants (Kd) were calculated using a 1-site–specific binding curve fitting with GraphPad Prism v5.0b (GraphPad Software). Conversion to nanomolar was done using the integrated densities of known amounts of ligand in buffer C, electrophoresed, and blotted as described in the Methods, and values were normalized for cell numbers. Depicted curves for each protein are fitted from 3 independent experiments. Data points are shown as mean ± standard deviation (SD). The y-axis of all graphs depicts FVIIa bound material.
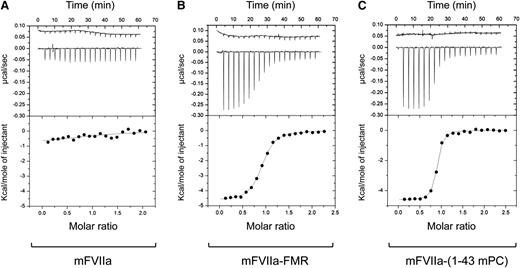
To further validate the interaction between mFVIIa chimeras and mEPCR, isothermal titration calorimetry was used to measure enthalpy and entropy (ΔH and ΔS,35 respectively) and determine Kd and stoichiometry of interaction (n) in solution. Titration of msEPCR into mFVIIa resulted in weak heat measurements that did not allow for the accurate determination of thermodynamic terms (Figure 4A), indicating a weak interaction between the 2 proteins (Kd > 8 µM). In contrast, mFVIIa-FMR and mFVIIa-(1-43mPC) exhibited significant heat flow as a result of specific binding with msEPCR, as shown by the resulting isotherms (Figure 4B-C, respectively). Moreover, the Kd values [0.48 ± 0.02 µM for mFVIIa-FMR and 0.11 ± 0.01 µM for mFVIIa-(1-43mPC)] were in good agreement with our findings on CHO-K1-mEPCR cells. Collectively, our data confirm that binding to mEPCR either when it is presented on the cell surface or in solution can be engineered as a gain of function to mFVIIa by modification of its Gla domain. This can be achieved by a small change in the ω-loop of the protease (mFVIIa-FMR), thus suggesting that the mEPCR minimal binding region is located in the first 11 residues of mPC Gla domain.
Recombinant mFVIIa chimeras bind to msEPCR in contrast to mFVIIa. Isothermal titration calorimetry was used to study the interaction of (A) mFVIIa, (B) mFVIIa-FMR, and (C) mFVIIa-(1-43 mPC) with msEPCR. Upper portion of each panel: heat flow tracings were obtained upon successive injections (2 µL) of msEPCR into the sample cell containing 25 µM mFVIIa or mFVIIa chimeras in dialysate at 25°C. The first injection (0.5 µL) was eliminated from the analysis. The heat flow profile from an identical injection of msEPCR into dialysate was used as a reference and is shown offset (top of upper panel) for clarity. Bottom portion of each panel: corrected integrated heats (kilocalories per mole of injectant) were analyzed. The line is drawn according to parameters listed in Table 1.
Recombinant mFVIIa chimeras bind to msEPCR in contrast to mFVIIa. Isothermal titration calorimetry was used to study the interaction of (A) mFVIIa, (B) mFVIIa-FMR, and (C) mFVIIa-(1-43 mPC) with msEPCR. Upper portion of each panel: heat flow tracings were obtained upon successive injections (2 µL) of msEPCR into the sample cell containing 25 µM mFVIIa or mFVIIa chimeras in dialysate at 25°C. The first injection (0.5 µL) was eliminated from the analysis. The heat flow profile from an identical injection of msEPCR into dialysate was used as a reference and is shown offset (top of upper panel) for clarity. Bottom portion of each panel: corrected integrated heats (kilocalories per mole of injectant) were analyzed. The line is drawn according to parameters listed in Table 1.
