Abstract
The present review describes the biology of human leukocyte antigen haplotype mismatched (“haploidentical”) transplantation, its translation to clinical practice to cure leukemia, and the results of current transplantation protocols. The 1990s saw what had been major drawbacks of haploidentical transplantation, ie, very strong host-versus-graft and graft-versus-host alloresponses, which led respectively to rejection and graft-versus-host disease (GVHD), being overcome through transplantation of a “mega-dose” of T cell–depleted peripheral blood hematopoietic progenitor cells and no posttransplant pharmacologic immunosuppression. The absence of posttransplant immunosuppression was an opportunity to discover natural killer cell alloreactions that eradicated acute myeloid leukemia and improved survival. Furthermore, it also unveiled the benefits of transplantation from mother donors, a likely consequence of the mother-to-child interaction during pregnancy. More recent transplantation protocols use unmanipulated (without ex vivo T-cell depletion) haploidentical grafts combined with enhanced posttransplant immunosuppression to help prevent GVHD. Unmanipulated grafts substantially extended the use of haploidentical transplantation with results than even rival those of matched hematopoietic transplantation. In T cell–depleted haploidentical transplantation, recent advances were made by the adoptive transfer of regulatory and conventional T cells.
Allogeneic hematopoietic stem cell transplantation is the most powerful therapy for high-risk acute leukemia.1 Donor T cells recognize host alloantigens on leukemic cells and eradicate leukemia (graft-versus-leukemia [GVL] effect). They also attack nonhematopoietic tissues (such as skin, gut, and liver) and thus mediate graft-versus-host disease (GVHD), a major cause of morbidity and mortality. Matching donor and recipient human leukocyte antigens (HLA) is considered crucial for optimal transplant outcomes with acceptable nonrelapse mortality (NRM).1 However, only 25% of individuals have an HLA-identical sibling who could serve as donor. Alternative stem cell sources (which of course carry increased risk of NRM due to diverse combinations of graft failure, GVHD, veno-occlusive disease, and infections) are matched unrelated volunteers, unrelated umbilical cord blood, and HLA haplotype mismatched (“haploidentical”) family members.1,2 In fact, recent years have witnessed tremendous progress in the field of haploidentical transplantation.3-10
The experience of T cell–depleted haploidentical transplantation and the discovery of donor features that improve outcomes
Haploidentical transplantation has become a widely used treatment of patients without a matched donor.2-15 Indeed, a haploidentical donor is immediately available in the family for almost all patients, without delay due to registry search. Because of multiple class I and II HLA disparities between donor and recipient, T cell–mediated host-versus-graft and graft-versus-host alloresponses (leading, respectively, to rejection and GVHD) are very strong after haploidentical transplantation. As anticipated by murine transplantation models,16 pioneering clinical studies showed that a “mega-dose” of haploidentical T cell–depleted granulocyte colony-stimulating factor (G-CSF)–mobilized peripheral blood hematopoietic progenitor cells engrafted in 95% of leukemia patients.11-13 T-cell depletion was achieved by ex vivo CD34+ cell selection and in vivo antithymocyte globulin administration, and it was the only form of GVHD prophylaxis. Less than 10% of patients developed acute and chronic GVHD. With >15 years of follow-up, relapse incidence was 18% in patients with acute myeloid leukemia (AML) and 30% in patients with acute lymphoblastic leukemia (ALL) who were transplanted in any complete remission (CR).3,6,11-13 In a retrospective European Bone Marrow Transplantation registry–based survey of T cell–depleted haploidentical transplantation in adults with acute leukemia, 2-year relapse incidence was 21% in patients with AML and 27% in patients with ALL in any CR at transplant.17 T-cell depletion was associated with slow posttransplant immune reconstitution. Indeed, these studies reported ∼40% NRM, mainly caused by infections.11-13,17
Effectiveness of donor-versus-recipient natural killer–cell alloreactivity
In T cell–depleted haploidentical transplantation, the absence of any posttransplant immunosuppression favored the emergence of natural killer (NK)-cell alloreactions that reduced relapse rates and improved survival.18-22 NK-cell function is regulated by a balance of signals mediated by activating and inhibitory receptors.23-27 Human NK cells express inhibitory receptors for HLA class I molecules, such as killer cell immunoglobulin-like receptors (KIRs) and the C-type lectin-like CD94/NKG2A receptor. Inhibitory KIRs recognize epitopes shared by groups of HLA class I alleles (KIR ligands). KIR2DL1 recognizes HLA-C group 2 alleles, KIR2DL2 and KIR2DL3 recognize HLA-C group 1 alleles, and KIR3DL1 is the receptor for HLA-Bw4 alleles.25-27 NK cells become fully functional (“licensed/educated”) on interaction with self-HLA molecules.23 Educated NK cells react to the lack of self-HLA expression on allogeneic targets (so-called “missing self-recognition”).28-31 After hematopoietic transplantation, reconstituting NK cells are educated by HLA KIR ligands on donor hematopoietic cells.32 In