Key Points
SATB1 is specifically overexpressed in the CD30+ lymphoma cells in cutaneous CD30+ lymphoproliferative disease.
SATB1 promotes proliferation of CD30+ lymphoma cells by direct transcriptional repression of cell cycle inhibitor p21.
Abstract
Cutaneous CD30+ lymphoproliferative disease (CD30+LPD), characterized by the presence of CD30+ anaplastic large T cells, comprises the second most common group of cutaneous T-cell lymphoma (CTCL). However, little is known about the pathobiology of the CD30+ lymphoma cells, as well as the mechanisms of disease progression. Here we report that Special AT-rich region binding protein 1 (SATB1), a thymocyte specific chromatin organizer, is over-expressed in CD30+ lymphoma cells in most CD30+LPDs, and its expression is upregulated during disease progression. Our findings show that SATB1 silencing in CD30+LPD cells leads to G1 cell cycle arrest mediated by p21 activation. Using chromatin immunoprecipitation, luciferase assays, and mutational analysis, we demonstrate that SATB1 directly regulates the transcription of p21 in a p53-independent manner. Moreover, DNA demethylation on a specific CpG-rich region of the SATB1 promoter is associated with the upregulation of SATB1 during disease progression. These experiments define a novel SATB1-p21 pathway in malignant CD30+ T lymphocytes, which provides novel molecular insights into the pathogenesis of CD30+LPDs and possibly leads to new therapies.
Introduction
Primary cutaneous CD30+ lymphoproliferative diseases (CD30+LPDs) are the second most common form of cutaneous T-cell lymphomas (CTCLs) and represent a spectrum of disorders ranging from lymphomatoid papulosis (LyP) to primary cutaneous anaplastic large-cell lymphoma (PCALCL).1 They are characterized by the common phenotypic hallmark of CD30+ anaplastic T cells.2 Although LyP has a chronic course with recurrent self-healing papulonodular lesions, PCALCL manifests as rapidly growing and ulcerating skin tumors,2 and >20% of patients with multifocal skin involvement die within 5 years.3 Despite its usual indolent clinical behavior, LyP is associated with the development of second malignant lymphoma in nearly 20% of patients, including PCALCL and mycosis fungoides (MF).4 Although not considered to be a CD30+LPD, MF may develop CD30+ T cells, especially in the setting of large-cell transformation (MF-LCT), which manifests a more aggressive clinical course.1
The clonal relationships between LyP and PCALCL, as well as MF have been firmly established.5,6 However, the mechanisms of tumor progression of CD30+LPDs remain to be elucidated. The current hypothesis focuses on the growing resistance of CD30+ lymphoma cells to extrinsic inhibitory stimuli7 : transforming growth factor (TGF)-β receptor mutation was shown to contribute to unregulated cell growth via abrogation of TGF-β–mediated growth inhibition.8 Escape from CD30L/CD30 ligation–mediated growth regulation was associated with the progression to PCALCL.9 Meanwhile, the intrinsic oncogenic drivers in CD30+ lymphoma cells during disease progression are less known.
To address this critical gap, we focus on SATB1 (special AT-rich sequence–binding protein 1), an important thymocyte nuclear protein that plays a crucial role in T-cell development.10,11 Previously we reported that SATB1 deficiency was responsible for the pathogenesis of a leukemic form of CTCL-Sézary syndrome by inducing resistance to activation-induced cell death of Sézary cells.12 SATB1, as a chromatin organizer, binds to matrix attachment region of DNA and participates in the maintenance and compaction of chromatin.10,13 By dynamically altering the organization and epigenetic status of the chromatin, SATB1 functions as a highly pleiotropic regulator of gene expression.14 Ectopic SATB1 expression has been associated with varied malignancies14-17 ; however, its status in CTCLs other than Sézary syndrome is unknown.
Given the heterogeneity within CTCLs, we speculate that there may be different SATB1 expression patterns and functions among CTCL subtypes. Here we identified SATB1 overexpression in CD30+ lymphoma cells of CD30+LPDs and MF-LCTs with CD30+ cells. Moreover, increased SATB1 expression, associated with demethylation of the gene promoter, contributes to disease progression via transcriptional inhibition of cell-cycle inhibitor p21. Our findings demonstrate that epigenetic activation of SATB1 is a critical mechanism that promotes malignant progression in CD30+LPDs.
Methods
Patient recruitment and clinical features
Lesional skin biopsies were obtained from 50 patients with CD30+ LPDs (26 LyP, 18 PCALCL, and 6 MF-LCT) recruited from the Skin Lymphoma Clinic of Peking University First Hospital (N = 30), West China Hospital (N = 6), and Roger Williams Cancer Center (N = 14), with approval from the Clinical Ethics Board of each institution, in accordance with the Declaration of Helsinki principles. Patients were diagnosed based on previously described criteria2 (supplemental Table 1, available on the Blood Web site). Control skin biopsies were obtained from 10 patients with nontransformed MF (5 plaque stage, 5 tumor stage) and 22 patients with benign inflammatory dermatoses (5 lichen planus, 5 eczema, 3 psoriasis, 3 eosinophilic dermatoses, 3 atopic dermatitis, and 3 molluscum contagiosum).
Cell culture and treatment
PCALCL cell lines Mac-1, Mac-2A, and Mac-2B were cultured in RPMI-1640 medium with 10% fetal bovine serum, 100 U/mL penicillin, and 0.1 mg/mL streptomycin. 3 × 105/mL Mac-1 cells were cultured in the presence of 10 μM 5-Aza-2′-Deoxycytidine (DAC; Sigma-Aldrich) or 0.3 nM Trichostatin A (TSA; Sigma-Aldrich), using solvent DMSO as a control. Cells were harvested at 48 hours and subjected to DNA and RNA extraction.
