Key Points
MM cell lines and primary MM cells can be engrafted and grown in vivo in Casper zebrafish larvae.
The zebrafish MM in vivo xenograft model can be used as a pretreatment drug-sensitivity prediction platform for MM patients.
Abstract
Patient-derived multiple myeloma (MM) cells are difficult to establish in culture or propagate in vivo in murine model. Here, we describe a zebrafish xenograft model that permits rapid, reliable growth of human MM cells injected into the perivitelline space of albino zebrafish (Casper) embryos 48 hours postfertilization. MM1S and MM1R MM cell lines and primary CD138+ MM cells were stained with CM-Dil red fluorescent dye and suspended in Matrigel prior to their injection. The cells grew at the site of injection and disseminated throughout the developing embryos and larvae. Tumor size was quantified by fluorescent microscopy, and cell fate was followed for 4 days. All of the cell line xenografts showed responses similar to those previously observed with in vitro assays. CD138+ plasma cell xenografts derived from MM patients also grew and were inhibited by the same drugs patients had responded to clinically. Using this technique, we can assess drug sensitivity or resistance with a small number of MM cells in a short period. This raises the possibility that one might be able to assess drug sensitivity in real time with readily obtainable clinical samples.
Introduction
The gene expression profile and other genomic data have so far failed to predict individual responses to therapy.1,2 Newer murine multiple myeloma (MM) models that recapitulate human disease have been developed to some extent, but their ability to predict clinical responses remains limited.2-5 The limitations of the murine tumor xenografts model include the need for human fetal bone fragments to support tumor growth, low efficiency resulting in the generation of a limited number of tumor-bearing mice, and a 2-month lag period for tumor growth. These limitations highlight the need for a new model that allows for the efficient and rapid growth of human MM cells in an animal model applicable for pretreatment drug-response prediction and screening. Here, we describe a novel zebrafish model that supports the growth of both MM cell lines and primary MM cells and could provide an efficient, rapid, and inexpensive platform for drug screening and studying the biology of MM.
Methods
Animal care and handling
The Casper strain of zebrafish (Danio rerio) were raised at 28.5°C in E3 buffer with cycles of 14/10 hours of light/darkness and fed in accordance with guidelines established by the Committee on the Ethics of Animal Experiments of Children’s Hospital Boston (No. 09-09-1540R).
MM cell lines and patient cell microinjection
All MM cell lines used and their culture conditions have been previously described.6 Primary plasma cells from MM patients were obtained after informed consent and isolated as described previously.7 The CM-Dil (ThermoFisher Scientific)–labeled MM cell lines and primary CD138+ MM cells were washed in phosphate-buffered saline and resuspended in Matrigel at 10 000 cells/µL. Casper embryos were dechorionated for 5 to 10 minutes with 1 mg/mL pronase 36 hours postfertilization (hpf). Embryos were anesthetized by placement in 0.04 mg/mL ethyl 3-aminobenzoate methanesulfonate tricaine 48 to 96 hpf. MM cells (100 cells in 10 nL, 5- to 20-nL injection volume per embryo) were injected into the perivitelline space with a pneumatic Pico-pump injector using glass microinjection needles.
Drug treatment and efficiency evaluation
We evaluated tumor xenografts by fluorescence microscopy 24 hours postinjection (hpi). Embryos with red fluorescence at the injection site were moved to 24-well uncoated plates (1 embryo per well), and freshly prepared E3 medium containing drugs or solvent was added to each well according to the experimental group. Stock solutions of the drugs to be tested were prepared as previously described7-10 and then diluted at least at 1:1000 with E3 medium before adding to embryos in the 24-well plates. Fresh drug containing E3 medium was replaced daily. We evaluated tumor growth and fish survival at 24, 48, and 72 hours posttreatment (hpt) by fluorescence microscopy. We measured the area and density of fluorescence on photomicrographs with ImageJ software and estimated tumor xenografts volume in control and drug-treated animals.