the presence of KIR ligand mismatches in the graft-versus-host direction (ie, the donor possesses an HLA class I KIR ligand that is absent in the recipient), NK cells that express a KIR for an HLA class I group that is absent on allogeneic targets (as their only inhibitory receptor for self) sense the missing expression of the self-HLA and mediate alloreactions.18-21 Combined evidence from in vitro studies, murine models, and clinical trials indicated donor-versus-recipient alloreactive NK cells killed myeloid-lineage cells, including AML, and mature B-lineage cells (such as chronic lymphocytic leukemia and multiple myeloma). In contrast, B-cell precursor ALL was not susceptible.18-21 Remarkably, not only do alloreactive NK cells not cause GVHD, but even protect from T cell–mediated GVHD as they kill recipient antigen presenting cells.19,33 After haploidentical transplantation, recipients display an NK-cell repertoire of donor origin that contains alloreactive NK cells that kill recipient hematopoietic targets. Such cells persist for up to 12 months.20 In AML patients, donor-versus-recipient NK-cell alloreactions exerted a marked GVL effect, with a very low relapse incidence (3%) in patients transplanted in any CR. This translated into a remarkable survival advantage that is 67% leukemia-free survival in patients in any CR at transplant.20 These findings were confirmed in T cell–depleted haploidentical transplantation in children with acute leukemia.34,35 The results in T cell–depleted haploidentical transplantation prompted clinical trials that proved safety of NK-cell infusions and documented promising clinical responses in leukemia patients.36-40
Additional benefit of NK alloreactive donors with concomitant activating KIRs
The effects of donor KIR gene diversity were recently investigated in a series of T cell–depleted haploidentical transplants.22 Approximately 25% of whites are homozygous for group A KIR haplotypes that contain the main inhibitory KIR genes. The others are either heterozygous or homozygous for group B KIR haplotypes that also carry various combinations of activating KIR genes (KIR2DS1, 2, 3, 5, and 3DS1).27 It is well established that KIR2DS1 binds HLA-C group 2 molecules with less affinity compared with its inhibitory counterpart KIR2DL1.41 Little is known about the ligands of other activating KIRs. Interestingly, several studies reported associations between activating KIR genes and the outcome of viral infections.42 HIV-infected individuals who were homozygous for KIR3DS1 and expressed the HLA-Bw480I allele progressed slowly to AIDS.43 The presence of activating KIR genes was associated with less cytomegalovirus reactivation after solid organ and HLA-identical hematopoietic transplantation.44-47 Finally, expansion of NK cells expressing activating KIRs was documented in cytomegalovirus-seropositive individuals.48 Haploidentical transplantation from NK alloreactive donors with concomitant KIR2DS1 and/or KIR3DS1 activating genes was associated with reduced incidence of infectious mortality and improved survival.22 Apparently, when KIR–ligand mismatches release NK cells from recipient HLA blockade, activating KIRs are allowed to enhance NK-cell functions on binding to their ligands on recipient cells. Indeed, in vitro analyses showed that KIR2DS1 binding to its HLA-C group 2 ligand upregulated interferon γ production by alloreactive NK cells in response to infectious challenge.22
Improving outcomes using the mother as donor
The donor–recipient relationship is another key factor that influences outcomes of T cell–depleted haploidentical transplantation.49,50 Trans-placental trafficking of maternal and fetal cells during pregnancy establishes long-term, reciprocal micro-chimerism in both mother and child.51 As a consequence, the immune system of the mother may become sensitized to paternal histocompatibility antigens. In fact, antibodies directed against paternal HLA antigens and T lymphocytes directed against paternal major and minor histocompatibility antigens were detected in multiparous women.52-54 It was hypothesized that mother’s “exposure” to paternal HLA haplotype antigens during pregnancy may affect transplantation outcomes when the mother acts as donor for the child. Indeed, survival after T cell–depleted haploidentical hematopoietic transplantation was improved using the mother as donor (vs all other family members).49 More recently, a retrospective European Bone Marrow Transplantation registry–based study in 438 haploidentical transplants (78% of patients received T cell–depleted grafts, 22% received unmanipulated grafts) for acute leukemia patients (pediatric and adult) showed that transplantation from mother donors, compared with transplantation from any other family member, is an independent factor predicting lower relapse, lower NRM, and better survival.50 One may hypothesize that T-cell depletion removed enough T cells to prevent GVHD but allowed transfer of sufficient numbers of tolerant/educated T cells that protected the child from relapse and infections. Whether and how such a tolerant/educated T-cell repertoire is generated is under investigation. Thus, NK alloreactive donors (preferentially those with activating KIRs) and/or mother donors should be preferred when selecting a donor for T cell–depleted haploidentical transplantation.18-22,49,50,55 Studies are needed to establish a hierarchy among such donor selection criteria.
