Key Points
Expression of caudal-related homeobox gene 2 (CDX2) and angiotensin-converting enzyme (ACE) correlates during hematopoietic emergence.
This emergence occurs in human and mouse embryos and in human acute myeloid leukemia; CDX2 homeoprotein also binds to the ACE promoter.
Introduction
During vertebrate, definitive hematopoietic stem cells (HSCs) are first generated in the aorta-gonad-mesonephros (AGM) region of the posterior embryo.1 We previously showed that the angiotensin-converting enzyme (ACE) is a cell-surface marker of human HSCs in both adult bone marrow (BM)2 and all hematopoietic niches developmentduring human ontogeny.3 In the embryo, ACE identifies HSCs associated with the ventral side of the aortic endothelium (in the AGM region) and of vitelline and umbilical arteries, both in humans3 and mice.4 ACE is a key component of the renin-angiotensin system (RAS), catalyzing the production of angiotensin II (AngII),5 which we have recently shown to be involved in hematopoietic emergence during ontogeny.4 ACE overexpression, leading to AngII increase, has also been reported in the BM of patients with acute myeloid leukemia (AML), influencing malignancy6,7 ; however, its mechanisms of regulation are still unknown.8 The caudal-related homeobox gene 2 (CDX2) encodes an important transcription factor involved in tissue expansion and patterning of the posterior embryo.9,10 At midgestation, it becomes limited to the presumptive mid-/hindgut endoderm and maintains selectively in the adult intestinal epithelium11 where it has tumor-suppressor activity.12-14 Conversely, although CDX2 is not expressed in adult HSCs, ectopic expression occurs in 80% of acute leukemia, which is considered a driver of leukemogenesis.15-17 Here, we investigated the ACE and CDX2 expression patterns during human and mouse hematopoietic development, as well as in AML, and we conducted functional assays supporting that CDX2 participates in ACE regulation.
Methods
Human embryos were obtained from voluntary abortions and staged using anatomic criteria and the Carnegie classification as described.18 Umbilical cord blood was collected from healthy newborns. BM of AML patients (n = 29) and healthy subjects (n = 6) was collected after obtaining written consent according to the Declaration of Helsinki and the guidelines of the French National Ethics Committee. C57BL/6 mice were provided by Charles River and housed in a certified animal facility (number H-67-482-21). Murine embryos were staged by somite counting. Para-aortic splanchnopleura (P-Sp) or AGM regions were dissected as described.19
Additional methods are described in supplemental Material and methods.
Results and discussion
Although CDX2 is crucial for the development of the posterior embryonic structures and ACE expression also emerges in the posterior region during hematopoietic ontogenesis, the first aim of this study was to perform a comparative study of CDX2 and ACE expression patterns in both human and mouse embryos. At early stages of murine development (embryonic day 9 [E9]), both proteins were synthetized in the extraembryonic YS endoderm and in the intraembryonic P-Sp (including the gut endoderm): the region endowed with hematopoietic ability20,21 (supplemental Figure 1). Because our previous observations indicated ACE as a marker of HSCs in the AGM region,3,4 we focused on CDX2 at this site. By reverse transcription–polymerase chain reaction (RT-PCR), CDX2 transcripts were detected in the AGM region that also expressed ACE (supplemental Figure 2). Indeed, CDX2 protein was found in intra-aortic hematopoietic clusters (IAHCs), also positive for ACE, in 33- to 36-day human embryos and in E10.5 mouse embryos (Figure 1A). This observation was strengthened by RNAscope in situ hybridization, which localized CDX2 transcripts in IAHCs, in associated endothelial cells (hemogenic endothelium), as well as in scattered mesodermal cells (Figure 1B). To further document the spatial evolution of ACE and CDX2 messenger RNA (mRNA) patterns in this blood-forming site at early stages of mouse development (E10.5-E11.5), mesenchymal cells (MCs), endothelial cells (ECs), and IAHC cells were sorted from the AGM region and processed for quantitative RT-PCR (RT-qPCR) analyses (Figure 1C-D). At E10.5, CDX2 and ACE transcripts were expressed in all 3 cell populations. Compared with the bulk of AGM cells, the proportion of CDX2 transcripts in ECs and HSCs was lower than that of ACE, but it was higher in MCs, consistent with the wider function of CDX2 in posterior morphogenesis at this stage.9,10 Nevertheless, this pattern was rapidly evolving because, at E11.5, both transcripts were still detected in ECs and MCs, but were turned off in IAHCs. The loss of CDX2 expression observed in murine IAHCs at midgestation is in line with the absence of CDX2 in fetal liver– and BM-derived HSCs and is also confirmed in humans because, despite its expression in IAHCs, CDX2 was absent in CD34+ progenitors isolated from human fetal liver and cord blood (supplemental Table 1). Furthermore, ACE+ MCs isolated from early human embryos also expressed CDX2, which by contrast is absent in ACE− vascular endothelial cells derived from the same embryos (supplemental Figure 3).
