Abstract
High-titer, HIV-1–based lentiviral vector particles were found to transduce cytokine-mobilized rhesus macaque CD34+ cells and clonogenic progenitors very poorly (< 1%), reflecting the postentry restriction in rhesus cells to HIV infection. To overcome this barrier, we developed a simian immunodeficiency virus (SIV)–based vector system. A single exposure to a low concentration of amphotropic pseudotyped SIV vector particles encoding the green fluorescent protein (GFP) resulted in gene transfer into 68% ± 1% of rhesus bulk CD34+ cells and 75% ± 1% of clonogenic progenitors. Polymerase chain reaction (PCR) analysis of DNA from individual hematopoietic colonies confirmed these relative transduction efficiencies. To evaluate SIV vector–mediated stem cell gene transfer in vivo, 3 rhesus macaques underwent transplantation with transduced, autologous cytokine-mobilized peripheral blood CD34+ cells following myeloablative conditioning. Hematopoietic reconstitution was rapid, and an average of 18% ± 8% and 15% ± 7% GFP-positive granulocytes and monocytes, respectively, were observed 4 to 6 months after transplantation, consistent with the average vector copy number of 0.19 ± 0.05 in peripheral blood leukocytes as determined by real-time PCR. Vector insertion site analysis demonstrated polyclonal reconstitution with vector-containing cells. SIV vectors appear promising for evaluating gene therapy approaches in nonhuman primate models.
Introduction
The ability to transfer genes into repopulating hematopoietic stem cells ex vivo and to achieve regulated expression in specific lineages following hematopoietic reconstitution would create many therapeutic opportunities.1 Although the initial use of murine oncoretroviral vectors to transfer genes into primitive murine hematopoietic cells was reported 20 years ago,2 translation of this approach to clinical application has been slow and has required considerable effort. Despite progress being achieved in the murine system with correction of single gene defects in murine models of human immunodeficiencies3-6 and chronic granulomatous disease,7,8 the much lower efficiency of gene transfer into human stem cells1 has hampered success. The necessity for high-level oncoretroviral vector gene transfer to achieve therapeutic benefit, however, was circumvented in 2 recent clinical trials designed to cure severe combined immunodeficiency due to a deficiency of the common γ-chain of the lymphoid cytokine receptor9 or adenosine deaminase.10 In these trials, a potent selective repopulating advantage of the gene-corrected lymphoid cells resulted in therapeutically relevant numbers of functional lymphocytes.
Despite this success, 2 barriers appear to limit the ability of murine oncoretroviral vectors to achieve adequate transduction of primitive hematopoietic stem cells for treatment of other disorders in which the gene-corrected cells do not have a selective advantage. Because the human homolog of the receptor for murine ecotropic vector particles does not interact with the ecotropic envelope protein, amphotropic particles have been used in both human studies and in large animal models. The amphotropic receptor, however, is expressed at low levels on human stem cells.11 Alternative envelopes have been tested, such as those derived from the gibbon ape leukemia virus,12 feline endogenous virus (RD114),13 or feline leukemia virus type C,14 the receptors for which are expressed at higher levels on primitive hematopoietic cell populations. However, large animal studies have failed to clearly identify a superior pseudotype that consistently yields high-level stem cell gene transfer. These data suggest that a second barrier—namely, the requirement for mitosis to allow integration of the oncoretroviral vector genome15 —along with the relative instability of the preintegration complex16 may be the main limitations of oncoretroviral-mediated stem cell gene transfer. In an effort to induce stem cell cycling, cytokines such as stem cell factor (SCF), Flt-3 ligand (Flt3-L), and megakaryocyte growth and development factor (MGDF)17,18 are added to culture medium and a fragment of fibronectin is used to colocalize vector particles and target cells,19 leading to improved stem cell transduction efficiency in large animal models.12,20,21 Nonetheless, there remains significant variability among animals, with many animals having low marking and only occasional animals having proportions of genetically modified blood cells in the 10% to 20% range.
Lentiviral vectors such as human immunodeficiency virus (HIV) have inherent advantages in transducing cells without cell division, because their preintegration complex enters the nucleus without cellular mitosis and is relatively stable compared with that of oncoretroviral vectors.16,22 Fully quiescent, G0 cells are poorly transduced by lentiviral vectors23,24 ; progression to at least the G1b phase cell cycle is necessary for reverse transcription to occur in T cells.23 Under equivalent conditions of cytokine stimulation and use of a fibronectin fragment to colocalize vector particles and target cells, lentiviral vectors have proved superior to oncoretroviral vectors at transducing human primitive hematopoietic cells, including those that are capable of establishing hematopoiesis in immunodeficient mice.25-27
Despite the theoretical advantages and the encouraging data from in vitro culture systems and the immunodeficient mouse assays, the use of lentiviral vectors based on HIV-1 to transduce reconstituting stem cells in nonhuman primate models has given disappointing results.28-32 The proportion of genetically modified cells in peripheral blood of transplant recipients was generally less than 5% long term, with only one animal having marking in the myeloid lineages of 8%.32 Our experiments were designed to evaluate the hypothesis that this low level of transduction of nonhuman primate stem cells reflects the well-recognized postentry block to transduction with HIV-1 virus or vectors that has been observed with other cell types of Old World monkey species.33-36 We found that simian immunodeficiency virus (SIV) vector particles were more efficient at transducing rhesus CD34+ cells and the progenitors within that population than were HIV vector particles. Importantly, SIV vector particles efficiently transduced rhesus hematopoietic repopulating stem cells as assessed in the autologous transplantation model.
