Abstract
Cryopreservation of hematopoietic stem cells (HSCs) and hematopoietic progenitor cells (HPCs) is crucial for cord blood (CB) banking and transplantation. We evaluated recovery of functional HPC cryopreserved as mononuclear or unseparated cells for up to 23.5 years compared with prefreeze values of the same CB units. Highly efficient recovery (80%-100%) was apparent for granulocyte-macrophage and multipotential hematopoietic progenitors, although some collections had reproducible low recovery. Proliferative potential, response to multiple cytokines, and replating of HPC colonies was extensive. CD34+ cells isolated from CB cryopreserved for up to 21 years had long-term (≥ 6 month) engrafting capability in primary and secondary immunodeficient mice reflecting recovery of long-term repopulating, self-renewing HSCs. We recovered functionally responsive CD4+ and CD8+ T lymphocytes, generated induced pluripotent stem (iPS) cells with differentiation representing all 3 germ cell lineages in vitro and in vivo, and detected high proliferative endothelial colony forming cells, results of relevance to CB biology and banking.
Introduction
The first cord blood (CB) transplantation saved the life of a young patient with Fanconi anemia using HLA-matched sibling CB cells,1 a procedure made possible by identification and cryopreservation of transplantable hematopoietic progenitor cells (HPCs) and hematopoietic stem cells (HSCs) in CB.2 More than 20 000 CB transplantations have treated the same malignant and nonmalignant disorders as bone marrow (BM).3-8 CB transplantation is possible because of CB banks, and how long CB can be stored in a cryopreserved state with efficient recovery of HSCs and HPCs is critical for CB banking. We reported highly efficient recovery of CB HPCs after 5,9 10,10 and 1511 years, and recovery of HSCs after 15 years.11 We now report efficient recovery of functional HPCs up to 21-23.5 years, with more in depth studies on CB HSC engraftment in immune deficient mice, recovery of responsive T cells, generation of induced pluripotent stem (iPS) cells,12-14 and detection of endothelial colony forming cells (ECFCs).15
Methods
CB cells were scheduled for discard.2 The study was approved by the Institutional Review Board of Indiana University (IU). Cryopreservation, thawing, and plating were as reported.2,9-11 CB was assessed within 36 hours of collection. Cells were either separated into a mononuclear (MNC) fraction (Ficoll-Hypaque; Pharmacia) and aliquoted into cryotubes (Nalge Nunc) or left unseparated and aliquoted into cryo-freezer bags,2,16,17 in 10% Dimethylsulfoxide and 10% autologous plasma for eventual analysis of HPC recovery. Percent recovery from MNC or unseparated cryopreserved cells was based on total prefreeze cells per volume of the exact same CB unit.2,9-11 After thaw of unseparated cells, CD34+ cells were magnetic-bead separated11 for HSC engraftment and iPS cell generation studies. CD4+ and CD8+ T lymphocytes were separated from the CD34+-depleted cells and stimulated on plates precoated with anti-CD3 (OKT3, 0.5 μg/mL) and anti-CD28 (clone CD28.2, 1 μg/mL) with 10% FBS, 50μM 2ME and 10ng/mL IL-15 as described.18 Immune-deficient mouse assay for human CB donor chimerism was as reported,11 except that recipients were NOD/SCID/IL-2Rgnull (NSG).19
iPS cell generation
At IU, CD34+ cells isolated from thawed, unseparated cells were grown with 10% FBS, 10 ng/mL human (h) SCF, 10 ng h Flt3-ligand, and 10 ng h Thrombopoietin/mL for 3 days. At day 4, cells were spin-infected (2200 rpm; 45 minutes) with concentrated lentiviral vectors Sox2-Oct4-EGFP and cMyc-Klaf4 (pc DNA-HIV-CS-CGW, provided by Dr P. Zoltick, Children's Hospital, Philadelphia; supplemental Figure 1, available on the Blood Web site; see the Supplemental Materials link at the top of the online article) in α-MEM medium with polybrene (Sigma-Aldrich). Medium was replaced at 6 days with the cytokines noted in this paragraph. At day 7, cells were transferred to mitotically inactivated murine embryonic fibroblasts (MEFs) and cultured as for human embryonic stem cells (hESCs).20 iPS cells were also generated at Johns Hopkins using retroviruses expressing Oct4, Sox2, Klf4, and c-Myc.12 ECFC assay was performed with MNCs isolated from thawed, unseparated CB.15
