Abstract
To investigate the mechanism by which macrophage inflammatory protein-1α (MIP-1α) affects graft-versus-host disease (GVHD), the expression and function of MIP-1α in 2 murine models of GVHD were evaluated. In irradiated class I and class II disparate recipients, the expression of messenger RNA (mRNA) and protein for MIP-1α was significantly increased in GVHD target organs after transfer of allogeneic lymphocytes compared to syngeneic lymphocytes. When lymphocytes unable to make MIP-1α were transferred, there was a decrease in the production of MIP-1α in the liver, lung, and spleen of bm1 (B6.C-H2bm1/By) and bm12 (B6.C-H2bm12/KhEg) recipients compared to the transfer of wild-type splenocytes. At day 6 there was a 4-fold decrease in the number of transferred CD8+ T cells in the lung and approximately a 2-fold decrease in the number of CD8+ T cells in the liver and spleen in bm1 recipients after transfer of MIP-1α–deficient (MIP-1α−/−) splenocytes compared to wild-type (MIP-1α+/+) splenocytes. These differences persisted for 13 days after splenocyte transfer. In contrast, the number of donor CD4+ T cells found in the liver and lung was significantly increased after the transfer of MIP-1α−/− compared to wild-type splenocytes in bm12 recipients from day 6 through day 10. Thus, the transfer of allogeneic T cells was associated with the enhanced expression of MIP-1α in both a class I and class II mismatch setting. However, the increased expression only led to enhanced recruitment of CD8+, but not CD4+, donor T cells. Production of MIP-1α by donor T cells is important in the occurrence of GVHD and functions in a tissue-dependent fashion.
Introduction
Graft-versus-host disease (GVHD) limits the availability of allogeneic bone marrow transplantation for the treatment of bone marrow failure syndromes,1,2 acute and chronic leukemias,3-10 and inborn errors of metabolism.11 GVHD involves the recognition by donor lymphocytes of peptides presented by host major histocompatability complex (MHC) class I and class II molecules.12-15 This process leads to the release of preformed cytolytic proteins such as perforin16-18 and proinflammatory cytokines such as tumor necrosis factor (TNF)-α.19 In murine models of GVHD, the production of these same proteins is critical to the effector phase.
Acute GVHD in both humans and mouse models typically involves specific target organs. The reasons for the occurrence of GVHD in these organs are not well understood. In an MHC-matched sibling transplant, the tissue specificity of GVHD may relate to tissue-specific expression of minor antigens.20 However, in MHC-mismatched transplantation, current data does not support the fact that alloreactive T cells recognize tissue-specific peptides.21
Chemokines are a large family of proteins that recruit specific populations of effector cells to sites of inflammation.22-31 Chemokines are classified based on the motif of amino acids around the N-terminal cysteine (Cys). Currently, there are 4 known chemokine subfamilies, denoted C, C-C, C-X-C, and C-X3-C. Although chemokines have been implicated in the recruitment of inflammatory cells in autoimmune diseases and graft rejection,30,32-36 until now there has been little data to show the involvement of chemokines in the pathogenesis of GVHD. Recent work suggests that some chemokines have roles in inflammation, whereas others function in basal cell trafficking. The C-C chemokine, macrophage inflammatory protein (MIP)-1α, is among a group of chemokines induced during inflammation.37 Thus, the specific expression and production of these proteins in target organs involved in GVHD may be important in the recruitment of alloreactive lymphocytes. In support of this, we and others have shown recently that the production of MIP-1α is important in determining the severity of GVHD in mouse models.38 39
We investigated the expression and function of MIP-1α in the pathogenesis of GVHD in class I and class II disparate recipients. In both models MIP-1α was expressed and produced early in multiple organs in which GVHD occurred, and in certain organs, T cells were a critical source of MIP-1α production. Previously we have shown that the enhanced expression of MIP-1α, however, was only associated with the occurrence of GVHD across a class I MHC barrier. To investigate this we evaluated the trafficking of donor CD4+ and CD8+ T cells after transfer of either MIP-1α–deficient (MIP-1α−/−) or wild-type (MIP-1α+/+) splenocytes into bm1 or bm12 recipients (bm1 or bm12). T-cell production of MIP-1α increased the recruitment of donor CD8+ T cells by approximately 2- to 4-fold in a subset of GVHD target organs. On the contrary, an increase in the number of donor CD4+ T cells was found after the transfer of splenocytes from MIP-1α−/− donors.
Materials and methods
Mice
C57BL/6, B6.C-H2bm1/By (bm1), and B6.C-H2bm12/KhEg (bm12) mice were purchased (Jackson Laboratory, Bar Harbor, ME). B6-129-Scya3tm1unc(MIP-1α−/−) mice were backcrossed 10 generations onto C57BL/6 and have been previously described.38 Transgenic mice expressing the enhanced green fluorescent protein (eGFP) were generated by one of us (S.A.L.) using a transgene (Dr M. Okabe, Osaka University, Japan). The eGFP expression is driven by the chicken β-actin promoter and a CMV immediate early enhancer and is expressed in all tissues except red blood cells and hair. These mice are phenotypically identical to mice previously described by Okabe et al.40 Cells expressing eGFP fluoresce after excitation using ultraviolet light, which can be detected by flow cytometry at 488 nm. This allows for tracking of eGFP-expressing transgenic cells after transfer to non-transgenic hosts in vivo. The eGFP mice were crossed initially to MIP-1α−/− B6 mice to produce eGFP+ MIP-1α−/+ B6 mice. These mice were then backcrossed to MIP-1α−/− B6 and C57BL/6 controls to produce the mice used in these experiments. All mice were housed under microisolator cage tops and maintained in a specific pathogen–free environment. Male and female mice between 7 and 12 weeks of age were used. All experiments were performed under approved protocols of the Division of Laboratory Medicine at the University of North Carolina School of Medicine, Chapel Hill, NC.