Haploidentical transplant protocols using unmanipulated grafts
In recent years, the haploidentical transplantation field saw the development of unmanipulated grafts (ie, without ex vivo T-cell depletion) combined with new strategies to attenuate/modulate donor T-cell alloreactivity and help prevent GVHD, eg, posttransplant high-dose cyclophosphamide,10,56-67 in vivo T-cell depletion of G-CSF–mobilized peripheral blood hematopoietic grafts,68,69 or G-CSF–primed blood and/or marrow grafts and intensified posttransplant immunosuppression.9,70-74 Unmanipulated hematopoietic transplants relay on the high T-cell content of the graft for engraftment and immune reconstitution. Unfortunately, the large number of T cells in the graft and the consequent need for posttransplant immunosuppressive GVHD prophylaxis can obscure the benefits of both donor-versus-recipient NK-cell alloreactivity and transplantation from mother donors.55,75-77
Posttransplant high-dose cyclophosphamide
In murine models, it was shown that posttransplant cyclophosphamide, when given within a narrow, 2- to 3-day, window after T-cell receptor (TCR) stimulation (at the time of peak DNA synthesis), targets recipient and donor activated alloreactive T cells (thus reducing the risk of rejection and GVHD).10 After a nonmyeloablative conditioning regimen comprised of cyclophosphamide, fludarabine, and low-dose (2-Gy) total body irradiation (TBI) and bone marrow transplantation, the Johns Hopkins and the Fred Hutchinson Cancer Research groups gave high-dose cyclophosphamide (on days +3 and +4) for GVHD prophylaxis (followed by mycophenolate mofetil and tacrolimus).56 In a recent retrospective study of 372 adult patients with a variety of hematological malignancies, 1-year NRM incidence was 11%, and incidences of acute GVHD and 2-year chronic GVHD were 32% and 13%, respectively. Three-year relapse incidence was 46% and progression-free survival was 40%.57 The Genoa group used a myeloablative conditioning regimen that comprised thiotepa, busulfan, and fludarabine or TBI and fludarabine.58,59 In 148 adult patients with a variety of hematologic malignancies, they reported 4-year NRM was 14%, and incidences of acute and chronic GVHD were 18% and 20%, respectively. Incidence of relapse-related death was 27%. It was 11% in patients in first complete remission (CR1) at transplant, 26% in patients in second complete remission (CR2) at transplant, and 40% in patients transplanted with active disease. With a median follow-up of 430 days, overall survival was 77% in patients in CR1 at transplant, 49% in patients in CR2 at transplant, and 38% in patients transplanted with active disease.59 Other groups used G-CSF–mobilized peripheral blood hematopoietic cell grafts with similar results.10,60-65 A recent study reported results from a cohort of AML patients (74% in CR1 or CR2 and 26% in relapse at transplant), who received bone marrow or peripheral blood hematopoietic grafts.66 Incidence of 3-year NRM was 14% after myeloablative conditioning regimens and 9% after reduced intensity conditioning regimens. Incidence of acute GVHD was 16% after myeloablative conditioning regimens and 19% after reduced intensity conditioning regimens. Incidence of 3-year chronic GVHD was 30% after myeloablative conditioning regimens and 34% after reduced intensity conditioning regimens. Three-year relapse incidence was 44% after myeloablative conditioning regimens and 58% after reduced intensity conditioning regimens. Three-year overall survival was ∼45% after both myeloablative and reduced intensity conditioning regimens. Three-year chronic GVHD-free/leukemia-free survival was 24% after myeloablative conditioning regimens and 18% after reduced intensity conditioning regimens.66 In another study, 5-year overall survival was 43% in AML and 32% in ALL patients.67 Therefore, the main advantages of haploidentical unmanipulated hematopoietic grafts with posttransplant high-dose cyclophosphamide appear to be the relatively low incidences of NRM and severe GVHD.