Expression patterns of CDX2 and ACE in the mouse and human AGM region. (A) Representative cross sections through the aorta (Ao) of 34-day human embryos (Carnegie stage [CS], 15) (n = 3) and E10.5 mouse embryos (36 somite pairs) (n = 4), immunostained with anti-CD34, anti-CD31, anti-CDX2, and anti-ACE antibodies (brown staining). All intra-aortic hematopoietic cluster (IAHC) cells (black arrowhead) stained by CD34 (for human) and CD31 (for mouse) express ACE, which is also expressed by the endothelial cells underlining the ventral side of the aorta (white arrowhead). Transcription factor CDX2 is expressed by the hematopoietic cell of the intra-aortic clusters. Scale bars represent 10 µm. (B) Representative images of CDX2 RNASCOPE in situ hybridization in cross sections of the aorta (Ao) of 36-day human embryos (CS, 15) (n = 4) and E10.5 mouse embryos (n = 5) (brown staining). CDX2 mRNA is detected in hematopoietic cell clusters (black arrowheads), in some endothelial cells (white arrowheads), and in rare mesenchymal cells (stars). Scale bars represent 10 µm. Higher magnification views of highlighted regions are shown below each figure. (C-D) RT-qPCR analysis of ACE (C) and CDX2 (D) expression in IAHC cells, endothelia cells (EC), and mesenchymal cells (MC) isolated from E10.5 and E11.5 murine AGM. Results are normalized to glyceraldehyde-3-phosphate dehydrogenase (Gapdh), and expressed relative to the E10.5 AGM samples. Values are represented as mean plus standard error of the mean (SEM) (n = 7). *P < .05, **P < .01, ***P < .001, ****P < .0001.
Expression patterns of CDX2 and ACE in the mouse and human AGM region. (A) Representative cross sections through the aorta (Ao) of 34-day human embryos (Carnegie stage [CS], 15) (n = 3) and E10.5 mouse embryos (36 somite pairs) (n = 4), immunostained with anti-CD34, anti-CD31, anti-CDX2, and anti-ACE antibodies (brown staining). All intra-aortic hematopoietic cluster (IAHC) cells (black arrowhead) stained by CD34 (for human) and CD31 (for mouse) express ACE, which is also expressed by the endothelial cells underlining the ventral side of the aorta (white arrowhead). Transcription factor CDX2 is expressed by the hematopoietic cell of the intra-aortic clusters. Scale bars represent 10 µm. (B) Representative images of CDX2 RNASCOPE in situ hybridization in cross sections of the aorta (Ao) of 36-day human embryos (CS, 15) (n = 4) and E10.5 mouse embryos (n = 5) (brown staining). CDX2 mRNA is detected in hematopoietic cell clusters (black arrowheads), in some endothelial cells (white arrowheads), and in rare mesenchymal cells (stars). Scale bars represent 10 µm. Higher magnification views of highlighted regions are shown below each figure. (C-D) RT-qPCR analysis of ACE (C) and CDX2 (D) expression in IAHC cells, endothelia cells (EC), and mesenchymal cells (MC) isolated from E10.5 and E11.5 murine AGM. Results are normalized to glyceraldehyde-3-phosphate dehydrogenase (Gapdh), and expressed relative to the E10.5 AGM samples. Values are represented as mean plus standard error of the mean (SEM) (n = 7). *P < .05, **P < .01, ***P < .001, ****P < .0001.