Splenocyte transfers
Recipient animals were given 675 cGy of irradiation from a cesium 137 (137Cs) source (dose rate, 105 cGy/min) (Atomic Energy of Canada, Ottawa, ON, Canada) 6-12 hours prior to the splenocyte transfer. Donor mice were killed by cervical transection, and the splenocytes were harvested and then depleted of red blood cells using ACK lysis buffer. The splenocytes were resuspended in nonbacteriostatic saline, and 5 × 106 splenocytes were injected intravenously in a volume of 100-150 μL into a lateral tail vein. The animals were followed for the occurrence of GVHD using a previous protocol for the grading of GVHD.41
Splenocyte transfers using eGFP+ MIP-1α−/−or eGFP+ control mice were performed as stated above. On the indicated day after transfer bm1 or bm12 mice were killed, and the cells were isolated from various tissues using an established protocol.38 Briefly, the mice were killed with Avertin (2,2,2-Tribromoethanol; Aldrich, Milwaukee, WI) and injected interperitoneally with heparin to prevent red cell clotting. We infused 10 mL phosphate-buffered saline (PBS) into the right and left ventricle by cardiac puncture. The tissues were excised and minced in Roswell Park Memorial Institute medium (RPMI) with 5% fetal calf serum (FCS). The tissue was incubated in 15 mL digestion solution (15 mL RPMI with 5% FCS), 1 mg/mL Collagenase A (Boehringer Mannheim, Indianapolis, IN), and 25 units deoxyribonuclease (DNAse) (Sigma Chemical Co, St Louis, MO). The tissue was washed, and the red cells were removed using ACK lysis buffer. The cells were washed 3 times with PBS, layered onto a 40% Percoll gradient, and spun at 1500g for 20 minutes. The cells were resuspended in PBS with 2% fetal bovine serum (FBS) and 0.5% sodium azide. The cells were analyzed by 2-color flow cytometry using phycoerythrin (PE)-conjugated anti-CD4 (clone GK 1.5 rat immunoglobulin G [IgG2b]) and anti-CD8 (clone 53-6.7 rat IgG2a) monoclonal antibodies (PharMingen, La Jolla, CA) at 488 nm for the expression of eGFP.
RNA isolation
On the indicated day after splenocyte transfer, the mice were killed by cervical transection. The tissues were removed and immediately homogenized in Trizol reagent (Gibco BRL Life Technologies, Grand Island, NY). RNA was extracted according to the manufacturer's instructions.
Ribonuclease protection assay
RNA probes (PharMingen) were synthesized according to the manufacturer's instructions. We used 40 μg RNA per sample, and the assays were run according to the protocol given by the manufacturer. RNA was separated on a 20- by 1.5-cm 5% acrylamide per 8 mol/L urea gel then transferred to a Hy-bond membrane (Amersham Pharmacia Biotech, Arlington Heights, IL) using a vertical submarine transfer unit (CBS Scientific, Del Mar, CA). The membrane was dried in an oven at 80°C for 2 hours.
The membrane was washed twice with blocking buffer (0.2% I-Block Reagent; Tropix, Bedford, MA), 1 × PBS and 0.5% sodium dodecyl sulfate (SDS) (Sigma), then incubated in avidin-alkaline phosphatase conjugate (Tropix) at a concentration of 1:10 000 in blocking buffer. The blot was washed in wash buffer (1 times PBS and 0.5% SDS) and incubated with CDP-Star substrate (Tropix) for 5 minutes prior to exposure onto chemiluminescent hyperfilm (Amersham Pharmacia Biotech). The MIP-1α expression was normalized by analyzing the ratio of glyceraldehyde-3-phosphate dehydrogenase (GAPDH) to MIP-1α using a densitometer with the National Institutes of Health (NIH) Image Software Program (NIH, Bethesda, MD).
Enzyme-linked immunosorbent assay
Enzyme-linked immunosorbent assays (ELISAs) were performed on hepatic, pancreatic, colonic, and splenic tissue. Briefly, mice were killed on the indicated day after transplantation, and the liver, colon, pancreas, and spleen were removed. The tissue was homogenized using a tissue grinder (Polytron; Kinematica AG, Switzerland) in the presence of the following protease inhibitors: 2.5 μg/mL leupeptin, 1 mmol/L phenylmethylsulfonylflouride, and 2.5 μg/mL aprotinin. ELISA was performed on supernatants from the tissue homogenate according to the manufacturer's instructions (R&D Systems, Minneapolis, MN).
Histopathology
The organs were removed at the time of sacrifice, placed in OCT or omnifix, and sectioned with a microtome. The sections were stained with hematoxylin and eosin. Individual sections were evaluated for evidence of GVHD using a quantitative assessment, as previously described.41 The sections were evaluated by one of us (A.P.-M.) who was blinded to the treatment given.
T-cell lines
H2Kbm1-specific T-cell lines were generated by transferring 5 × 106 splenocytes from bm1 mice into irradiated (650 cGy) MIP-1α−/− recipients. The mice were killed 10 days after splenocyte transfer, splenocytes were isolated, and CD8 T cells were selected using a VarioMacs magnet and an anti–CD8-α monoclonal antibody coupled to magnetic beads (Miltenyi Biotech GmbH, Bergisch Gladbach, Germany). More than 98% of the population expressed CD8 as measured by flow cytometry (data not shown). The CD8+ splenocytes were incubated with irradiated (2500 cGy) splenocytes from MIP-1α−/− mice. At day 6, 25% concavalin A (con A) supernatant was added to the T cells, which were stimulated weekly in this manner. CTL assays were performed using MIP-1α−/− B6 con A blasts and bm1 con A blasts (lines termed 150A-C) as previously reported.