In vivo T-cell depletion of G-CSF–mobilized peripheral blood hematopoietic grafts
In vivo T-cell depletion of G-CSF–mobilized peripheral blood hematopoietic grafts with anti–T-cell antibodies is an alternative strategy to prevent GVHD.68,69 One study used fludarabine, cyclophosmamide, and the humanized anti-CD52 monoclonal antibody alemtuzumab as a preparative regimen.68 Alemtuzumab also exerted in vivo T-cell depletion of the graft (additional GVHD prophylaxis consisted of mycophenolate mofetil with or without cyclosporine). In 49 patients with hematologic malignancies or marrow failure, the NRM rate was 31%, and rates of acute and chronic GVHD were 16% and 14%, respectively. One-year overall survival was 31%.68 Another study in 83 adult patients with acute leukemia or myelodysplastic syndrome (MDS) used busulfan, fludarabine, and antithymocyte globulin for in vivo T-cell depletion (and cyclosporine and methotrexate as additional GVHD prophylaxis).69 Incidence of NRM was 18%, and incidences of acute and chronic GVHD were 20% and 34%, respectively. Incidence of relapse was 27% in acute leukemia patients in CR1 at transplant, 32% in patients in CR ≥2 at transplant, 79% in patients transplanted with refractory acute leukemia, and 20% in patients with MDS. After a median follow-up of 2 years, disease-free survival was 60% in acute leukemia patients in CR1 at transplant, 53% in patients in CR ≥2 at transplant, 9% in patients transplanted with refractory acute leukemia, and 53% in patients with MDS.69
G-CSF–primed hematopoietic grafts combined with in vivo T-cell depletion
Several studies showed that G-CSF attenuated the risk of GVHD because it decreased production of inflammatory cytokines, increased production of interleukin-10, and induced T-helper-2 and regulatory T-cell immune deviation.9,78 The Beijing group designed a transplant protocol consisting of G-CSF administration (“priming”) before marrow and blood harvesting. GVHD prophylaxis consisted of in vivo T-cell depletion of the graft with antithymocyte globulin and posttransplant cyclosporine, mycophenolate mofetil, and methotrexate.70-72 In a study of 1210 patients with hematologic malignancies, 3-year incidence of NRM was 17%, and incidences of acute and chronic GVHD were 40% and 50%, respectively. Three-year relapse incidence was 17%, and progression-free survival was 67%.70 In a cohort of pediatric and adult patients with acute leukemia, NRM was ∼25%, and incidences of acute GVHD and 3-year chronic GVHD were 45.8% and 53.9%, respectively.71 The incidence of relapse was 11.9% in standard-risk AML and 24.3% in standard-risk ALL (standard risk denotes patients in CR1 or CR2 at transplant, without Philadelphia chromosome),71 and 20.2% in high-risk AML and 48.5% in high-risk ALL (high risk denotes patients in CR >2 or nonremission at transplant, or acute leukemia with Philadelphia chromosome in any disease status at transplant).71 Three-year leukemia-free survival was 70.7% in standard-risk AML, 59.7% in standard-risk ALL, 55.9% in high-risk AML, and 24.8% in high-risk ALL.71 In another series of 620 acute leukemia patients, the authors found 3-year leukemia-free survival was 64% in AML and 57% in ALL.72 Such results appear to be better than those of other studies. Younger age and fewer high-risk leukemia patients might have played a role.71,72,79 The Rome Transplant Network used bone marrow alone and intensified GVHD prophylaxis with the addition of anti-CD25 antibody administration.73,74 In a cohort of patients with a variety of hematologic malignancies, 5-year NRM incidence was 34%, acute GVHD incidence was 31%, and 2-year chronic GVHD incidence was 25%.74 Five-year relapse incidence was 26% in patients in CR1 or CR2 at transplant, and 36% in patients in CR >2 or with active disease at transplant. Five-year disease-free survival was 48% in patients in CR1 or CR2 at transplant and 22% in patients in CR >2 or with active disease at transplant.74
Haploidentical vs matched hematopoietic transplantation
Several retrospective studies compared haploidentical transplantation with posttransplant high-dose cyclophosphamide with matched sibling and unrelated donor transplantation.66,80-84 In 475 patients with a variety of hematologic malignancies, Bashey et al showed matched sibling, optimally (10 of 10 HLA allele) matched unrelated donor, and haploidentical transplants all had similar 2-year disease-free survival (ie, 56% vs 50% vs 54%).80 In 459 patients, Raiola et al showed 4-year disease-free survival was 43% after haploidentical transplantation, 32% after matched sibling transplantation, 36% after matched unrelated donor transplantation, 34% after mismatched unrelated donor transplantation, and 33% after unrelated cord blood transplantation.81 In 227 AML/MDS patients, Di Stasi et al showed that 