Results and discussion
HPC recovery
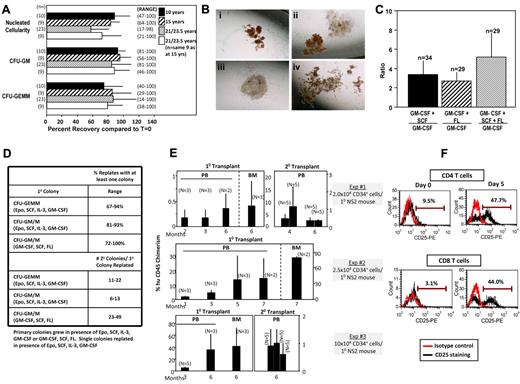
We show efficient recovery of HPCs from 23 different collections of MNCs thawed from vials after 21-23.5 years (Figure 1A) compared with the exact same unit's precryopreservation numbers, a recovery similar to 10- and 15-year thaws, that assessed the same plus additional CB units. A range of recoveries was evident, but values were similar whether the same samples were assessed 3 times over 3 years, 2 times over 1-3 years, or twice on the same day (data not shown). Recovery of CFU-GM and CFU-GEMM from unseparated cryopreserved cells (N = 3) was greater than 80% (data not shown), and consistent with recovery from MNCs. It is not clear why some samples resulted in low-efficiency recovery, but assessing the recovery of stored cells by thawing a small sample before their possible use in a clinical transplantation setting could help identify low-recovery CB units, and a decision made as to whether or not to use that unit. Proliferation of HPCs was high (Figure 1B) and within range for fresh CB.2,9-12 Thawed CB is highly responsive to increased colonies from immature HPCs when GM-CSF plus SCF and/or FL are used to stimulate them, compared with that of only GM-CSF (Figure 1C) demonstrating retention of immature HPCs.9 Thawed CB contains HPC colonies that can be replated (Figure 1D), suggesting maintenance of HPCs with limited self-renewal capacity.21 Secondary CFU-GM/M colonies formed from single replated CFU-GM/M colonies. CFU-GEMM colonies gave rise to secondary colonies of CFU-GEMM, erythroid progenitors, CFU-GM, and CFU-M.
Recovery of nucleated cellularity, HPCs, HSCs, and immune cells after cryopreservation and long-term frozen storage of CB. (A) Comparative percent recovery of nucleated cells, CFU-GM, and CFU-GEMM compared with prefreeze numbers for 10, 15, and 21-23.5 years of the exact sample frozen. n = number of different samples thawed for analysis. Results shown as mean ± 1 SEM with range of recoveries shown in parentheses. (B) Representative examples of colonies grown from CB thawed after 21-23.5 years in frozen state: (i) CFU-GEMM colony; (ii) CFU-GEMM (left) and CFU-GM (right) colonies; (iii) CFU-GM colony; (iv) CFU-GEMM colony. A Nikon TMS microscope was used with a PLAN objective 4× and projection lens magnification of 2.5× for a total magnification of 10× at 25°C. We used 35mm film (ASA200), and pictures were made with a Nikon HFX-DX automatic 35mm camera system. (C) Ratio of CFU-GM colonies formed after stimulation of CB cells with GM-CSF plus either SCF, FL, or SCF plus FL, divided by number of colonies formed by stimulation of same cells with only GM-CSF. n = number of different CB samples analyzed from cells frozen from 21-23.5 years before thaw and analysis. (D) Replating capacity of single CFU-GEMM or CFU-GM plus CFU-M (macrophage) colonies recovered from thawed CB cells stored frozen for up to 21 years. Results shown are from a total of over 1000 separately replated colonies each, and designated percent of secondary plates with at least 1 colony (% replates = top) and range of colonies in secondary dish per single replated colony (= bottom). (E) Analysis of engrafting capacity in sublethally irradiated NOD-SCID IL-2 receptor γ chain null (NSG) mice of cells (≥ 90% CD34+) purified from unseparated CB stored frozen for 18-21 years before thawing. Each experiment shows chimerism data from a different frozen CB unit. Analysis of human CD45+chimerism in peripheral blood (PB) or bone marrow (CB) of primary (1°) mouse recipients with N = number of mice per group, or CD45+ cell chimerism in secondary (2°) recipients of the same mouse strain given the same sublethal irradiation dose. For 2° recipients, each bar represents the number of secondary recipients per pooled BM of 1° recipient mice (experiments 1 and 2 only). Results are given as mean ± 1 SEM. (F) Response of CD4+ and CD8+ T lymphocytes, (≥ 98% Pure) isolated from thawed unseparated CB cells stored frozen for 21 years, as assessed by flow cytometry, to anti-CD3/CD28 stimulation, as determined by induced expression of CD25, a T-cell activation antigen. Shown are experiments from 1 of 2 experiments in which different frozen CB units were assessed.