In vitro T-cell activation
Peripheral blood mononuclear cells (PBMCs) were obtained from 5 healthy donors by Ficoll-Paque PLUS (GE Healthcare). Naïve T cells were purified from PBMCs using a naïve CD4+ T cell isolation kit (Miltenyi Biotec). Naïve T cells and PBMCs were washed and cultured at 1 × 106/mL in supplemented RPMI-1640 media with plate-bound anti-CD3 mAbs (30 ng/mL; BD Pharmingen) plus anti-CD28 mAbs (10 ng/mL; BD Pharmingen). Cells were harvested on day 6 for CD30 and SATB1 phenotype analysis by flow cytometry with respective antibodies (BD Pharmingen), as previously described.18
Lentivirus shRNA vector mediated gene knockdown
Four lentivirus shRNA vectors were constructed by ligating 4 independently designed oligonucleotides encoding shRNAs against SATB1 to mU6-MCS-Ubi-EGFP vector (GV118) between HpaI and XhoI sites. Lentivirus shRNA vector against CDKN1A were constructed with hU6-MCS-CMV-RFP vector (GV113) between AgeI and EcoRI sites. Oligonucleotides against SATB1 and CDKN1A are listed in supplemental Table 2. Constructs with oligonucleotides encoding scrambled shRNA were used as controls. All constructs were verified by DNA sequencing. Lentiviral production and transduction was performed as previously described.19
Cell viability, cell cycle, and apoptosis assay
Cell growth was measured with an MTS-based cell viability assay using the Cell Viability Colorimetric Assay Kit (Promega) as previously described.12 Cell-cycle composition was determined by PI-mediated flow cytometric analysis; cell apoptosis was analyzed by Annexin V–based apoptosis detection, following the manufacturer’s instructions (BD Pharmingen). Flowcytometry data were analyzed with CellQuest Pro and ModFit software (BD Bioscience).
Colony-forming cell assay
Colony-forming cell assays were performed in methylcellulose cultures (H4230; StemCell Technologies). Colony counts were performed, using standard scoring criteria, on the basis of the ability to produce colonies containing a minimum of 20 cells after 12 to 14 days.20
Gene expression profile analysis
Total RNA was extracted and reverse-transcribed into cDNA. Agilent Whole Human Genome Oligo Microarray (4 × 44 K) (Agilent Technologies) was used for transcriptome analysis as previously described.12 Processed signals were normalized and analyzed with the SBC Analysis System (Ebioservice) to identify differentially expressed genes (data accessible at the National Center for Biotechnology Information Gene Expression Omnibus database, #GSE50916). The Genomatix Pathway System (GePS, Genomatix software suite, v3.0) was adopted to evaluate the contribution of molecular pathways to the transcriptional differentiation of samples.21 All pathways were ranked by enrichment scores and Benjamini adjusted P value.
Chromatin immunoprecipitation
Immunoprecipitation was performed using the EZ-CHIP Kit (Millpore) with ChIP-grade antibodies against SATB1 (BD Pharmingen), RNA polymerase II, or IgG control, according to the manufacture’s protocol. DNA was eluted and purified, followed by polymerase chain reaction (PCR) amplification using primers specific for CDKN1A promoter. Primers used for PCR amplification are listed in supplemental Table 2.
Luciferase reporter assay
Wild-type CDKN1A promoter (from −698 to +236 relative to the transcription start site [TSS]) was amplified by PCR from the Mac-1 cell genome. The mutated CDKN1A promoter was generated by inducing point mutation to the key nucleotides in SATB1 binding motif using the Fast Mutagenesis System Kit (TansGen Biotech) according to the manufacturer’s protocol. Wild-type and the mutated promoter were cloned to pGL3-Basic luciferase vector (Promega) between MluIand BglII sites separately. Mac-1 cells were electroporated with dual-luciferase reporter assay system (Promega) following the manufacturer’s instruction. Relative luciferase activity was quantified with normalization to Renilla luciferase activity, which served as an internal control for transfection efficiency.
Bisulfite sequencing and mass spectrometry
Bisulfite sequencing was used to generate DNA-methylation maps with single-base resolution. The method is based on the selective deamination of cytosine to uracil (and subsequent conversion to thymine via PCR), whereas 5-methylcytosine residues remain unchanged.22 Genomic DNA from Mac-1 and Mac-2A cells were bisulfite-converted with the EpiTect Bisulfite Kit (QIAGEN) according to the manufacturer’s instruction, followed by PCR designed to amplify the SATB1 gene promoter (supplemental Table 2). For each cell, at least 3 PCR product clones were randomly selected for DNA sequencing and following methylation analysis by MethTools.22 For PCR amplicons from tissue samples that consist of mixed-cell types besides tumor cells, quantitative methylation analysis was performed by Sequenom’s MassARRAY platform. This system uses MALDI-TOF mass spectrometry (MS) in combination with RNA base–specific cleavage (MassCLEAVE).23,24 The methylation level on individual an CpG site refers to the percentage of methylated CpG dinucleotides in the settings of total DNA (1 refers to 100%).
Statistics
If not specified, all experimental data were analyzed and compared for statistically significant differences by nonparametric Mann-Whitney U tests. A P value < .05 was considered statistically significant.
Additional methods are listed in the supplemental Methods.