3-year progression free-survival was 36% after matched sibling, 27% after matched unrelated donor, and 30% after haploidentical transplantation.82 In AML patients, Ciurea et al reported 3-year overall survival was similar (∼45%) in 1982 matched unrelated donor transplants and 192 haploidentical transplants.66 Two studies in lymphoma patients showed similar outcomes after haploidentical transplants with high-dose cyclophosphamide and matched sibling or unrelated donor transplants.83,84 In a nonrandomized, prospective, multicenter trial in 450 adult AML patients in CR1 at transplant, the Beijing group found 3-year disease-free survival was 74% after haploidentical transplantation and 78% after matched sibling transplantation.79 A retrospective pair-matched study in adult AML patients in CR1 showed similar results after haploidentical transplants performed in China and matched unrelated donor transplants performed in Europe.85 A randomized, multicenter study in 210 patients with Philadelphia-negative ALL in CR1 showed 3-year disease-survival was ∼65% after matched sibling and haploidentical transplantation.86 Albeit nonrandomized, these studies suggest equivalence of haploidentical transplantation with matched sibling and unrelated donor transplantation.
Enhancing the potency of T cell–depleted haploidentical transplants
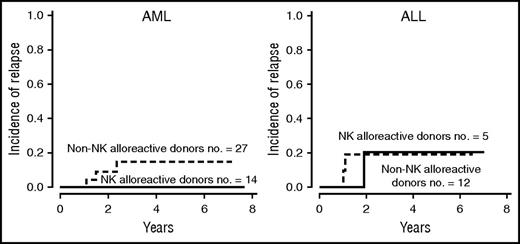
Recently, further advances were made to enhance the effectiveness of T cell–depleted haploidentical transplants by transferring donor T-cell immunity safely. An example is the multicenter randomized phase 3 clinical trial evaluating the efficacy of posttransplant infusion of donor T cells transduced with the herpes simplex virus–derived thymidine kinase suicide gene.8,87 TCR-α/β+ T-cell depletion of the hematopoietic graft is another option. TCR-α/β+ T cells are depleted as they contain alloreactive T cells and may therefore cause GVHD. TCR-γ/δ+ T and NK cells are retained in the graft because they do not cause GVHD and may instead exert beneficial effects.88,89 TCR-α/β+ T cell–depleted grafts were used in children with acute leukemia. Three-year relapse incidence was 21.9% and probability of leukemia-free survival was 73.1%.90 Another promising strategy is adoptive immunotherapy with thymic-derived/natural CD4+CD25+FoxP3+ regulatory T cells (Tregs)91,92 in combination with conventional T cells (Tcons).3,6,7,93-95 In major histocompatibility complex mismatched bone marrow transplantation murine models, coinfusion of Tregs and Tcons prevented lethal GVHD without impairing Tcon alloreactivity and leukemia killing.94-97 The mechanisms whereby Tregs protect from GVHD while preserving the Tcon-mediated GVL effect are suggested by experiments in immunodeficient NOD-scidIL2rgtm mice infused with human leukemia and HLA-mismatched Tregs/Tcons.95 Human peripheral blood Tregs preferentially migrated to GVHD target organs (ie, liver and gut) where they prevented GVHD. As they did not to migrate to the bone marrow, coinfused human Tcons were free to exert unopposed alloantigen recognition and leukemia killing. In fact, the infused human peripheral blood Tregs displayed a memory CD45RO+ phenotype and expressed low levels of the bone marrow homing receptor CXCR4.95,98 In a recent clinical trial, 60 adult patients with high-risk AML and ALL in any CR at transplant received Treg/Tcon-based haploidentical transplantation (Alessandra Carotti, Franca Falzetti, L.R., and A.V., unpublished data, June 30, 2016).93-95 The Treg infusion was the only form of GVHD prophylaxis. Fifty-eight patients were engrafted. The infusion of 2 × 106/kg freshly isolated CD4+CD25+FOXP3+ Tregs controlled the alloreactivity mediated by the subsequent infusion of up to 1 × 106/kg Tcons (which is about 2 log more than the threshold dose for GVHD).93,94 Eighty-three percent of patients were protected from acute and chronic GVHD. Five-year NRM incidence was 35%. The first 23 patients were conditioned with TBI, thiotepa, fludarabine, and cyclophosphamide (35 mg/kg per day for 2 days). This protocol was associated with extra-hematologic toxicity and a 52% NRM. To reduce NRM, the subsequent 35 patients received a lower dose of cyclophosphamide (15 mg/kg per day for 2 days) or anti-T cell antibodies (in the place of cyclophosphamide). NRM fell from 52% to 24%.93,94 In the entire series of 58 patients, the 5-year relapse incidence was 12%. None of the 14 AML patients transplanted from an NK alloreactive donor relapsed (Figure 1). Consequently, in the entire series, 5-year probability of chronic GVHD-free/leukemia-free survival was 50% (Alessandra Carotti, Franca Falzetti, L.R., and A.V., unpublished data, 30 June 2016).93-95 Table 1 summarizes results of haploidentical transplantation for acute leukemia performed under different protocols.
