Older unrelated donor age and PBM mismatching confer similarly adverse effects on OS and the impacts are additive.
Younger donors appear to negate the detrimental effect of PBM mismatching in the unrelated-donor HCT setting.
Visual Abstract
An HLA-mismatched unrelated donor who is class I peptide-binding motif (PBM)-matched is preferred over a PBM-mismatched donor. We hypothesized that using a younger donor (aged ≤35 years vs >35 years) could compensate for the inferior overall survival (OS) associated with PBM mismatches. We compared 6 groups: HLA-matched/younger donor (n = 10 531), HLA-matched/older donor (n = 3572), PBM-matched/younger donor (n = 357), PBM-matched/older donor (n = 257), PBM-mismatched/younger donor (n = 616), and PBM-mismatched/older donor (n = 339) in patients undergoing transplantation with conventional graft-versus-host disease prophylaxis. In multivariate analysis, HLA-matched/younger donors were associated with superior OS relative to any other group. Pairwise comparisons showed that donor age significantly impacted OS in both HLA-matched and HLA-mismatched groups. Moreover, younger donors appeared to negate the detrimental effect of PBM mismatching: the PBM-matched/younger donor group had similar OS as the HLA-matched/older donor group and the PBM-mismatched/younger donor group had similar OS as the PBM-matched/older donor group. Our study suggests that older unrelated donor age and PBM mismatching confer similarly adverse effects on OS and the impacts are additive, a finding which may widen the “acceptable” donor pool. The best OS is observed with HLA-matched/younger donors and the worst with PBM-mismatched/older donors. These findings should be validated with other data sets and with posttransplantation cyclophosphamide-based prophylaxis.
Introduction
A novel donor selection algorithm based on HLA class I peptide-binding motif (PBM) matching was recently proposed for the mismatched unrelated donor (MMUD) selection for hematopoietic cell transplantation (HCT).1 A study using the Center for International Blood and Marrow Transplant Research data found that as compared with the 10/10 HLA matched unrelated donor group, there was no statistically significant difference in overall survival (OS) of patients that were either PBM matched with their donors or had unidirectional PBM mismatches in the host-versus-graft direction, whereas PBM mismatches in the graft-versus-host (GVH) direction appeared to be associated with significantly worse OS.1
Although these observations suggest that PBM-matches or the host-versus-graft PBM mismatches are better tolerated than GVH PBM mismatches, consideration for donor age is also clinically relevant2-7, and frequently the choice of donors is not optimal for both HLA and donor age concurrently. To expand on this, we analyzed the impact of donor age on outcomes in both HLA matched and mismatched groups Specifically, we tested 2 hypotheses: (a) OS with younger donors would be superior to that with older donors in HLA-matched, HLA-mismatched/PBM-matched, and HLA-mismatched/PBM-mismatched groups; and (b) using a younger donor could compensate for the inferior OS associated with HLA/PBM mismatches; that is, OS with PBM-matched/younger donors would be similar to that with HLA-matched/older donors, and OS with PBM-mismatched/younger donors would be similar to PBM-matched/older donors.
Methods
We performed a secondary analysis of the data previously published by Crivello et al1 using the publicly available Center for International Blood and Marrow Transplant Research data set.8 Our study population includes patients aged ≥18 years with acute myeloid leukemia, acute lymphoblastic leukemia, or myelodysplastic neoplasm who underwent a 10/10 HLA–matched unrelated donor or 9/10 HLA–MMUD HCT (mismatched at HLA-A, -B, or -C) between 2008 and 2018. All patients received calcineurin inhibitor–based prophylaxis. Posttransplant cyclophosphamide–based prophylaxis was excluded.
The assignment of the PBM groups and HLA-DPB1 matching was defined as previously published.1 The local Institutional Review Board (FHIRB0020181) granted the study permission, which was conducted in accordance with the Declaration of Helsinki. We defined donors aged >35 years as “older” and those aged ≤35 years as “younger” based on summary statistics of the receiver operating characteristic curve, which revealed a cutoff of 35.5 years as a predictor of mortality. This is also consistent with several previous publications that showed a survival advantage with donors aged <30 to 36 years vs older donors.2-7 The statistical methods are elaborated in the supplemental Material.