Results
SATB1 is overexpressed in cutaneous CD30+LPDs and upregulated during disease progression
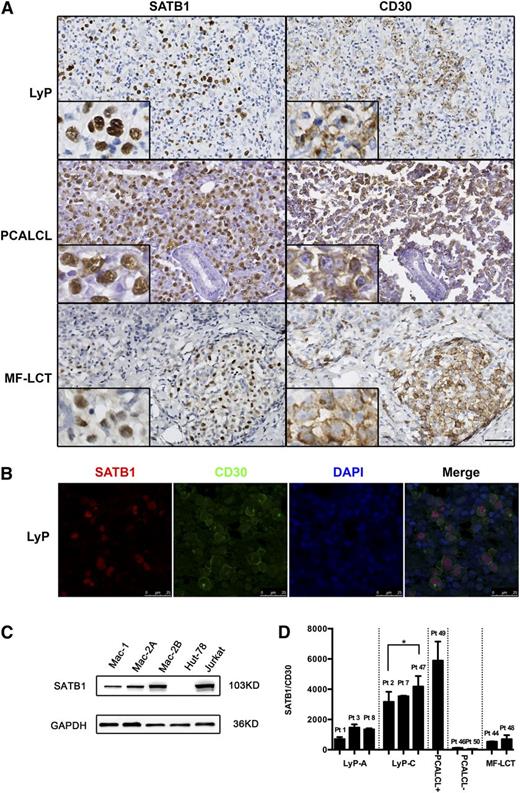
To investigate SATB1 expression in cutaneous CD30+ LPDs, 45 skin biopsy samples (including 25 LyP, 15 PCALCL, and 5 MF-LCT) were subjected to immunohistochemistry with SATB1 and CD30 antibodies, whereas biopsies from 10 nontransformed MF (5 plaque stage and 5 tumor stage) and 13 benign inflammatory dermatoses (5 lichen planus, 5 eczema, and 3 psoriasis) served as controls. As is shown in Figure 1A, bright diffuse nuclear SATB1 staining occurred in the CD30 large atypical lymphocytes in 21 of 25 LyP (84%), 11 of 15 PCALCL (73%), and 5 of 5 MF-LCT (100%) samples, whereas no or only dim focal staining was observed in control samples. Patient characteristics and the staining results are summarized in supplemental Table 1. Of note, the distribution of SATB1+ cells coincided with CD30 staining in all samples: The 4 LyP type B samples, characterized by absence of CD30+ T cells, were negative for SATB1 staining. We further confirmed co-expression of SATB1 and CD30 by double staining on LyP samples (Figure 1B).
SATB1 and CD30 expression in cutaneous CD30+LPDs. (A) Paraffin-embedded tissues from LyP, PCALCL, and MF-LCT patients were analyzed with immunohistochemistry. The large, atypical tumor cells showed bright diffuse nuclear staining of SATB1, and they coincided with CD30 staining (original magnification ×400, scale bar = 20 µm). (B) Immunofluoresence with double staining demonstrated specific SATB1 expression (red) in CD30+ cells (green) in LyP, whereas the surrounding reactive small T cells were negative for SATB1 or CD30. DAPI (blue) was used for visualize cell nuclei (scale bar = 25 µm). (C) SATB1 protein expression was detected in all 3 PCALCL cell lines with Western blot. Jurkat and Hut78 cell lines, derived from T lymphoblasts and Sézary cells, respectively, served as controls for high and low SATB1 expression in CD30+ T cells. (D) SATB1 expression levels relative to CD30 in CD30+LPD clinical samples. PCALCL+, PCALCLs positive for SATB1; PCALCL–, PCALCLs negative for SATB1; Pt, Patient no. *P < .05. Biological replicates for each cell were repeated 3 times.
SATB1 and CD30 expression in cutaneous CD30+LPDs. (A) Paraffin-embedded tissues from LyP, PCALCL, and MF-LCT patients were analyzed with immunohistochemistry. The large, atypical tumor cells showed bright diffuse nuclear staining of SATB1, and they coincided with CD30 staining (original magnification ×400, scale bar = 20 µm). (B) Immunofluoresence with double staining demonstrated specific SATB1 expression (red) in CD30+ cells (green) in LyP, whereas the surrounding reactive small T cells were negative for SATB1 or CD30. DAPI (blue) was used for visualize cell nuclei (scale bar = 25 µm). (C) SATB1 protein expression was detected in all 3 PCALCL cell lines with Western blot. Jurkat and Hut78 cell lines, derived from T lymphoblasts and Sézary cells, respectively, served as controls for high and low SATB1 expression in CD30+ T cells. (D) SATB1 expression levels relative to CD30 in CD30+LPD clinical samples. PCALCL+, PCALCLs positive for SATB1; PCALCL–, PCALCLs negative for SATB1; Pt, Patient no. *P < .05. Biological replicates for each cell were repeated 3 times.
Although the biological counterparts of the CD30+ lymphoma cells are largely unknown, CD30 can be a marker of activated T cells. To exclude the possibility that SATB1 expression in CD30+ lymphoma cells was merely the consequence of T-cell activation, we detected SATB1 expression in inflammatory dermatoses frequently harboring reactive CD30+ T cells (including atopic dermatitis, eosinophilic dermatoses, and molluscum contagiosum) and observed negative SATB1 expression in the majority of CD30+ cells (supplemental Figure 1). We further verified this finding using in vitro–generated CD30+ T cells from PBMCs and naïve CD4+ T cells isolated from healthy donors. Although SATB1 was slightly upregulated in activation-generated CD4+CD30+ T cells, expression levels were far lower than in PCALCL cell line Mac-1 (supplemental Figure 1), consistent with our observations of dim SATB1 expression of infiltrating T cells in CD30+LPD sections.
In addition, we observed increasing SATB1+ cells from LyP type A to LyP type C and PCALCL (Figure 1A), corresponding to the increase of tumor cells from scattered CD30+ anaplastic T cells in LyP type A, to sheets of CD30+ cells in type C, and to >75% CD30+ cells in PCALCL.2 To further quantify SATB1 expression during disease progression, we first studied cells established from a patient who progressed from LyP to PCALCL.5,25 In this case, the Mac-1 cell line was derived during an indolent course of disease, whereas the Mac-2A and Mac-2B cells were derived from separate rapidly growing skin tumors 3 years later in disease progression. These cells are representative of the primary tumors and retain the features of the original lymphoma.26 All 3 PCALCL cells highly expressed SATB1, and Mac-2A and -2B cells showed significantly higher expression (Figure 1C), whereas the other CD30+ T-cell lines, Jurkat and Hut78, derived from T lymphoblasts and Sézary cells, respectively, served as controls for high and low SATB1 expression in CD30+ T cells (supplemental Figure 2). Second, we quantified the mRNA expression of SATB1 in 11 CD30+LPD samples (including 3 LyP type A, 3 LyP type C, 1 SATB1+ PCALCL, 2 SATB1– PCALCL, and 2 MF-LCT samples). To exclude the possibility that SATB1 expression only reflected the CD30+ tumor cell burden, CD30 was adopted as the internal control for SATB1 mRNA levels. As expected, relative SATB1 expression increased with disease progression in SATB1+ samples (Figure 1D).