Cumulative incidence of leukemia relapse after T cell–depleted haploidentical transplantation with regulatory and conventional T-cell immunotherapy in the presence or absence of donor-versus-recipient NK-cell alloreactivity. Transplantation from NK alloreactive donors reduced AML relapse but not ALL relapse (as previously demonstrated in the standard T cell–depleted haploidentical protocol).18-22,93,94
Cumulative incidence of leukemia relapse after T cell–depleted haploidentical transplantation with regulatory and conventional T-cell immunotherapy in the presence or absence of donor-versus-recipient NK-cell alloreactivity. Transplantation from NK alloreactive donors reduced AML relapse but not ALL relapse (as previously demonstrated in the standard T cell–depleted haploidentical protocol).18-22,93,94
Recent results of different haploidentical hematopoietic transplant protocols in adult acute leukemia patients
| Transplant protocol . | Patient age, median (range) . | Sample size and disease, no. (%) . | Disease status at transplant, no. (%) . | Incidence of GVHD, % . | Incidence of NRM, % . | Incidence of relapse, % . | Probability of survival, % . |
|---|---|---|---|---|---|---|---|
| Unmanipulated BM or G-CSF–mobilized PB graft with high-dose posttransplant cyclophosphamide*66 | NR (21-70) | 192 AML | CR1: 91 (47) | Acute grade II-IV GVHD: | 14-9† (at 3 years) | 44-58† (at 3 years) | Overall survival: 45-46† |
| CR2: 52 (27) | 16-19† | ||||||
| Relapse: 49 (26) | Chronic GVHD: 30-34† | Chronic GVHD-free/ | |||||
| (at 3 years) | leukemia-free survival: | ||||||
| 24-18† (at 3 years) | |||||||
| Unmanipulated G-CSF–mobilized PB graft with in vivo T-cell depletion69 | 40 (16-70) | 83 | CR1: 15 (18) | Acute grade II-IV GVHD: 20 | 18 | CR1: 27 | Leukemia-free survival: |
| AML: 52 (63) | CR ≥2: 19 (23) | Chronic GVHD: 34 | CR≥2: 32 | CR1: 60 | |||
| ALL: 16 (19) | Refractory disease: 34 (41) | Refractory disease: 79 | CR ≥2: 53 | ||||
| MDS: 15 (18) | Low-risk MDS: 6 (7) | MDS: 20 | Refractory disease: 9 | ||||
| High-risk MDS: 9 (11) | MDS: 53 | ||||||
| Unmanipulated G-CSF–primed BM and PB graft with in vivo T-cell depletion72 | 25 (3-57) | 756 | Standard risk‡: 484 (64) | Acute grade II-IV GVHD: 43 | 18% (at 3 years) | AML: 22 | Leukemia-free survival: |
| AML: 321 (42) | High risk§: 136 (18) | Chronic GVHD: 53 | ALL: 20 | AML: 64 | |||
| ALL: 299 (40) | First chronic phase CML: 77 (10) | (at 2 years) | CML: 9 (at 3 years) | ALL: 57 | |||
| CML: 136 (18) | Beyond first chronic phase CML: 59 (8) | CML: 73 (at 3 years) | |||||
| Unmanipulated BM and/or PB graft with in vivo T-cell depletion or high-dose posttransplant cyclophosphamide||99 | 42 (18-66) | 229 | CR1: 77 (34) | Acute grade II-IV GVHD: 32 | 28 (at 3 years) | CR1: 32 | Leukemia-free survival: |
| AML: 156 (68) | CR2: 56 (24) | Chronic GVHD: 34 | CR2: 24 | CR1: 44 | |||
| ALL: 73 (32) | Advanced disease (CR3/active disease): | (at 3 years) | Advanced disease: 61 | CR2: 42 | |||
| 96 (42) | (at 3 years) | Advanced disease: 12 | |||||
| (at 3 years) | |||||||
| Unmanipulated BM or PB graft with in vivo T-cell depletion or high-dose posttransplant cyclophosphamide¶100 | 32 (18-76) | 208 ALL | CR1: 90 (44) | Acute grade II-IV GVHD: 31 | CR1: 26 | CR1: 26 | Leukemia-free survival: |
| CR ≥2: 59 (28) | Chronic GVHD: 27 | CR ≥2: 36 | CR ≥2: 32 | CR1: 45 | |||