First, we assessed the independent effects of HLA match and donor age on OS. In multivariate analysis, older donors (hazard ratio [HR], 1.21; 95% confidence interval [CI], 1.15-1.27; P < .001 vs younger donors) and the HLA match (PBM matched: HR, 1.21; 95% CI, 1.09-1.35; P = .001; and PBM mismatched: HR, 1.49; 95% CI, 1.37-1.62; P < .001 vs HLA matched) were both noted to be significant predictors of OS. No interaction was found between the HLA match and donor age. Similarly, older donors (HR, 1.27; 95% CI, 1.17-1.38; P < .001 vs younger donors) and the HLA match (PBM matched: HR, 1.31; 95% CI, 1.09-1.57; P = .003; and PBM mismatched: HR, 1.63; 95% CI, 1.41-1.88; P < .001 vs HLA matched) were both noted to be significant independent predictors of nonrelapse mortality (NRM).
Next, using a combination of HLA/PBM matching and donor age, 6 groups were created: HLA-matched/younger donor (n = 10 531), HLA-matched/older donor (n = 3572), PBM-matched/younger donor (n = 357), PBM-matched/older donor (n = 257), PBM-mismatched/younger donor (n = 616), and PBM-mismatched/older donor (n = 339). All statistical analyses were performed using STATA/MP 18 (College Station, TX; StataCorp LLC).
Results and discussion
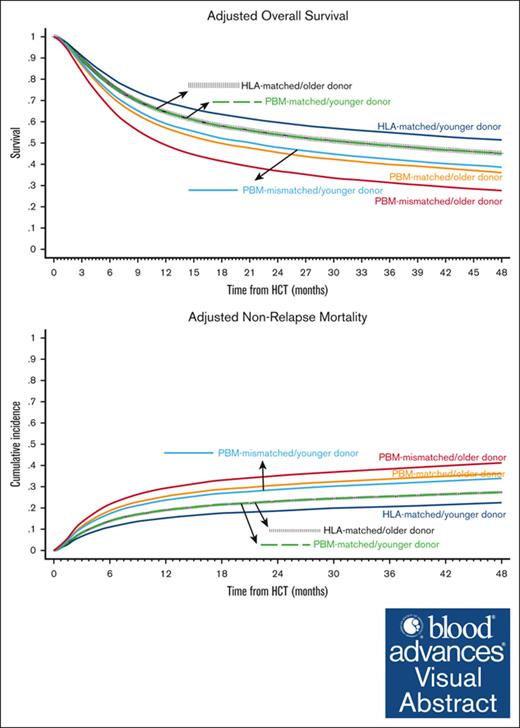
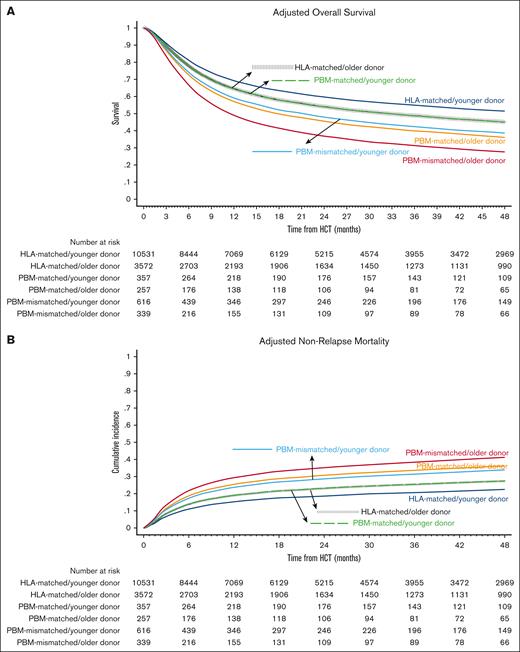
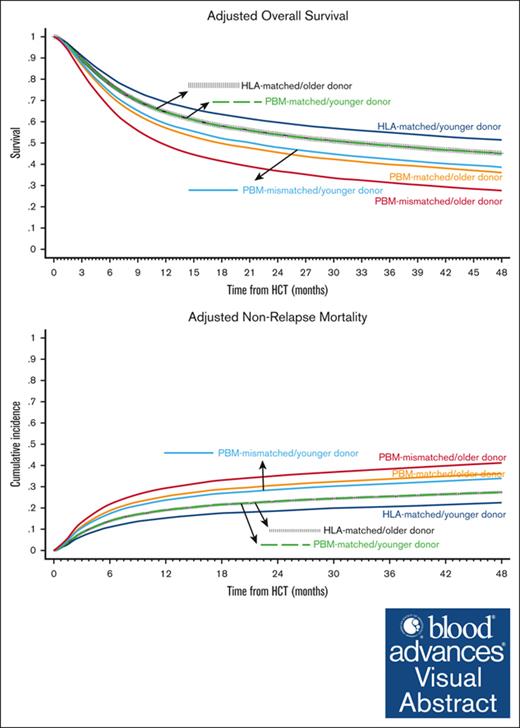
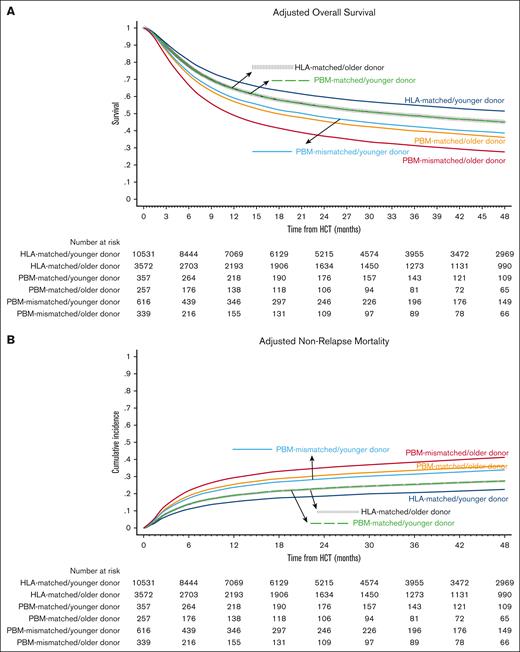