Recovery of nucleated cellularity, HPCs, HSCs, and immune cells after cryopreservation and long-term frozen storage of CB. (A) Comparative percent recovery of nucleated cells, CFU-GM, and CFU-GEMM compared with prefreeze numbers for 10, 15, and 21-23.5 years of the exact sample frozen. n = number of different samples thawed for analysis. Results shown as mean ± 1 SEM with range of recoveries shown in parentheses. (B) Representative examples of colonies grown from CB thawed after 21-23.5 years in frozen state: (i) CFU-GEMM colony; (ii) CFU-GEMM (left) and CFU-GM (right) colonies; (iii) CFU-GM colony; (iv) CFU-GEMM colony. A Nikon TMS microscope was used with a PLAN objective 4× and projection lens magnification of 2.5× for a total magnification of 10× at 25°C. We used 35mm film (ASA200), and pictures were made with a Nikon HFX-DX automatic 35mm camera system. (C) Ratio of CFU-GM colonies formed after stimulation of CB cells with GM-CSF plus either SCF, FL, or SCF plus FL, divided by number of colonies formed by stimulation of same cells with only GM-CSF. n = number of different CB samples analyzed from cells frozen from 21-23.5 years before thaw and analysis. (D) Replating capacity of single CFU-GEMM or CFU-GM plus CFU-M (macrophage) colonies recovered from thawed CB cells stored frozen for up to 21 years. Results shown are from a total of over 1000 separately replated colonies each, and designated percent of secondary plates with at least 1 colony (% replates = top) and range of colonies in secondary dish per single replated colony (= bottom). (E) Analysis of engrafting capacity in sublethally irradiated NOD-SCID IL-2 receptor γ chain null (NSG) mice of cells (≥ 90% CD34+) purified from unseparated CB stored frozen for 18-21 years before thawing. Each experiment shows chimerism data from a different frozen CB unit. Analysis of human CD45+chimerism in peripheral blood (PB) or bone marrow (CB) of primary (1°) mouse recipients with N = number of mice per group, or CD45+ cell chimerism in secondary (2°) recipients of the same mouse strain given the same sublethal irradiation dose. For 2° recipients, each bar represents the number of secondary recipients per pooled BM of 1° recipient mice (experiments 1 and 2 only). Results are given as mean ± 1 SEM. (F) Response of CD4+ and CD8+ T lymphocytes, (≥ 98% Pure) isolated from thawed unseparated CB cells stored frozen for 21 years, as assessed by flow cytometry, to anti-CD3/CD28 stimulation, as determined by induced expression of CD25, a T-cell activation antigen. Shown are experiments from 1 of 2 experiments in which different frozen CB units were assessed.