| Active disease: 59 (28) | Active disease: 35 | Active disease: 60 | CR≥2: 32 | ||||
| (at 2 years) | (at 2 years) | Active disease: 5 | |||||
| (at 2 years) | |||||||
| Ex vivo T cell–depleted G-CSF–mobilized PB graft with Treg/Tcon immunotherapy93-95 (Alessandra Carotti, Franca Falzetti, L.R., and A.V., unpublished data, June 30, 2016) | 38.5 (20-59) | 60 | CR1: 38 (63) | Acute grade II-IV GVHD: 15 | 35 (at 5 years) | 12 (at 5 years) | Leukemia-free survival: 53 |
| AML: 42 (70) | CR ≥2: 22 (37) | Chronic GVHD: 3 | Chronic GVHD-free/ | ||||
| ALL: 18 (30) | (at 5 years) | leukemia-free survival: 50 (at 5 years) | |||||
| Transplant protocol . | Patient age, median (range) . | Sample size and disease, no. (%) . | Disease status at transplant, no. (%) . | Incidence of GVHD, % . | Incidence of NRM, % . | Incidence of relapse, % . | Probability of survival, % . |
|---|---|---|---|---|---|---|---|
| Unmanipulated BM or G-CSF–mobilized PB graft with high-dose posttransplant cyclophosphamide*66 | NR (21-70) | 192 AML | CR1: 91 (47) | Acute grade II-IV GVHD: | 14-9† (at 3 years) | 44-58† (at 3 years) | Overall survival: 45-46† |
| CR2: 52 (27) | 16-19† | ||||||
| Relapse: 49 (26) | Chronic GVHD: 30-34† | Chronic GVHD-free/ | |||||
| (at 3 years) | leukemia-free survival: | ||||||
| 24-18† (at 3 years) | |||||||
| Unmanipulated G-CSF–mobilized PB graft with in vivo T-cell depletion69 | 40 (16-70) | 83 | CR1: 15 (18) | Acute grade II-IV GVHD: 20 | 18 | CR1: 27 | Leukemia-free survival: |
| AML: 52 (63) | CR ≥2: 19 (23) | Chronic GVHD: 34 | CR≥2: 32 | CR1: 60 | |||
| ALL: 16 (19) | Refractory disease: 34 (41) | Refractory disease: 79 | CR ≥2: 53 | ||||
| MDS: 15 (18) | Low-risk MDS: 6 (7) | MDS: 20 | Refractory disease: 9 | ||||
| High-risk MDS: 9 (11) | MDS: 53 | ||||||
| Unmanipulated G-CSF–primed BM and PB graft with in vivo T-cell depletion72 | 25 (3-57) | 756 | Standard risk‡: 484 (64) | Acute grade II-IV GVHD: 43 | 18% (at 3 years) | AML: 22 | Leukemia-free survival: |
| AML: 321 (42) | High risk§: 136 (18) | Chronic GVHD: 53 | ALL: 20 | AML: 64 | |||
| ALL: 299 (40) | First chronic phase CML: 77 (10) | (at 2 years) | CML: 9 (at 3 years) | ALL: 57 | |||
| CML: 136 (18) | Beyond first chronic phase CML: 59 (8) | CML: 73 (at 3 years) | |||||
| Unmanipulated BM and/or PB graft with in vivo T-cell depletion or high-dose posttransplant cyclophosphamide||99 | 42 (18-66) | 229 | CR1: 77 (34) | Acute grade II-IV GVHD: 32 | 28 (at 3 years) | CR1: 32 | Leukemia-free survival: |
| AML: 156 (68) | CR2: 56 (24) | Chronic GVHD: 34 | CR2: 24 | CR1: 44 | |||
| ALL: 73 (32) | Advanced disease (CR3/active disease): | (at 3 years) | Advanced disease: 61 | CR2: 42 | |||
| 96 (42) | (at 3 years) | Advanced disease: 12 | |||||
| (at 3 years) | |||||||
| Unmanipulated BM or PB graft with in vivo T-cell depletion or high-dose posttransplant cyclophosphamide¶100 | 32 (18-76) | 208 ALL | CR1: 90 (44) | Acute grade II-IV GVHD: 31 | CR1: 26 | CR1: 26 | Leukemia-free survival: |
| CR ≥2: 59 (28) | Chronic GVHD: 27 | CR ≥2: 36 | CR ≥2: 32 | CR1: 45 | |||
| Active disease: 59 (28) | Active disease: 35 | Active disease: 60 | CR≥2: 32 | ||||
| (at 2 years) | (at 2 years) | Active disease: 5 | |||||
| (at 2 years) | |||||||
| Ex vivo T cell–depleted G-CSF–mobilized PB graft with Treg/Tcon immunotherapy93-95 (Alessandra Carotti, Franca Falzetti, L.R., and A.V., unpublished data, June 30, 2016) | 38.5 (20-59) | 60 | CR1: 38 (63) | Acute grade II-IV GVHD: 15 | 35 (at 5 years) | 12 (at 5 years) | Leukemia-free survival: 53 |