To evaluate the expression of chemokines by the T-cell lines, T cells were incubated with bone marrow–derived macrophages from either MIP-1α−/− or bm1 mice. At 24 hours after incubation the supernatants were harvested, the cells were lysed in Trizol reagent, and RNA was isolated as previously described. To verify that production of MIP-1α was from the T cells, the T cells were incubated with bone marrow–derived macrophages from MIP-1α−/− animals in all experiments. Chemokine production in this model was evaluated by performing ELISAs on supernatants.
Statistics
Expression of messenger RNA (mRNA) for MIP-1α using the ribonuclease (RNAse) protection assay, protein production by ELISA, and the differences in the quantitative GVHD score were compared using the Student t test. All tests were 2-tailed.P ≤ .05 was considered significant.
Results
MIP-1α expression in tissues after syngeneic and allogeneic T-cell transfer
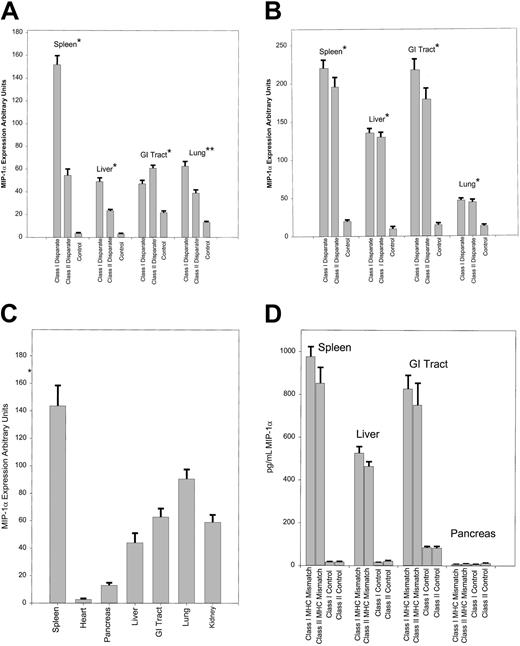
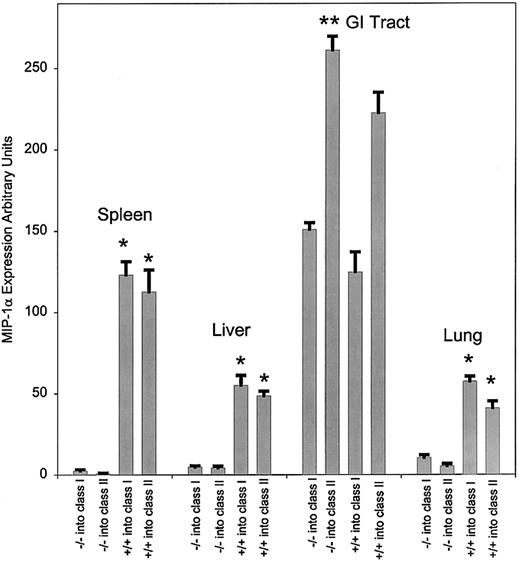
To evaluate whether the expression of MIP-1α was enhanced after allogeneic splenocyte transfer, we compared the expression of MIP-1α from the gastrointestinal (GI) tract, kidney, liver, lung, and spleen in bm1 (class I disparate) and bm12 (class II disparate) recipients after splenocyte transfer from syngeneic (bm1 or bm12) or allogeneic C57BL/6 mice. The expression of MIP-1α was not detected until day 3 and then only in the spleen after the transfer of allogeneic T cells (data not shown). By day 6 there was a significant increase in the expression of MIP-1α in the GI tract (P = .03) and in the liver, lung, and spleen (P < .001; Figure1A) only after the transfer of allogeneic splenocytes. This increase persisted through days 12-13 in both bm1 and bm12 recipients (P < .001; Figure 1B). There was no difference in the expression of MIP-1α in the kidney after allogeneic or syngeneic T-cell transfer at day 6 (data not shown) or day 12 (Figure 1C), which suggests an effect of irradiation on the production of MIP-1α at this site. The increase in MIP-1α was specific to certain organs, as we did not find an increase in the expression of MIP-1α in the heart or pancreas, 2 organs not involved with GVHD (Figure 1C).
Expression of mRNA and protein for MIP-1α at days 6 and 13 after syngeneic and allogeneic T-cell transfer.
RNA was isolated from the individual organs, and RPA was performed as described in the text. Expression of MIP-1α was analyzed by densitometry using the NIH Image Software Program, then normalized to the expression of GAPDH for each condition. The mean expression with SE is given. Background activity was subtracted using an internal control for each lane. We evaluated 4 mice for each condition. Each experiment was repeated 3 times, and the data were pooled from the 3 experiments. Control animals received irradiation and syngeneic splenocytes. (A) Data are shown for expression at day 6 from the GI tract, liver, lung, and spleen. (B) The same analysis was performed as described above except that the mice were killed 13 days after splenocyte transfer. We evaluated 3 mice per condition, and each experiment was repeated twice. The results are given as the mean expression and SE and are pooled from 2 separate experiments. (C) Expression of MIP-1α at day 13 in GVHD target organs was evaluated as above and compared to the expression in the kidney, heart, and pancreas. (D) ELISA was performed on colonic, hepatic, pancreatic, and splenic tissue from bm1 and bm12 recipients on day 6 following the transfer. The limit of detection of MIP-1α for the assay is 3.75 pg/mL. We evaluated 3 mice per condition. *P < .001; **P = .02 for comparison between class I and class II only.
Expression of mRNA and protein for MIP-1α at days 6 and 13 after syngeneic and allogeneic T-cell transfer.