Baseline characteristics of the comparison groups are shown in Table 1 and univariate analyses are shown in supplemental Table 1. The best-estimated 4-year OS was noted in the HLA-matched/younger donor group (49.9%; 95% CI, 48.8-50.9) and the worst in the PBM-mismatched/older donor group (29.0%; 95% CI, 24.1-34.1). In multivariate analysis, the hazards of overall mortality increased in a stepwise fashion from the HLA-matched/younger donor (reference group) to HLA-matched/older donor and the PBM-matched/younger donor (HR, 1.18 in both groups), to PBM-matched/older donor and the PBM-mismatched/younger donor (HR, 1.48 and 1.42, respectively), and finally to the PBM-mismatched/older donor group (HR, 1.91; Figure 1A; Table 2). The notable findings of the pairwise comparisons were as follows: first, donor age significantly affected OS in both HLA-matched and HLA-mismatched groups, and the negative impact of older donors relative to younger donors appeared to be the highest in the worst HLA category (PBM-mismatched: HR, 1.35) and the lowest in the best HLA category (HLA-matched: HR, 1.18; supplemental Table 2; pairwise comparison group 1). Secondly, younger donors appeared to negate the detrimental effect of HLA/PBM mismatching. Specifically, the PBM-matched/younger donor group had a similar OS (HR, 1.00) as that of the HLA-matched/older donor group, and the PBM-mismatched/younger donor group had a similar OS (HR, 0.96) as that of the PBM-matched/older donor group (pairwise comparison group 2).
Overall Survival and Nonrelapse Mortality. Outcomes of HLA-matched/younger donor (navy blue), HLA-matched/older donor (vertical dashed black); PBM-matched/younger donor (dashed green); PBM-matched/older donor (dark orange); PBM-mismatched/younger donor (light blue), and PBM-mismatched/older donor (maroon). (A) Adjusted OS, and (B) adjusted NRM.
