Abstract
The treatment of mice with anti-CD4 and anti-CD8 monoclonal antibodies (mAbs) on day −5, plus 3 Gy whole body irradiation (WBI) and 7 Gy thymic irradiation (TI) on day 0, allows fully major-histocompatibility-complex–mismatched allogeneic bone marrow engraftment and the induction of immunologic tolerance. TI is required in this model to overcome alloreactivity and possibly to make “space” in the recipient thymus so that lasting central tolerance can be achieved. In addition to suppressing mature T cells in the periphery, Cyclosporine A (CYA) and glucocorticoids have a powerful influence on the thymus. In this study, we evaluated whether the administration of CYA to recipient mice for 12 days prior to bone marrow transplant (BMT), of glucocorticosteroids on the day of BMT, or a combination of both, could create space and overcome alloresistance in the thymus by specifically depleting immature and mature thymocytes prior to BMT. High levels of multilineage donor hematopoietic repopulation and specific transplantation tolerance were achieved in mice treated from days −15 to −3 with CYA (20 mg/kg/d subcutaneously), anti-CD4/CD8 mAbs on day −5, followed by 3 Gy WBI and 15 × 106 allogeneic bone marrow cells on day 0. Vβ analysis suggested a central deletional tolerance mechanism. The same treatment without CYA pretreatment allowed only transient chimerism, without tolerance. Corticosteroid treatment abolished the engraftment-promoting and tolerance-inducing effects of CYA. These results demonstrate a novel pretransplantation-only application of CYA, which facilitates allogeneic marrow engraftment with minimal conditioning, by creating thymic space and/or overcoming intrathymic alloresistance.
The induction of immunological tolerance has become a major goal of transplantation research because modern immunosuppressive therapy has not improved chronic rejection rates and is associated with significant side effects. A state of donor-specific tolerance would obviate the risks of both acute and chronic rejection while eliminating the need for chronic immunosuppressive therapy, thereby permitting normal immunocompetence. Successful allogeneic bone marrow engraftment results in a state of specific transplantation tolerance. However, most applications of this approach to tolerance induction have involved the use of supralethal whole body irradiation (WBI) or high doses of total lymphoid irradiation followed by injection of allogeneic bone marrow cells (BMCs).1-3 Unfortunately, the severe toxic side effects of such regimens have precluded their use in humans as a means of inducing tolerance.
Several years ago, Cobbold et al4 developed a nonmyeloablative regimen for induction of permanent chimerism and transplantation tolerance that involved host treatment with depleting anti-CD4 and CD8 monoclonal antibodies (mAbs) and 6 Gy of WBI. Our group subsequently showed that similar results could be achieved with an even lower WBI dose, if thymic irradiation (TI) was added to the regimen.5 This nonmyeloablative regimen includes administration of depleting doses of anti-CD4 and anti-CD8 mAbs on day −5, 3 Gy WBI and 7 Gy TI on day 0, followed by injection of 1.5 × 107 fully major-histocompatibility-complex (MHC)–mismatched allogeneic BMCs on day 0. Mice treated in this way develop permanent mixed chimerism and donor-specific skin allograft tolerance.5 We have shown that the mechanism of tolerance in these animals primarily involves central deletion of donor-reactive T-cell clones resulting from the presence of donor-type antigen-presenting cells in the thymus, and that the maintenance of tolerance in this model depends upon the continuous intrathymic presence of donor hematopoietic cells.6-8 While animals receiving this conditioning regimen without BMT show no mortality and maintain excellent health, the mixed chimerism/central deletion approach to tolerance induction would have greater appeal if irradiation, with its associated risks, could be eliminated from the conditioning treatment.
We have recently reported that an additional treatment with depleting anti-CD4 and anti-CD8 mAbs can overcome the requirement for TI in order to achieve consistent allogeneic marrow engraftment, mixed chimerism, and specific transplantation tolerance.9 When 2 mAb injections were given before transplantation and 3 Gy WBI was given on day 0, most animals demonstrated high early levels of donor T-cell repopulation, lasting mixed chimerism, and specific skin graft acceptance.9 In contrast, when a single mAb injection was administered either on day −1 or on day −5, administration of TI was essential for the reliable induction of lasting chimerism and tolerance.5,6,9 A single administration of mAbs 5 days before BMT leads to almost complete depletion of T cells in the periphery of mice. However, alloreactive cells persist in the thymus and are thought to be responsible for the loss of chimerism and tolerance if no further treatment is given10 (also, B.N. and M.S., unpublished data, 1996). Consistent with this hypothesis, animals receiving a single mAb injection without TI have shown high early levels of chimerism in the periphery,5with an absence of chimerism in the thymus and a failure to delete donor-reactive host thymocytes.10 These animals may undergo intrathymic rejection of donor progenitors. Despite the presence of an initially successful hematopoietic graft, these animals later lose peripheral chimerism, presumably owing to the emergence of nontolerant host T cells from the thymus. The administration of a second mAb injection or TI seems to effectively overcome the persisting intrathymic alloresistance, thus allowing long-term chimerism to be achieved.