Therefore, our data showed that SATB1 is specifically overexpressed in CD30+ T cells in most cutaneous CD30+ LPDs, and its expression is further upregulated during disease progression.
SATB1 suppression confers a growth disadvantage to PCALCL cells by inducing G1 cell-cycle arrest
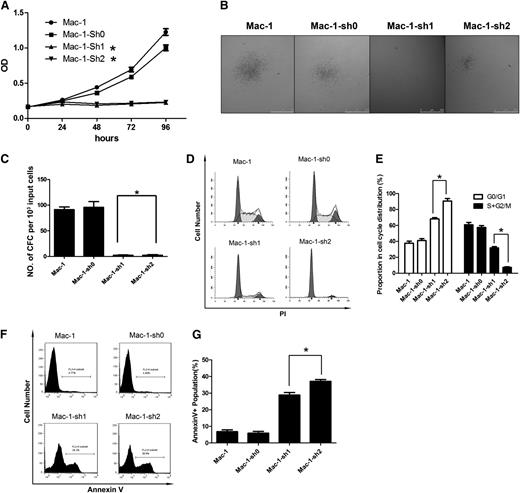
To define the pathological function of SATB1 in PCALCL, we suppressed SATB1 expression in Mac-1 cells via lentivirus-mediated RNAi (supplemental Figure 1) and observed marked inhibition of cell expansion, with flat and low growth curves indicating growth arrest of SATB1-suppressed cells (Figure 2A). Then we examined the ability of Mac-1 cells to form tumors in vitro with colony-forming cell (CFC) assay in semisolid cultures as described previously.20 Mac-1 with SATB1 suppression showed a more than 10-fold reduction in CFC output, in terms of the number and size of the colonies (Figure 2B-C). Next, we identified SATB1 suppression–induced G1 cell-cycle arrest by demonstrating a significantly increased G0/G1 phase and decreased S and G2/M phase in Mac-1 cells 3 days after the transduction (P < .05), whereas the sub-G1 phase representing apoptotic cells was not affected (Figure 2D-E). Arrest eventually resulted in cell apoptosis during longer-term culture as evidenced by Annexin V–based apoptosis assay (Figure 2F-G). These results suggested that inhibition of SATB1 can suppress tumor progression in PCALCL through cell-cycle interruption.
SATB1 suppression in PCALCL cells induced cell-cycle arrest at the G0/G1 stage. (A) MTS-based cell viability assay demonstrated a lack of increase in viable cells in 2 SATB1-silenced lines, whereas control Mac-1 cells demonstrated a normal growth curve. (B) The size of colonies formed in the CFC assay was decreased in SATB1-suppressed Mac-1 cells compared with control cells. (C) The number of colonies formed in semisolid culture (CFC output) decreased more than 10-fold upon SATB1 suppression. (D-E) Accumulation of the G0/G1 population and decrease of the S and G2/M populations were observed in Mac-1 cells with SATB1 silencing via PI cell-cycle analysis. (F-G) Long-term culture of the transduced cells revealed an increase in the Annexin V+ population in SATB1-silenced cells, which indicates increased cell apoptosis. Mac-1-sh1 to Mac-1-sh2, SATB1-silenced Mac-1 cell lines with 2 independent hairpins against SATB1. Mac-1 cells transduced with scrambled shRNA (Mac-1-sh0) and parental cells served as controls. *P < .05. Each experiment was repeated 2 times with 3 biological replicates.
SATB1 suppression in PCALCL cells induced cell-cycle arrest at the G0/G1 stage. (A) MTS-based cell viability assay demonstrated a lack of increase in viable cells in 2 SATB1-silenced lines, whereas control Mac-1 cells demonstrated a normal growth curve. (B) The size of colonies formed in the CFC assay was decreased in SATB1-suppressed Mac-1 cells compared with control cells. (C) The number of colonies formed in semisolid culture (CFC output) decreased more than 10-fold upon SATB1 suppression. (D-E) Accumulation of the G0/G1 population and decrease of the S and G2/M populations were observed in Mac-1 cells with SATB1 silencing via PI cell-cycle analysis. (F-G) Long-term culture of the transduced cells revealed an increase in the Annexin V+ population in SATB1-silenced cells, which indicates increased cell apoptosis. Mac-1-sh1 to Mac-1-sh2, SATB1-silenced Mac-1 cell lines with 2 independent hairpins against SATB1. Mac-1 cells transduced with scrambled shRNA (Mac-1-sh0) and parental cells served as controls. *P < .05. Each experiment was repeated 2 times with 3 biological replicates.
