To the editor:
Congenital combined vitamin K–dependent coagulation factors deficiency (VKCFD) is a rare autosomal recessive bleeding disorder.1 Patients with VKCFD have decreased activity in multiple vitamin K–dependent coagulation factors due to genetic mutations that limit the ability of the proteins to be carboxylated in a vitamin K–dependent manner. Since the discovery of the 2 vitamin K cycle enzymes GGCX (γ-glutamyl carboxylase)2 and VKOR (vitamin K epoxide reductase),3,4 >30 GGCX mutations and 1 VKOR mutation have been identified from VKCFD patients.5
The first clinical case of VKCFD was reported in 1966.6 The activity of the patient’s vitamin K–dependent coagulation factors (factors II, VII, IX, and X) was undetectable. High doses of vitamin K administration (15 mg/d) partially corrected the patient’s clotting activity.6 Thirteen years later, this patient’s clotting factors were reevaluated with immunologic assays,7 which suggested that abnormal forms of the clotting factors were present. As these studies were carried out before the identification of any of the enzymes in the vitamin K cycle, the molecular mechanism of the patient’s clinical phenotypes remained unclear.
Here, we examined the genotype of the first reported VKCFD patient via exome sequencing. Notably, 2 potential deleterious mutations were identified in the GGCX gene (supplemental Table 1, available on the Blood Web site). One of the mutations (c.1657delA) had not previously been described. This mutation encodes for a frameshift mutant (caused by the deletion of an adenine base in exon 12) that prematurely terminates the GGCX translation at residue I553 with 7 additional amino acid residues (I553*; Figure 1E). The other mutation (c.1889-6G>A) is an intronic mutation in intron 13, near the splicing-acceptor site at the intron-exon conjunction of intron 13 and exon 14. Sequencing results of exon 12 to 14 from the patient’s genomic DNA showed that these 2 mutations are compound heterozygous mutations and are thus located on 2 different alleles.
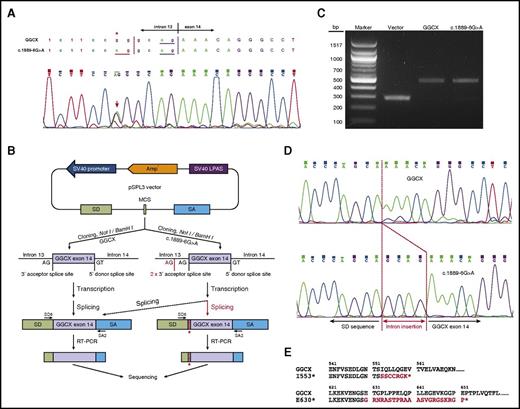
The intronic mutation c.1889-6G>A affects GGCX splicing. (A) Chromatograms of the sequencing results of the patient’s genomic DNA near the junction of intron 13 and exon 14. A heterozygous mutation of c.1889-6G>A was identified; it is indicated by an asterisk and an arrow. The splicing-acceptor site (AG) of the wild-type and the mutant sequences is underlined. (B) Schematic diagram of the minigene splicing assay. Polymerase chain reaction (PCR) fragments of exon 14 flanked by intronic sequences were cloned into a pSPL3 minigene splicing assay vector. These constructs were transfected into HEK293 cells, and the splicing products were amplified by reverse transcription polymerase chain reaction (RT-PCR) for sequencing analysis. (C) DNA electrophoresis of the splicing products from the empty vector, the wild-type GGCX, and the c.1889-6G>A mutant. (D) Chromatograms of the sequencing results of the splicing products of the wild-type GGCX and the c.1889-6G>A mutant. Intronic insertion sequences are indicated in red. (E) Amino acid sequences of the C-terminus of the 2 identified GGCX truncation mutants. Additional sequences added to the C-terminus of GGCX due to reading frame shift are highlighted in red. bp, base pair; LPAS, late poly(A) signal; SA, splice acceptor; SD, splice donor; SV40, simian virus 40.
The intronic mutation c.1889-6G>A affects GGCX splicing. (A) Chromatograms of the sequencing results of the patient’s genomic DNA near the junction of intron 13 and exon 14. A heterozygous mutation of c.1889-6G>A was identified; it is indicated by an asterisk and an arrow. The splicing-acceptor site (AG) of the wild-type and the mutant sequences is underlined. (B) Schematic diagram of the minigene splicing assay. Polymerase chain reaction (PCR) fragments of exon 14 flanked by intronic sequences were cloned into a pSPL3 minigene splicing assay vector. These constructs were transfected into HEK293 cells, and the splicing products were amplified by reverse transcription polymerase chain reaction (RT-PCR) for sequencing analysis. (C) DNA electrophoresis of the splicing products from the empty vector, the wild-type GGCX, and the c.1889-6G>A mutant. (D) Chromatograms of the sequencing results of the splicing products of the wild-type GGCX and the c.1889-6G>A mutant. Intronic insertion sequences are indicated in red. (E) Amino acid sequences of the C-terminus of the 2 identified GGCX truncation mutants. Additional sequences added to the C-terminus of GGCX due to reading frame shift are highlighted in red. bp, base pair; LPAS, late poly(A) signal; SA, splice acceptor; SD, splice donor; SV40, simian virus 40.