HSC activity
Using different CB collections cryopreserved as unseparated cells, isolated CD34+ cells efficiently engrafted NSG mice for 6-7 months (Figure 1E). In 2 experiments, BM cells from engrafted chimeric mice repopulated secondary mice for 6 months. While we demonstrated engrafting capability of thawed CB after 15 years of storage using first generation NOD/SCID mice,11 those mice did not allow long-term primary engraftment or secondary repopulation. Thus, the current study greatly extends previous findings, and demonstrates recovery of long-term repopulating and self-renewing HSCs. We could not calculate percent recovery of HSCs as this assay was not available when cells were cryopreserved, but this engraftment is similar to fresh CB HSCs.11
T-cell activity
Attaining vigorous T-cell responses against common viral pathogens is critical for survival after CB transplantation.22 CB T cells are almost exclusively naive cells, with few effector or memory cells.18 CB T cells are immature compared with adult T cells because of impaired cytokine production and diminished lytic activity.22,23 To verify immune capability, CD4+ and CD8+ T lymphocytes, purified from unseparated CB stored up to 21 years, were activated as assessed by CD3/CD28-induced expression of CD25 (Figure 1F). This demonstrated recovery of functional T-cell subsets.
iPS cells
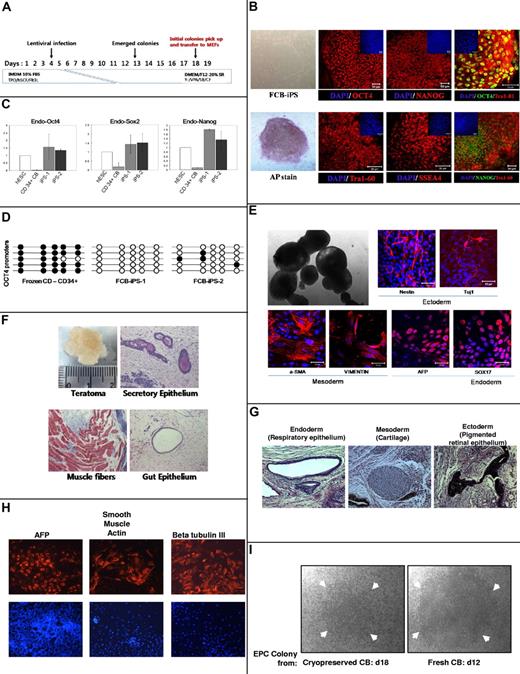
iPS cells are generated from different cell sources,24,25 including fresh CB,12-14 and CB cryopreserved for 5-8 years.12,14 We generated iPS cells from CB cryopreserved for up to 21 years using Oct4, KLF-4, Sox2, and c-Myc reprogramming with lentiviral vector transduction of CD34+ cells at IU (Figure 2A). iPS cell colonies stained positive for OCT4, NANOG, TRA-1-60, SSEA4, and alkaline phosphatase (Figure 2B). Quantitative RT-PCR demonstrated reprogramming via expression of endogenous OCT4, SOX2, and NANOG in comparison to H9 ESC cell line and CD34+ cells from which iPS cells were generated (Figure 2C). Unmethylated OCT4 promoter in 2 iPS cell lines generated from thawed CB in comparison to enhanced methylation for CD34+ cells from which iPS cell colonies were derived (Figure 2D), demonstrates early stages of produced cells. Embryoid bodies developed from iPS cells after removal from MEFs (Figure 2E), and expressed ectodermal, mesodermal, and endodermal proteins (Figure 2E). Moreover, injection of iPS cell colonies into testis capsules of immune-deficient mice demonstrated teratomas with ectoderm, mesoderm, and endoderm, confirming reprogramming. Generation of iPS cells12 at Johns Hopkins with 21-year frozen CB from a different collection produced cells expressing TRA-1-60, SSEA4, NANOG, and OCT4 (data not shown), and produced teratomas12 with expression of endoderm, mesoderm, and ectoderm markers (Figure 2G). These CB-derived iPS cells were differentiated in vitro (Figure 2H). Efficiency of iPS cell generation from thawed CB ranged from 0.027%-0.05% per CD34+ cell, similar to cultured CD34+ cells from freshly isolated or shorter-term frozen CB.12 This reprogramming efficiency appears higher than from human adult blood or fibroblastic cells.12,14 If iPS cells are found to be of clinical utility, which is not yet clear,24,25 HLA-typed CB stored in banks could serve as a source of such typed cells.