p21 mediates SATB1 suppression–induced cell cycle arrest
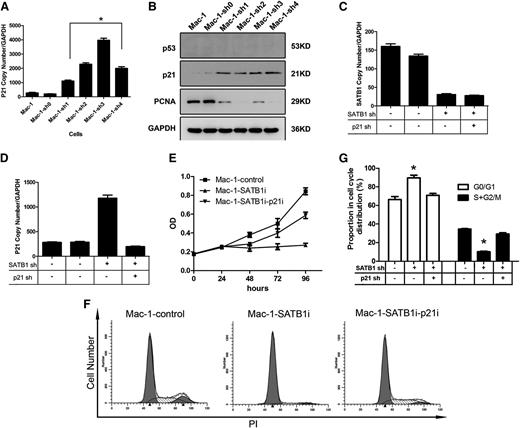
To elucidate molecular mechanisms underlying the growth arrest resulting from SATB1 knockdown, we subjected SATB1-suppressed Mac-1 cells and control cells to transcriptome analysis. As expected, SATB1 suppression led to marked gene expression alterations in Mac-1 cells. With the criteria of fold change >2, P < .05, 1100 transcripts were dysregulated, in which 562 transcripts were upregulated and 538 were downregulated in SATB1 suppressed Mac-1 cells. Pathway analysis of differentially expressed genes using the Genomatix Pathway System identified 13 significantly enriched molecular pathways (supplemental Table 3),21 and nine of them were related to cell-cycle regulation, including CDK regulation of DNA replication, p53 signaling pathway, and cell-cycle G1/S checkpoint, etc. Among those affected by SATB1, CDKN1A was one of the most remarkably upregulated genes (Figure 3A). Accordingly, p21 protein, a cyclin-dependent kinase (CDK) inhibitor encoded by CDKN1A, was also upregulated (Figure 3B). As a key regulator of G1/S transition, p21 mediates p53-dependent cell-cycle arrest and functions as a tumor suppressor when expressed in the cell nucleus.27 We then confirmed the nuclear localization of p21 in Mac-1 cells upon SATB1 suppression (supplemental Figure 2).
p21 activation in SATB1-suppressed PCALCL cells. (A) mRNA expressions of the CDKN1A gene were significantly upregulated in SATB1-suppressed Mac-1 cells. (B) Protein expressions of p21 in Mac-1 cells were markedly upregulated in SATB1-suppressed Mac-1 cells, whereas the expression of PCNA was inhibited in these cells. The p53 protein was absent in control Mac-1 cells and SATB1-suppressed Mac-1 cells. (C-D) Real-time reverse-transcriptase PCR demonstrated successful inhibition of p21 (C) expression in SATB1 (D)-suppressed Mac-1 cells with additional lentiviral vector harboring hairpin against p21. SATB1 sh, SATB1 knockdown; p21 sh, p21 knockdown. (E) A cell viability assay showed that blockage of p21 expression rescued cell proliferation, which was halted by SATB1 suppression. (F-G) Restored cell-cycle progression with decreased G0/G1 phase and increased S and G2/M phase was shown in cells with SATB1 and p21 double suppression. Mac-1-control, Mac-1 cells transduced with 2 lentivirus vectors (GV118 and GV113), both harboring scrambled shRNA; Mac-1-SATB1i, Mac-1 cells transduced with lentivector GV118 harboring shRNA against SATB1 and GV113 harboring scrambled shRNA; Mac-1-p21i, Mac-1 cells transduced with lentivector GV118 harboring scrambled shRNA and GV113 harboring shRNA against p21. *P < .05. Each experiment was repeated 2 times with 3 biological replicates.
p21 activation in SATB1-suppressed PCALCL cells. (A) mRNA expressions of the CDKN1A gene were significantly upregulated in SATB1-suppressed Mac-1 cells. (B) Protein expressions of p21 in Mac-1 cells were markedly upregulated in SATB1-suppressed Mac-1 cells, whereas the expression of PCNA was inhibited in these cells. The p53 protein was absent in control Mac-1 cells and SATB1-suppressed Mac-1 cells. (C-D) Real-time reverse-transcriptase PCR demonstrated successful inhibition of p21 (C) expression in SATB1 (D)-suppressed Mac-1 cells with additional lentiviral vector harboring hairpin against p21. SATB1 sh, SATB1 knockdown; p21 sh, p21 knockdown. (E) A cell viability assay showed that blockage of p21 expression rescued cell proliferation, which was halted by SATB1 suppression. (F-G) Restored cell-cycle progression with decreased G0/G1 phase and increased S and G2/M phase was shown in cells with SATB1 and p21 double suppression. Mac-1-control, Mac-1 cells transduced with 2 lentivirus vectors (GV118 and GV113), both harboring scrambled shRNA; Mac-1-SATB1i, Mac-1 cells transduced with lentivector GV118 harboring shRNA against SATB1 and GV113 harboring scrambled shRNA; Mac-1-p21i, Mac-1 cells transduced with lentivector GV118 harboring scrambled shRNA and GV113 harboring shRNA against p21. *P < .05. Each experiment was repeated 2 times with 3 biological replicates.
To examine whether p21 upregulation is responsible for the G1 arrest upon SATB1 suppression, we first demonstrated that cell proliferation marker PCNA, a downstream effector inhibited by p21, was downregulated in SATB1-suppressed cells (Figure 3B). Further, we blocked p21 upregulation in SATB1-suppressed Mac-1 cells via additional lentivirus vector–harboring shRNA against the CDKN1A gene (Figure 3C-D) and observed marked restoration of cell proliferation in SATB1-silenced Mac-1 cells (Figure 3E). Consistently, p21 blockade partially restored G1 to S progression in SATB1-suppressed cells (Figure 3F-G). These results indicate that p21 is at least partially responsible for G1 cell-cycle arrest induced by SATB1 suppression in PCALCL cells.