Partial substitution of the Gla domain of mFVIIa does not affect its in vitro activity
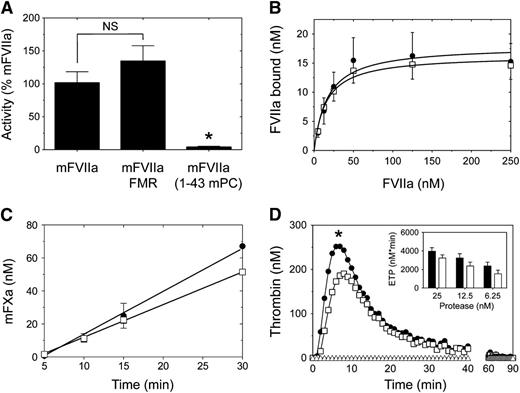
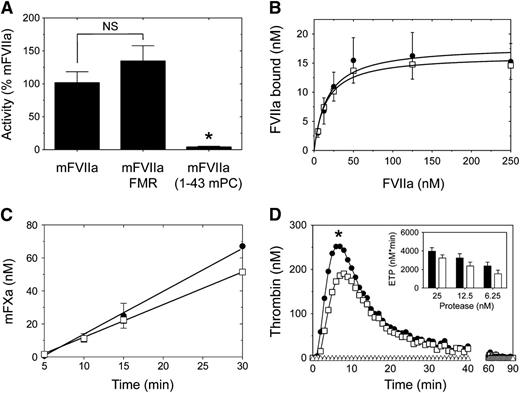
The Gla domain of human FVII contributes to the interaction with TF,36 thus affecting function within the initiation of coagulation. Using a clotting-based assay, we found that the partial Gla domain modification in mFVIIa-FMR did not affect activity (Figure 5A). In contrast, full transplant of the mPC Gla domain to mFVIIa in the mFVIIa-(1-43 mPC) chimeric molecule abolished activity (Figure 5A; P < .05 vs mFVIIa or mFVIIa-FMR). Therefore, we decided to further characterize the mFVIIa-FMR molecule only, relative to mFVIIa.
Comparison of mTF-dependent binding and in vitro activity of mFVIIa and mFVIIa chimeras. (A) Using a PT clotting assay, mFVIIa and mFVIIa-FMR exhibit similar activity. Asterisk indicates P < .05 vs mFVIIa or mFVIIa-FMR. Data are expressed as mean ± SD. NS, nonstatistically significant difference. (B) Binding of mFVIIa (●) or mFVIIa-FMR (☐) on CHO-K1 cells expressing mTF. Increasing concentration of ligand (10-250 nM) was incubated on CHO-K1 or CHO-K1-mTF cells, and the bound fraction was analyzed as described in Figure 3. Both ligands exhibit similar mTF binding. Depicted curves for each protein are fitted from 3 independent experiments. Data points are shown as mean ± SD. (C) Generation of mFXa over time on CHO-K1 cells expressing mTF following incubation of mFVIIa (●) or mFVIIa-FMR (☐). Both ligands exhibit similar rates of mFXa generation. Depicted curves for each protein are fitted from 3 independent experiments. Data points are shown as mean ± SD. (D) Mouse thrombin generation assay following the addition of 25 nM mFVIIa (●) or mFVIIa-FMR (☐) or buffer (△) in mouse hemophilia B plasma. *P < .05 vs mFVIIa of the entire fitted data. (Inset) Endogenous thrombin potential (ETP) following addition of different concentration of mFVIIa (black bar) or mFVIIa-FMR (white bar).
Comparison of mTF-dependent binding and in vitro activity of mFVIIa and mFVIIa chimeras. (A) Using a PT clotting assay, mFVIIa and mFVIIa-FMR exhibit similar activity. Asterisk indicates P < .05 vs mFVIIa or mFVIIa-FMR. Data are expressed as mean ± SD. NS, nonstatistically significant difference. (B) Binding of mFVIIa (●) or mFVIIa-FMR (☐) on CHO-K1 cells expressing mTF. Increasing concentration of ligand (10-250 nM) was incubated on CHO-K1 or CHO-K1-mTF cells, and the bound fraction was analyzed as described in Figure 3. Both ligands exhibit similar mTF binding. Depicted curves for each protein are fitted from 3 independent experiments. Data points are shown as mean ± SD. (C) Generation of mFXa over time on CHO-K1 cells expressing mTF following incubation of mFVIIa (●) or mFVIIa-FMR (☐). Both ligands exhibit similar rates of mFXa generation. Depicted curves for each protein are fitted from 3 independent experiments. Data points are shown as mean ± SD. (D) Mouse thrombin generation assay following the addition of 25 nM mFVIIa (●) or mFVIIa-FMR (☐) or buffer (△) in mouse hemophilia B plasma. *P < .05 vs mFVIIa of the entire fitted data. (Inset) Endogenous thrombin potential (ETP) following addition of different concentration of mFVIIa (black bar) or mFVIIa-FMR (white bar).