| AML: 42 (70) | CR ≥2: 22 (37) | Chronic GVHD: 3 | Chronic GVHD-free/ | ||||
| ALL: 18 (30) | (at 5 years) | leukemia-free survival: 50 (at 5 years) | |||||
BM, bone marrow; CML, chronic myeloid leukemia; NR, not reported; PB, peripheral blood.
Graft source was bone marrow in 84% of patients and G-CSF–mobilized peripheral blood hematopoietic cells in 16% of patients.
Outcomes after myeloablative or reduced intensity conditioning regimens, respectively.
Standard risk: CR1 or CR2 at transplant, without Philadelphia chromosome.
High risk: CR ≥3 or nonremission at transplant, or acute leukemia with Philadelphia chromosome in any disease status at transplant.
Graft source was bone marrow in 44% of patients, G-CSF–mobilized peripheral blood hematopoietic cells in 53% of patients, and a combination of the two in 3% of patients. GVHD prophylaxis was based on in vivo T-cell depletion with anti–T-cell antibodies (75%) or posttransplant high-dose cyclophosphamide (13%) or other (12%).
Graft source was bone marrow in 44% of patients and G-CSF–mobilized peripheral blood hematopoietic cells in 56% of patients. GVHD prophylaxis was based on posttransplant high-dose cyclophosphamide (52%) or in vivo T-cell depletion with antithymocyte globulin (48%).
Conclusions
The 1990s saw what had been major drawbacks of haploidentical transplantation, ie, very strong host-versus-graft and graft-versus-host alloresponses, which led, respectively, to rejection and GVHD, being overcome through transplantation of a mega-dose of T cell–depleted peripheral blood hematopoietic progenitor cells.3-7,11-13 Immune recovery in the absence of any posttransplant immunosuppression created an opportunity for discovering innovative forms of immunotherapy.18-22,87-90 It favored NK-cell development and revealed donor-versus-recipient NK-cell alloreactions that eradicated AML and improved survival.18-22 More recent transplantation protocols use unmanipulated haploidentical grafts combined with enhanced posttransplant immunosuppression to help prevent GVHD.9,10,56-74 Because of their results and because they do not need dedicated laboratory facilities and personnel, they substantially extended the use of haploidentical transplantation. A more recent, promising strategy is the infusion of donor Tregs to protect against otherwise lethal doses of Tcons add-backs.3-7,93-95 The Tcon infusion reduced leukemia relapse rate and infections, leading to a chronic GVHD-free/leukemia-free survival that compares favorably with results of unmanipulated haploidentical transplantation.
Acknowledgments
The authors’ data discussed in this review were obtained with the support of grants by the Italian Ministry of Health (European Research Area Net Translational Cancer Research), the Italian Association for Cancer Research, the Italian Ministry of Further Education, and the Leukemia and Lymphoma Society.
Authorship
Contribution: A.M. and A.V. wrote the manuscript; and L.R. reviewed and edited the manuscript.
Conflict-of-interest disclosure: The authors declare no competing financial interests.
Correspondence: Antonella Mancusi, Division of Hematology and Clinical Immunology, Department of Medicine, University of Perugia, Piazza L. Severi 1/8, 06132 Perugia, Italy; e-mail: antonella.mancusi@unipg.it.