SATB1 regulates p21 expression by directly inhibiting CDKN1A transcription
The best known regulator of p21 is the tumor suppressor p53. However, previous studies and our own revealed an absence of p53 in resting Mac-1 cells,28 and it was not affected by SATB1 suppression (Figure 3B). Given the high potency of SATB1 as a transcription factor, we postulate that SATB1 may directly regulate CDKN1A transcription in PCALCL cells. In support of this hypothesis, chromatin immunoprecipitation (ChIP) analysis of Mac-1 cells revealed binding of SATB1 to a region of CDKN1A promoter (−520 to −368 upstream of TSS; Figure 4A). Transcription factor binding analysis using 2 databases (MatInspector29 and the JASPAR CORE database30 ) predicted that the only SATB1 binding site on CDKN1A promoter would be located within the aforementioned region (−434 to −421; Figure 4B). To determine the transcriptional outcome of the binding of SATB1 to CDKN1A promoter, we cloned the CDKN1A promoter from −698 to +236 and fused it to a luciferase reporter vector, as well as 2 promoter mutants with the key SATB1 binding motif TATT progressively mutated to CACT or CGCG (Figure 4B). Dual luciferase assay in Mac-1 cells revealed a dose-dependent increase of luciferase activity with mutated promoters, indicating an inhibitory effect of SATB1 binding on CDKN1A transcription (Figure 4B). These results show that SATB1 inhibits p21 expression by directly binding to the specific locus of CDKN1A promoter and negatively regulating its transcription, although other regulation mechanisms cannot be excluded.
SATB1 binds to CDKN1A promoter and regulates its transcription. (A) PCR products after Chromatin immunoprecipitation (ChIP) with anti-SATB1 antibody showed enrichment of SATB1 in a region within the CDKN1A promoter (–520 to −368 relative to TSS, 152 bp) in Mac-1 cells. M, DNA marker; input, cell lysate before immunoprecipitation; S, cell lysate immunoprecipitated with anti-SATB1 antibody; +, positive control cell lysate immunoprecipitated with anti-RNA polymerase II; –, negative control cell lysate immunoprecipitated with normal mouse IgG. (B) The CDKN1A promoter (WT) and its mutants with deficient SATB1 consensus binding site are shown schematically. Dual luciferase assay demonstrated that the luciferase (LUC) activity of the CDKN1A promoter mutants were significantly higher than that of wide-type in Mac-1 cells. *P < .05. Each experiment was repeated 2 times with 3 biological replicates.
SATB1 binds to CDKN1A promoter and regulates its transcription. (A) PCR products after Chromatin immunoprecipitation (ChIP) with anti-SATB1 antibody showed enrichment of SATB1 in a region within the CDKN1A promoter (–520 to −368 relative to TSS, 152 bp) in Mac-1 cells. M, DNA marker; input, cell lysate before immunoprecipitation; S, cell lysate immunoprecipitated with anti-SATB1 antibody; +, positive control cell lysate immunoprecipitated with anti-RNA polymerase II; –, negative control cell lysate immunoprecipitated with normal mouse IgG. (B) The CDKN1A promoter (WT) and its mutants with deficient SATB1 consensus binding site are shown schematically. Dual luciferase assay demonstrated that the luciferase (LUC) activity of the CDKN1A promoter mutants were significantly higher than that of wide-type in Mac-1 cells. *P < .05. Each experiment was repeated 2 times with 3 biological replicates.
p21 expression is inversely correlated with SATB1 expression in CD30+ lymphoma cells
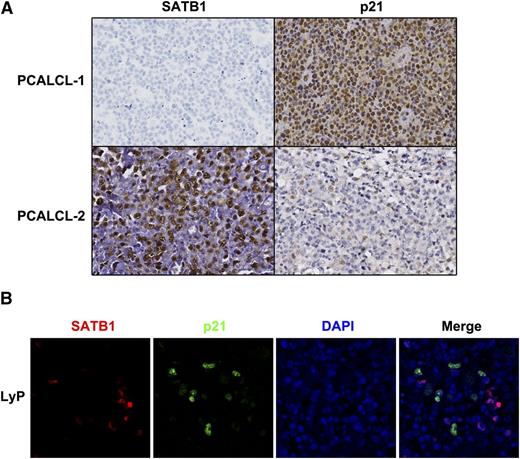
To validate the negative regulatory relationships between SATB1 and p21 in clinical samples, we performed p21 staining in the 4 SATB1– PCALCL samples, compared with 4 SATB1+ PCALCLs. Consistent with in vitro data, inverse correlation of SATB1 and p21 expression was observed in vivo. In PCALCLs negative for SATB1, strong p21 nuclear staining was detected in almost all atypical lymphocytes, whereas in PCALCLs expressing high levels of SATB1, <20% of atypical cells expressed p21 (Figure 5A). Double staining on LyP samples confirmed negative p21 expression in SATB1+ lymphoma cells (Figure 5B). Paradoxically, we only observed scattered p21 staining in SATB1– reactive lymphocytes (Figure 5B). Similar results were obtained in benign inflammatory dermatoses (data not shown), indicating distinct p21 regulatory machinery in normal lymphocytes, which awaits further exploration.
SATB1 and p21 expression in CD30+LPD clinical samples. (A) Immunohistochemistry shows diffuse p21 expression in SATB1– PCALCLs (PCALCL-1), whereas in SATB1+ PCALCLs (PCALCL-2), p21+ cells were <20%. (B) Double staining on LyP samples shows that, among large atypical lymphocytes, SATB1 (red) and p21 (green) detect distinct populations of atypical cells. Only a small portion of SATB1– reactive lymphocytes was positive for p21 staining. DAPI (blue) was used to visualize cell nuclei.
SATB1 and p21 expression in CD30+LPD clinical samples. (A) Immunohistochemistry shows diffuse p21 expression in SATB1– PCALCLs (PCALCL-1), whereas in SATB1+ PCALCLs (PCALCL-2), p21+ cells were <20%. (B) Double staining on LyP samples shows that, among large atypical lymphocytes, SATB1 (red) and p21 (green) detect distinct populations of atypical cells. Only a small portion of SATB1– reactive lymphocytes was positive for p21 staining. DAPI (blue) was used to visualize cell nuclei.