To investigate the mFVIIa-FMR affinity for the natural FVII/FVIIa cofactor (TF), we generated CHO-K1 cells stably expressing full-length mTF. We found that mFVIIa-FMR and mFVIIa exhibited similar affinity for mTF (Figure 5B), as demonstrated by the estimated Kd of 14.9 ± 11.1 (mFVIIa-FMR) and 16.9 ± 8.9 nM (mFVIIa). To determine the effect of Gla domain modifications on phospholipid binding, we determined the affinity binding constants using phospholipid binding assays (supplemental Materials). Under these experimental conditions, mFVIIa-FMR affinity was slightly reduced compared with mFVIIa (supplemental Figure 1). To verify that mFVIIa-FMR and mFVIIa can form functional complexes with mTF in the context of a cell membrane, we determined the rate of activation of mouse FX (mFX) following incubation of CHO-K1-mTF cells with either mFVIIa or mFVIIa-FMR. As shown in Figure 5C, both mFVIIa and mFVIIa-FMR exhibited similar rates of activated mFX (mFXa) generation (2.20 ± 0.07 and 2.00 ± 0.03 nM/minute, respectively). Last, to characterize the overall potential of the mFVIIa-FMR relative to mFVIIa on thrombin production in mouse plasma, we used a thrombin generation assay. To simulate hemophilia conditions, we supplemented mouse hemophilia B plasma with 25 nM mFVIIa or mFVIIa-FMR because this is sufficient to induce hemostasis in hemophilic patients.37 Addition of either mFVIIa or mFVIIa-FMR resulted in mouse thrombin generation, with mFVIIa-FMR exhibiting a modest but statistically significant (P < .05) reduction in peak of thrombin compared with mFVIIa (Figure 5D, main panel). However, both mFVIIa-FMR and mFVIIa exhibited similar endogenous thrombin potential, indicative of a similar amount of total mouse thrombin generated during the assay at all doses tested (Figure 5D, inset; P > .05 for mFVIIa vs mFVIIa-FMR at each dose shown).
Collectively, our data suggest that the modifications in the Gla domain present in mFVIIa-FMR do not affect its interaction with mTF or the function of the molecule compared with mFVIIa. Therefore, mFVIIa-FMR is functionally equivalent to mFVIIa but with the added ability to bind mEPCR.
mFVIIa-FMR molecule binds mEPCR in vivo
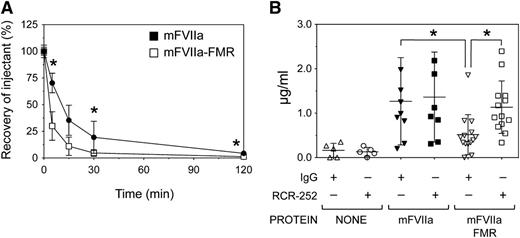
To address whether mFVIIa-FMR can bind mEPCR in vivo, we monitored protease levels in plasma over time, after intravenous bolus administration in C57BL/6 adult male mice. Animals received a dose of 500 µg/kg of mFVIIa or mFVIIa-FMR via a single tail vein injection, and blood plasma levels of administered protease (recovery, as percentage of injectant) were determined at different time points. As shown in Figure 6A, remarkably, within 5 minutes after administration, mFVIIa-FMR rapidly disappeared from the plasma, in contrast to mFVIIa (30.4 ± 6.8% vs 53.2 ± 19.3% recovery, respectively, P < .01). This lower recovery of mFVIIa-FMR vs mFVIIa was also evident at 30 and 120 minutes after administration (P < .05 for each time point).
The chimeric mFVIIa-FMR molecule can bind mEPCR in vivo. (A) C57BL/6 mice (n = 5 per group per time point) were infused via the tail vein with 500 µg/kg of mFVIIa or mFVIIa-FMR, and blood was collected at various time points thereafter. Plasma protein concentration was measured by antigen and activity assays and shown as recovery (percentage of the initial amount of injectant, mean ± SD). Recovery of mFVIIa-FMR is reduced compared with mFVIIa. Asterisk indicates P < .05 vs mFVIIa-FMR. (B) Cohorts of C57BL/6 mice were initially infused via the tail vein with 50 µg of either mEPCR blocking antibody (RCR-252) or isotype control IgG. The type of antibody administered in each cohort is indicated by the + or − symbol. One hour later, 500 µg/kg of mFVIIa, mFVIIa-FMR, or buffer was infused, and antigen levels at 5 minutes after injection were determined. *P < .05 for each indicated comparison. Data are expressed as mean ± SD.
The chimeric mFVIIa-FMR molecule can bind mEPCR in vivo. (A) C57BL/6 mice (n = 5 per group per time point) were infused via the tail vein with 500 µg/kg of mFVIIa or mFVIIa-FMR, and blood was collected at various time points thereafter. Plasma protein concentration was measured by antigen and activity assays and shown as recovery (percentage of the initial amount of injectant, mean ± SD). Recovery of mFVIIa-FMR is reduced compared with mFVIIa. Asterisk indicates P < .05 vs mFVIIa-FMR. (B) Cohorts of C57BL/6 mice were initially infused via the tail vein with 50 µg of either mEPCR blocking antibody (RCR-252) or isotype control IgG. The type of antibody administered in each cohort is indicated by the + or − symbol. One hour later, 500 µg/kg of mFVIIa, mFVIIa-FMR, or buffer was infused, and antigen levels at 5 minutes after injection were determined. *P < .05 for each indicated comparison. Data are expressed as mean ± SD.