Additional studies in the Ly-5 congenic system suggest that thymic “space” for hematopoietic engraftment may be regulated independently from hematopoietic space11 and that specific measures are required to permit donor marrow to repopulate the recipient thymus early, even when very large numbers of donor marrow cells are administered. Therefore, it is possible that TI or a second mAb injection might be necessary not only to eliminate preexisting donor-reactive thymocytes, but also to create “thymic space,” which assures the intrathymic engraftment of large numbers of donor-derived cells and an early high level of donor-derived T-cell recovery.
Repeated injections of Cyclosporine A (CYA) have been shown to induce marked but reversible thymic dysfunction.12,13 CYA treatment blocks differentiation of immature CD4+CD8+ thymocytes into single-positive (SP) cells and induces thymic medullary involution with destruction of the medullary dendritic cells.12-15 After discontinuation of CYA, the immigration of dendritic cell precursors to the thymus is facilitated.16 Similarly, steroid hormones, such as glucocorticoids, have been shown to selectively deplete double-positive (DP) thymocytes and to affect several T lymphocyte functions.17 We hypothesized that combined steroid and CYA treatment might help to create thymic space by depleting the majority DP population, enhance the early migration of donor dendritic cells to the thymus, and also help to overcome alloresistance by specifically depleting mature SP thymocytes prior to BMT. Therefore, we hypothesized that host CYA pretreatment, injection of hydrocortisone on the day of BMT, or a combination of both treatments would enhance donor thymic repopulation and allow induction of permanent tolerance without TI or a second mAb injection.
Materials and methods
Animals
Female C57BL/6 (B6: H-2b; Kb, I-Ab, I-E-, Db), B10.A (B10.A: H-2a; Kk, I-Ak, I-Ek, Dd), and SJL (H-2s) mice were purchased from Harley-Sprague Dawley via Frederick Cancer Research Center, Frederick, MD. All mice were maintained in sterilized microisolator cages, in which they received autoclaved feed and autoclaved acidified drinking water. Recipients in each experiment were age-matched and were 10 to 12 weeks old.
Conditioning and BMT
Recipient B6 mice were treated with mAbs intraperitoneally on the indicated days (see below). Each injection18,19 consisted of 1.1 mg of purified rat antimouse CD4 immunoglobulin (Ig) G2b mAb GK1.5 and 0.7 mg of purified rat antimouse CD8 IgG2b mAb 2.43. On day 0, we administered 3 Gy WBI to mAb-treated animals, as described.5 At 4 to 6 hours after completion of conditioning on day 0, we intravenously administered 1.5 × 107 untreated BMCs from B10.A mice.
Pretreatment with Cyclosporine A and corticosteroids
Recipient B6 mice were pretreated with daily subcutaneous injections containing 20 mg/kg CYA (Sandimmune purchased from Sandoz, East Hanover, NJ) dissolved in sterile olive oil from day −15 to day −3 prior to BMT. Two groups of mice received 1 intraperitoneal injection of 20 mg/kg hydrocortisone acetate (Hydrocortone purchased from Merck Sharp & Dohme, West Point, PA) 4 hours prior to irradiation.
Flow cytometric analysis of chimerism
The level of allogeneic donor T-cell and non–T-cell reconstitution was evaluated by 2-color flow cytometric analysis on a FACScan or FACSort (Becton Dickinson, Mountain View, CA) as previously described.20 Briefly, forward-angle and 90° light-scatter properties were used to distinguish lymphocytes, granulocytes, and monocytes in peripheral white blood cells (WBCs). Two-color fluorescence cytometry (FCM) was used to distinguish donor and host cells expressing particular lineage markers, and the percentage of donor cells was calculated by subtracting control staining from quadrants containing donor and host cells expressing a particular lineage marker and by dividing the net percentage of donor cells by the total net percentage of donor plus host cells of that lineage. Nonspecific FcγR binding was blocked21 with 10 μL of undiluted culture supernatant containing rat antimouse FcγR mAb 2.4G2. Biotinylated anti–H-2Dd mAb 34-2-12 and control mAb HOPC1 were incubated22 with phycoerythrin-streptavidin (PEA). Fluorescein isothiocyanate (FITC)-conjugated mAbs were anti-CD4, anti-CD8, and anti-B220 (all purchased from Pharmingen, San Diego, CA) and MAC1 (Caltag, San Francisco, CA). Negative control mAb HOPC1-FITC, with no reactivity to mouse cells, was prepared in our laboratory. Dead cells were excluded by gating out low forward-angle light scatter/high propidium iodide-retaining cells.
T-cell receptor analysis
Peripheral blood lymphocytes (PBLs) and spleen cells were stained with FITC-conjugated anti–T-cell receptor (TCR) Vβ11, Vβ8.1/8.2, and Vβ5.1/5.2 mAbs (Pharmingen). For 2-color analysis, PBLs were labeled with FITC-conjugated HOPC-1 or anti-TCR Vβ11, Vβ8.1/8.2, or Vβ5.1/5.2 mAbs, and phycoerythrin-conjugated antimouse CD4 and CD8 mAbs (Pharmingen). At least 5000 gated CD4+cells were collected for Vβ analysis.