Materials and methods
Assembly of the SIV vector system
The SIV vector and helper plasmids were derived from corresponding components of the HIV-1–based system, SJ1, developed in our laboratory.27 The HIV-1–based transfer vector, pCL20c MSCV-GFP, was derived by replacing the 321-bp NotI-KpnI fragment between the GFP coding sequences and the 3′ long-terminal repeat (3′LTR) of pCL10.1 MSCVc GFP with oligos 5′-GGCCGCATCGATGCCGTATACGGTAC-3′ and 5′CGTATACGGCATCGATGC-3′. The SIV transfer vector, pCL20cSLFR MSCV-GFP, was derived from pCL20c MSCV-GFP by replacing the HIV-1 sequences with corresponding segments of the plasmid, SIVmac1A11,37 a nonpathogenic lentivirus that was obtained from the National AIDS Reagent Repository (NARR, Washington, DC). The open reading frames for each of the accessory proteins were eliminated from the vector. The gag/pol expression plasmid, pCAG-SIVgprre, was assembled by sequential replacement of the gag/pol and rev responsive element (RRE) sequences from pCAGkGP1R as follows: (1) ligation of the BsrB1IEcl136II SIVmac1A11 gag/pol fragment into a BstEII (blunt) EcoNI (blunt) fragment from pCAG-kGP1R designated pCAG-SIVgp and (2) ligation of a PmlI-NheI SIVmac1A11 RRE fragment into an MfeI (blunt)–NheI fragment from pCAG-SIVgp. The rev/tat expression plasmid pCAG4-RTR-SIV was made by a 3-way ligation of the following fragments: (1) the rev/tat 5′ exon generated by polymerase chain reaction (PCR) amplification with primers 5′-CGGAATTCTCCTCGCTTGCTAACTGCACTT-3′ and 5′-GGCATCGATACTTACTTGTTTGATGCAGAAGATGT-3′ using the plasmid SIVmac1A11 as template followed by digestion with EcoRI and ClaI; (2) a ClaI-SalI fragment from SIVmac1A11 that contains the RRE and the 3′ rev/tat exon; and (3) an EcoRI-XhoI fragment from pCAG4-RTR2.
Vector production and characterization
Conditioned medium containing vector particles was produced as described27 with minor modifications. Briefly, 4 × 106 293T cells were seeded onto 10 cm dishes and transfected with a total of 20 μg DNA composed as follows: 6 μg pCAGkGP1R (or pCAG-SIVgprre), 2 μg pCAG4-RTR2 (or pCAG4-RTR-SIV), 2 μg pCAG4-Amph (or pCAG-VSV-G), and 10 μg pCL20c MSCV-GFP (or pCL20cSLFR MSCV-GFP). Eighteen hours after transfection, the 293T cells were washed with phosphate-buffered saline (PBS) twice and then cultured for an additional 24 hours in Dulbecco modified eagle medium (DMEM) supplemented with 10% fetal bovine serum (FBS) and containing 50 U/mL penicillin G and 50 μg/mL streptomycin. Conditioned medium containing vector particles was harvested after 24 hours, cleared by centrifugation, filtered, snap frozen in aliquots, and stored at -80° C. Vector preparations were tested for replication competent lentivirus (RCL) with a serial passage assay in which 5 × 104 HeLa cells were exposed to an equivalent amount of SIV or HIV vector particles and then grown for 2 to 3 weeks with splitting of cultures every 3 days. Conditioned media from the passaged cultures were then tested for the presence of SIV p27 or HIV p24 using commercially available enzyme-linked immunosorbent assay (ELISA) kits (Coulter, Miami, FL). No preparations tested positive. SIV and HIV vectors encoding GFP were titered by transducing HeLa cells, as described below, with serial dilutions of the vector preparations. Three to 4 days following transduction, gene transfer to cells was determined using flow cytometry (fluorescence-activated cell sorting [FACS]) to enumerate the proportion of GFP-positive cells. Titers were routinely determined from a volume of vector preparation that yielded linear, dose-dependent transduction of target cells with a level not in excess of 20%.
Transduction of cell lines and hematopoietic cells
HeLa cells, owl monkey kidney (OMK; CRL-1556) cells, and rhesus monkey kidney (LLC-MK2; CCL-7.1) cells were purchased from the American Type Culture Collection (Manassas, VA) and cultured using DMEM supplemented with FBS and antibiotics as noted above. A total of 5 × 104 cells per well were seeded into 6-well plates and, after 24 hours of culture, transduced with vector particles in a total volume of 2 mL in the presence of 7.5 μg/mL Polybrene. After 24 hours, the transduction medium was replaced with growth medium and culture was continued for an additional 2 to 5 days before FACS analysis for GFP expression.
Healthy human volunteers received granulocyte colony-stimulating factor (G-CSF) after which peripheral blood mononuclear cells were collected by apheresis and CD34+ cells purified.27 Informed consent was obtained for participation in the protocol approved by the Institutional Review Board of St Jude Children's Research Hospital (SJCRH). Purified human CD34+ cells were stored frozen prior to use. Rhesus monkeys were given G-CSF 10 μg/kg and SCF 200 μg/kg for 5 doses, and peripheral blood cells were collected by leukapheresis and CD34+ cells purified20 (see the next section). Rhesus CD34+ cells were shipped overnight from the National Institutes of Health (NIH) to SJCRH either frozen or in DMEM with 10% FBS containing 100 ng/mL of each of the following cytokines: SCF, Flt3-L, and MGDF. CD34+ cells were transduced on RetroNectin- (20 μg/cm2) coated plates in DMEM supplemented with 10% FBS and antibiotics and containing SCF, Flt3-L, and MGDF (100 ng/mL each) and 4 μg/mL protamine sulfate. The cell number, total volume of medium, and vector amounts were as follows: 12-well plate: 2 × 105 cells, 1 mL, and 2.4 × 105 transducing unit (TU); 6-well plate: 5 × 105 cells, 2.5 mL, and 6 × 105 TU. After culture for 24 hours, the vector-containing medium was replaced and culture continued for 5 days with medium changed every other day after which CD34+ cells were subjected to FACS or seeded into Methocult GF H4434 (1000 cells per milliliter) for analysis of progenitor-derived colonies 14 days later. GFP-positive colonies were scored by fluorescent microscopy. DNA was prepared from hematopoietic colonies as described.38 PCR analysis for the presence of GFP sequences was performed using the HotStarTaq Master Mix Kit (Qiagen, Valencia, CA) according to the manufacturer's protocol; 10 μL DNA solution was used as template. Primers were 5′-AGCGGATCGCTCACAACCAG-3′ and 5′-GCCGGTGGTGCAGATGAACT-3′ for detection of the HIV-1 or SIV vector provirus band of 569 bp. As a control for DNA integrity, a 232-bp band was generated with β-actin primers. The PCR cycle started with an initial activation step of 95° C for 15 minutes, followed by 40 cycles of a 3-step amplification cycle (94° C, 1 minute; 60° C, 1 minute; 72° C, 1 minute), and ending with a final extension at 72° C for 10 minutes. PCR products were visualized with ethidium bromide after electrophoresis in 2% agarose.