RNA was isolated from the individual organs, and RPA was performed as described in the text. Expression of MIP-1α was analyzed by densitometry using the NIH Image Software Program, then normalized to the expression of GAPDH for each condition. The mean expression with SE is given. Background activity was subtracted using an internal control for each lane. We evaluated 4 mice for each condition. Each experiment was repeated 3 times, and the data were pooled from the 3 experiments. Control animals received irradiation and syngeneic splenocytes. (A) Data are shown for expression at day 6 from the GI tract, liver, lung, and spleen. (B) The same analysis was performed as described above except that the mice were killed 13 days after splenocyte transfer. We evaluated 3 mice per condition, and each experiment was repeated twice. The results are given as the mean expression and SE and are pooled from 2 separate experiments. (C) Expression of MIP-1α at day 13 in GVHD target organs was evaluated as above and compared to the expression in the kidney, heart, and pancreas. (D) ELISA was performed on colonic, hepatic, pancreatic, and splenic tissue from bm1 and bm12 recipients on day 6 following the transfer. The limit of detection of MIP-1α for the assay is 3.75 pg/mL. We evaluated 3 mice per condition. *P < .001; **P = .02 for comparison between class I and class II only.
To confirm that our expression data correlated with the production of MIP-1α, we performed an ELISA evaluation from the GI tract, liver, pancreas, and spleen after syngeneic and allogeneic T-cell transfer in bm1 and bm12 recipients at days 6, 13, and 15. These data are shown for day 13 samples (Figure 1D). We found that the enhanced expression of mRNA for MIP-1α after allogeneic T-cell transfer correlated quite closely with protein production in this set of organs. This was also true for production on days 6 and 15 after T-cell transfer (data not shown). Thus, the transfer of alloreactive T cells mediated the expression of MIP-1α in GVHD target organs.
T-cell expression of chemokines in vivo
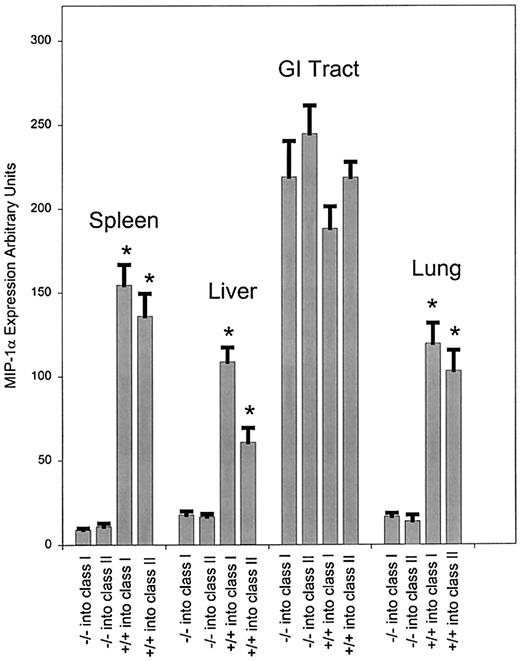
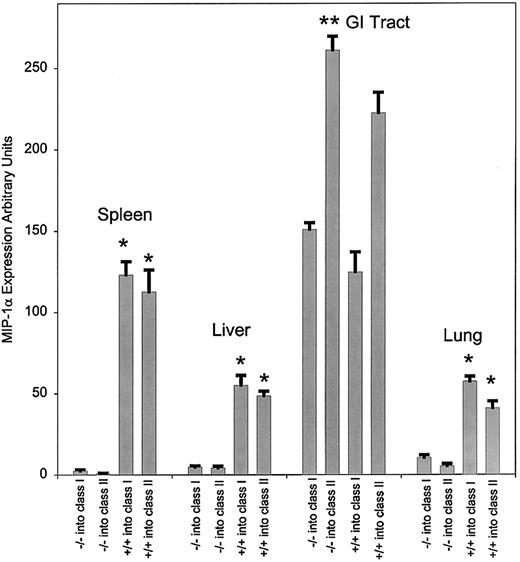
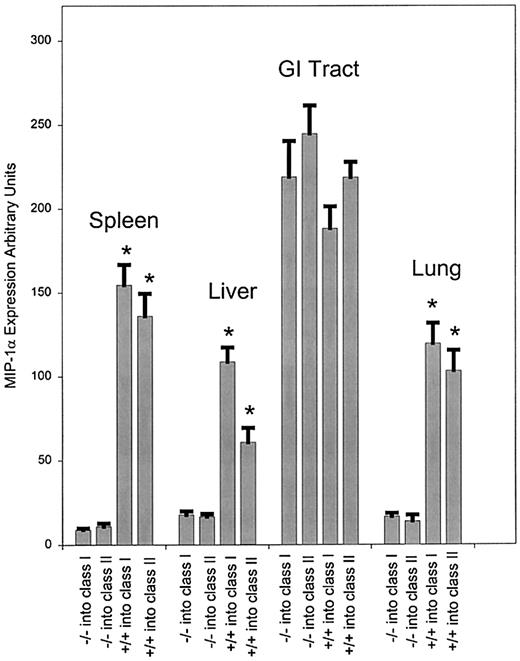
To explore the contribution of the transferred T-cell population to the production of MIP-1α in the bm1 and bm12 models, we transferred splenocytes from either MIP-1α–deficient (MIP-1α−/−) or wild-type (MIP-1α+/+) mice. Prior to day 3, little expression of MIP-1α was detected in the spleen (data not shown). At day 3 after transfer (data not shown), there was decreased expression of MIP-1α in the spleen when using MIP-1α−/− splenocytes compared to using MIP-1α+/+ splenocytes (P < .001 for both class I and class II mismatch). Starting on day 6 (Figure2) and through day 13 (Figure3), we observed a significant decrease in expression of MIP-1α in the liver, lung, and spleen (P < .001 for both class I and class II mismatch) in mice receiving MIP-1α−/− splenocytes compared to those receiving MIP-1α+/+ splenocytes. By contrast, there was no difference in the expression of MIP-1α in the GI tract in either bm1 or bm12 recipients after transfer of MIP-1α−/− or MIP-1α+/+ splenocytes. Thus the expression of MIP-1α by donor cells was important in the overall production of MIP-1α, but only in a subset of GVHD target organs (liver, lung, and spleen). Although the transfer of allogeneic T cells was critical to the expression of MIP-1α in the GI tract in both bm1 and bm12 recipients, production of MIP-1α did not diminish after the transfer of splenocytes unable to generate MIP-1α.