To determine the percentage of mature host thymocytes that were Vβ11+, Vβ8.1/8.2+, or Vβ5.1/5.2+, we collected 5000 gated H-2 Kbclass Ihigh cells (distinguished by bright staining above negative control biotinylated mAb HOPC-1/PEA) for analysis of staining with FITC-conjugated anti-Vβ mAb. Thymocytes were also stained with negative control FITC-conjugated HOPC-1 versus biotinylated anti-Kb mAb23 5F1. The percentage of gated H-2 Kb class Ihigh cells staining with control mAb HOPC-1 was subtracted from the percentage of gated H-2 class Ihigh cells staining with anti-Vβ11, Vβ8.1/8.2, or Vβ5.1/5.2 mAbs or with FITC-conjugated anti-panTCRβ mAb, respectively.
Immunohistochemical staining
Indirect immunoperoxidase staining of thymic sections (4 μm) was performed as we have previously described.10 For detection of mouse I-Ab+ cells, mouse IgG2aanti-I-Ab mAb 25-9-17 was used24with a biotinylated rat antimouse IgG2a mAb as secondary reagent. For detection of mouse I-E+ cells, mouse IgG2b anti-I-E mAb ISCR-3 was used25 with a biotinylated rat antimouse IgG2b mAb as secondary reagent. In each case, staining was compared with that using isotype-matched negative control mAbs HOPC-1 (mouse IgG2a isotype control) and 74-11-10 (mouse IgG2b isotype control) with the same secondary reagent. Staining was developed with the use of the Vectastain ABC kit (Vector Corp, Burlingame, CA), as described.10
Skin grafting
Initial skin grafting was performed 9 weeks following BMT as previously described.5 Square full-thickness tail skin grafts (1 cm2) were prepared from donors and grafted onto the right and left lateral thoracic wall of recipient mice. The first inspection was carried out on the seventh day, followed by daily inspection for the first month and then 2 to 3 times per week thereafter. Grafts were defined as rejected at the time of complete sloughing or when they formed a dry scab.
Statistical analysis
Statistical significance was determined by means of the Studentt test. A P value less than .05 was considered to be statistically significant.
Results
Pretreatment with CYA from day −15 to day −3 reduces the number of SP thymocytes
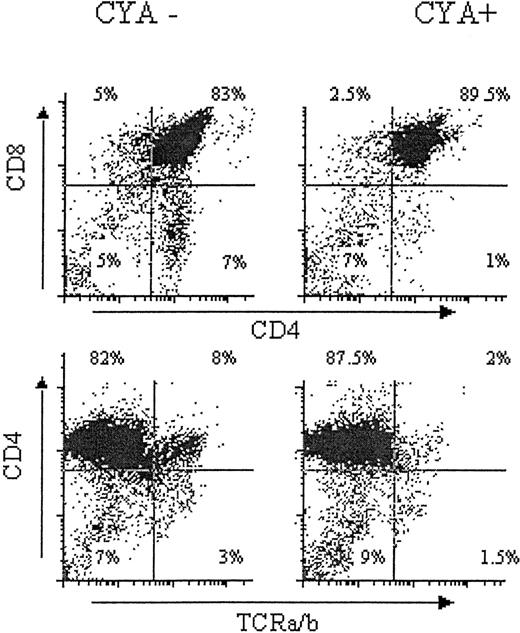
A 2- to 7-week course of treatment with CYA has been shown to decrease the absolute number of thymocytes and to most markedly decrease the number of SP mature thymocytes.13,14 To confirm this result with our treatment regimen, B6 mice were pretreated with daily subcutaneous injections containing 20 mg/kg CYA dissolved in sterile olive oil from day −15 to day −3 with respect to the time of BMT planned for future studies. In order to avoid the impairment of negative selection associated with BMT plus CYA treatment,14,26-28 we stopped the administration of CYA on day −3 prior to BMT. It was anticipated that this period of time would allow the clearance of CYA from the circulation prior to BMT. Mice treated only with olive oil were compared at the time of sacrifice on day 0 to mice that received CYA from days −15 to −3. A greater than twofold reduction in total thymocyte number, a 14-fold reduction in the absolute number of SP CD4+ thymocytes (sevenfold reduction in percentage of CD4 SP, Figure1), and a fourfold reduction in the absolute number of SP CD8+ cells thymocytes (twofold reduction in percentage of CD8 SP, Figure 1) were detected in the thymuses of CYA-treated mice compared with animals treated only with olive oil (Figure 1). Consistent with previous data, the majority of cells that disappeared with CYA treatment were mature SP T cells expressing high levels of TCRs (Figure 1, lower left and right) and high levels of class I MHC (not shown).12 13
Effects of CYA on developing thymocytes.
Mice were treated with daily subcutaneous injections containing sterile olive oil alone (upper and lower left, CYA−), or 20mg/kg CYA dissolved in sterile olive oil from day −15 to day −3 (upper and lower right, CYA+). Mice were killed at day 0. Age- and sex-matched experimental and control mice were treated and analyzed simultaneously. Unfractionated thymocytes were harvested and stained as indicated.
Effects of CYA on developing thymocytes.