Given the mEPCR binding capacity of mFVIIa-FMR, the lower recovery of mFVIIa-FMR suggests a potential mEPCR-dependent sequestration from circulation. To test this, we infused the RCR-252 mEPCR-blocking antibody38 or isotype control IgG 1 hour prior to tail vein administration of 500 µg/kg mFVIIa or mFVIIa-FMR, in C57BL/6 mice (Figure 6B). We assessed plasmatic levels of the proteases at 5 minutes after injection to maximize protein detection and differences in protein recovery. Because mFVIIa shows negligible affinity to mEPCR, administration of either mEPCR blocking or isotype control IgG prior to mFVIIa infusion had no effect on plasmatic levels. Mice that received mFVIIa-FMR after isotype control IgG showed reduced plasmatic levels compared with mFVIIa (P < .05), as expected. In contrast, blocking mEPCR prior to administration of mFVIIa-FMR resulted in increased plasmatic levels, similar to those observed for mFVIIa. Collectively, these data suggest that the modified Gla domain in mFVIIa-FMR allows it to bind specifically to mEPCR in vivo. As a result, changes observed in the mFVIIa-FMR recovery vs mFVIIa are dependent on specific binding to mEPCR.
EPCR binding enhances FVIIa hemostatic activity in vivo
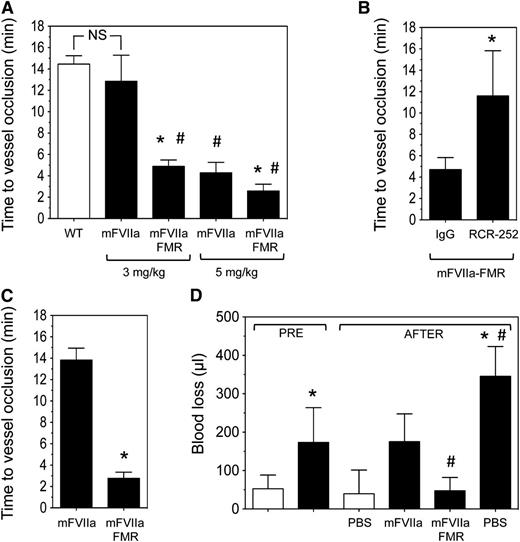
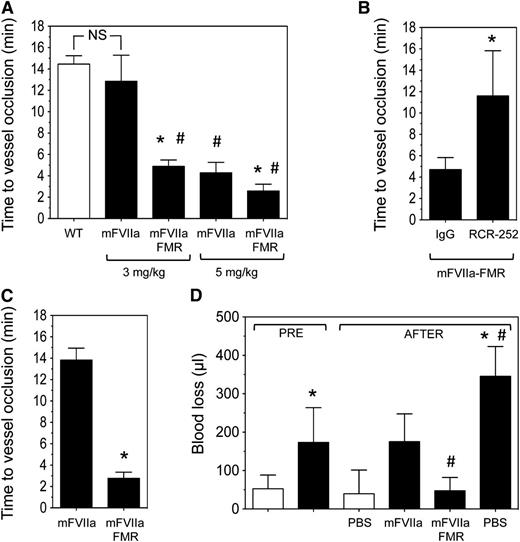
As EPCR expression is higher in large vessels compared with the microcirculation,39 we chose hemophilia mice that underwent a 7.5% FeCl3 injury on their carotid artery. The mice were subsequently infused via the jugular vein with mFVIIa, mFVIIa-FMR, or phosphate-buffered saline (PBS), and time to vessel occlusion as a result of thrombus formation was determined. Under these experimental conditions, a dose of 3 mg/kg of mFVIIa restored the hemostatic response in hemophilia B mice, resulting in vessel occlusion times comparable to those observed in C57BL/6 mice (Figure 7A). Remarkably, at the same dose, mFVIIa-FMR treated mice occluded ∼3 times faster compared with mFVIIa (4.9 ± 0.6 vs 12.9 ± 1.4 minutes, respectively; P < .05). The enhanced procoagulant properties of mFVIIa-FMR were also observed at 5 mg/kg, resulting in vessel occlusion after 2.6 ± 0.6 vs 4.3 ± 1.0 minute for mFVIIa (Figure 7A; P < .05).