Expression of mRNA for MIP-1α at day 6 after transfer of either MIP-1α−/− or wild-type splenocytes.
Expression of MIP-1α was evaluated as given previously after transfer of splenocytes from C57BL/6 or MIP-1α−/− B6 donors into bm1 recipients. The bm1 mice were killed on day 6 after the transfer, and RNA was isolated and quantitated as described. We evaluated 3 mice per condition, and the data were pooled from 2 separate experiments. P values were analyzed using the Student t test. *P < .001, which compares the difference of MIP-1α−/− and MIP-1α+/+ splenocytes in class I and class II.
Expression of mRNA for MIP-1α at day 6 after transfer of either MIP-1α−/− or wild-type splenocytes.
Expression of MIP-1α was evaluated as given previously after transfer of splenocytes from C57BL/6 or MIP-1α−/− B6 donors into bm1 recipients. The bm1 mice were killed on day 6 after the transfer, and RNA was isolated and quantitated as described. We evaluated 3 mice per condition, and the data were pooled from 2 separate experiments. P values were analyzed using the Student t test. *P < .001, which compares the difference of MIP-1α−/− and MIP-1α+/+ splenocytes in class I and class II.
Expression of mRNA for MIP-1α at day 13 after transfer of either C57BL/6 or MIP-1α−/− B6 splenocytes.
RNA was extracted from the various tissues as indicated in the text and evaluated by RPA. The quantity of mRNA for each condition was normalized to the expression of GAPDH. We evaluated 3 mice per condition, and the data were pooled from 3 separate experiments. P values were analyzed using the Studentt test. *P < .001, which compares the difference of MIP-1α−/− and MIP-1α+/+ for class I and class II; **P = .03.
Expression of mRNA for MIP-1α at day 13 after transfer of either C57BL/6 or MIP-1α−/− B6 splenocytes.
RNA was extracted from the various tissues as indicated in the text and evaluated by RPA. The quantity of mRNA for each condition was normalized to the expression of GAPDH. We evaluated 3 mice per condition, and the data were pooled from 3 separate experiments. P values were analyzed using the Studentt test. *P < .001, which compares the difference of MIP-1α−/− and MIP-1α+/+ for class I and class II; **P = .03.
T-cell expansion/recruitment is dependent on expression of MIP-1α
Despite our findings that the MIP-1α expression was increased in both bm1 and bm12 recipients after allogeneic T-cell transfer, we had previously shown that blocking T-cell production of MIP-1α only affected GVHD across a class I MHC barrier.38 This suggested that MIP-1α is involved specifically in CD8+T-cell engraftment, trafficking, or function. To evaluate this we transferred splenocytes from eGFP+MIP-1α−/− and eGFP+ mice with an intactMIP1A gene into bm1 (class I disparate) recipients and bm12 (class II disparate) recipients and followed the trafficking of CD4+ or CD8+ T cells. In these models both bm1 and bm12 recipients developed clinical evidence of GVHD by 7 days after transfer. Death was due to GVHD involving the liver, lung, and GI tract in bm1 and bm12 recipients. Additionally, both bm1 and bm12 recipients developed significant marrow aplasia due to donor destruction of host class I–positive or class II–positive hematopoietic cells in the absence of the transfer of donor marrow.42-45 In both bm1 and bm12 recipients we observed a similar level of engraftment of the transferred cells in the spleen, independent of the production of MIP-1α, at day 1 after transfer (data not shown). At day 2 there was a 1.6-fold increase in the number of donor CD8+ T cells in the spleen in bm1 recipients after transfer of MIP-1α+/+splenocytes compared to MIP-1α−/− splenocytes (data not shown). Interestingly, despite the lack of a difference in class II MHC molecules in this model, there was an increase in the number of donor CD4+ T cells and monocytes in the spleen after transfer of MIP-1α−/− splenocytes. We found no difference in the number of donor CD4+ lymphocytes in the spleen of bm12 recipients at day 2 (data not shown). From day 2 to day 6 there were significantly less donor CD8+ T cells in bm1 recipients after transfer of MIP-1α splenocytes compared to wild-type splenocytes. There was no difference in the number of donor cells found in bm12 recipients of either MIP-1α or wild-type splenocytes (data not shown).