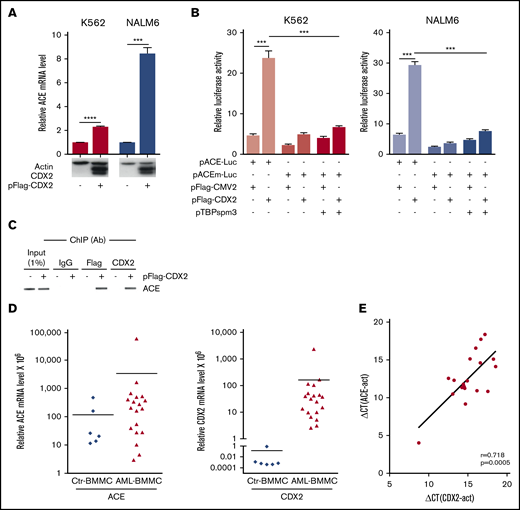
Based on the similarities between ACE and CDX2 patterns during embryonic hematopoiesis, we investigated whether CDX2 could participate in the gene regulation of ACE and/or of other RAS components. For this purpose, 2 leukemia cell lines, Nalm6 and K562, were transfected with plasmid pFlag-CDX2 encoding the human CDX2 homeoprotein.22 CDX2 increased the endogenous expression of ACE mRNA (Figure 2A), whereas no effect was observed on any other RAS element (supplemental Figure 4). To get further insight into the underlying molecular mechanism, we constructed the reporter luciferase plasmid pACE-Luc containing the 461-bp promoter and the first 37 bp of exon 1 of the human ACE gene up to the translation start site. Luciferase activity in Nalm6 and K562 cells transfected with pACE-Luc was stimulated by cotransfection with pFlag-CDX2 (Figure 2B). The ACE promoter contains a putative consensus CDX-type binding site23 overlapping the TATA box and conserved in mice in the configuration: TTTATAA (CDX-type site in bold; TATA box underlined). Changing the TTTATA sequence into TTTGTA in pACEm-Luc strongly compromised the CDX2-stimulatory effect (Figure 2B). This was not due to destroying the TATA box because cotransfection with plasmid pTBPspm3, which encodes a mutant TBP able to bind the TGTA sequence instead of the classical TATA box,24 did restore the basal activity of the promoter but not the stimulatory effect of CDX2 (Figure 2B). In addition, chromatin immunoprecipitation with anti-Flag or anti-CDX2 antibodies revealed the CDX2 protein linked to chromatin containing the CDX-type binding site of the ACE promoter in nuclear extracts of pFlag-CDX2–transfected cells (Figure 2C).
Correlation between ACE and CDX2 and expression. (A) Endogenous ACE mRNA expression by RT-qPCR in K562 and Nalm6 cells transfected with the control vector pFlag-CMV2 (−) or with the CDX2-expressing plasmid pFlag-CDX2 (+). (n = 3). Western blots for CDX2 and actin as control are illustrated below the graphic. (B) Luciferase activity in K562 and Nalm6 cells cotransfected with the reporter plasmid pACE-Luc containing the ACE promoter, or with the mutated form pACEm-Luc, together with the control plasmid pFlag-CMV2, with pFlag-CDX2 and/or with pTBPspm3. Values are expressed relative to the luciferase activity measured in cells transfected with the control luciferase plasmid pGL3-basic and presented as mean plus or minus SEM (n = 3). (C) Chromatin immunoprecipitation (ChIP) with anti-CDX2 or anti-Flag antibodies (Ab) in K562 cells transfected with pFlag-CDX2 (+) or pFlag-CMV2 (−), followed by PCR amplification of the ACE promoter fragment overlapping the CDX2-binding element. Specificity of the chromatin immunoprecipitation was assessed using immunoglobulin G (IgG). (D) ACE and CDX2 mRNA levels measured by RT-qPCR in the BMMCs from AML patients (AML-BMMC) and from healthy individuals (Ctr-BMMC). (E) Pearson correlation coefficient (r) and P value (p) between CDX2 and ACE expressions measured by RT-qPCR on BMMCs of AML patients. ***P < .001, ****P < .0001.