SATB1 upregulation during disease progression is associated with promoter DNA demethylation
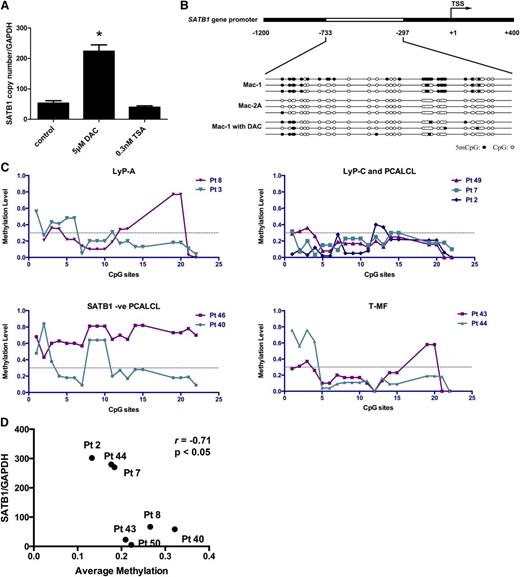
Increasing evidence suggests that SATB1 transcription can be regulated epigenetically.31,32 To test whether epigenetic mechanisms contribute to the SATB1 upregulation during disease progression, we treated Mac-1 cells with DNA methylation inhibitor DAC or histone deacetylase inhibitor TSA and found marked SATB1 upregulation upon DNA demethylation, rather than histone deacetylase inhibition (Figure 6A). There were several CpG dinucleotide-rich regions around and upstream of SATB1 TSS locus.32 Thus methylation analysis on the SATB1 promoter from −1200 to +400 relative to TSS was performed on Mac-1 and Mac-2A cells, which represented different disease stages and SATB1 expression levels. Bisulfite sequencing revealed specific demethylation at a CpG-rich region (−733 to −297) in Mac-2A cells compared with Mac-1 cells, whereas the methylation status of the remaining promoter was comparable between those 2 cells (Figure 6B). Corresponding to SATB1 upregulation, Mac-1 cells under DAC treatment showed demethylation in this region and demonstrated a comparable DNA methylation status to Mac-2A cells (Figure 6B), supporting the negative association of SATB1 expression and DNA methylation in this CpG-rich locus.
SATB1 expression levels in different disease stages of PCALCL are associated with the methylation status on the SATB1 promoter. (A) SATB1 mRNA expression in Mac-1 cells treated with a DNA demethylation reagent DAC was significantly upregulated, compared with cells treated with a histone deacetylase inhibitor TSA or the control DMSO solvent. *P < .05. Each experiment was repeated 2 times with 3 biological replicates.) (B) Schematic description of the SATB1 promoter and the sequencing results after bisulfite conversion. Mac-2A cells showed specific demethylation in the −733 to −297 region in the SATB1 promoter, compared with Mac-1 cells. DAC treatment in Mac-1 cells resulted in demethylation in this region, corresponding to upregulation of SATB1. Three independent PCR product clones were demonstrated for each cell. Solid dot, methylated CpG dinucleotide; circle, unmethylated CpG dinucleotide. (C) Quantitative methylation analysis on each CpG site in clinical samples with different stages of diseases. Methylation level 1 represents 100% methylated CpG dinucleotides on this site. Dotted line, 0.4. (D) Correlation between SATB1 mRNA expression and the average methylation level on all CpG sites. Pt, Patient no.
SATB1 expression levels in different disease stages of PCALCL are associated with the methylation status on the SATB1 promoter. (A) SATB1 mRNA expression in Mac-1 cells treated with a DNA demethylation reagent DAC was significantly upregulated, compared with cells treated with a histone deacetylase inhibitor TSA or the control DMSO solvent. *P < .05. Each experiment was repeated 2 times with 3 biological replicates.) (B) Schematic description of the SATB1 promoter and the sequencing results after bisulfite conversion. Mac-2A cells showed specific demethylation in the −733 to −297 region in the SATB1 promoter, compared with Mac-1 cells. DAC treatment in Mac-1 cells resulted in demethylation in this region, corresponding to upregulation of SATB1. Three independent PCR product clones were demonstrated for each cell. Solid dot, methylated CpG dinucleotide; circle, unmethylated CpG dinucleotide. (C) Quantitative methylation analysis on each CpG site in clinical samples with different stages of diseases. Methylation level 1 represents 100% methylated CpG dinucleotides on this site. Dotted line, 0.4. (D) Correlation between SATB1 mRNA expression and the average methylation level on all CpG sites. Pt, Patient no.
To validate this correlation in clinical settings, we used MS-based methylation quantification23,24 to determine the methylation status around this CpG-rich region on SATB1 promoter in 9 clinical samples. PCALCL and LyP-type C samples demonstrated marked demethylation on almost every individual CpG site within this region compared with LyP A samples (Figure 6C; corresponding promoter sequence and CpG sites in supplemental Table 4). Accordingly, PCALCL samples negative for SATB1 showed very high methylation levels, whereas MF-LCT samples, with variable SATB1 expression, showed variable methylation levels. With 7 samples available for both mRNA and bisulfited DNA, we demonstrated negative correlation between SATB1 expression and average methylation levels within this promoter region by Pearson correlation analysis (r = −0.71, P < .05; Figure 6D). Collectively, these results suggest a strong correlation of SATB1 promoter demethylation with its upregulation during disease progression in CD30+LPDs.
Discussion
Distinct from its nodal counterpart, the pathobiology of cutaneous CD30+LPD remains largely unknown. In this report we demonstrate aberrant SATB1 expression by CD30+ lymphomatous T cells in the majority of cutaneous CD30+LPDs. CD30+ lymphoma cells are highly proliferative cells expressing high levels of cell-proliferating marker PCNA and Ki-67.33,34 Although clonality has been detected in 22% to 50% of LyP, most if not all CD30+ cells in PCALCL are clonally related.35,36 Our findings of increasing SATB1+/CD30+ cells from LyP to PCALCL and upregulation of SATB1 in a series of CD30+LPD clinical samples strongly suggest a correlation between SATB1 aberrancy and clonal expansion of CD30+ lymphoma cells.