Mice were treated with daily subcutaneous injections containing sterile olive oil alone (upper and lower left, CYA−), or 20mg/kg CYA dissolved in sterile olive oil from day −15 to day −3 (upper and lower right, CYA+). Mice were killed at day 0. Age- and sex-matched experimental and control mice were treated and analyzed simultaneously. Unfractionated thymocytes were harvested and stained as indicated.
CYA but not corticosteroid pretreatment permits engraftment of donor pluripotent hematopoietic stem cells (PPHSCs)
Groups of recipient B6 mice were prepared as indicated in Table1. Allogeneic repopulation was evaluated 2 weeks and later after BMT by staining peripheral WBCs with a mAb (34-2-12; anti-H-2Dd) that recognizes donor class I MHC. Early and lasting chimerism was observed in 4 of 5 recipients treated with anti-CD4 plus anti-CD8 mAbs on days −1 and −6, and 3 Gy WBI prior to injection of B10.A BMCs (Table 1, Group A), as described previously.9 In contrast, exclusion of the second mAb injection was associated with the initial development of chimerism in 4 of 5 animals, but none of the animals developed long-term chimerism (Table 1, Group B). These results confirmed previous results indicating that a second mAb injection was essential for the consistent induction of lasting chimerism in mice that did not receive TI prior to BMT.10
CYA pretreatment permits engraftment of B10.A bone marrow cells in B6 mice conditioned without thymic irradiation or a second mAb injection
| Group* . | Day mAbs administered . | CYA† . | Cortisone‡ . | Fraction of chimeric animals (mean % donor cells + SD)1-153 . | ||||
|---|---|---|---|---|---|---|---|---|
| 2 weeks . | 4 weeks . | 6 weeks . | 8 weeks . | 22 weeks . | ||||
| A | −1, −6 | − | − | 5/5 | 5/5 | 4/5 | 4/5 | 2/3 |
| (25 ± 2) | (32 ± 6) | (21 ± 16) | (23 ± 18) | (56 ± 18) | ||||
| B | −5 | − | − | 4/5 | 4/5 | 4/5 | 2/5 | 0/5 |
| (13 ± 5) | (25 ± 7) | (30 ± 15) | (19 ± 8) | (0) | ||||
| C1-155 | −5 | + | − | 10/10 | 9/9 | 9/9 | 9/9 | 9/9 |
| (12 ± 2) | (41 ± 5) | (48 ± 7) | (55 ± 6) | (70 ± 11) | ||||
| D | −5 | − | + | 3/5 | 1/5 | 1/5 | 1/5 | 1/5 |
| (8 ± 4) | (64) | (63) | (63) | (47) | ||||
| E | −5 | + | + | 3/4 | 2/4 | 1/4 | 1/4 | 1/4 |
| (12 ± 6) | (11 ± 17) | (35) | (31) | (27) | ||||
| Group* . | Day mAbs administered . | CYA† . | Cortisone‡ . | Fraction of chimeric animals (mean % donor cells + SD)1-153 . | ||||
|---|---|---|---|---|---|---|---|---|
| 2 weeks . | 4 weeks . | 6 weeks . | 8 weeks . | 22 weeks . | ||||
| A | −1, −6 | − | − | 5/5 | 5/5 | 4/5 | 4/5 | 2/3 |
| (25 ± 2) | (32 ± 6) | (21 ± 16) | (23 ± 18) | (56 ± 18) | ||||
| B | −5 | − | − | 4/5 | 4/5 | 4/5 | 2/5 | 0/5 |
| (13 ± 5) | (25 ± 7) | (30 ± 15) | (19 ± 8) | (0) | ||||
| C1-155 | −5 | + | − | 10/10 | 9/9 | 9/9 | 9/9 | 9/9 |
| (12 ± 2) | (41 ± 5) | (48 ± 7) | (55 ± 6) | (70 ± 11) | ||||
| D | −5 | − | + | 3/5 | 1/5 | 1/5 | 1/5 | 1/5 |
| (8 ± 4) | (64) | (63) | (63) | (47) | ||||
| E | −5 | + | + | 3/4 | 2/4 | 1/4 | 1/4 | 1/4 |
| (12 ± 6) | (11 ± 17) | (35) | (31) | (27) | ||||
CYA indicates Cyclosporine A; mAb, monoclonal antibody.
Each group received 3 Gy of whole body irradiation on day 0.
CYA given as described, from day −15 to day −3.
Cortisone was given as described, on day 0.
Animals defined as chimeric if level of donor class I+cells in white blood cells was above 2%. Mean percentage of donor class I+ cells in chimeric animals.
Animals pooled from 2 experiments.
We evaluated the possibility that the requirement for a second mAb injection or TI9 might be overcome by pretreating recipient mice with CYA, corticosteroids, or a combination of both. As is shown in Table 1, pretreatment of mice with CYA from day −15 to day −3, a single injection of anti-CD4 plus anti-CD8 mAbs on day −5, and 3 Gy WBI prior to BMT resulted in the induction of mixed chimerism in all animals, and this state persisted longer than 22 weeks (Table 1, Group C). The addition of CYA pretreatment led to a higher level of donor chimerism (from 4 to 22 weeks) compared with recipients of the protocol that includes a second mAb injection on day −1.