Correlation between ACE and CDX2 and expression. (A) Endogenous ACE mRNA expression by RT-qPCR in K562 and Nalm6 cells transfected with the control vector pFlag-CMV2 (−) or with the CDX2-expressing plasmid pFlag-CDX2 (+). (n = 3). Western blots for CDX2 and actin as control are illustrated below the graphic. (B) Luciferase activity in K562 and Nalm6 cells cotransfected with the reporter plasmid pACE-Luc containing the ACE promoter, or with the mutated form pACEm-Luc, together with the control plasmid pFlag-CMV2, with pFlag-CDX2 and/or with pTBPspm3. Values are expressed relative to the luciferase activity measured in cells transfected with the control luciferase plasmid pGL3-basic and presented as mean plus or minus SEM (n = 3). (C) Chromatin immunoprecipitation (ChIP) with anti-CDX2 or anti-Flag antibodies (Ab) in K562 cells transfected with pFlag-CDX2 (+) or pFlag-CMV2 (−), followed by PCR amplification of the ACE promoter fragment overlapping the CDX2-binding element. Specificity of the chromatin immunoprecipitation was assessed using immunoglobulin G (IgG). (D) ACE and CDX2 mRNA levels measured by RT-qPCR in the BMMCs from AML patients (AML-BMMC) and from healthy individuals (Ctr-BMMC). (E) Pearson correlation coefficient (r) and P value (p) between CDX2 and ACE expressions measured by RT-qPCR on BMMCs of AML patients. ***P < .001, ****P < .0001.
Next, we analyzed the expression of ACE and CDX2 in human BM mononuclear cells (BMMCs) derived from healthy individuals and AML patients (Figure 2D). RT-qPCR revealed a basal expression of ACE transcripts in the BMMCs of healthy individuals (cycle threshold [Ct] = 33 cycles), and a significant increase in AML patients. Unlike ACE, CDX2 was virtually absent in BMMCs of healthy individuals (no RT-qPCR signal was detected below Ct = 50 cycles), but aberrant expression was detected at various levels in AML patients. Statistical analysis revealed a significant positive correlation (r = 0.718; P = .0005) between ACE and CDX2 levels in AML patients (Figure 2E). Moreover, a coexpression pattern between ACE and CDX2 was observed in leukemic blasts sorted from 7 AML patients (supplemental Figure 5).
Altogether, these data suggest that homeotic transcription factor CDX2, a major determinant of growth and patterning of the posterior embryo, is a part of the regulator network involved in ACE regulation during hematopoietic emergence. Strikingly, although absent in normal BM-derived HSCs, CDX2 becomes abnormally expressed in AML cells, at levels that correlate with the increase of ACE expression. This opens new perspectives for understanding the deleterious effect exerted by CDX2 upon abnormal reexpression in leukemia through the proliferative effect exerted by AngII on hematopoietic cell emergence and growth.4,25
Original data can be requested through the corresponding author, Manuela Tavian, at manuela.tavian@inserm.fr.
Acknowledgments
The authors thank T. Ayoub for technical support.
This work was supported by INSERM and by grants from the University of Strasbourg, the Ligue Contre le Cancer Bourgogne France Comté, the Fondation ARC pour la Recherche sur le Cancer (PJA 20131200190), the Région Grand-Est (program OMAGE), and ANR (ANR-14-CE11-0008). E.J. was awarded a fellowship from INSERM-Alsace and from the Fondation ARC pour la Recherche sur le Cancer. K.B. was awarded a fellowship from INSERM-Alsace and from the Société Française d'Hématologie. R.E.O. was supported by a grant from ANR.