We have previously shown a dramatic decrease in the inflammatory response in the lung of bm1 mice that received splenocytes from MIP-1α−/− mice compared to wild-type mice.38 As a result we evaluated the number of donor CD8+ T cells in the lung, liver, and spleen and compared this to the expression in the kidney, an organ where MIP-1α expression was not dependent on the transfer of alloreactive T cells. Prior to day 3 we were unable to detect eGFP+ donor cells in the GI tract, kidney, liver, or lung from either bm1 or bm12 recipients. By day 6 there was a 4-fold decrease (P < .001) in the number of transferred CD8+T cells in the lung and approximately a 2-fold decrease in CD8+ T cells in the liver and spleen (P < .001), which was determined comparing eGFP+ MIP-1α−/− splenocytes with eGFP+ MIP-1α+/+ splenocytes (Figure4 and Table1). The remainder of the eGFP+ cells in the lung after the transfer of MIP-1α−/− splenocytes were monocytes and CD4+ lymphocytes (data not shown). Interestingly, in the kidney (P = .8), where there was no observed difference in the production of MIP-1α after transfer of wild-type or MIP-1α−/− splenocytes, there was also no difference in the number of CD8+ donor T cells after transfer of the GFP+ MIP-1α−/− or MIP-1α+/+splenocytes (Figure 4). Similarly there was no difference found in the number of donor CD8+ T cells in the GI tract after transfer of MIP-1α−/− splenocytes compared to MIP-1α+/+ splenocytes (Table 1). These findings persisted through day 12 (Table 1).
Two-color histograms at day 6 evaluating recruitment of CD8+ T cells to the kidney, liver, lung and spleen.
Analysis of the proportion of CD8+ T cells in the kidney, liver, lung, and spleen at day 6 after transfer of MIP-1α−/− or MIP-1α+/+ T cells. Significant differences were noted in the number of CD8+ T cells in the liver, lung, and spleen (P < .001 for both; Table 1) as a function of T-cell production of MIP-1α. Due to the significant expression of eGFP in the donor cells, the population of CD8-expressing cells has been shifted down on the y axis, resulting in the majority of cells not expressing CD8 falling below the y axis.
Two-color histograms at day 6 evaluating recruitment of CD8+ T cells to the kidney, liver, lung and spleen.
Analysis of the proportion of CD8+ T cells in the kidney, liver, lung, and spleen at day 6 after transfer of MIP-1α−/− or MIP-1α+/+ T cells. Significant differences were noted in the number of CD8+ T cells in the liver, lung, and spleen (P < .001 for both; Table 1) as a function of T-cell production of MIP-1α. Due to the significant expression of eGFP in the donor cells, the population of CD8-expressing cells has been shifted down on the y axis, resulting in the majority of cells not expressing CD8 falling below the y axis.
In contrast, there was a statistically significant increase in the number of CD4+ donor T cells in the liver and lung of irradiated class II disparate bm12 recipients after the transfer of MIP-1α−/− splenocytes compared to wild-type splenocytes (Table 2). Thus, in the liver, lung, and spleen, inhibiting T-cell production of MIP-1α markedly diminished MIP-1α expression and similarly CD8+ T-cell recruitment. In the GI tract and kidney, where host cell production of MIP-1α was important, blocking the generation of MIP-1α by donor cells had no effect on the recruitment of donor CD8+ T cells (Figure 4and Table 1). However, the transfer of T cells unable to produce MIP-1α enhanced the recruitment of CD4+ T cells in bm12 recipients.
Histology
Previously we had shown a marked increase in the inflammatory response in the lung of wild-type compared to MIP-1α−/−splenocytes.38 To assess the grading of GVHD using a slightly higher dose of irradiation, in which mice have died earlier of classical GVHD, a quantitative scoring system was used to assess GVHD in the GI tract, lung, and liver after the transfer of MIP-1α−/− and MIP-1α+/+ splenocytes. On day 6 after transfer we found a significant decrease (P = .03) in the quantitative assessment of GVHD in the liver after transfer of MIP-1α−/− splenocytes compared to MIP-1α+/+ splenocytes (Table3). We did not find that the decrease in the number of donor CD8+ T cells in the first 2 weeks after transplantation affected the inflammatory response in the lung, which was consistent with previous findings that GVHD occurs later at this site than it does in other organs. Correlating with our findings that donor T cells were not a significant source of MIP-1α in the GI tract, we found that inhibition of the T-cell production of MIP-1α had little effect on the scoring of GVHD at this site. Additionally, we found that blocking T-cell production of MIP-1α had no effect on the severity of GVHD in the GI tract, liver, or lung in class II mismatched (bm12) recipients (Table 3).
T-cell expression and production of chemokines in vitro
To evaluate if the presence of antigens in local tissues could trigger T-cell expression and production of chemokines, we generated H2Kbm1-restricted T-cell lines. Initially we confirmed that the T-cell lines we isolated were antigen-specific and MHC-restricted (data not shown). We found that the expression of chemokines was dependent on the presence of antigens by antigen-presenting cells (APCs) (Figure 5). In the presence of antigens, there was a 40-fold increased expression of MIP-1α and an 8- and 4-fold increase in MIP-1β and RANTES (regulated on activation normal T expressed and secreted), respectively. We confirmed that the increased expression correlated with the enhanced production of protein for MIP-1α by ELISA (Table 4). Chemokine expression or production was not found in the absence of antigens or when using nonsyngeneic APCs.
Chemokine expression by GVHD-inducing T cells.
T cells from bm1 donors were transferred into irradiated MIP-1α−/− recipients. Ten days after transfer, the recipient mice were killed, and the splenoctyes were removed, selected for CD8+ cells, and stimulated twice in vitro with irradiated (2500 cGy) MIP-1α−/− splenocytes. The isolated T cells were then incubated with bone marrow–derived macrophages from MIP-1α−/− B6 or bm1 mice. The supernatants were harvested after 24 hours, and RNA was prepared from the cells as indicated. Lane 1: Macrophages from MIP-1α−/− mice; Lane 2: macrophages from bm1 mice; Lane 3: T cells incubated with syngeneic bm1 macrophages; and Lane 4: T cells incubated with class I disparate MIP-1α−/− B6 macrophages. Enhanced expression is found for RANTES, MIP-1β, and MIP-1α in the presence of an antigen (compare expressions in lane 3 vs lane 4).
Chemokine expression by GVHD-inducing T cells.