The administration of a single dose of corticosteroids on day 0, in addition to mAbs on day −5 and 3 Gy WBI prior to BMT, resulted in early chimerism in 3 of 5 animals at 2 weeks post-BMT, with lasting chimerism in only 1 of 5 animals (Table 1, Group D). The addition of corticosteroids to CYA pretreatment, anti-CD4 plus anti-CD8 mAbs, and 3 Gy WBI prior to BMT resulted in lasting chimerism in only 1 of 4 animals (Table 1, group E), suggesting that corticosteroid treatment had a deleterious effect on donor marrow engraftment.
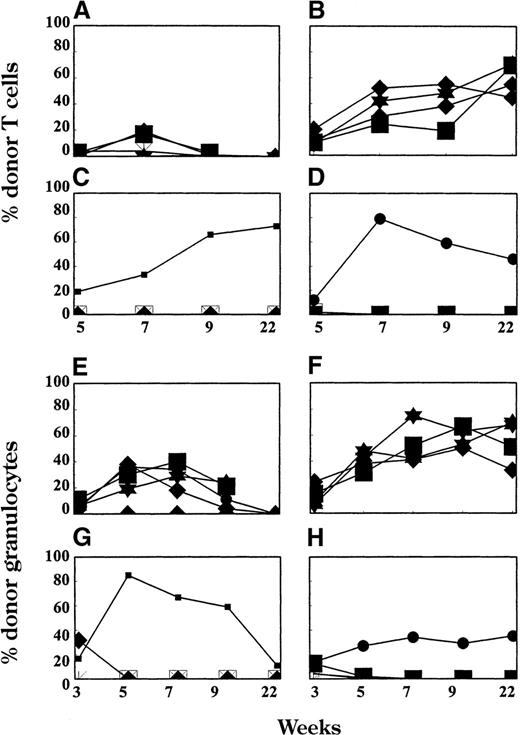
We followed the levels of donor chimerism in the T-cell (Figure2A-D), granulocyte (Figure 2E-H), and monocyte (not shown) lineages. Animals treated with only 1 injection of mAbs on day −5 (Group B from Table 1) developed only initial, transient T-cell chimerism with fewer than 20% donor-derived cells (Figure 2A). Of 5 animals that received a second mAb injection, 4 developed stable long-lasting chimerism in the T-cell, granulocyte, and monocyte lineages (not shown). Animals receiving pretreatment with CYA in addition to 1 mAb injection (Group C in Table1) showed high levels of early donor T-cell repopulation, which increased over time (Figure 2B). The addition of 1 dose of corticosterids on day 0 to 1 dose of mAbs on day −5 (Figure 2C) or to 1 dose of mAbs on day −5 and CYA on days −15 to −3 (Figure 2D) resulted in a complete lack of T-cell chimerism in all, except for 1 mouse per group. The lower panel of Figure 2 shows the level of donor chimerism in the granulocyte lineage, which in most of the animals followed the same pattern as T-cell chimerism.
T-cell (A-D) and granulocyte (E-H) chimerism in peripheral blood.
Donor representation among various hematopoietic lineages was determined at multiple time points after BMT by FACS. Each line represents 1 animal. Results from 1 of 2 similar experiments are shown. All mice received TCD mAbs on day −5, 3 Gy WBI (day 0), and 15 × 106 BMCs (day 0). Animals in Group B from Table 1 (n = 4; panels A and E) received no additional treatment. Group C (n = 5; panels B and F) received pretreatment with CYA from day −15 to day −3; Group D (n = 5; panels C and G) received 1 dose of corticosteroids on day 0; and Group E (n = 4; panels D and H) received pretreatment with CYA from day −15 to day −3 and 1 dose of corticosteroids on day 0.
T-cell (A-D) and granulocyte (E-H) chimerism in peripheral blood.
Donor representation among various hematopoietic lineages was determined at multiple time points after BMT by FACS. Each line represents 1 animal. Results from 1 of 2 similar experiments are shown. All mice received TCD mAbs on day −5, 3 Gy WBI (day 0), and 15 × 106 BMCs (day 0). Animals in Group B from Table 1 (n = 4; panels A and E) received no additional treatment. Group C (n = 5; panels B and F) received pretreatment with CYA from day −15 to day −3; Group D (n = 5; panels C and G) received 1 dose of corticosteroids on day 0; and Group E (n = 4; panels D and H) received pretreatment with CYA from day −15 to day −3 and 1 dose of corticosteroids on day 0.
At 41 weeks after BMT, the animals were killed and examined for chimerism. Long-term chimeras conditioned with CYA pretreatment, mAbs on day −5, and 3 Gy of WBI, had high levels of multilineage donor cell chimerism in both the spleen (52.9% to 63% donor-derived class I+ cells) and the bone marrow (40% to 70% donor-derived class I+ cells), whereas animals pretreated with only 1 mAb injection with or without corticosteroids had no detectable chimerism by FACS (not shown).