Reprogramming of 21-year-old frozen human CB CD34+ cells to iPS cells (A-H) and recovery of ECFCs (I). The phase contrast images in panels B (FCB-iPS and AP stain), E (top left image), and I were viewed using a Zeiss Axiovert 25 CFL inverted microscope with a 5× CP-ACHROMAT/0.12 NA objective. Images were acquired using a SPOT RT color camera (Diagnostic Instruments) with the manufacturer's software. Phase contrast images were taken with air objectives. The confocal images in panels B and E were viewed with a Zeiss Axiovert 100 LSM 510 confocal microscope using as objective a C-Apochromat (at 40×/1.2W corr) or Plan-Apochromat (at 10×/0.45). Images were taken by epifluorescence detector and transillumination detector and processed using Zeiss LSM Image Browser Version 3.5.0.376 software. All the images were taken at room temperature. Cells were mounted in proLong Gold antifade mounting reagent with DAPI (Invitrogen) and confocal images were taken using water immersion objectives. (A) Schematic representation of strategy used at IU to reprogram CD34+ cells from frozen human CB. TPO indicates Thrombopoietin; hSCF, stem cell factor; Flt2L, fms-like tyrosine kinase 3 ligand; Y, Y-27632; VPA, valporic acid; and SB, SB202190. (B) Immunocytochemistry for pluripotency markers OCT4 (Alexa 448 or 546 nm), NANOG (Alexa 488 or 564 nm), TRA-1–60 (Alexa 564 nm), SSEA4 (Alexa564 nm) and alkaline phosphatase (AP). Small panels on top part of subpanels show total cell content per field stained with DAPI. Scale bars represent 50 μm. (C) Quantitative RT-PCR analysis for expression of endogenous ES cell-marker genes OCT4, SOX2, and NANOG in hESC line H9, 2 iPS cell lines generated, and parental CD34+ cells. Specific primers were designed to probe for 3′ untranslated region (Endo) to measure expression of the endogenous gene. Individual PCR reactions were normalized against β-actin and plotted relative to expression levels in hESCs. (D) Each horizontal row of circles represents an individual sequencing reaction for a given amplicon. Open and filled circles, respectively, represent unmethylated and methylated CpGs dinucleotides. (E) Morphology of 10-day embryoid body (EB) under phase contrast microscopy (top left). Immunostaining of frozen CB (FCB)–iPS derivatives on day 10 of differentiation revealed expression of ectodermal (Nestin and Tuj1), mesodermal (α-SMA and Vimentin), and endodermal (AFP and Sox17) marker proteins (all with Alexa 546nm). Nuclei are stained with DAPI (blue). Scale bars represent 50 μm. (F) Teratomas derived from immunodeficient mice injected with FCB-iPS cells shows tissues representing all 3 embryonic germ layers, including secretory epithelium (ectoderm), muscle fibers (mesoderm), and gut epithelium (endoderm). Samples from the teratomas were paraffin-embedded and serially sectioned (5 μm) using a microtome (Leica Microsystems). To analyze the 3 germ-layer lineage cells derived from injected FCB-iPS cells in the teratomas, sectioned slides were histologically examined by hematoxylin and eosin for the gut epithelium and special stains as follows: PAS stain for the secretory epithelium, and Masson trichrome stain for muscle fibers. Images were taken with a Nikon Eclipse 50i equipped with a digital camera (Infinty 2 Megapixel; Lumenera Corp) at a magnification of Plan Fluor 10×/0.30 and analyzed using the i-solution image analysis program (iMT; i-Solution Inc). Teratoma images were taken with air objectives. Results in panels B through F were from studies performed at IU. Results in panels G and H were from another 21-year cryopreserved CB sample sent to, thawed, and separately reprogrammed at Johns Hopkins Medical Center12 after isolation of CD34+ cells. For studies in panels G and H, a Nikon Eclipse TE2000-U microscope was used, with ELWD Plan Fluor, NA:0.45 at 25°C. For panel G, fixed slides were used with H&E staining, and for panel H, PBS plus 0.1% BSA was used with Alexa 555 and DAPI. Images were taken with Imaging Micropublisher 5.0 (Q Imaging) camera with Q Capture Version 3.1.2 (Q Imaging) software. Markers for teratoma formation in vivo (G) and for differentiation of EBs from reprogrammed iPS cells in vitro (H). (I) Representative ECFC colony from MNCs isolated from 21-year CB defrosts and cultured for 18 days (left) compared with colony from freshly obtained CB cultured for 12 days. Arrows denote size of colonies. Note that even after 18 days culture of frozen CB-derived colonies, the colony size is not as large as that of colony from fresh CB at 12 days of culture.