Overall Survival and Nonrelapse Mortality. Outcomes of HLA-matched/younger donor (navy blue), HLA-matched/older donor (vertical dashed black); PBM-matched/younger donor (dashed green); PBM-matched/older donor (dark orange); PBM-mismatched/younger donor (light blue), and PBM-mismatched/older donor (maroon). (A) Adjusted OS, and (B) adjusted NRM.
The differences in OS were not explained by relapse, which did not differ significantly between the groups, but by NRM. We observed similar patterns for NRM as for OS, that is, the lowest 4-year NRM was noted in the HLA-matched/younger donor group (21.6%; 95% CI, 20.8-22.4) and the highest in the PBM-mismatched/older donor group (41.3%; 95% CI, 36-46.5). In multivariate analysis, the hazards of NRM increased in an almost stepwise fashion from the HLA-matched/younger donor (reference group) to the HLA-matched/older donor and PBM-matched/younger donor (HR, 1.18 in both groups), to the PBM-matched/older donor and PBM-mismatched/younger donor (HR, 1.48 and 1.42, respectively), and the PBM-mismatched/older donor group (HR, 1.91; Figure 1B; Table 2). In the direct pairwise comparisons, the risk of NRM was numerically higher with older donors than with younger donors in all HLA/PBM groups, although it did not reach statistical significance in the HLA-mismatched groups. Younger donors appeared to negate the higher risk of NRM with HLA/PBM mismatching. Specifically, the PBM-matched/younger donor group had a similar risk of NRM (HR, 1.00) as that of the HLA-matched/older donor group; and the PBM-mismatched/younger donor had a similar risk of NRM (HR, 0.92) as that of the PBM-matched/older donor group (supplemental Table 2).
The reason for differences in the NRM between the groups could not be determined because of the lack of data on cause of death but it may be related to infections or other causes and less likely because of GVH disease (GVHD). Among bone marrow graft recipients, older donors were associated with a significantly higher risk of chronic GVHD in the HLA-matched (HR, 1.23) and the PBM-matched (HR, 1.83) groups as than in the HLA-matched/younger donor group, and a numerically higher risk but without statistical significance in the PBM-mismatched group (HR, 1.44). The risk of chronic GVHD did not differ significantly between the groups among peripheral blood graft recipients. The reason for this unexpected finding could not be explained; however, it may suggest that the unfavorable impact of peripheral blood grafts on chronic GVHD is greater than the potential protective effects of donor age. The risk of grade 3/4 acute GVHD did not differ significantly by donor age in the HLA-matched group with either graft source, whereas both PBM-mismatched groups had a significantly higher risk irrespective of donor age than the HLA-matched donor groups. The impact of our main variable (donor age/HLA matching) on all outcomes was independent of other predictors (supplemental Table 3). Although in vivo T-cell depletion was associated with a significantly lower risk of GVHD and NRM, it was also associated with a significantly higher risk of relapse, which translated to no significant impact on OS.
Because NRM was the primary driver for worse outcomes, we performed exploratory analyses to study the relative impact of older vs younger donors based on patient characteristics that were significant predictors of NRM in the multivariate analysis. For these analyses, we dichotomized patient age as ≤40 vs >40 years because the risk of NRM increased in patients aged >40 years in the multivariate analysis. The goal of these analyses was to assess the effect of donor age in patients with the lowest risk features (eg, age of <40 years, HCT-specific comorbidity index score of 0-1, and Karnofsky performance status of ≥90) and in those with the highest risk features (eg, age of ≥40 years, HCT-specific comorbidity index score of >2, and Karnofsky performance scale score of <90), after fitting the multivariate model adjusted for covariates. We found that the older donor group had an increased hazard of NRM as compared with the younger donor group in both the patients at lowest risk (HR, 1.28; 95% CI, 1.15-1.42) and those at highest risk (HR, 1.94; 95% CI, 1.46-2.56), but the effect appeared to be more pronounced in patients at highest risk. These findings suggest that although selecting a younger donor, in general, is important whenever possible, the need becomes even more critical in patients with baseline increased risk of NRM because of older patient age, high comorbidity index, and low performance status. The risk of NRM was higher in the older donor group than the younger donor group irrespective of whether in vivo T-cell depletion was used (HR, 1.57; 95% CI, 1.28-1.92; P < .001) or not (HR, 1.68; 95% CI, 1.34-2.09; P < .001).