T cells from bm1 donors were transferred into irradiated MIP-1α−/− recipients. Ten days after transfer, the recipient mice were killed, and the splenoctyes were removed, selected for CD8+ cells, and stimulated twice in vitro with irradiated (2500 cGy) MIP-1α−/− splenocytes. The isolated T cells were then incubated with bone marrow–derived macrophages from MIP-1α−/− B6 or bm1 mice. The supernatants were harvested after 24 hours, and RNA was prepared from the cells as indicated. Lane 1: Macrophages from MIP-1α−/− mice; Lane 2: macrophages from bm1 mice; Lane 3: T cells incubated with syngeneic bm1 macrophages; and Lane 4: T cells incubated with class I disparate MIP-1α−/− B6 macrophages. Enhanced expression is found for RANTES, MIP-1β, and MIP-1α in the presence of an antigen (compare expressions in lane 3 vs lane 4).
Discussion
GVHD is the limiting factor in the widespread application of allogeneic stem cell transplantation. Despite the presence of class I MHC molecules in all organs, GVHD typically involves a limited number of target organs. The reason(s) for the occurrence of GVHD in a limited tissue distribution is not well understood. We have focused on the role of MIP-1α, a C-C chemokine, in the recruitment of alloreactive lymphocytes to these sites. Here we show a marked increase in production and expression of MIP-1α after allogeneic T-cell transfer compared to syngeneic T-cell transfer. We show that the expression of MIP-1α in the liver, lung, and spleen is dependent on expression by the transferred T cells. Finally, using eGFP transgenic mice with an inactivated gene for MIP-1α, we demonstrate that recruitment of donor CD8+ T cells to the liver and lung is decreased significantly when T cells that do not produce MIP-1α are transferred and that this correlated with decreased inflammation in the liver secondary to GVHD. Interestingly, recruitment of CD4+ T cells was statistically increased after the transfer of MIP-1α−/− splenocytes compared to wild-type splenocytes in bm12 recipients.
Our data suggest that the expression of MIP-1α by donor cells is critical to the expression of this chemokine in GVHD target organs. Using the parent B6 into a B6 X DBA/2 F1 GVHD model, Murai et al39 showed that MIP-1α and its receptor CCR5 were important in mediating inflammation in the liver. Using immunohistochemistry they demonstrated that MIP-1α was produced by a number of different cell types in the liver. Our findings do not dispute the fact that in GVHD target organs, MIP-1α is made by different types of cells including both those of host and donor origin. The previous model, however, did not allow investigators to determine whether production of MIP-1α by any of these specific populations was critical to the pathogenesis of GVHD. Our data suggest that T-cell production of MIP-1α is critical to the initiation of inflammation in specific GVHD target organs. Recently we have begun to evaluate this further by transferring bm1 splenocytes into MIP-1α−/−B6 recipients. In this model, in which only donor cells make MIP-1α, a substantial concentration of MIP-1α was generated in the liver and spleen (J.S.S., unpublished data, January 2000).
Thus, we believe that our data are consistent with the role of T-cell production MIP-1α in the initiation of inflammation and that T-cell production of MIP-1α contributed significantly to the overall production of this chemokine in the liver, lung, and spleen. At other sites, such as the GI tract, our data suggest that the expression of MIP-1α was initially dependent on the transfer of allogeneic T cells, as we found a significant decrease in the concentration of MIP-1α after the transfer of syngeneic compared to allogeneic splenocytes. In the GI tract, however, the majority of the production of MIP-1α was not affected by the transfer of T cells unable to make this chemokine.
By producing MIP-1α−/− mice transgenic for eGFP, we were able to evaluate the recruitment of CD8+ alloreactive cells as a function of the elaboration of MIP-1α. We have shown that the number of transferred cells in the spleen at day 1 is similar in bm1 recipients of wild-type and MIP-1α−/− splenocytes. This strongly suggests that the decreased incidence of GVHD found using splenocytes from MIP-1α−/− mice in bm1 recipients was not due to deficient T-cell engraftment. By day 2 we found a 1.6-fold decrease in the number of eGFP+ CD8+ T cells in the spleen in bm1 recipients after transfer of MIP-1α−/− splenocytes compared to MIP-1α+/+ splenocytes. Interestingly, this correlated with the earliest expression of MIP-1α, which was found at day 2 in the spleen (data not shown). The total number of donor cells was not different at day 2, as there was a 2-fold increase in the number of CD4+ T cells in the spleen in bm1 recipients of MIP-1α−/− splenocytes. This implies that the recruitment of CD4+ T cells was influenced by other chemokines that were up-regulated in the absence of MIP-1α.
Previously we had shown a marked difference in survival of bm1 recipients after the transfer of MIP-1α−/− splenocytes compared to wild-type splenocytes. There was no difference in survival in bm12 recipients in this model. There are several possibilities for this finding. One possibility is the fact that MIP-1α is not expressed in the setting of a class II mismatch and thus does not play a role in the occurrence of GVHD. Alternatively, MIP-1α may be expressed in bm12 recipients, but it is not required for activation or recruitment of CD4+ T cells. Our data here are consistent with the second hypothesis. Although the levels of MIP-1α were less in bm12 recipients compared to bm1 recipients, a significant increase in the production of MIP-1α was shown in bm12 recipients after the transfer of allogeneic splenocytes compared to syngeneic splenocytes. This would refute the possibility that MIP-1α was not produced in the setting of a class II MHC mismatch. Additionally, despite a significant decrease in the expression of MIP-1α in the spleen of bm1 recipients early after splenocyte transfer from MIP-1α−/− donors, a greater number of eGFP+ CD4+ donor cells were found here, which strongly suggests, as has been previously reported,46 that the concentration of MIP-1α found in these models had little effect on the recruitment of CD4+ T cells. Finally, we show a statistically significant increase in the number of donor CD4+ T cells after the transfer of MIP-1α−/− as compared to MIP-1α+/+splenocytes in the liver and lung in bm12 recipients.