Reprogramming of 21-year-old frozen human CB CD34+ cells to iPS cells (A-H) and recovery of ECFCs (I). The phase contrast images in panels B (FCB-iPS and AP stain), E (top left image), and I were viewed using a Zeiss Axiovert 25 CFL inverted microscope with a 5× CP-ACHROMAT/0.12 NA objective. Images were acquired using a SPOT RT color camera (Diagnostic Instruments) with the manufacturer's software. Phase contrast images were taken with air objectives. The confocal images in panels B and E were viewed with a Zeiss Axiovert 100 LSM 510 confocal microscope using as objective a C-Apochromat (at 40×/1.2W corr) or Plan-Apochromat (at 10×/0.45). Images were taken by epifluorescence detector and transillumination detector and processed using Zeiss LSM Image Browser Version 3.5.0.376 software. All the images were taken at room temperature. Cells were mounted in proLong Gold antifade mounting reagent with DAPI (Invitrogen) and confocal images were taken using water immersion objectives. (A) Schematic representation of strategy used at IU to reprogram CD34+ cells from frozen human CB. TPO indicates Thrombopoietin; hSCF, stem cell factor; Flt2L, fms-like tyrosine kinase 3 ligand; Y, Y-27632; VPA, valporic acid; and SB, SB202190. (B) Immunocytochemistry for pluripotency markers OCT4 (Alexa 448 or 546 nm), NANOG (Alexa 488 or 564 nm), TRA-1–60 (Alexa 564 nm), SSEA4 (Alexa564 nm) and alkaline phosphatase (AP). Small panels on top part of subpanels show total cell content per field stained with DAPI. Scale bars represent 50 μm. (C) Quantitative RT-PCR analysis for expression of endogenous ES cell-marker genes OCT4, SOX2, and NANOG in hESC line H9, 2 iPS cell lines generated, and parental CD34+ cells. Specific primers were designed to probe for 3′ untranslated region (Endo) to measure expression of the endogenous gene. Individual PCR reactions were normalized against β-actin and plotted relative to expression levels in hESCs. (D) Each horizontal row of circles represents an individual sequencing reaction for a given amplicon. Open and filled circles, respectively, represent unmethylated and methylated CpGs dinucleotides. (E) Morphology of 10-day embryoid body (EB) under phase contrast microscopy (top left). Immunostaining of frozen CB (FCB)–iPS derivatives on day 10 of differentiation revealed expression of ectodermal (Nestin and Tuj1), mesodermal (α-SMA and Vimentin), and endodermal (AFP and Sox17) marker proteins (all with Alexa 546nm). Nuclei are stained with DAPI (blue). Scale bars represent 50 μm. (F) Teratomas derived from immunodeficient mice injected with FCB-iPS cells shows tissues representing all 3 embryonic germ layers, including secretory epithelium (ectoderm), muscle fibers (mesoderm), and gut epithelium (endoderm). Samples from the teratomas were paraffin-embedded and serially sectioned (5 μm) using a microtome (Leica Microsystems). To analyze the 3 germ-layer lineage cells derived from injected FCB-iPS cells in the teratomas, sectioned slides were histologically examined by hematoxylin and eosin for the gut epithelium and special stains as follows: PAS stain for the secretory epithelium, and Masson trichrome stain for muscle fibers. Images were taken with a Nikon Eclipse 50i equipped with a digital camera (Infinty 2 Megapixel; Lumenera Corp) at a magnification of Plan Fluor 10×/0.30 and analyzed using the i-solution image analysis program (iMT; i-Solution Inc). Teratoma images were taken with air