Results and discussion
Casper zebrafish in vivo MM xenograft model
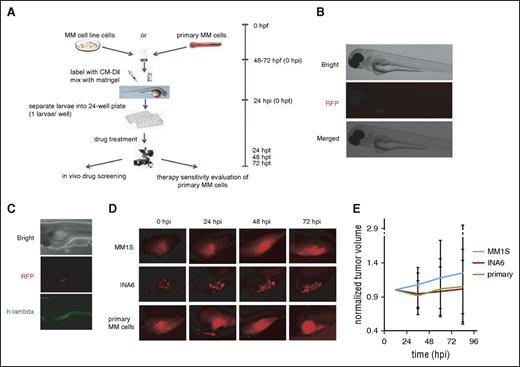
We used transparent Casper zebrafish early-stage embryos (48 hpf) because they allow in vivo observation of growth changes in fluorescent-labeled myeloma cell xenografts (Figure 1A)11 and are immune deficient,12,13 allowing the growth of human cells. We first used MM1S and MM1R MM cell lines to demonstrate MM cell growth in zebrafish embryos and determine whether the addition of anti-MM drugs to the embryo water would inhibit cell growth. As seen in Figure 1B, CM-Dil–labeled MM cells injected in the perivitelline area of Casper zebrafish could be observed by fluorescence microscopy 24 hpi and >80% of larvae retained injected MM cells for 9 days, providing an adequate window for drug screening. These xenografts were confirmed by fluorescence microscopy (Figure 1C).
Casper zebrafish support MM cell growth. (A) Schematic chart depicting the procedures used to establish the zebrafish MM xenograft model (hpf, hours postfertilization; hpi, hours postinjection; hpt, hours posttreatment). (B) Fluorescence microscopy images of the CM-Dil–stained MM cells injected into the perivitelline area of Casper zebrafish larvae. Bright field of whole Casper zebrafish 24 hours after injection with human MM cells (top). Red fluorescence protein (RFP) channel of whole Casper zebrafish; the red fluorescence color under the microscope shows CM-Dil–stained MM cells (middle). Merged image from bright field and RFP field (bottom). All cells are located in the perivitelline area after mixing with Matrigel. (C) Whole-mount immunohistochemistry staining. Bright field of whole zebrafish 24 hpi with human multiple MM cells (top). RFP field shows red fluorescence from CM-Dil–stained MM cells (middle). GFP field showing the EGFP-labeled anti–human λ light-chain antibody colocalizing with CM-Dil–labeled MM1S cells (bottom). (D) Typical fluorescence microscopy images of the CM-Dil–stained MM cells xenograft growth of MM1S, INA-6, and primary patient MM cells without drug treatment in the perivitelline area of Casper zebrafish larvae at 24, 48, and 72 hpi. (E) Tumor growth curves of xenografts with these 3 cell types in Casper zebrafish larvae (n = 30 for each group).
Casper zebrafish support MM cell growth. (A) Schematic chart depicting the procedures used to establish the zebrafish MM xenograft model (hpf, hours postfertilization; hpi, hours postinjection; hpt, hours posttreatment). (B) Fluorescence microscopy images of the CM-Dil–stained MM cells injected into the perivitelline area of Casper zebrafish larvae. Bright field of whole Casper zebrafish 24 hours after injection with human MM cells (top). Red fluorescence protein (RFP) channel of whole Casper zebrafish; the red fluorescence color under the microscope shows CM-Dil–stained MM cells (middle). Merged image from bright field and RFP field (bottom). All cells are located in the perivitelline area after mixing with Matrigel. (C) Whole-mount immunohistochemistry staining. Bright field of whole zebrafish 24 hpi with human multiple MM cells (top). RFP field shows red fluorescence from CM-Dil–stained MM cells (middle). GFP field showing the EGFP-labeled anti–human λ light-chain antibody colocalizing with CM-Dil–labeled MM1S cells (bottom). (D) Typical fluorescence microscopy images of the CM-Dil–stained MM cells xenograft growth of MM1S, INA-6, and primary patient MM cells without drug treatment in the perivitelline area of Casper zebrafish larvae at 24, 48, and 72 hpi. (E) Tumor growth curves of xenografts with these 3 cell types in Casper zebrafish larvae (n = 30 for each group).