The GGCX intronic mutation c.1889-6G>A is 4 bases upstream of exon 14 (Figure 1A). This mutation introduces an extra splicing-acceptor site (AG), which might affect the splicing of GGCX’s pre–messenger RNA (mRNA). To test the effect of this presumptive pre-mRNA splicing mutation, wild-type and mutant alleles of the genomic DNA fragments containing exon 14 along with its upstream and downstream intronic sequences were cloned into the exon-trapping vector pSPL3 for minigene-splicing assays8 (supplemental Methods; Figure 1B). As expected, processing of the blank vector and the GGCX constructs resulted in ∼300-bp and ∼500-bp PCR fragments, respectively (Figure 1C). Sequencing results of the PCR products showed that the spliced mRNA from the c.1889-6G>A mutant retained 4 bases (GCAG from intron 13) in front of exon 14 of GGCX (Figure 1D). This result indicates that the intronic c.1889-6G>A mutation introduced a functional splicing-acceptor site that affects GGCX pre-mRNA splicing. Importantly, this alternative splicing leads to a reading frame shift at amino acid residue E630, thus introducing a premature stop codon with 22 additional amino acid residues (E630*; Figure 1E).
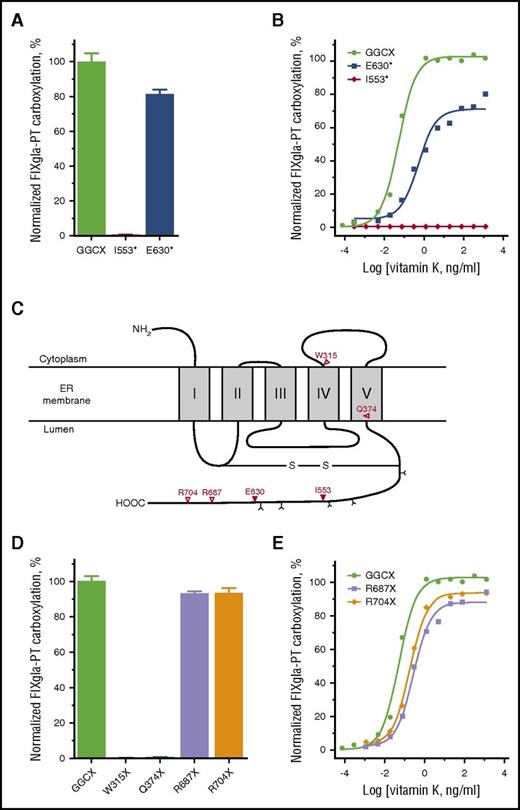
To gain insights on how these GGCX mutations impacted patient clinical phenotypes, we characterized these mutations using our recently established CRISPR-Cas9–mediated GGCX knockout cell-based assay.9 We transiently expressed each of the mutants in the GGCX-deficient reporter cells, cultured the cells with 5 µg/mL vitamin K, and measured how efficiently the reporter protein was carboxylated. Figure 2A shows that the I553* mutant abolished reporter-protein carboxylation; however, the E630* mutant retained ∼80% activity. The E630* mutant’s substantial residual activity cannot explain the patient’s clinical data, in which clotting activity of the vitamin K–dependent coagulation factors was decreased to undetectable levels.6
Characterization of the GGCX truncation mutations identified from patients/animals with bleeding disorders by cell-based activity assay. (A) Carboxylation activity of wild-type GGCX and the I553* and E630* mutants measured by GGCX-deficient cell-based assay with 5 µg/mL vitamin K. (B) Carboxylation of the reporter protein by wild-type GGCX and the I553* and E630* mutants in GGCX-deficient HEK293 reporter cells in response to increasing concentrations of vitamin K. (C) Proposed membrane topology of GGCX according to Tie et al.18 GGCX truncation mutations and the corresponding amino acid residues are indicated (∇). N-linked glycosylation sites are indicated by Y. (D) Carboxylation activity of wild-type GGCX and the W315X, Q374X, R687X, and R704X mutants measured by GGCX-deficient cell-based assay with 5 µg/mL vitamin K. (E) Carboxylation of the reporter protein by wild-type GGCX and the R687X and R704X mutants in GGCX-deficient HEK293 reporter cells in response to increasing concentrations of vitamin K.
Characterization of the GGCX truncation mutations identified from patients/animals with bleeding disorders by cell-based activity assay. (A) Carboxylation activity of wild-type GGCX and the I553* and E630* mutants measured by GGCX-deficient cell-based assay with 5 µg/mL vitamin K. (B) Carboxylation of the reporter protein by wild-type GGCX and the I553* and E630* mutants in GGCX-deficient HEK293 reporter cells in response to increasing concentrations of vitamin K. (C) Proposed membrane topology of GGCX according to Tie et al.18 GGCX truncation mutations and the corresponding amino acid residues are indicated (∇). N-linked glycosylation sites are indicated by Y. (D) Carboxylation activity of wild-type GGCX and the W315X, Q374X, R687X, and R704X mutants measured by GGCX-deficient cell-based assay with 5 µg/mL vitamin K. (E) Carboxylation of the reporter protein by wild-type GGCX and the R687X and R704X mutants in GGCX-deficient HEK293 reporter cells in response to increasing concentrations of vitamin K.