Transduction of rhesus repopulating hematopoietic stem cells
Young rhesus macaques (Macaca mulatta) were housed and handled in accordance with guidelines set by the Committee on Care and Use of Laboratory Animals of the Institute of Laboratory Animal Resources, National Research Council, and the protocol was approved by the Animal Care and Use Committee of the National Heart, Lung, and Blood Institute.20,38 The animals received SCF (200 mg/kg; Amgen, Thousand Oaks, CA) and G-CSF (10 mg/kg; Amgen) as subcutaneous injections for 3 days and twice daily on day 4. Mobilized peripheral blood cells were collected by apheresis on day 5 as described.39 Mononuclear cells were isolated using density gradient centrifugation over lymphocyte separation medium (Organon Teknika, Durham, NC), and enrichment of CD34+ cells was performed using a biotinylated anti-CD34 monoclonal antibody (12.8) and immunoselection using streptavidin-coated microbeads. Progenitor enrichment was confirmed by colony-forming unit (CFU) assays performed before and after column purification. CD34+ cells were prestimulated for 36 hours with SCF, Flt3-L, and MGDF (100 ng/mL) on RetroNectin-coated plates. The initial cell density was 1.0 × 105/mL to 2.0 × 105/mL, and the vector concentration was 2.4 × 105 TU/mL, resulting in a multiplicity of infection (MOI) of 1.2 to 2.4. The cells were exposed to amphotropic vector particles for 24 hours and then transferred into fresh medium containing cytokines and vector particles, and incubation was continued for another 12 hours. Prior to infusion of autologous cells, each animal received 2 doses of 500 cGy total body irradiation at a 24-hour interval. Recombinant human G-CSF (rhG-CSF) (10 mg/kg) was given beginning on day 1 after the cell infusion and continued until the total white cell count exceeded 6 × 109/L (6000/μL).
Quantitation of gene transfer following reconstitution
Forward and light scatter properties were used to gate on granulocytes, monocytes, lymphocytes, red cells, and platelets, allowing quantitation of the percentage of GFP-positive cells in these populations by FACS as previously described.40 These measurements were supplemented by use of specific monoclonal antibodies reactive with rhesus leukocytes to quantitate GFP-positive cells in immunophenotypically defined hematopoietic subsets in peripheral blood. The antibodies utilized were as follows: CD3, clone SP34-2; CD4, clone M-T477; CD8, clone RPA-T8; CD11b, clone ICRF44; CD14, clone M5E2; CD16, clone 3G8; and CD20, clone 2H7 (BD Biosciences PharMingen, Palo Alto, CA). The status of the vector genome was evaluated in total leukocyte DNA using Southern blot analysis. Purified peripheral blood DNA or DNA prepared from bone marrow progenitor-derived colonies was subjected to real-time PCR for estimation of vector copy number. The sequences of the primer/probe set for detection of the SIV provirus was as follows: forward primer, 5′-TACGGCTGAGTGAAGGCAGTAAG-3′; reverse primer, 5′-CTCCTCACGCCGTCTGGTA-3′; and the probe, 5′-6-FAM-AGGAACCAACCACGACGGAGTGCTC-TAMRA-3′. Taqman Ribosomal RNA control reagents were used to estimate the amount of DNA analyzed. A standard curve was established by analyzing triplicate aliquots of serial dilutions of DNA from a K562 clone having a single copy of the SIV vector genome (SIV-K562) for SIV provirus and rRNA sequences. Fifty nanograms or 0.2 to 0.5 μL purified DNA from an individual colony was used as templates. The real-time PCR was performed using the ABI PRISM 7700 Sequence Detector, and the data were analyzed using the Sequence Detection System software (Applied Biosystems, Foster City, CA). For each sample, the amount of SIV sequences, compared with the standard control curve, and the amount of ribosomal RNA sequences, again compared with the standard curve, were estimated, and the copy number of the sample was calculated using the following formula: copy number = (SIV [ng SIV-K562])/[(rRNA [ng SIV-K562]) × 1.5]. A coefficient of 1.5 was used in the denominator because the K562 cell line used was approximately triploid on average.
Evaluation of vector integration sites
Linear amplification mediated (LAM)–PCR analysis was performed on DNA extracted from peripheral blood (PB) leukocytes of reconstituted animals at different time points after transplantation as described before41 with the following modifications. For linear amplification of the 5′-SIV-LTR–genomic junction sites, 100 ng DNA was amplified in 6 different reactions using an SIV-LTR–specific 5′-biotinylated primer (LTR1), 5′-GCCAAGTGCTGGTGAGAGTC-3′. The digestion step was done using ApoI enzyme (New England BioLabs, Beverly, MA), and subsequently an asymmetric oligonucleotide linker cassette (5′-AATTCTCTAGTATGCTACTCGCACCGATTATCTCCGCTGTCAGT-3′ 5′-ACTGACAGCGGAGATAATCGGTGCGAGTAGCATACTAGAG-3′) was ligated to the end of ApoI-digested product. Nested PCR was done using the SIV-LTR–specific primer (LTR2) (5′-TACCTGCTAGTGCTGGAGAG-3′) and linker cassette (LC)–specific primer 1 (LC1) (5′-ACTGACAGCGGAGATAATCG-3′) for the outer exponential reaction and then LTR3 primer (5′-AGAGCGACTGACTACATAGC-3′) and LC2 primer (5′-GTGCGAGTAG CATACTAGAG-3′) for the inner reaction in a volume of 25 μL. The products were pooled together and then purified using the QIAquick PCR purification kit (Qiagen). Five microliters of the purified product were separated on a high-resolution Spreadex gel (Elchrom Scientific, Cham, Switzerland). The rest of purified product containing the mixture of bands was cloned into the TOPO TA kit vector (Invitrogen, Carlsbad, CA) to generate a library of insertion sites for sequencing. A sequence was considered as a true insertion site if it contained both the SIV-LTR sequence and the linker cassette sequence, with minimum of 90% similar identity to the National Center for Biotechnology Information (NCBI) build 33 of the human genome and with a unique best hit in the BLAST-like alignment tool (BLAT).