Infused mFVIIa-FMR shows enhanced hemostatic potential in hemophilic mice compared with mFVIIa. (A) Hemophilia B mice (n = 3-5 per group) were subjected to a 7.5% FeCl3 injury, and blood flow was monitored for 10 minutes. Subsequently, procoagulant (mFVIIa or mFVIIa-FMR) was administered at the indicated dose via the jugular vein, and time to vessel occlusion was determined. Wild-type (WT) mice (n = 4) are shown for reference (white bar). Data are shown as mean ± SD. *P < .05 vs mFVIIa for each procoagulant dose; #P < .05 vs WT; NS, nonsignificant difference. In all experiments, blood flow was monitored for 30 minutes following protein infusion, and the time to carotid artery occlusion was defined as the time following application of FeCl3 (or protease infusion in the treated mice) until the blood flow has decreased by >90%. Hemophilic mice did not show vessel occlusion over the time of observation (data not shown). (B) Hemophilia B mice (n = 3-4 per group) were injected with 50 µg of EPCR-blocking antibody (RCR-252) or control isotype IgG. Thirty minutes later, they were subjected to a 7.5% FeCl3 injury, and blood flow was monitored for 10 minutes. Subsequently, mFVIIa-FMR was administered at 3 mg/kg via the jugular vein, and time to vessel occlusion was determined. Data are shown as mean ± SD. *P < .05 vs IgG. In all experiments, blood flow was monitored for 30 minutes following protease infusion, and the time to carotid artery occlusion was defined as the time following protease infusion until the blood flow has decreased by >90%. (C) Hemophilia A mice (n = 4-5 per group) were subjected to the same experimental setup as in A. Data are shown as mean ± SD. *P < .05 vs mFVIIa. (D) Hemophilia B mice (black bar, n = 4-5 mice per group) or WT (white bar, n = 5 mice) were subjected to a tail transection, and blood loss (in µL) was measured for a period of 2 minutes (PRE period). Immediately after, 3 mg/kg of mFVIIa or mFVIIa-FMR was infused, and blood loss (in µL) was measured for an additional 8 minutes (AFTER period). Quantitative assessment of blood loss was determined by measuring total hemoglobin by absorbance at 575 nm following red cell lysis and converted to total blood loss (μL) using a standard curve. WT mice infused with PBS are shown as a reference. Data are shown as mean ± SD. *P < .05 vs WT mice; #P < .05 vs mFVIIa.
Infused mFVIIa-FMR shows enhanced hemostatic potential in hemophilic mice compared with mFVIIa. (A) Hemophilia B mice (n = 3-5 per group) were subjected to a 7.5% FeCl3 injury, and blood flow was monitored for 10 minutes. Subsequently, procoagulant (mFVIIa or mFVIIa-FMR) was administered at the indicated dose via the jugular vein, and time to vessel occlusion was determined. Wild-type (WT) mice (n = 4) are shown for reference (white bar). Data are shown as mean ± SD. *P < .05 vs mFVIIa for each procoagulant dose; #P < .05 vs WT; NS, nonsignificant difference. In all experiments, blood flow was monitored for 30 minutes following protein infusion, and the time to carotid artery occlusion was defined as the time following application of FeCl3 (or protease infusion in the treated mice) until the blood flow has decreased by >90%. Hemophilic mice did not show vessel occlusion over the time of observation (data not shown). (B) Hemophilia B mice (n = 3-4 per group) were injected with 50 µg of EPCR-blocking antibody (RCR-252) or control isotype IgG. Thirty minutes later, they were subjected to a 7.5% FeCl3 injury, and blood flow was monitored for 10 minutes. Subsequently, mFVIIa-FMR was administered at 3 mg/kg via the jugular vein, and time to vessel occlusion was determined. Data are shown as mean ± SD. *P < .05 vs IgG. In all experiments, blood flow was monitored for 30 minutes following protease infusion, and the time to carotid artery occlusion was defined as the time following protease infusion until the blood flow has decreased by >90%. (C) Hemophilia A mice (n = 4-5 per group) were subjected to the same experimental setup as in A. Data are shown as mean ± SD. *P < .05 vs mFVIIa. (D) Hemophilia B mice (black bar, n = 4-5 mice per group) or WT (white bar, n = 5 mice) were subjected to a tail transection, and blood loss (in µL) was measured for a period of 2 minutes (PRE period). Immediately after, 3 mg/kg of mFVIIa or mFVIIa-FMR was infused, and blood loss (in µL) was measured for an additional 8 minutes (AFTER period). Quantitative assessment of blood loss was determined by measuring total hemoglobin by absorbance at 575 nm following red cell lysis and converted to total blood loss (μL) using a standard curve. WT mice infused with PBS are shown as a reference. Data are shown as mean ± SD. *P < .05 vs WT mice; #P < .05 vs mFVIIa.