To better understand the details of how these mutations affect vitamin K–dependent carboxylation, we cultured the GGCX-deficient reporter cells expressing the GGCX mutant proteins with increasing concentrations of vitamin K. Figure 2B shows that the I553* mutant was unable to carboxylate the reporter protein at any of the vitamin K concentrations tested. At the vitamin K concentration where the wild-type GGCX is fully functional, the E630* mutant only exhibited ∼30% activity. However, higher concentrations of vitamin K ameliorated reporter protein carboxylation up to 80%. This result is consistent with the patient’s clinical phenotype, as the initial coagulation activity deficiency was partially corrected by the administration of high doses of vitamin K. These results together suggest that with a null allele of GGCX (ie, the I553* mutation), the E630* mutation on the other allele is the causative mutation for the patient’s clinical phenotype.
In addition to the GGCX truncation mutations identified in this study, 4 other truncation mutations in the GGCX gene have been identified in patients/animals with bleeding disorders. These mutations result in terminations of GGCX at residues W315 (W315X),5 Q374 (Q374X),10,11 R687 (R687X),12 and R704 (R704X)13 (Figure 2C). Because truncation of GGCX at residue I553 results in an inactive enzyme, we reasoned that termination of GGCX translation further toward the N-terminus (W315X and Q374X) would abolish GGCX activity. On the other hand, given that the GGCX molecule truncated at residue E630 retained ∼80% activity, we suspected that termination of GGCX translation further toward the C-terminus (R687X and R704X) would have only minor effects on GGCX activity. This hypothesis was confirmed by the cell-based GGCX activity assay, which shows that the W315X and Q374X mutants lacked GGCX activity, whereas the R687X and R704X mutants retained >95% activity (Figure 2D). The R687X and R704X mutants did require, however, 5.3- and 3.6-fold higher vitamin K concentrations, respectively, in order to reach half-maximal activity (Figure 2E). Together, these results suggest that in order for GGCX to be active, it must have at least 630 residues and that truncation of GGCX from its C-terminus can impact vitamin K binding, which is consistent with previous observations.14
GGCX is a glycoprotein with 5 functional glycosylation sites located at the C-terminus15 (Figure 2C). Truncation of GGCX at residue E630 not only removes the C-terminal sequences but also leaves the last 2 functional glycosylation sites (N605 and N627) at a position unfavorable for glycosylation.16,17 Consistent with our previous observation,15 a GGCX molecule with these 2 sites mutated (N605/627Q) migrates faster than the wild-type enzyme in sodium dodecyl sulfate–polyacrylamide gel electrophoresis (supplemental Figure 1A). Our cell-based activity assay results showed that this mutant protein exhibits ∼80% activity (supplemental Figure 1B) and required approximately twofold higher vitamin K concentration to reach half-maximal activity (supplemental Figure 1C). These results suggest that the last 2 glycosylation sites can have effects (albeit minor) on GGCX function.
GGCX has 5 transmembrane domains located at the N-terminus (residues 1-381), whereas its large C-terminus (residues 382-758) resides in the endoplasmic reticulum (ER) lumen (Figure 2C).18 Partial truncation of the C-terminal sequences of GGCX was expected thus to have only minor effects on ER targeting. Fluorescence confocal microscopy results showed that, as expected, GGCX and the E630* mutant clearly localized to the ER membrane (supplemental Figure 2). Under the same conditions, however, the I553* mutant showed significantly less fluorescent signal (indicating less intact protein expression). In addition, more fluorescent signal was observed outside the ER, suggesting mislocalization and probable misfolding of the mutant protein. The possible misfolding of the I553* mutant was further confirmed by the immunoblotting results (supplemental Figure 3), showing more protein bands for the mutant proteins (especially the I553* mutant) than the wild-type enzyme.
In summary, we identified 2 causative GGCX mutations from the first reported clinical case of a VKCFD patient. Functional characterization of these 2 mutations suggests that truncation of GGCX from its C-terminus affects not only substrate binding but also protein stability.
The online version of this article contains a data supplement.
Authorship
Acknowledgments: The authors thank the patient for her participation in this study. They also thank David Straight for helpful discussions and Heath Sledge for editing the manuscript.
This work was supported by a grant from the National Heart, Lung, and Blood Institute, National Institutes of Health (HL131690) (J.-K.T. and D.W.S.).
Contribution: D.-Y.J. created all the constructs and performed all the cell-based assays and GGCX mutation characterization; B.O.I. performed genomic DNA extraction, exome sequencing sample preparation, and data analysis; D.W.S. oversaw the whole project; and J.-K.T. designed the study, analyzed the data, and wrote the manuscript.
Conflict-of-interest disclosure: The authors declare no competing financial interests.
Correspondence: Jian-Ke Tie, Department of Biology, University of North Carolina at Chapel Hill, Chapel Hill, NC 27599; e-mail: jktie@e-mail.unc.edu.