Results
Efficient transduction of rhesus cells by SIV-based vector particles but not by HIV-1–based vector particles
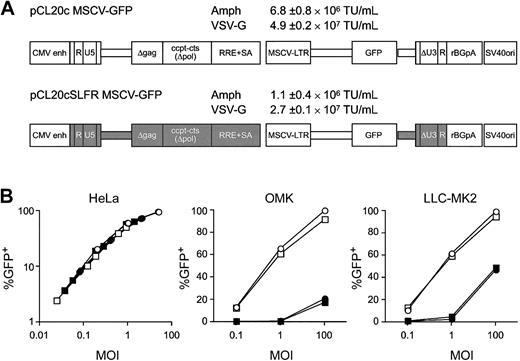
An SIV-based vector system highly analogous to our previously described HIV-1 SJ1 vector system was assembled using SIV sequences obtained from the nonpathogenic SIV macaque isolate 1A11. Both the SIV and HIV systems feature analogous self-inactivating transfer vectors containing GFP coding sequences driven by the murine stem cell virus (MSCV) enhancer/promoter (Figure 1A). Viral packaging functions are split on 3 plasmids with minimal homology to transfer vector sequences to reduce the possibility of generating RCL (see “Materials and methods”). Both vector systems generated amphotropic or vesicular stomatitis virus G protein (VSV-G) pseudotyped vector preparations with average titers more than 1 × 106 and 1 × 107 per milliliter, respectively, as titered on HeLa cells (Figure 1A). The HIV-1– and SIV-based vectors transduced human HeLa cells equivalently, whereas owl and rhesus monkey kidney cells were transduced much more efficiently with the SIV vector particles (Figure 1B). HIV vector preparations that transduced human CD34+ cells efficiently (80%) failed to transduce rhesus CD34+ cells (Figure 2A). A species-specific block to vector-mediated gene expression42 was excluded by PCR analysis of DNA from individual rhesus hematopoietic colonies that demonstrated the absence of HIV-1 vector sequences (Figure 2B). In contrast, the SIV vector particles efficiently transduced rhesus CD34+ cells and the progenitors within that population (Figure 2C-D).
Transduction of human and monkey cells with HIV-1–based or SIV-based lentiviral vectors. (A) Diagrams of the HIV-1–(pCL20c MSCV-GFP) or SIV-based (pCL20cSLFR MSCV-GFP) transfer vectors. Sequences from the SIVmac1A11 genome are indicated as solid boxes. Residual nef coding sequences upstream of a KpnI site (accession no. KO3455, nucleotide 9015) were removed from an earlier version of the HIV-1–based vector, pCL10.1 MSCVcGFP, to generate the vector designated pCL20c MSCV-GFP. The SIV-based transfer vector was constructed by replacing each element of the HIV-based vector with a corresponding fragment from the SIVmac1A11 genome; the fragment sizes of each element differ slightly. Helper plasmids encoding SIV-gag/pol or the SIV-rev/tat proteins were developed using the pCAGGS expression plasmid system as described for the HIV-1–based system, SJ1.6 The titers of amphotropic or VSV-G pseudotyped HIV-1 or SIV vectors are indicated as the means and standard errors of the means for 3 to 5 separate preparations. (B) Relative transduction of human (HeLa, left) cells, owl monkey kidney (OMK, middle), or rhesus monkey kidney (LLC-MK2, right) cells at various multiplicities of infection (MOIs). The experiments were performed in duplicate, and the averages are plotted. The vector/envelope for each titration was as follows: HIV-1/amphotropic (▪), HIV-1/VSV-G (•), SIV/amphotropic (□), or SIV/VSV-G (○).
Transduction of human and monkey cells with HIV-1–based or SIV-based lentiviral vectors. (A) Diagrams of the HIV-1–(pCL20c MSCV-GFP) or SIV-based (pCL20cSLFR MSCV-GFP) transfer vectors. Sequences from the SIVmac1A11 genome are indicated as solid boxes. Residual nef coding sequences upstream of a KpnI site (accession no. KO3455, nucleotide 9015) were removed from an earlier version of the HIV-1–based vector, pCL10.1 MSCVcGFP, to generate the vector designated pCL20c MSCV-GFP. The SIV-based transfer vector was constructed by replacing each element of the HIV-based vector with a corresponding fragment from the SIVmac1A11 genome; the fragment sizes of each element differ slightly. Helper plasmids encoding SIV-gag/pol or the SIV-rev/tat proteins were developed using the pCAGGS expression plasmid system as described for the HIV-1–based system, SJ1.6 The titers of amphotropic or VSV-G pseudotyped HIV-1 or SIV vectors are indicated as the means and standard errors of the means for 3 to 5 separate preparations. (B) Relative transduction of human (HeLa, left) cells, owl monkey kidney (OMK, middle), or rhesus monkey kidney (LLC-MK2, right) cells at various multiplicities of infection (MOIs). The experiments were performed in duplicate, and the averages are plotted. The vector/envelope for each titration was as follows: HIV-1/amphotropic (▪), HIV-1/VSV-G (•), SIV/amphotropic (□), or SIV/VSV-G (○).
Transduction of human and rhesus primitive hematopoietic cells with HIV-1– or SIV-based lentiviral vectors. (A) Transduction of cytokine-mobilized rhesus or human CD34+ cells with the HIV-1–based vector (thick line). Nontransduced cells (thin line) were used as a control in each experiment. CD34+ cells were cultured in the presence of cytokines (see “Materials and methods”) and exposed once to vector particles at an MOI of 1.2 to 2.4 for 24 hours and then cultured with a medium change every other day for an additional 5 days prior to FACS analysis for GFP expression. The percentages indicate the proportion of GFP-positive cells. (B) Transduction of rhesus (top panel) or human (bottom panel) progenitors with the HIV-1 vector as indicated above. Individual colonies were plucked from methylcellulose after culture for 2 weeks and DNA prepared for PCR analysis. A control for the integrity of the DNA was performed using primers specific for β-actin that generate a 232-bp band, whereas the primer pair for the vector fragment generated a band of 569 bp. DNA for each of the lanes were as follows: 2-3, nontransduced rhesus progenitors; 5-14, transduced rhesus progenitors; 16, H2O control; 17, DNA from 293T cells containing about 10 copies of plasmid pCL20c MSCV-GFP per cellular genome equivalent; 20-21, nontransduced human progenitors; and 23-32, transduced human progenitors. Lanes 1, 18, 19, and 33 contain marker DNA, whereas lanes 4, 15, and 22 were left blank. The upper band on the lower panel is derived from the HIV vector whereas the lower band on that panel and the band on the upper panel are derived from the β-actin gene. (C-D) Transduction of frozen (C) or fresh (D) cytokine-mobilized rhesus CD34+ cells with the HIV-1 or SIV vector. Previously frozen, cytokine-mobilized human CD34+ cells were used as a control in both experiments. In panel D, the black bars represent the percentage of GFP-positive CD34+ cells after a 24-hour exposure to vector particles and 6 days of subsequent culture, whereas the gray bars represent the percentage of hematopoietic colonies that were GFP positive after 2 weeks of culture. The results represent the mean and standard error of 2 (C) or 3 (D) experiments.