To demonstrate that the enhanced hemostatic effects of mFVIIa-FMR are due to specific binding to mEPCR, we administered either an EPCR-blocking (RCR-252) or isotype control antibody prior to FeCl3 injury and infusion of 3 mg/kg of mFVIIa-FMR in hemophilia B mice. As expected, mFVIIa-FMR was unaffected by the control IgG infusion (vessel occlusion at 4.7 ± 1.1 minutes; Figure 7B). In contrast, blocking mEPCR resulted in a significant increase in vessel occlusion time (11.6 ± 4.2 minutes; Figure 7B; P < .05 vs IgG), similar to what was observed with mFVIIa at the same dose (Figure 7A). Last, to confirm that EPCR effects on FVIIa activity are not disease specific, we administered 3 mg/kg of mFVIIa or mFVIIa-FMR in hemophilia A mice. Similar to hemophilia B mice, infusion of mFVIIa-FMR resulted in faster vessel occlusion than mFVIIa (2.8 ± 0.6 vs 13.8 ± 1.1 minutes, respectively; P < .05; Figure 7C).
To further confirm the increased hemostatic effects of mFVIIa-FMR, we used a mouse model of hemostasis following injury. Specifically, hemophilia B mice had their tail transected, and blood loss was measured for 2 minutes. Then, 3 mg/kg of procoagulant (mFVIIa or mFVIIa-FMR) or PBS (control) was infused via the jugular vein, and subsequent blood volume loss was measured for an additional 8 minutes (Figure 7D). In this experimental setting, infusion of this dose of either procoagulant in hemophilia B mice was hemostatic and led to a normalization of blood loss compared with PBS. Importantly, in agreement with our observations in the FeCl3 injury model (Figure 7A), mFVIIa-FMR infusion was approximately threefold more effective in reducing blood loss compared with mFVIIa (48 ± 34 vs 175 ± 72 µL, respectively; P < .05).
To define the contribution of endothelial EPCR vs EPCR expressed on the blood cell compartment in the enhanced hemostatic effects of mFVIIa-FMR, we performed ex vivo whole blood coagulation assays in the absence of endothelium using ROTEM. Hemophilia B blood had significantly extended clot time and formed a negligible clot, evidenced by the undetectable α angle of the clot and very modest maximum clot firmness (Table 2). Addition of 1 µg/mL of either mFVIIa or mFVIIa-FMR in hemophilia B blood normalized the ROTEM parameters to the same extent (Table 2). The similar hemostatic effects of mFVIIa or mFVIIa-FMR were also observed at lower concentrations (0.1 or 0.01 µg/mL, data not shown). Because the ROTEM analysis eliminated the endothelial contribution to clot formation, based on the properties of mFVIIa-FMR, our data suggest for the first time that its specific binding to EPCR on the endothelium enhances its procoagulant effect in vivo.
Discussion
Although the hemostatic effects of rhFVIIa administration in hemophilia are undisputed, its mechanism of action remains unclear. Moreover, the functional significance of the rhFVIIa-EPCR interaction as it relates to pharmacologic rhFVIIa administration has also remained unestablished. To study this interaction in a hemophilia animal model, we characterized a novel chimeric mFVIIa molecule (mFVIIa-FMR) that, in contrast to mFVIIa, merges normal TF-dependent binding/activity and EPCR binding and therefore mimics the properties of rhFVIIa. Using well-established injury models in hemophilic mice, we show that mFVIIa-FMR infusion exhibited a superior hemostatic capacity compared with mFVIIa that was dependent on its binding to endogenous EPCR. Based on the properties of mFVIIa-FMR, the results from our model demonstrate for the first time in vivo that EPCR contributes to the correction of hemostasis in the setting of pharmacologic rhFVIIa administration in hemophilia.