Our study findings apply to patients undergoing HCT who received calcineurin inhibitor–based prophylaxis. Whether similar findings would be seen with posttransplant cyclophosphamide–based or other novel GVHD prophylaxis regimens remains unknown. In addition to the shortcomings mentioned above and biases inherent to retrospective analysis, other limitations of our study are the lack of data on the molecular classification of disease and female-donor parity. Also, because of a lack of data on systemic therapy–requiring chronic GVHD, we were unable to compute GVHD-free relapse-free survival. Lastly, our study population predominantly comprised HLA-matched donor groups, whereas the number of patients in the HLA-MMUD groups was relatively small. The rationale for adverse outcomes with older donors may be because of several factors. These include changes in the DNA methylation patterns leading to an alteration of immune function9 or epigenetic aging,10,11 which is correlated with a higher risk of GVHD, infection-related deaths,10,12 and inferior survival.12 Other reasons could be age-related thymic involution13 or clonal hematopoiesis, which is associated with dysregulated cytokine signaling causing an inflammatory milieu in HCT recipients.14
Donor age, categorized by decades, was also shown to be an independent predictor of OS in the original analysis.1 It also showed no statistically significant differences in OS between the HLA-matched and the PBM-matched groups. Our study adds further granularity by showing that the HLA-matched and the PBM-matched groups may or may not differ in OS depending on the donor age and that younger donors can overcome the adverse effects of HLA/PBM mismatching. These findings should be validated with other independent data sets.
To summarize, older donor age and HLA/PBM mismatching result in similar independent adverse effects on OS after HCT, and the impacts are additive. Unrelated donor preference related to donor age and PBM matching should be HLA matched, followed by HLA mismatched/PBM matched, and finally HLA mismatched/PBM mismatched. Our data show that younger donors with inferior matching led to comparable survival as older donors with better matching, a finding that may widen the “acceptable” donor pool. The best OS is noted with HLA-matched/younger donors and the worst with HLA mismatched/PBM-mismatched/older donors. Further studies are needed to decipher the causes of worse outcomes with older donors and whether the use of novel GVHD prophylaxis regimens can alter these conclusions.
Acknowledgments
The authors thank the Center for International Blood and Marrow Transplant Research (CIBMTR) staff for providing this data set.
This work was supported by grants AI069197, CA218285, CA100019, and CA231838 from the National Institutes of Health (E.W.P). Also, this data set was collected by CIBMTR, which is supported primarily by the US Public Health Service U24CA076518 from the National Cancer Institute, the National Heart, Lung, and Blood Institute, and the National Institute of Allergy and Infectious Diseases; grant number 75R60222C00011 from the Health Resources and Services Administration; and grant numbers N00014-21-1-2954 and N00014-23-1-2057 from the Office of Naval Research; the National Marrow Donor Program/Be The Match; and the Medical College of Wisconsin.
Authorship
Contribution: R.S.M. conceptualized the study, performed the statistical analysis, interpreted data, and wrote the manuscript; E.W.P., S.R.S., and S.J.L. reviewed and interpreted the data, reviewed the manuscript, and provided critical feedback; R.S.M. had full access to the raw data, which are publicly available; all authors approved the manuscript; and the corresponding author had the final responsibility to submit for publication.
Conflict-of-interest disclosure: The authors declare no competing financial interests.
Correspondence: Rohtesh S. Mehta, Clinical Research Division, Fred Hutchinson Cancer Center, University of Washington School of Medicine, 1100 Fairview Ave N, D5-280, Seattle, WA 98109; email: rmehta@fredhutch.org.
References
Author notes
The data are publicly available and accessible at https://cibmtr.org/CIBMTR/Resources/Publicly-Available-Datasets.
The full-text version of this article contains a data supplement.