Transduction of human and rhesus primitive hematopoietic cells with HIV-1– or SIV-based lentiviral vectors. (A) Transduction of cytokine-mobilized rhesus or human CD34+ cells with the HIV-1–based vector (thick line). Nontransduced cells (thin line) were used as a control in each experiment. CD34+ cells were cultured in the presence of cytokines (see “Materials and methods”) and exposed once to vector particles at an MOI of 1.2 to 2.4 for 24 hours and then cultured with a medium change every other day for an additional 5 days prior to FACS analysis for GFP expression. The percentages indicate the proportion of GFP-positive cells. (B) Transduction of rhesus (top panel) or human (bottom panel) progenitors with the HIV-1 vector as indicated above. Individual colonies were plucked from methylcellulose after culture for 2 weeks and DNA prepared for PCR analysis. A control for the integrity of the DNA was performed using primers specific for β-actin that generate a 232-bp band, whereas the primer pair for the vector fragment generated a band of 569 bp. DNA for each of the lanes were as follows: 2-3, nontransduced rhesus progenitors; 5-14, transduced rhesus progenitors; 16, H2O control; 17, DNA from 293T cells containing about 10 copies of plasmid pCL20c MSCV-GFP per cellular genome equivalent; 20-21, nontransduced human progenitors; and 23-32, transduced human progenitors. Lanes 1, 18, 19, and 33 contain marker DNA, whereas lanes 4, 15, and 22 were left blank. The upper band on the lower panel is derived from the HIV vector whereas the lower band on that panel and the band on the upper panel are derived from the β-actin gene. (C-D) Transduction of frozen (C) or fresh (D) cytokine-mobilized rhesus CD34+ cells with the HIV-1 or SIV vector. Previously frozen, cytokine-mobilized human CD34+ cells were used as a control in both experiments. In panel D, the black bars represent the percentage of GFP-positive CD34+ cells after a 24-hour exposure to vector particles and 6 days of subsequent culture, whereas the gray bars represent the percentage of hematopoietic colonies that were GFP positive after 2 weeks of culture. The results represent the mean and standard error of 2 (C) or 3 (D) experiments.
Reconstitution of rhesus macaques with cytokine-mobilized peripheral blood stem cells transduced by SIV-based vector particles
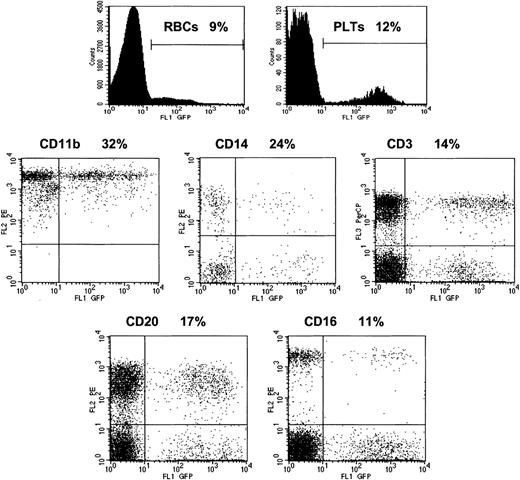
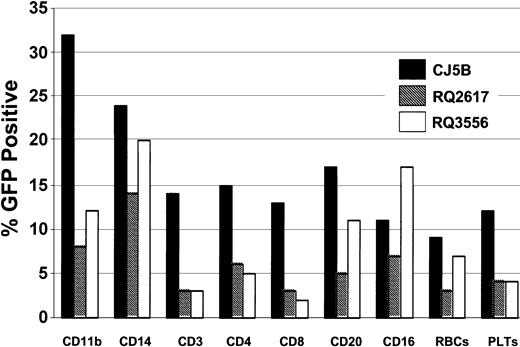
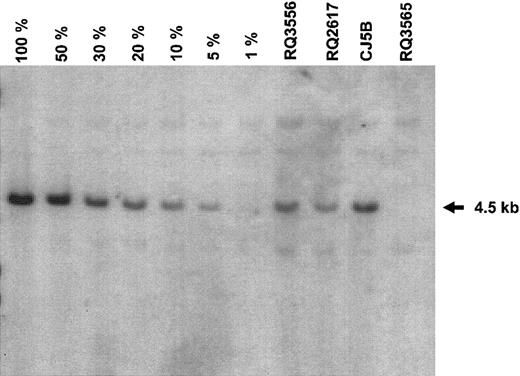
All 3 animals studied received more than 7 × 106 transduced CD34+ cells per kilogram after conditioning with 1000 cGy (Table 1). The animals received G-CSF for 8 to 13 days until the total leukocyte count exceeded 6 × 109/L (6000/μL). Hematopoietic reconstitution was prompt, with the total neutrophil count returning to more than 0.5 × 109/L (500/μL) by 7 or 8 days in each animal. These animals have been studied for periods ranging from 184 to 260 days following infusion of the transduced cells. The proportion of GFP-positive granulocytes and monocytes were 40% to 50% in 2 of the animals during the first month and 12% in the other animal. Over time, the proportion of genetically modified cells in these lineages tended to decline before stabilizing by 60 to 80 days following transplantation (Figure 3A-C). The percentage of GFP-positive granulocytes and monocytes of the 3 animals averaged 18% ± 8% and 15% ± 7%, respectively, while the copy number of the vector in genetically modified leukocytes, as determined by real-time PCR, averaged 0.19 ± 0.05, implying a single copy of the vector genome per transduced cell. The proportions of GFP-positive red cells, platelets, and lymphocytes were somewhat lower although significant proportions of genetically modified cells were detected in these subsets at all times following reconstitution. FACS analysis utilizing specific monoclonal antibodies that react with rhesus cells confirmed the presence of GFP-positive granulocytes (CD11b), natural killer cells (CD16), monocytes (CD14), B lymphocytes (CD20), and T lymphocytes (CD3, CD4, and CD8) (Figures 4 and 5). The unrearranged vector genome was detected in total leukocyte DNA from all 3 animals by Southern blot analysis at levels consistent with both the GFP expression and real-time PCR data for each animal (Figure 6). Finally, real-time PCR analysis of hematopoietic colony DNA derived from the 3 animals indicated that 32 of 34 genetically modified progenitors contained a single copy, with 1 colony containing 2 copies of the vector genome and the other 3 copies (data not shown). At 10 months after transplantation, 16% of the bone marrow CD34 cells were GFP-positive in animal CJ5B. The corresponding values for animals RQ2617 at 9 months and RQ3556 at 7.5 months were 12% and 11%, respectively. These data provide further evidence of long-term reconstitution with genetically modified repopulating stem cells.