We found a significant decrease in the number of donor CD8+T cells in the lung after the transfer of MIP-1α−/−splenocytes compared to wild-type splenocytes in bm1 recipients. As the lung has not been considered a standard site for GVHD in humans, this finding and its relevance to GVHD may be questioned. However, recently a number of investigators have shown that alloreactive T cells contribute significantly to pathology in the lung in murine GVHD models. Both Cooke et al47 and Shankar et al48 showed that significant lung inflammation occurred only after the transplantation of completely mismatched or haploidentical T cells into recipient mice, and this correlated with an influx of donor T cells into the lung 3-6 weeks after transfer. Using a CD45-specific alloreactive T-cell clone, Clark et al49demonstrated significant pulmonary vasculitis and pneumonitis in CD45-disparate host mice after intravenous infusion of the clone. Members of our group have shown that the early pneumonitis which occurs after administration of cyclophosphamide and total body irradiation is critically dependent on the infusion of alloreactive T cells, which enhance cytokine release and macrophage and T-cell recruitment to the lung.50 We have previously shown that after receiving 500 cGy of irradiation, pulmonary involvement with GVHD is a substantial cause of late death after the transfer of C57BL/6 splenocytes into bm1 recipients.38 Finally, we have preliminary data using CFSE-labeled lymphocytes which show that both MIP-1α−/−and B6 lymphocytes proliferate significantly on day 5 after splenocyte transfer in the lung of bm1 recipients (J.S.S., unpublished data, April 2000). This work strongly supports the fact that in mice, the lung is a site for inflammation induced by alloreactive T cells and that blockade of MIP-1α diminishes the accumulation of alloreactive T cells. As T-cell–induced damage in the lung typically occurs more than 3 weeks after transplantation, the early death found in bm1 recipients after the transfer of splenocytes from C57BL/6 donors limited our ability to show a significant difference in the inflammatory response induced by MIP-1α−/− or MIP-1α+/+ T cells in the lung.
The significant decrease observed by us in the number of donor CD8+ T cells in the liver and lung of bm1 recipients after transfer of MIP-1α−/− splenocytes compared to wild-type splenocytes may be due to 2 different effects of MIP-1α. Previously, Taub et al51 had shown that MIP-1α could enhance the proliferation of allospecific T cells in vitro. This process was enhanced by the presence of CD80 on the APCs. Thus, the production of MIP-1α by alloreactive lymphocytes may augment, in an autocrine manner, the proliferative activity of those lymphocytes. Additionally, MIP-1α has been shown by us52 and others23,25,26,46 53 to affect T-cell trafficking to specific sites of inflammation. Conceivably, the effects of MIP-1α on T-cell trafficking and proliferation may act together in an additive or synergistic manner to magnify the differences in the number of donor CD8+ T cells in the liver and lung in bm1 recipients after transfer of MIP-1α−/− and wild-type splenocytes. We are currently investigating this.
We found that MIP-1α was preferentially expressed after the transfer of allogeneic splenocytes in all organs except the kidney. Here we found that MIP-1α was expressed after syngeneic T-cell transfer, which suggests that the conditioning regimen affected the expression of MIP-1α. Interestingly, we also found that 15%-20% of the cells isolated from the kidney on day 6 are donor CD8+ T cells and that this correlated with significant inflammation found here (J.S.S and B.R.B, unpublished data, November 1999). Thus the production of MIP-1α in specific organs after total body irradiation may play a role in the recruitment of effector cells and may be related to organ damage, which is independent of that produced during GVHD. This would suggest that blocking the function of MIP-1α might also play a role in decreasing the complications associated with the administration of conditioning agents for stem cell transplantation such as total body irradiation.
From this and our previous work we suggest a model for the early events that is associated with GVHD. Previous investigators have shown that memory T cells circulate at very low numbers in the periphery.54 These donor T cells could sample alloantigens in the GI tract, liver, skin, and spleen. We and others have shown that these T cells can elaborate specific chemokines after an encounter with antigens.55-57 This interaction would lead to an increase in the expression of specific chemokines by T cells, which would recruit T cells as well as donor APCs to the site. The effects of chemotherapy and irradiation could influence this process by enhancing local chemokine expression in either a direct or indirect manner in other sites such as the GI tract. The overall effect would recruit a critical threshold of T cells, which upon activation by host APCs would lead to the effector arm of GVHD.58 Our previous work and the work here suggest that blocking the expression of specific chemokines (eg, MIP-1α) or the receptors that bind them (eg, CCR5) after allogeneic stem cell transplantation may offer a new avenue for decreasing the occurrence of GVHD and that this effect may be tissue dependent.
Acknowledgments
The authors thank Dr Jun-ichi Miyazaki for generously supplying the transgene for the production of eGFP+ mice and Drs Jeffrey Frelinger and Roland Tisch for critical reading of the manuscript.
Supported by grants AI 34495 and JL 55209 (B.R.B.) and CA 67715 (J.S.S.) from the National Institutes of Health, Bethesda, MD.
Submitted January 13, 2000; accepted July 7, 2000.
The publication costs of this article were defrayed in part by page charge payment. Therefore, and solely to indicate this fact, this article is hereby marked “advertisement” in accordance with 18 U.S.C. section 1734.
References
Author notes
Jonathan S. Serody, Lineberger Comprehensive Cancer Center, Campus Box 7295, University of North Carolina at Chapel Hill, Chapel Hill, NC 27599-7295; e-mail: serody@med.unc.edu.