We next examined the growth of the MM1s cell line, a huIL-6–dependent INA-6 MM cell line, and purified CD138+ primary human MM cells in Casper zebrafish and observed their growth up to 96 hpi without the addition of huIL-6 or bone marrow stromal cells (Figure 1D-E). Interestingly, the growth of MM cell lines and primary MM cells suggests that the zebrafish embryo perivitelline space recapitulates the growth support provided by the human bone marrow microenvironment.
Preclinical drug-sensitivity studies in the zebrafish model
We next evaluated whether this model could be used to assess efficacy of a known anti-MM agent. We first evaluated the effect of dexamethasone on both dexamethasone-sensitive (MM1S) and dexamethasone-resistant (MM1R) MM cell lines. We established 100 nM as the lowest concentration that inhibited the growth of MM1S cells. Up to 5 µM dexamethasone did not affect the survival of Casper zebrafish larvae for up to 3 days. We treated 11 MM1S and 11 MM1R xenografts in Casper zebrafish embryos with 100 nM dexamethasone, and the change in xenograft volume was estimated every 24 hours (Figure 2A-B). A 50% decrease in gross tumor volume from the size just prior to drug addiction was used as a “response” and indicator of drug sensitivity. The data are then presented as number of fish responding out of the whole group at 24, 48, and 72 hpt. According to this method of analysis, the response of MM1S cells to dexamethasone was >85%, whereas the response of MM1R to dexamethasone was 15%. This limited response was similar to that observed in the control group and is consistent with previously published in vitro and in vivo studies. It took 4 days from tumor inoculation to obtain these data, which demonstrates the potential of this model to rapidly predict the sensitivity of MM cells to added drug.
Response to standard and novel drugs in the zebrafish model. (A) Fluorescent microscopy images of CM-Dil–stained MM1S and MM1R xenografts at 0, 24, 48, and 72 hpi. (B) Tumor growth curves of MM1S and MM1R cells xenografts indicating that MM1S xenografts are responsive to dexamethasone treatment whereas MM1R xenografts are resistant. (C) Percentage of MM1S xenografts in zebrafish showing no response after treatment with bortezomib (Bor; n = 22), lenalidomide (Len; n = 22), dexamethasone (Dex; n = 22), AZD6244 (n = 18), 17-AAG (n = 22), rapamycin (Rapa; n = 18), AS703026 (n = 18), and dimethyl sulfoxide (control; n = 20), respectively. Error bars indicate standard errors. (D) Summary of percentage of MM1S, MM1R, OPM1, and RPMI8226 larvae without response with various indicated novel agents. (E) Individual fluorescence microscopy images of CD138+ MM cells from newly diagnosed MM patient xenografts in zebrafish before and after treatment with bortezomib (n = 8), lenalidomide (n = 8), dexamethasone (n = 8), and dimethyl sulfoxide (n = 8), at 0, 24, 48, and 72 hpt, respectively. (F) Percentage of primary patient MM cell xenografts without response to lenalidomide, bortezomib, and dexamethasone. Ctrl, control.
Response to standard and novel drugs in the zebrafish model. (A) Fluorescent microscopy images of CM-Dil–stained MM1S and MM1R xenografts at 0, 24, 48, and 72 hpi. (B) Tumor growth curves of MM1S and MM1R cells xenografts indicating that MM1S xenografts are responsive to dexamethasone treatment whereas MM1R xenografts are resistant. (C) Percentage of MM1S xenografts in zebrafish showing no response after treatment with bortezomib (Bor; n = 22), lenalidomide (Len; n = 22), dexamethasone (Dex; n = 22), AZD6244 (n = 18), 17-AAG (n = 22), rapamycin (Rapa; n = 18), AS703026 (n = 18), and dimethyl sulfoxide (control; n = 20), respectively. Error bars indicate standard errors. (D) Summary of percentage of MM1S, MM1R, OPM1, and RPMI8226 larvae without response with various indicated novel agents. (E) Individual fluorescence microscopy images of CD138+ MM cells from newly diagnosed MM patient xenografts in zebrafish before and after treatment with bortezomib (n = 8), lenalidomide (n = 8), dexamethasone (n = 8), and dimethyl sulfoxide (n = 8), at 0, 24, 48, and 72 hpt, respectively. (F) Percentage of primary patient MM cell xenografts without response to lenalidomide, bortezomib, and dexamethasone. Ctrl, control.