Reconstitution of rhesus macaques with autologous, genetically modified hematopoietic cells. Various cell populations were gated based on forward and side scatter properties using FACS. The percentages of GFP-positive granulocytes (▴), monocytes (○), lymphoid cells (▪), red cells (□), and platelets (▵) at various times after infusion of transduced, autologous CD34+ cells are shown. The vector copy number in DNA from peripheral blood leukocytes was estimated by real-time PCR (•, heavy line) using DNA from a K562 erythroleukemia cell clone having a single copy of the vector genome as a standard. Data are from the following animals: (A) CJ5B, (B) RQ2617, and (C) RQ3556.
Reconstitution of rhesus macaques with autologous, genetically modified hematopoietic cells. Various cell populations were gated based on forward and side scatter properties using FACS. The percentages of GFP-positive granulocytes (▴), monocytes (○), lymphoid cells (▪), red cells (□), and platelets (▵) at various times after infusion of transduced, autologous CD34+ cells are shown. The vector copy number in DNA from peripheral blood leukocytes was estimated by real-time PCR (•, heavy line) using DNA from a K562 erythroleukemia cell clone having a single copy of the vector genome as a standard. Data are from the following animals: (A) CJ5B, (B) RQ2617, and (C) RQ3556.
Multilineage reconstitution with genetically modified cells. The percentage of GFP-positive cells in immunophenotypically defined cell populations was determined by FACS. The top row shows the profiles of red cells (RBCs) and platelets (PLTs) as defined by forward and side scatter. The percentages of GFP-positive granulocytes (CD11b), monocytes (CD14), T lymphocytes (CD3), B lymphocytes (CD20), and natural killer cells (CD16) are shown in the other panels for animal CJ5B on day 198 following infusion of autologous, transduced CD34+ cells.
Multilineage reconstitution with genetically modified cells. The percentage of GFP-positive cells in immunophenotypically defined cell populations was determined by FACS. The top row shows the profiles of red cells (RBCs) and platelets (PLTs) as defined by forward and side scatter. The percentages of GFP-positive granulocytes (CD11b), monocytes (CD14), T lymphocytes (CD3), B lymphocytes (CD20), and natural killer cells (CD16) are shown in the other panels for animal CJ5B on day 198 following infusion of autologous, transduced CD34+ cells.
Summary of the percentages of GFP-positive cells in various lineages following reconstitution. Animal CJ5B was studied on day 198, RQ2617 on day 170, and animal RQ3556 on day 121 following infusion of autologous, transduced CD34+ cells. Peripheral blood cells were processed and characterized by FACS using standard techniques. Antibody specificities are as follows: CD11b, granulocytes; CD14, monocytes; CD3, T lymphocytes; CD4, helper T lymphocytes; CD8, suppressor T lymphocytes; CD20, B lymphocytes; and CD16, natural killer cells.
Summary of the percentages of GFP-positive cells in various lineages following reconstitution. Animal CJ5B was studied on day 198, RQ2617 on day 170, and animal RQ3556 on day 121 following infusion of autologous, transduced CD34+ cells. Peripheral blood cells were processed and characterized by FACS using standard techniques. Antibody specificities are as follows: CD11b, granulocytes; CD14, monocytes; CD3, T lymphocytes; CD4, helper T lymphocytes; CD8, suppressor T lymphocytes; CD20, B lymphocytes; and CD16, natural killer cells.
Presence of the intact, unrearranged vector genome in leukocyte DNA. Peripheral blood was obtained from animal CJ5B on day 183, from animal RQ2617 on day 135, and from animal RQ3556 on day 86 following infusion of autologous, transduced CD34+ cells. DNA was prepared, and a Southern blot was performed using standard techniques. For each reaction, 10 μg DNA was restricted with Bgl II, which cuts twice in the vector genome. The probe was a GFP fragment from the vector plasmid. Serial dilutions of control DNA were prepared by mixing DNA from a K562 erythroleukemia cell clone having a single copy of the vector genome with DNA from nontransduced K562 cells in the proportions indicated. Equivalency of loading of the DNA from lane to lane was verified by staining the gel with ethidium prior to DNA transfer (data not shown).
Presence of the intact, unrearranged vector genome in leukocyte DNA. Peripheral blood was obtained from animal CJ5B on day 183, from animal RQ2617 on day 135, and from animal RQ3556 on day 86 following infusion of autologous, transduced CD34+ cells. DNA was prepared, and a Southern blot was performed using standard techniques. For each reaction, 10 μg DNA was restricted with Bgl II, which cuts twice in the vector genome. The probe was a GFP fragment from the vector plasmid. Serial dilutions of control DNA were prepared by mixing DNA from a K562 erythroleukemia cell clone having a single copy of the vector genome with DNA from nontransduced K562 cells in the proportions indicated. Equivalency of loading of the DNA from lane to lane was verified by staining the gel with ethidium prior to DNA transfer (data not shown).