objectives. Results in panels B through F were from studies performed at IU. Results in panels G and H were from another 21-year cryopreserved CB sample sent to, thawed, and separately reprogrammed at Johns Hopkins Medical Center12 after isolation of CD34+ cells. For studies in panels G and H, a Nikon Eclipse TE2000-U microscope was used, with ELWD Plan Fluor, NA:0.45 at 25°C. For panel G, fixed slides were used with H&E staining, and for panel H, PBS plus 0.1% BSA was used with Alexa 555 and DAPI. Images were taken with Imaging Micropublisher 5.0 (Q Imaging) camera with Q Capture Version 3.1.2 (Q Imaging) software. Markers for teratoma formation in vivo (G) and for differentiation of EBs from reprogrammed iPS cells in vitro (H). (I) Representative ECFC colony from MNCs isolated from 21-year CB defrosts and cultured for 18 days (left) compared with colony from freshly obtained CB cultured for 12 days. Arrows denote size of colonies. Note that even after 18 days culture of frozen CB-derived colonies, the colony size is not as large as that of colony from fresh CB at 12 days of culture.
ECFCs
High proliferative ECFCs have been identified in CB.15 MNCs from thawed, unseparated CB stored frozen for up to 21 years formed ECFC colonies, but their size was smaller than colonies from fresh CB (Figure 2I). ECFC colony numbers from thawed CB (2-5/107 mononuclear cells) were 1/5 to 1/10 numbers from fresh CB, even when colonies from fresh cells versus those frozen and stored for up to 3-6 months were assayed. Thus, the freezing procedure that works well for efficient recovery of HPCs may not be optimal for storage of ECFCs. However, ECFCs that can be cryopreserved and recovered may be of value for regenerative medicine, if clinical applicability is proven.24 Thus, recovery of HSCs, HPCs, and other early cell types bodes well for CB banking and use.
The online version of this article contains a data supplement.
The publication costs of this article were defrayed in part by page charge payment. Therefore, and solely to indicate this fact, this article is hereby marked “advertisement” in accordance with 18 USC section 1734.
Acknowledgments
These studies were supported by National Institutes of Health Public Health Service grants NIH R01 HL56416 and NIH R01 HL67384, and a project in NHLBI PO1 HL053586 to H.E.B., NIH R01 HL073781 to L.C., and a grant from the Riley Children's Foundation to M.C.Y. Z.Y. was supported by NIH T32 grant HL007525. The IU production of lentiviral vector was funded by NIH P40RR024928 to K.C., and S.W. is funded on a faculty recruitment grant from NIH (P30 HL101337).
National Institutes of Health
Authorship
Contribution: H.E.B., M.-R.L., N.P., Z.Y., S.W., K.C., L.C., and M.C.Y. designed experiments; H.E.B., M.-R.L., G.H., S.C., N.P., Y.-J.K., C.M., Z.Y., S.W., K.C., L.C., and M.C.Y. performed research and analyzed and interpreted data; H.E.B. wrote the paper, and H.E.B., M.-R.L., N.P., Y.-J.K., Z.Y., S.W., K.C., L.C., and M.C.Y. edited the paper.
Conflict-of-interest disclosure: H.E.B. is on the Medical Scientific Advisory Board of Corduse, a cord blood banking company. M.C.Y. is a cofounder and consultant to EndGenitor Technologies Inc. The remaining authors declare no competing financial interests.
Correspondence: Hal E. Broxmeyer, PhD, Indiana University School of Medicine, Department of Microbiology and Immunology, 950 West Walnut St, R2-302, Indianapolis, IN 46202-5181; e-mail: hbroxmey@iupui.edu.