Authorship
Contribution: R.E.O. and E.J. performed experiments and analysis, and contributed to interpretation of results and the writing of the manuscript; K.B. designed and conducted experiments, and analyzed and interpreted the data; I.G., C.D.-D., and B.G. conducted experiments and analyzed the data; C.G. and F.L. contributed to data interpretation and final discussion; B.L. and L.V. provided materials and contributed to data interpretation; M.N. designed and performed plasmid constructions and mutagenesis; J.-N.F. contributed to experiment design, interpretation of results, and the writing of the manuscript; and M.T. supervised the overall project, designed experiments, analyzed the data, and wrote the manuscript.
Conflict-of-interest disclosure: The authors declare no competing financial interests.
The current affiliation for R.E.O. is Ingénierie Moléculaire et Physiopathologie Articulaire (IMoPA), UMR7365 CNRS–University of Lorraine, Vandœuvre Les Nancy, France.
Correspondence: Manuela Tavian, University of Strasbourg, INSERM, IRFAC/UMR-S1113, Strasbourg, France; e-mail: manuela.tavian@inserm.fr.
References
Author notes
The full-text version of this article contains a data supplement.

![Expression patterns of CDX2 and ACE in the mouse and human AGM region. (A) Representative cross sections through the aorta (Ao) of 34-day human embryos (Carnegie stage [CS], 15) (n = 3) and E10.5 mouse embryos (36 somite pairs) (n = 4), immunostained with anti-CD34, anti-CD31, anti-CDX2, and anti-ACE antibodies (brown staining). All intra-aortic hematopoietic cluster (IAHC) cells (black arrowhead) stained by CD34 (for human) and CD31 (for mouse) express ACE, which is also expressed by the endothelial cells underlining the ventral side of the aorta (white arrowhead). Transcription factor CDX2 is expressed by the hematopoietic cell of the intra-aortic clusters. Scale bars represent 10 µm. (B) Representative images of CDX2 RNASCOPE in situ hybridization in cross sections of the aorta (Ao) of 36-day human embryos (CS, 15) (n = 4) and E10.5 mouse embryos (n = 5) (brown staining). CDX2 mRNA is detected in hematopoietic cell clusters (black arrowheads), in some endothelial cells (white arrowheads), and in rare mesenchymal cells (stars). Scale bars represent 10 µm. Higher magnification views of highlighted regions are shown below each figure. (C-D) RT-qPCR analysis of ACE (C) and CDX2 (D) expression in IAHC cells, endothelia cells (EC), and mesenchymal cells (MC) isolated from E10.5 and E11.5 murine AGM. Results are normalized to glyceraldehyde-3-phosphate dehydrogenase (Gapdh), and expressed relative to the E10.5 AGM samples. Values are represented as mean plus standard error of the mean (SEM) (n = 7). *P < .05, **P < .01, ***P < .001, ****P < .0001.](https://ash.silverchair-cdn.com/ash/content_public/journal/bloodadvances/5/7/10.1182_bloodadvances.2020003563/2/m_advancesadv2020003563f1.png?Expires=1764989144&Signature=QXfiQJcYEBzOqX9W4KiU8qjAI4QhL31NMzhbR27s0nrVbhC~XEvP1rPSXNiVaK8vtZBAHyJ~4gu2Y~NX0tRuPUfUPGq-hLlkxdrFf6B1ThUsmMPID~Xfr0zH-XMQX~vDigUEf~2UO1-CiLh~8fNRjr2jDVGdHQCMVz~O7GLNjLQ5~57xFjCz14VLnbU4Eft3BGsAwvyRZ-cyLLW-BPtO03w7zLOQk~IjRp2LqD-PXIz136WncTfSRFgAKWhjQQ95I0r-yZD7gwkaOYRNJqsD49Ag29ha5iOxH-wK8pfzQzeE2wQXMnDvxBazrM66ZRj9XDH0jcnx90P9Cqb8HOWMgQ__&Key-Pair-Id=APKAIE5G5CRDK6RD3PGA)