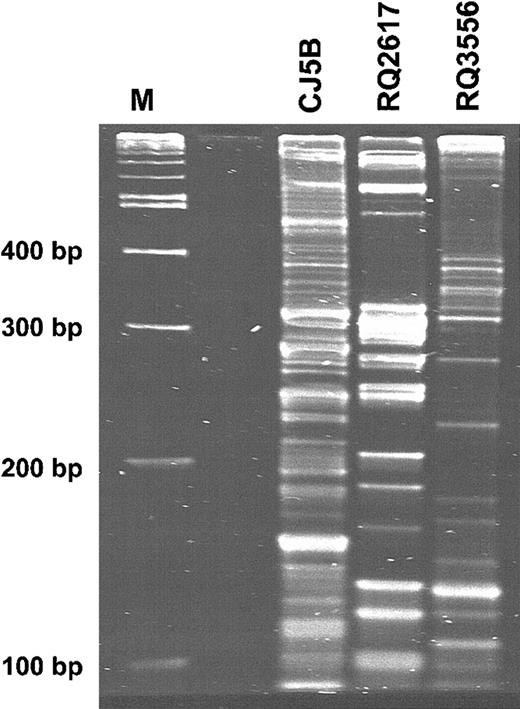
Polyclonal reconstitution with stem cells genetically modified with SIV-based vector particles
Using LAM-PCR and Spreadex gel analysis, multiple presumptive proviral integration bands were detected in peripheral blood DNA from each recipient (Figure 7). To further validate the inference that reconstitution was polyclonal, 35 unique integration sites in leukocyte DNA from animal RQ2617 obtained at day 46 or 3 months after transplantation, and 19 unique insertions from animal CJ5B at 7 months were sequenced. Most of these unique insertion sites were within introns of genes with other insertions found in intergenic regions and various repetitive elements. Finally, in an independent experiment to validate the interpretation that reconstitution was polyclonal, Southern blot analysis of leukocyte DNA from the 3 animals at 3, 5, and 6 months after transplantation, respectively, was performed using an enzyme that cuts once in the integrated provirus. No discernible hybridizing bands were observed (data not shown). Based on the signals of the hybridizing band generated by digesting DNA with an enzyme that cuts twice in the integrated provirus (Figure 6), the absence of hybridizing bands generated by an enzyme that cuts once within the provirus supports the inference that at least 13 or 33 different insertions were present in animal RQ2617 or CJ5B, respectively.
Clonal heterogeneity in peripheral blood following reconstitution with autologous, transduced CD34+ cells. Granulocyte DNA was subjected to LAM-PCR, and the products were resolved on a Spreadex gel. Peripheral blood samples were obtained at 7.5 months after reconstitution from animal CJ5B, at 6.5 months from animal RQ2617, and at 5 months from animal RQ3556.
Clonal heterogeneity in peripheral blood following reconstitution with autologous, transduced CD34+ cells. Granulocyte DNA was subjected to LAM-PCR, and the products were resolved on a Spreadex gel. Peripheral blood samples were obtained at 7.5 months after reconstitution from animal CJ5B, at 6.5 months from animal RQ2617, and at 5 months from animal RQ3556.
Discussion
Lentiviral vectors based on HIV-1 that efficiently transduced primitive human hematopoietic cells transduced the corresponding rhesus population very poorly. In contrast, vector particles based on SIVmac1A11, a nonpathogenic lentivirus, efficiently transduced rhesus cells as assayed in vitro. This efficiency translated into an average of 16% of peripheral blood leukocytes containing the SIV vector genome in 3 animals reconstituted with autologous CD34+ cells transduced with SIV vector particles. Reconstitution was polyclonal in that numerous integration bands were detected in leukocyte DNA that were shown to represent unique insertion sites by DNA sequencing. Overall, our results establish that lentiviral vectors transduce primitive repopulating stem cells efficiently and suggest that such vectors could be useful for developing gene therapy approaches for many human genetic disorders of the blood.
Several SIV-based vector systems have been developed (reviewed by Ruggieri et al43 and Negre et al44 ). Our system incorporates several features previously described, including the self-inactivating design of the vector, the distribution of coding sequences for the required viral proteins on separate plasmids, the elimination of coding sequences for accessory proteins from vector and helper plasmids, and the inclusion of the central polypurine tract in the vector genome. Our vectors compare favorably with those reported by others.45-47 For example, an MOI of 15 or greater was required to achieve transduction of 60% or 30% of human or cynomolgus CD34+ cells, respectively, with SIV vector particles pseudotyped with the RD114 envelope protein,47 whereas we achieved transduction of 50% or 75% of human or rhesus CD34+ cells, respectively, at an MOI of 1.2 to 2.4. Furthermore, we provide the first evidence that SIV-based vectors can be used to successfully transduce repopulating hematopoietic cells in a large animal model.
The early postentry restriction on infection of simian cells by HIV vector particles involves an interaction with the capsid protein.33,34 A cellular protein behaves as a saturable inhibitor of HIV infection prior to reverse transcription.35 Recent studies have shown that substitution of the cyclophilin A binding domain of an HIV-1–based lentiviral vector with a corresponding region from a macrophage tropic strain abrogated the early postentry restriction although an MOI of 50 was required to transduce 20% of baboon CD34+ cells.48 In initial experiments we found that substituting SIV for HIV gag/pol coding sequences did not ensure high titer vector production (data not shown), leading us to develop an SIV system fully analogous to our HIV system.27 Highly efficient transduction of primitive hematopoietic cells was achieved at a low MOI.