Consistent with other reports in hemophilic mice with mFVIIa,14,30 the procoagulant doses used in this study were higher compared with humans, which may be related to the nature of injuries used. The affinity of mFVIIa-FMR for mEPCR is somewhat lower than that reported for human FVIIa or human PC and human EPCR.16,17 This may be reflective of methodological differences with previously published work and is further complicated by the fact that the strength of interaction of mEPCR with its natural ligand mPC is unknown. Nevertheless, the specific properties of mFVIIa and mFVIIa-FMR allowed for a comparative analysis that provide the first in vivo support for the coagulant effector function of an FVIIa-EPCR interaction. A plausible explanation for our observations is that engagement of EPCR-binding capacity of mFVIIa-FMR is an endothelial recruiter of the molecule in proximity to and/or the site of the injury where it participates in procoagulant reactions. Previous studies suggest that the activated endothelium can provide TF and support prothrombinase complex assembly and the early stages of thrombus formation including platelet accumulation and activation.40,41 Because TF-FVIIa-EPCR can form a complex with FX/FXa,19 EPCR tethering of mFVIIa-FMR on the endothelium may provide an extended locale of procoagulant reactions, leading to the observed enhanced procoagulant effects of mFVIIa-FMR vs mFVIIa. A role for the endothelium is supported by our ex vivo ROTEM data. EPCR occupancy by mFVIIa-FMR may also down-regulate activated mPC generation, and hence anticoagulation, resulting in the apparent enhancement of hemostatic function of mFVIIa-FMR. Such a hypothesis relies on effective competition of mPC-mEPCR binding by mFVIIa-FMR and is supported by 2 observations: (1) endogenous EPCR in mice appears to be a limiting factor for activated PC (aPC) generation42 ; and (2) the dose of mFVIIa-FMR administered (3 mg/kg) would likely reach plasmatic levels well beyond those of mPC. Such competition will likely occur on the endothelial surface, given that EPCR-dependent aPC generation occurs on the nonhematopoietic cell compartment.43 However, an EPCR-dependent additive FXa generation and aPC competition are nonexclusive hypotheses and can occur concurrently. Ultimately, real-time thrombus formation imaging will add the extent, temporal, and topographical information of the procoagulant reactions following mFVIIa-FMR infusion, including the contribution of the blood cell compartment.
Although our data suggest an EPCR role in FVIIa-mediated hemostasis in hemophilic mice, it is unclear why the mouse system lacks an endogenous FVII/FVIIa-EPCR interaction. In humans, support for an EPCR role in the engagement of FVIIa in inflammation has been previously provided.44 Additionally, an EPCR role in the FVII/FVIIa tissue distribution and, possibly, its catabolism has also been suggested.18 Such EPCR-dependent processes are not expected to be present in the mouse. Whether this is an evolutionary adaptation is unclear, given the lack of data on the FVII/FVIIa-EPCR interaction in other species, and more experiments need to be conducted.
Previously, enhancing the function of rhFVIIa has focused on amino acid modification relating to phospholipid membrane binding or catalytic function. Chemical modifications to increase the half-life of rhFVIIa have also been explored, as well as fusion to albumin or monomeric Fc.45 Based on our data, limited mutagenesis within the Gla domain of human FVIIa could reveal a novel class of rhFVIIa molecules with enhanced binding to EPCR and increased hemostatic function. Recombinant human FVIIa has also been used in prophylaxis. Notably, in a study of rhFVIIa secondary prophylaxis in hemophilic patients with inhibitors, patients receiving daily rhFVIIa infusions for 3 months, followed by a 3-month postprophylaxis period, showed clinical benefits even in the postprophylaxis period.46 Previous mouse studies suggested that rhFVIIa is transported via an EPCR-dependent mechanism to the extravascular space,18 where it can persist for extended periods of time in complex with TF,47 potentially preventing minor bleeds before they escalate. Such mechanism may explain the prolonged clinical benefits of rhFVIIa prophylaxis. In this context, enhancement of EPCR binding of rhFVIIa could also benefit prophylaxis-based therapies. This includes gene-based FVIIa approaches (as we have previously shown in mice and dogs22,25,48,49 ) directing continuous expression of FVIIa that simulate a prophylaxis setting. Of course, efficacy and any potential adverse effects (immunologic or thrombotic) of such novel therapies will need to be properly evaluated.
In conclusion, using a minimal modification of the Gla domain of mFVIIa, we generated and characterized a novel mFVIIa molecule (mFVIIa-FMR) with the capacity to interact with mEPCR in vitro and in vivo as a true gain of function. Using the mouse as a relevant platform, we demonstrate that EPCR on the endothelium is a previously unestablished component of the mechanism of action of rhFVIIa in hemophilia. This effector function of EPCR immediately suggests pathways to design novel protein or gene-based FVIIa therapeutics with increased efficacy. Moreover, given that FVIIa is involved in signaling through protease-activated receptors, our work may enable the use of the mouse to further understand physiologic or pathologic processes other than hemostasis that involve FVII/FVIIa and EPCR.
The online version of this article contains a data supplement.
The publication costs of this article were defrayed in part by page charge payment. Therefore, and solely to indicate this fact, this article is hereby marked “advertisement” in accordance with 18 USC section 1734.
Acknowledgments
The authors thank the following colleagues: Dr Sriram Krishnaswamy (University of Pennsylvania) for assistance with the analytical ultracentrifugation of msEPCR and insights throughout the course of the study; Dr Rodney M. Camire (University of Pennsylvania) for valuable help and providing the recombinant mouse FX and FXa; Harlan Bradford (University of Pennsylvania) for the γ-cabroxyglutamic analysis of recombinant proteins; and Dr Charles Esmon (University of Oklahoma) for the mouse hemangioendothelioma cell line.