We further demonstrated marked cell-cycle arrest at the G0/G1 stage upon SATB1 suppression in PCALCL cells. The process of cell division is highly ordered and regulated. Cell cycle checkpoints, which can be activated as a result of DNA damage or exogenous stress signals are cell-intrinsic mechanisms that prevent cells from developing a malignant phenotype.37 Previous cytogenetic studies revealed an accumulation of chromosome aberrations during the progression from LyP to PCALCL and MF,25,38,39 indicating an impaired cell-cycle checkpoint during the malignant progression. Our study suggests that the cytogenetic abnormalities during disease progression may be attributed to SATB1 aberrancy–induced G1/S checkpoint disruption in CD30+ lymphoma cells.
The consequence of SATB1 overexpression seems to at least partially rely on its ability to modulate p21 expression. p21 belongs to the CIP and KIP family of CDK inhibitors.27 Initially identified as a mediator of p53-dependent G1 cell-cycle arrest,40 p21 has been shown to be a master effector of multiple tumor suppressor pathways by promoting antiproliferative activities independent of p53,41 and by abrogating S-phase progression via inhibition of PCNA.42 Our results confirmed the pivotal role of p21 in PCALCL progression and suggested the p53-independent regulation of p21 by SATB1 direct binding. SATB1 regulates gene transcription either by binding to the target gene locus or by targeting chromatin remodeling epigenetically, allowing it to regulate genes up to 10 kb away from its binding site.10,13 Our findings suggest a direct transcriptional repression on CDKN1A by SATB1, although indirect regulation mechanisms via chromatin remodeling or other transcription factors such as MYC and E2F1 cannot be excluded. The minimal p21 expression in inflammatory T cells, in agreement with a previous report,33 indicates the heterogeneity and complexity of p21 regulation in lymphocytes. Although our results suggest an apical role for p21 in SATB1-mediated tumor progression, most of the functional data were derived from the only available PCALCL cell model Mac cells, which were derived from a single patient; thus additional analyses are needed to comprehensively define the downstream effectors of SATB1 in CD30+LPDs.
SATB1 and p21 are nuclear proteins that are difficult to target. An alternative way to explore potential therapeutic targets is to elucidate the regulation mechanism of SATB1 itself during disease progression. IL-443,44 and/or polycomb group protein that inactivates acetylated histone H3K945 were previously defined as regulators of SATB1 expression. However, Mac-1 and Mac-2A cells lack IL-446,47 and did not respond to TSA treatment in terms of SATB1 expression. Instead, we found that demethylation of a CpG-rich region in the SATB1 promoter was associated with SATB1 upregulation during disease progression in cell lines and CD30+LPD clinical samples. This CpG-rich locus on the SATB1 promoter is an evolutionarily conservative region and harbors binding sites of numerous transcription factors (http://genome.ucsc.edu). Our data indicate that DNA methylation manipulation may help to control the progression of SATB1+ PCALCLs.
Together with the deficiency of SATB1 in Sézary cells observed by our group12 and others,48 SATB1 seems to have distinct expression patterns and functions in different subtypes of CTCLs, which is in agreement with the previous findings that Sézary syndrome and MF derive from distinct subsets of T cells.49 Although the bona fide biological counterpart of CD30+ lymphoma cells and its SATB1 expression level remain unknown, we postulate that rather than being an oncogene or a tumor suppressor, SATB1 plays a heterogeneous role in mature T cells, depending on the origin and context of cells.
It is noteworthy that 4 of 15 PCALCL samples in our study were negative for SATB1, which reveals intrinsic heterogeneity within PCALCL. We did not observe clinical or histological distinctions by SATB1 expression, which might be a result of the limit of the sample size and limited long-term follow-up. Of note, a previous study on systemic ALCL demonstrated SATB1 gene expression in ALK+ ALCLs and the absence of SATB1 in ALK− samples.50 However, LyP and PCALCL do not harbor the ALK fusion protein.1 This discrepancy strengthens the notion that systemic ALCL and PCALCL are distinct diseases. It is possible that PCALCLs without SATB1 expression may acquire features of Sézary cells, consistent with the plasticity of CD4+ T cells.51
In summary, this study highlights a novel SATB1-p21 axis that plays an important role in the disease progression of cutaneous CD30+LPDs, which provides novel molecular insights into this disease and possibly leads to new therapies in the future. Further investigations on this pathway and its significance in disease control are warranted.
The online version of this article contains a data supplement.
The publication costs of this article were defrayed in part by page charge payment. Therefore, and solely to indicate this fact, this article is hereby marked “advertisement” in accordance with 18 USC section 1734.
Acknowledgments
This study was supported by grants from National Nature Science Foundation of China (81072233 [P.T.] and 81201228 [Y.W.]), and by grant FRN#115138 from the Canadian Institute of Health Research (Y.Z.).
Authorship
Contribution: Y. Wang, X.G., and G.Z. performed research and analyzed data; L. Wang and T. Wang recruited eligible patients for this study and collected clinical information; Y.Z. and X.Z. performed promoter luciferase assay and critically revised the manuscript; Y.Z. codesigned the study and critically revised the manuscript; M.K. provided Mac cell lines, recruited eligible patients, coanalyzed the data, and critically revised the manuscript; and Y. Wang and P.T. designed the study, analyzed data, and wrote the paper.
Conflict-of-interest disclosure: The authors declare no competing financial interests.
Correspondence: Ping Tu, Department of Dermatology and Venerology, Peking University First Hospital, No. 8 Xishiku St, Beijing 100034, China; e-mail: tup0207@sina.com; or Yang Wang, Department of Dermatology and Venerology, Peking University First Hospital, No. 8 Xishiku St, Beijing 100034, China; e-mail: yangwang_dr@bjmu.edu.cn.
References
Author notes
Y.W., X.G., and G.Z. contributed equally to this study.


This feature is available to Subscribers Only
Sign In or Create an Account Close Modal