To further evaluate the utility of the xenograft model for drug screening, we chose 2 first-line anti-MM drugs (10 nM bortezomib and 2 μM lenalidomide) and 4 novel agents (400 nM AZD6244, 800 nM 17-AAG, 800 nM AS703026, and 40 nM rapamycin), which have been tested in MM. We used 4 MM cell lines (MM1S, MM1R, OPM1, and RPMI8226) for this evaluation. MM1S was sensitive to bortezomib, lenalidomide, and dexamethasone (with >60% of larvae showing a response) and relatively sensitive to the other 4 agents (30% to 60% of tested fish had a response) (Figure 2C). Relative sensitivity and resistance of other cell lines to the standard agents as well as investigational agents are shown in Figure 2D.
Next, we assessed the effect of these novel agents on primary MM cells from 2 newly diagnosed and 4 relapsed/refractory MM patients. The efficiency of engraftment of primary cells is similar to that of cell lines, which is ∼80%. We treated embryos engrafted with patient cells with these agents and observed that both newly diagnosed patients responded to bortezomib and lenalidomide with a >50% reduction at 72 hpt (Figure 2E-F and data not shown). We observed that cells from those patients with resistance to bortezomib and/or lenalidomide were also resistant to the same agents in our zebrafish model, validating the reliability of our system and confirming that primary MM xenografts can be used to predict patient responses to chemotherapy in vivo.
We here describe a zebrafish embryo xenograft model that supports the growth of MM cell lines and primary patient cells, can be used to predict drug sensitivity, and can potentially be used for preclinical drug screening. The advantages of the system are the ability to use patient cells, the small number of MM cells required, the reduced variability between animals, the ability to perform medium-throughput in vivo drug screening using primary MM patient cells, and a short latency that may permit quick screening in real time. The limitations of this model include the smaller number of embryos that could be injected at single sitting, growth within a unique microenvironment, growth at a lower temperature required by fish, and undervaluation of the influence on drug resistance induced by the bone marrow microenvironment. Variability between different fish can be observed, but this can be overcome by injecting a large number of larvae. The ability of zebrafish to support MM cell growth may provide insight into the unique microenvironment in fish that supports MM cell growth, even though it may not have all the characteristics of the actual bone marrow microenvironment. This easy, quick, and economical model overcomes many of the limitations of murine xenograft models and should be further evaluated as a tool to screen and predict drug sensitivity in MM.
The publication costs of this article were defrayed in part by page charge payment. Therefore, and solely to indicate this fact, this article is hereby marked “advertisement” in accordance with 18 USC section 1734.
Acknowledgments
This work was supported in part by grants from the Veterans Administration (I01-BX001584), National Institutes of Health, National Cancer Institute (RO1-124929; P50-100007, PO1-78378, and PO1-155258 [N.C.M. and K.C.A.]; and RO1-50947 [K.C.A.]), and Leukemia Society of America (N.C.M.); and by a Dana-Farber/Harvard Cancer Center Specialized Programs of Research Excellence in Multiple Myeloma Career Development Award (J.L.). K.C.A. is an American Cancer Society Clinical Research Professor.
Authorship
Contribution: J.L. and N.C.M. designed experiments, analyzed data, and wrote the paper; J.L., W.Z., J.-J.Z., A.H.K., C.Y., D.M., and X.R. performed experiments and analyzed data; Y.-T.T., and K.C.A. provided clinical samples; and R.I.H. critically revised the paper.
Conflict-of-interest disclosure: The authors declare no competing financial interests.
Correspondence: Nikhil C. Munshi, Dana-Farber Cancer Institute, 450 Brookline Ave, Boston, MA 02215; e-mail: nikhil_munshi@dfci.harvard.edu; or Robert I. Handin, Hematology Division, Brigham and Women’s Hospital, Karp Family Research Building, Room 6210, One Blackfan Circle, Boston, MA 02115; e-mail: rhandin@partners.org.
References
Author notes
J.L. and W.Z. contributed equally to this study.


This feature is available to Subscribers Only
Sign In or Create an Account Close Modal