Skin graft survival
To evaluate tolerance in chimeric mice, donor B10.A and third-party SJL (H-2s) skin was grafted 9 weeks post-BMT. All grafted animals rejected third-party SJL skin grafts within 30 days (Figure 3B). In 4 of 4 recipients prepared with the regimen that included mAbs on day −5 only and 3 Gy WBI, B10.A skin grafts were rejected within 20 days (Figure 3A). In contrast, in 3 of 4 recipients prepared with the regimen that included mAbs on both day −5 and day −1, plus 3 Gy WBI, B10.A skin grafts were accepted for longer than 100 days. In 5 of 5 recipients of CYA pretreatment and mAbs only on day −5, and 3 Gy WBI, B10.A skin grafts were accepted for longer than 100 days, indicating that donor-specific skin allograft tolerance had been induced (Figure 3B). The administration of corticosteroids in addition to CYA pretreatment prevented the induction of skin graft acceptance in the majority of animals, with an outcome similar to that achieved in mice receiving corticosteroids without CYA (Figure 3A).
Donor and third-party skin graft survival.
(A) Donor B10.A skin graft survival for groups that received CYA pretreatment and a single mAb injection (n = 5, ▴); a single mAb injection and corticosteroids (n = 4, ▿); CYA pretreatment, a single mAb injection, and corticosteroids (n = 4, ⧫); 2 mAb injections (mAbs 2 ×, n = 4, ○); and a single mAb injection only (n = 4, ▪). (B) Third-party skin graft survival for the same groups. Donor B10.A and third-party SJL (H-2s) skin was grafted 9 weeks post-BMT. Results from the first of 2 similar experiments are shown.
Donor and third-party skin graft survival.
(A) Donor B10.A skin graft survival for groups that received CYA pretreatment and a single mAb injection (n = 5, ▴); a single mAb injection and corticosteroids (n = 4, ▿); CYA pretreatment, a single mAb injection, and corticosteroids (n = 4, ⧫); 2 mAb injections (mAbs 2 ×, n = 4, ○); and a single mAb injection only (n = 4, ▪). (B) Third-party skin graft survival for the same groups. Donor B10.A and third-party SJL (H-2s) skin was grafted 9 weeks post-BMT. Results from the first of 2 similar experiments are shown.
Clonal deletion of donor-reactive host thymocytes in mice receiving CYA pretreatment, mAbs on day −5, and 3 Gy WBI
The marrow donor used in our studies, B10.A, expresses I-E, the class II MHC molecule that most efficiently presents Mtv8- and Mtv9-associated superantigens that are endogenous to the C57BL background. As a result, deletion of developing thymocytes whose TCRs contain Vβ11 and Vβ5, which binds to these superantigens, is observed in B10.A mice.29,30 The B6 strain, on the other hand, cannot express the I-E molecules to which these superantigens bind and therefore does not delete Vβ11+ and Vβ5+ T cells from its repertoire.31 To determine whether or not intrathymic clonal deletion could also be the basis for T-cell tolerance in B6 mice receiving B10.A BMCs after pretreatment with CYA on days −15 to −3, anti-CD4 and anti-CD8 mAbs on day −5, and 3 Gy WBI on day 0, we measured the percentages of Vβ11+ and Vβ5+ host (ie, B6) mature thymocytes at 10 months following BMT. To select for mature host-type T cells, we measured these Vβ for gated H-2Kb high thymocytes, since this population consists mainly of mature, SP recipient T cells. When B10.A control mice were studied, gated H-2Dd highthymocytes were analyzed instead. As a nondeleted TCR family control, Vβ8.1/2-bearing thymocytes were also studied. Consistent with the maturity of thymocyte populations selected in this manner, only high-intensity staining with anti-Vβ mAbs was observed, indicating that TCRlow immature DP thymocytes had been gated out.
Mature thymocytes from B10.A mice were appropriately deleted, with Vβ11+ T cells composing only 0.33%, and Vβ5+ T cells composing only 0.38%, of thymocytes. In contrast, Vβ11+ and Vβ5+ cells composed 3% and 5.3%, respectively, of mature thymocytes in normal B6 mice (Figure4A). Thymuses from B6 mice receiving CYA pretreatment, anti-CD4 plus anti-CD8 mAbs on day −5, and 3 Gy WBI prior to B10.A BMT showed marked deletion of Vβ11+(average of 0.34%) and Vβ5+ (average of 0.8%) cells among mature B6 thymocytes (Figure 4A). In contrast to these results, intrathymic clonal deletion of Vβ11+ and Vβ5+ B6 thymocytes was not observed in B6 mice that had received B10.A BMT after being conditioned with 1 injection of anti-CD4 plus anti-CD8 mAbs, and 3 Gy WBI; lasting mixed chimerism was not successfully induced in these mice. Similarly, intrathymic clonal deletion of Vβ11+ and Vβ5+ T cells was not observed in B6 mice that had received corticosteroids alone or in combination with CYA (not shown).
Deletion at 41 weeks post-BMT of (A) donor-reactive mature host thymocytes and (B) CD4+ splenocytes.
Results are shown as mean ± standard deviation. Values are for normal age-matched B6 mouse (□), normal B10.A mouse (▨), day −5 mAb-injected group (▪, n = 4), and day −5 mAb-injected group receiving CYA pretreatment ( , n = 2). Values are compared with those of age-matched B6 mice. *P < .05. **P < .005, ***P < .0005.