A recent report has described induction of cytotoxic T-lymphocyte responses to enhanced green (GFP) and yellow (YFP) fluorescent proteins after myeloablative conditioning in baboons that had received primitive hematopoietic cells transduced with HIV-1–based lentiviral vectors.49 The transduced CD34+ cells appear to engraft normally in 5 animals that were studied based on neutrophil and platelet recovery. During the early days after engraftment, the percentages of GFP- and/or YFP-positive cells in one animal were approximately 20%, in a second, 12%; in a third, 4% to 5%; and in the other 2, 1%. However, genetically modified cells disappeared from the circulation in all animals between 26 and 54 days following transplantation. The authors attributed this disappearance to the presence of cytotoxic T lymphocytes, which was demonstrated in 4 animals, and speculated that induction of the immune response in these animals that had received myeloablative irradiation reflected residual T-lymphocyte function. These results contrast with the general experience that there is no significant immune response to GFP in animals receiving oncoretroviral vector–transduced cells following lethal irradiation.28,40,41,50 Indeed, a recent study documented tolerance to the GFP protein in rhesus macaques that harbored lentiviral vector, modified cells expressing the protein even when subsequently challenged with GFP-soluble protein.32 To account for the results in the baboon model, the authors49 speculate that the low level of transduction achieved with HIV-based lentiviral vectors resulted in the induction of an immune response as opposed to tolerance that occurs when higher levels of transduction are achieved. As noted earlier, this low level of transduction is likely to be due to the postentry block to HIV infection of simian cells.33,34,48 In fact, this postentry block to HIV was specifically observed in baboon CD34+ cells. In our studies, use of an SIV-based lentiviral vector system circumvented this block. The persistence of genetically modified cells for 227 to 304 days after transplantation indicates that no significant immune response to GFP was induced in our experiments.
Our initial results support the previous prediction that lentiviral vector particles will be superior to oncoretroviral vector particles at transducing repopulating stem cells. All 3 animals studied displayed genetically modified cells in peripheral blood of more than 10%, with 1 animal consistently having a level of more than 20%. In 3 earlier studies in which a total of 11 rhesus macaques received autologous CD34+ cells transduced with amphotropic oncoretroviral vector particles, the percentage of genetically modified cells in peripheral blood averaged 4% with a range of 0.1% to 20%.51-53 In the baboon model, in recent studies of a total of 11 animals that received autologous CD34+ cells transduced with oncoretroviral vector particles derived from a human cell line and pseudotyped with the gibbon ape leukemia virus (GALV) envelope, the percentage of genetically modified cells averaged 10% with a range of 1% to 40%.28,54,55 Further optimization of SIV vector transduction by using particles pseudotyped with alternative envelope proteins12-14 or adding factors such as those in the Wnt family56 or HoxB457 to the culture medium is likely to further improve transduction efficiency.
For the first time, we have a vector system that provides efficient gene transfer into primate stem cells and is of the type that is capable of transferring a globin gene with regulatory elements required to achieve high-level expression in maturing erythroblasts. Lentiviruses have effective, rev-dependent mechanisms for nuclear to cytoplasmic transport of unspliced RNA,58 which uniquely facilitate the assembly of vectors having the extended globin promoter sequences and elements from the locus control region (LCR) that are necessary to achieve significant expression of globin genes in mouse models of hemoglobin disorders.59-64 Recently, we have shown that a vector genome containing larger LCR fragments consistently gave higher levels of γ-globin gene expression, particularly when expressed on a per vector copy basis, and this vector was shown to be less susceptible to position effect variegation by analysis of secondary spleen CFU (CFU-S)–derived colonies.65 An effective strategy for selectively amplifying genetically modified populations of stem cells in vivo66-72 may obviate the need for complete myeloablation, thereby increasing the safety of gene therapy for hemoglobin disorders. Using a variant O6-benzylguanine–resistant methylguanine methyltransferase, we have successfully treated murine β thalassemia by in vivo selection of genetically modified, drug-resistant hematopoietic stem cells.71
Lentiviral vectors have a propensity to insert into transcriptionally active genes,73,74 and target cells often undergo multiple integration events, particularly at high multiplicities of infection.75 Two of 10 children with severe combined immunodeficiency (SCID) due to common γ-chain receptor deficiency developed lymphoid leukemia, and in each the leukemic cells contained an oncoretroviral vector insertion within or upstream of the LMO2 gene, leading to its activation.76 Factors unique to the SCID common γ-chain–deficient children may have also contributed to the development of the lymphoproliferative process.77,78 The risk of insertional gene activation in the context of gene therapy trials had been estimated to be relatively low (10-6 to 10-7) based on theoretical considerations as well as data from various experimental systems, but revised estimates suggest that the risk may be higher, in the range 10-2 to 10-3.79 The self-inactivating design of our lentiviral vectors reduces the risk of insertional activation.
Ongoing studies are focused on defining the pattern of lentiviral integration long-term in the 3 animals reported here and on comparing these data with earlier results reported for cell lines with lentiviral vectors73 and for both cell lines74 and primary hematopoietic cells80 with oncoretroviral vectors. Oncoretroviral vectors exhibit a predilection for integration into genes of long-term repopulating cells from rhesus macaques, and recurrent integration loci have been identified,80 one of which is near the MDS1 gene, which is involved in chromosomal translocations in human leukemias. To date, no evidence of progressive enrichment of such clones has been noted during long-term follow-up. A comparison of the patterns of lentiviral versus oncoretroviral integration in stem cells in the rhesus model will be of considerable interest.
Overall, we believe that the SIV vector system represents a significant advance with respect to efforts to evaluate gene transfer in a nonhuman primate model. The ability to efficiently transduce rhesus repopulating cells will facilitate evaluation of drug selection systems and potential globin therapeutic vectors in this large animal model. Attention to the elements of vector design that may influence expression of genes surrounding integration sites and tracking of clonal populations in which the integrated vector genome is near a protooncogene will be critical in evaluating the potential safety of gene therapy for the treatment of human genetic diseases.
Prepublished online as Blood First Edition Paper, February 19, 2004; DOI 10.1182/blood-2004-01-0045.
Supported by NHLBI Program Project Grant P01 HL 53749, Cancer Center Support CORE Grant P30 CA 21765, the Hartwell Center for Bioinformatics and Biotechnology at St Jude Children's Research Hospital, and American Lebanese Syrian Associated Charities (ALSAC).
H.H. and P.H. contributed equally to this study. D.A.P. and A.W.N. contributed equally to this study.
The publication costs of this article were defrayed in part by page charge payment. Therefore, and solely to indicate this fact, this article is hereby marked “advertisement” in accordance with 18 U.S.C. section 1734.
We are grateful for the help of Jean Johnson in preparing this manuscript, the veterinary and animal support staff for help in caring for the animals, Aylin Bonifacino for her help in the flow analysis, and Earl West for clinical laboratory support.