This work was supported by the Bayer Hemophilia Awards Program through an Early Career Investigator Award (P.M.).
Authorship
Contribution: G.P. designed and performed research, analyzed data, and wrote the manuscript; L.I. performed research and analyzed data; A.F. performed research; O.A.M.-C. performed research and analyzed data; P.M. conceived the study, designed and performed research, analyzed data, and wrote the manuscript; and all authors commented on the final manuscript.
Conflict-of-interest disclosure: P.M. received research funding through a competitive grant from Bayer Hemophilia Awards Program, additional research funding from NovoNordisk A/S, and salary (spouse) from Shire Inc and Bristol-Myers Squibb. G.P. and P.M. are inventors on a pending Patent Cooperating Treaty (PCT) application. The remaining authors declare no competing financial interests.
Correspondence: Paris Margaritis, 5024 Colket Translational Research Building, 3501 Civic Center Blvd, The Children’s Hospital of Philadelphia, Philadelphia, PA 19104; e-mail: margaritis@email.chop.edu.

![Figure 1. Structure of the recombinant proteins used in this study. All proteins have a Gla domain, 2 epidermal growth factor (EGF) domains (EGF-1 and EGF-2), and a catalytic domain. A recognition site (Arg-Lys-Arg-Arg-Lys-Arg [RKRRKR]) for a paired amino acid cleaving enzyme (PACE)/furin type protease is inserted after the Arg at position 152. The sequence of the Gla domain is shown and highlighted in blue for each protein. Changes in the Gla domain compared with mFVIIa taken from the mouse PC Gla domain are highlighted in orange. Numbering refers to the mature form of each protein.](https://ash.silverchair-cdn.com/ash/content_public/journal/blood/124/7/10.1182_blood-2014-04-567297/5/m_1157f1.jpeg?Expires=1767734538&Signature=YHo9V0eYQ1BJchOiwfEfAxOhrkuIYgPwtiVYjd08O0xIg80i~ktKGl3R0PA0Kq6ScA03PwlcCUDLYMcYWoOGWPyiX3dABX1WL~GueYROUfeP1-Yxrxi8mQexhRwQJmP2yjpm2CWiWYR8K0hM8t5UfTp2CNUX7mRQrWD~8vkr-XWKLHZdvirhlmh~VESDWSY7f5t4hZitVsls2JDnfj-AuvonYkWKB2FXelmXULwVKvHBijRcBWNtFwNKEjvUQ2pwQz1NnEFusyKoh6zUoz5qkGyoImfY7WHLxfQ4ZXFtaOayalD8udQtNAMK6HuSFLFZ4Nnwg3yY8EqAheFtNWNEow__&Key-Pair-Id=APKAIE5G5CRDK6RD3PGA)



![Figure 1. Structure of the recombinant proteins used in this study. All proteins have a Gla domain, 2 epidermal growth factor (EGF) domains (EGF-1 and EGF-2), and a catalytic domain. A recognition site (Arg-Lys-Arg-Arg-Lys-Arg [RKRRKR]) for a paired amino acid cleaving enzyme (PACE)/furin type protease is inserted after the Arg at position 152. The sequence of the Gla domain is shown and highlighted in blue for each protein. Changes in the Gla domain compared with mFVIIa taken from the mouse PC Gla domain are highlighted in orange. Numbering refers to the mature form of each protein.](https://ash.silverchair-cdn.com/ash/content_public/journal/blood/124/7/10.1182_blood-2014-04-567297/5/m_1157f1.jpeg?Expires=1767806110&Signature=D-ALnue3jkuxCiGUHBlyTJf~D5TdDU1iWK9h~KknD9qoDLlA1NNDmekQWEtBn5D6M9wg1TvcknEKJLehQm32eNGYINmtXvCFfOiTja2FvmbsGlQwO-lTxtQ7jiSUvPVsJTdIJSR1Zad1LNtSYzRo-x2lLzodSm0mAUfBH-IGCB6lfe4RKqRltTTW-KZQmbU6J8o9klAJRZTBnSJvS3qXXWpfP45HSF~cn0H675zh6b6SX~6ve09fYJIoaB6wmlEqrT7TtYTrHzGzg8VtW-lvXW3oNS-REaPqMBAQcGnOvrhoMcQ0H~u6PipT1mO0zQ~CETAaCa2FjEkNLKB0s5YSfg__&Key-Pair-Id=APKAIE5G5CRDK6RD3PGA)