, n = 2). Values are compared with those of age-matched B6 mice. *P < .05. **P < .005, ***P < .0005.
Deletion at 41 weeks post-BMT of (A) donor-reactive mature host thymocytes and (B) CD4+ splenocytes.
Results are shown as mean ± standard deviation. Values are for normal age-matched B6 mouse (□), normal B10.A mouse (▨), day −5 mAb-injected group (▪, n = 4), and day −5 mAb-injected group receiving CYA pretreatment ( , n = 2). Values are compared with those of age-matched B6 mice. *P < .05. **P < .005, ***P < .0005.
, n = 2). Values are compared with those of age-matched B6 mice. *P < .05. **P < .005, ***P < .0005.
Figure 4B shows that this intrathymic deletion was reflected among the CD4 SP T cells in the spleen. An absence of Vβ11+ and Vβ5+ CD4 T cells was observed in the spleens of untreated B10.A mice and in long-term chimeras prepared with CYA pretreatment, anti-CD4 plus anti-CD8, and 3 Gy WBI (Figure 4B). However, there was no deletion of these Vβ families in control B6 mice, which received only conditioning without BMT, or in mice treated with mAbs on day −5 and with 3 Gy WBI and B10.A BMCs, which demonstrated only early, not lasting chimerism. Similarly, deletion of splenic Vβ11+and Vβ5+ CD4+ T cells was not observed in transplanted B6 mice that had received corticosteroids alone or in combination with CYA (not shown).
At the time of sacrifice (41 weeks post-BMT), immunohistochemical staining was performed to examine the presence of donor-derived class II+ cells in the thymuses of chimeras. In all chimeras that received CYA pretreatment from day −15 to day −3, anti-CD4 plus anti-CD8 on day −5, and 3 Gy WBI prior to BMT, a large number of donor-derived class II+ cells with a dendritic morphology was detected in the corticomedullary and medullary regions (not shown). Mice pretreated with only 1 injection of mAbs on day −5 and 3 Gy WBI, and a control mouse receiving the conditioning without BMT, were negative for donor class II+ cells (not shown). The presence of donor class II+ cells correlated with the deletion of donor-reactive cells in the thymuses of chimeras, further supporting the conclusion that intrathymic deletion was a major mechanism of tolerance.
Discussion
We demonstrate here that pretreatment of recipients with CYA can overcome the requirement for thymic irradiation or for an additional dose of depleting anti-CD4 and anti-CD8 mAbs in order to achieve consistent allogeneic marrow engraftment, mixed chimerism, and specific transplantation tolerance in mice receiving 3 Gy WBI and a peripheral T-cell–depleting mAb treatment. This, to our knowledge, is the first such application of CYA, in which the drug is not used to suppress the immune response after donor grafting, but is instead used to condition the thymus to make it permissive for donor engraftment. These results advance our effort to develop a more specific, targeted approach to recipient conditioning that uses the minimal amount of myelosuppression and immunosuppression required to permit partial reconstitution by an allogeneic marrow donor.
CYA is an important immunosuppressive drug that is widely used in transplantation medicine. In addition to suppressing mature T cells in the periphery, CYA has a powerful influence on the thymus. Short treatment with CYA decreases the absolute number of thymocytes (approximately 50%) and reduces the SP thymocyte number by 90%.12 CYA exerts many of its effects via specific inhibition of TCR-mediated T-cell activation. The precise point at which CYA blocks thymocyte maturation is unclear. However, the relative lack of CYA-mediated effects on the development of CD4−8− (double negative) and CD4+8+ (DP) cells,12,14 as well as the lack of a CYA-mediated effect on TCR gene rearrangement,12 suggests that the inhibition occurs at a later stage in thymocyte development, perhaps at the transition of DP to SP cells.12
With TI, intrathymic engraftment of large numbers of donor-derived progenitors and, hence, high initial levels of donor-derived T-cell reconstitution are achieved. TI can be omitted if a second injection of anti-CD4 and anti-CD8 mAbs is administered 1 day prior to BMT.9 In support of the hypothesis that administration of a second mAb injection depletes or inactivates residual host thymocytes that are capable of causing intrathymic rejection of donor hematopoietic cells even when peripheral engraftment is achieved, we have detected residual cytotoxic lymphocyte and mixed lymphocyte reaction alloreactivity in thymuses of animals receiving a single mAb injection and 3 Gy of WBI. In contrast, no alloreactivity was detected in thymuses of animals receiving mAbs on days −6 and −1 and 3 Gy WBI (B.N. and M.S., unpublished data). Therefore, CYA pretreatment–induced depletion or inactivation of mature alloreactive SP thymocytes may enhance early donor thymic repopulation and induction of permanent tolerance by depleting the SP thymocytes that are presumably responsible for this alloreactivity in recipients of a single mAb injection.
In addition to suppressing positive selection and SP thymocyte maturation, CYA treatment blocks the deletion of potentially autoreactive T-cell clones among the small number of αβ T cells that do mature in its presence.12,14 Clonal deletion is prevented by interference with TCR-mediated signal transduction and by reduction of I-A molecule expression on bone marrow–derived elements.12,32 A short course of CYA treatment induces thymic involution with destruction of the medullary dendritic cells (DCs) and medullary epithelium. In contrast, the cortex of CYA-treated rodents is relatively intact, and the thymus consists almost entirely of cortical tissue.32 The autoreactivity of T cells that escape clonal deletion is not manifested until the withdrawal of CYA, which relieves the block in lymphokine production and initiates the autoimmune process.33 Indeed, irradiated hosts transplanted with syngeneic bone marrow and then treated with and withdrawn from CYA develop autoimmunity. This phenomenon, which is observed in rats,27 mice,26,28 and humans,34,35 is characterized by a graft-versus-host disease (GVHD)-like syndrome that is transferable to naive recipients by T cells.36 To prevent failure of negative selection of donor and host-reactive thymocytes in our model, we stopped the administration of CYA on day −3 prior to BMT. Also, the administration of mAbs, which remain detectable in the circulation for the first 2 weeks post-BMT,10 and of 3 Gy of WBI on day 0, makes the occurrence of clinical GVHD or antidonor reactivity as a consequence of our treatment very unlikely. We have followed animals for a year after BMT, and no signs of GVHD or loss of donor-specific tolerance have been observed.
The development of central tolerance depends on the appropriate presentation of antigens to developing thymocytes. The DCs represent the principal blood-borne antigen-presenting cells in the thymus. A short course of CYA depletes the thymus of the medullary DCs. In one study, Brown Norway (BN) rats received (LEW × BN) F1 splenocytes followed by a short course of CYA.16 After CYA was stopped, the F1 cells were rapidly recruited to the thymus, and by 10 days after CYA, they were localized to the corticomedulary junction, the natural location of thymic dendritic cells.16 We hypothesized that the facilitated recruitment of new donor-derived DCs into the host thymus might therefore enhance deletion of alloreactive host-derived T cells and tolerance induction to alloantigens. Since the host and not the donor hematopoietic progenitors are subjected to 3 Gy WBI, donor progenitors might have an advantage over those of the host for early repopulation of the thymus. This is suggested by the increased levels of donor T-cell reconstitution observed at relatively early time points in CYA-pretreated mice.
Although corticosteroids are known to enhance immunosuppression and to deplete DP thymocytes, therefore potentially creating thymic space, they did not mediate any beneficial effect on donor marrow engraftment or tolerance induction. Surprisingly, the addition of corticosteroids to CYA pretreatment impeded the engraftment of donor cells and the induction of tolerance. In a recent study, dexamethasone, which induces cortical involution, did not lead to thymic recruitment of (LEWxBN) F1 DCs when administered to BN rats.16 CYA, which induces medullary involution, had the opposite effect.16 Accordingly, by depleting the medulla of DCs, CYA may create space for the homing of new DCs to the medulla during thymic regeneration. In contrast, the cortical involution induced by steroids fails to create space in the medulla for new DCs.
Our data suggest that the detrimental effect of corticosteroids on tolerance induction may be due to impairment of engraftment of donor hematopoietic cells. As shown in Figure 2, the animals that received corticosteroids showed a lack of chimerism as early as 5 weeks post-BMT, suggesting an initial failure to engraft. We do not know of any reason to expect such an effect or of any precedent for it, but the result emphasizes the necessity for a careful analysis of the effects of administering of corticosteroids around the time of BMT.
Overall, our studies indicate that specific measures are required to permit donor progenitors to repopulate the recipient thymus at high levels. CYA pretreatment could be a valuable, relatively nontoxic, and highly specific treatment for creating a permissive thymic environment for donor-derived cells. Instead of receiving CYA for a prolonged period, a recipient would receive only a short course of CYA followed by BMT from a living donor as a lifelong source of donor-derived DCs. Donor hematopoietic stem cell engraftment would result in a robust state of central deletional tolerance to organ grafts from the same donor. This approach might also be potentially applicable to xenotransplantation, in which the host could be pretreated with CYA for several weeks in anticipation of marrow and organ xenotransplantation.
Acknowledgments
The authors thank Diane Plemenos for assistance in preparing the manuscript; Drs Dave Anderson, Hugh Auchincloss, and Joey Kurtz for helpful review of the manuscript; and Dr David H. Sachs for helpful advice.
Supported by National Institutes of Health Grant ROI HL49915 and by a sponsored research agreement between Massachusetts General Hospital and BioTransplant, Inc. Supported in part (B.N.) by The Daland Fellowship for Research in Clinical Medicine (American Philosophical Society) and an American Society of Transplant Physicians–Novartis Fellowship in Transplantation.
Reprints:Megan Sykes, Bone Marrow Transplantation Section, Transplantation Biology Research Center, Massachusetts General Hospital, MGH East, Building 149-5102, 13th St, Boston, MA 02129; e-mail: megan.sykes@tbrc.mgh.harvard.edu.
The publication costs of this article were defrayed in part by page charge payment. Therefore, and solely to indicate this fact, this article is hereby marked “advertisement” in accordance with 18 U.S.C. section 1734.



This feature is available to Subscribers Only
Sign In or Create an Account Close Modal