Key Points
In relapsed or refractory AML, mutated NPM1 has no impact on the risk of relapse or death.
The addition of venetoclax to salvage treatment for NPM1-mutated AML is associated with improved outcomes.
Abstract
Mutations in Nucleophosmin 1 (NPM1) are associated with a favorable prognosis in newly diagnosed acute myeloid leukemia (AML), however, their prognostic impact in relapsed/refractory (R/R) settings are unknown. In a retrospective analysis, we identified 206 patients (12%) with mutated NPM1 (NPM1c) and compared their outcomes to 1516 patients (88%) with NPM1 wild-type (NPM1wt). NPM1c was associated with higher rates of complete remission or complete remission with incomplete count recovery compared with NPM1wt following each line of salvage therapy (first salvage, 56% vs 37%; P < .0001; second salvage, 33% vs 22%; P = .02; third salvage, 24% vs 14%; P = .02). However, NPM1 mutations had no impact on relapse-free survival (RFS) and overall survival (OS) with each salvage therapy with a median OS following salvage 1, 2 or 3 therapies in NPM1c vs NPM1wt of 7.8 vs 6.0; 5.3 vs 4.1; and 3.5 vs 3.6 months, respectively. Notably, the addition of venetoclax to salvage regimens in patients with NPM1c improved RFS and OS (median RFS, 15.8 vs 4.6 months; P = .05; median OS, 14.7 vs 5.9 months; P = .02). In conclusion, NPM1 mutational status has a minimal impact on prognosis in relapsed or refractory AML; therefore, novel treatment strategies are required to improve outcomes in this entity.
Introduction
Mutations in the Nucleophosmin 1 (NPM1) gene are the most common genetic alterations in acute myeloid leukemia (AML), occurring in 20% to 30% of adults with this disease.1,2 AML with mutated NPM1 is considered a distinct entity according to the World Health Organization classification and included in the European LeukemiaNet (ELN) 2017 classification owing to its biological and prognostic significance.3,4 These mutations frequently occur in exon 12 of NPM1, causing truncation of the protein and disruption of shuttling between the cytoplasm and nucleus, thereby leading to persistence of NPM1 in the cytoplasm (thus termed NPM1c).5,NPM1c frequently co-occur with FMS-like tyrosine kinase 3 (FLT3), isocitrate dehydrogenase (IDH) 1 and IDH2 or DNA methyltransferase 3 alpha (DNMT3A) mutations.6 In newly diagnosed patients, AML with NPM1c without a FLT3-ITD mutation is associated with high response rates and a favorable prognosis.7,8 However, this prognosis is significantly affected by the presence of co-occurring mutations.1,4,6,9 Given that NPM1c is a leukemia initiating event, multiple studies have demonstrated the value of detecting NPM1c as a measurable disease marker, albeit growing evidence that these mutations can be lost at relapse.10-12 Despite better understanding of the disease course associated with NPM1c following first-line treatment, very little is known about the prognostic impact and response to various therapies in the relapsed or refractory (R/R) settings.
We conducted a retrospective analysis of patients with R/R AML and NPM1c to characterize the clinical presentation, prognosis, and response to various lines of therapy.
Methods
Study design and patient selection
We screened 1722 adult patients with R/R AML treated at The University of Texas MD Anderson Cancer Center between 1 September 2012 and 1 December 2020. Targeted next-generation sequencing was performed using panels of genes recurrently mutated in hematologic malignancies (panels of either 28, 53, or 81 genes were used at our center during this time as described in previous publications).13 Measurable residual disease (MRD) assessment was performed on bone marrow samples using multicolor flow cytometry (sensitivity 10−4 to 10−5) as previously described by our group.14
Various treatment strategies were used, depending on factors such as age, performance status, comorbidities, and comutations (supplemental Table 1). The treatment consisted of either high- or low-intensity regimens based on age and comorbidities. High-intensity (HI) regimens included combinations of cytarabine and idarubicin with or without the addition of a nucleoside analog (ie, cladribine, fludarabine, or clofarabine). Low-intensity (LI) regimens included either hypomethylating agents (5-azacitidine or decitabine) or low-dose cytarabine, with the addition of venetoclax more recently (starting in 2018) or investigational agents. Targeted therapies (ie, FLT3, IDH1, and IDH2 inhibitors) were used as single agents or in combination, as indicated. This study was approved by the institutional review board and was performed in accordance with the Declaration of Helsinki.
Statistical methods
Patient characteristics were summarized using medians and ranges for continuous variables, and frequencies or percentages for categorical variables. Continuous variables were compared using the Wilcoxon rank-sum test for pairwise comparisons and Kruskal-Wallis test for multiple comparisons. Categorical variables were compared using the Fisher exact test. Responses were defined according to the International Working Group recommendations.15 Overall survival (OS) was calculated from the treatment start date in patients with relapsed disease to the time of death or the last follow-up. Relapse-free survival (RFS) was calculated from the time of complete remission (CR)/complete remission with incomplete count recovery (CRi) until relapse or death and censored if the patient was alive at the last follow-up. The Kaplan–Meier method was used to estimate the probability of OS or RFS and was compared using the log-rank test. We assessed the independent effect of variables on prognosis in a multivariate analysis, where all variables with P < .1 in the univariate analysis were included. Analyses were performed using GraphPad Prism version 8.0 and SPSS statistics version 26.0.
Results
Baseline characteristics
We identified 1722 patients with R/R AML treated between 2012 and 2020, of whom 206 (12%) had NPM1c. The baseline characteristics of the patients are summarized in Table 1. Most patients (63%) in this cohort received their first salvage therapy (S1) at our institution, whereas the remaining patients received 2 or more previous lines of therapy (S2+). The median number of therapy lines administered in this cohort was 2 (range, 2 to 15 lines of therapy). The median age of all patients was 64 years (range, 16 to 91 years).
Among patients with R/R AML, NPM1c occurred more commonly in women than in NPM1 wild-type (NPM1wt) (58% vs 38%; P < .0001), was associated with a higher white blood cell count at presentation (P < .0001), and higher percentages of circulating blasts (P < .0001), and bone marrow blasts (P < .0001). This was likely owing to the significantly higher co-occurrence of FLT3 mutations in patients with NPM1c. Similar to what has been previously described in the newly diagnosed setting, patients with R/R NPM1c AML more commonly had a diploid karyotype than NPM1wt patients (61% vs 31%; P < .0001). At relapse, patients with NPM1c had a significantly lower incidence of therapy-related AML (t-AML) or AML secondary to an antecedent hematological malignancy (s-AML) than those with NPM1wt (8% vs 15%, P = .007; 3% vs 16%, P < .0001, respectively). The proportion of patients with early vs late first relapse did not differ according to the NPM1 mutational status (Table 1).
Mutational landscape
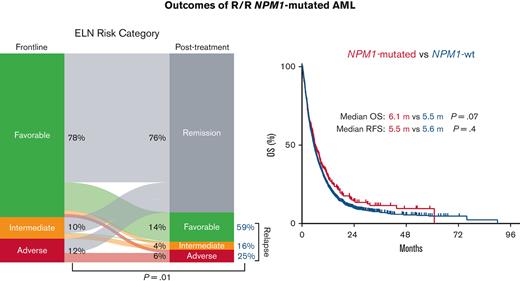
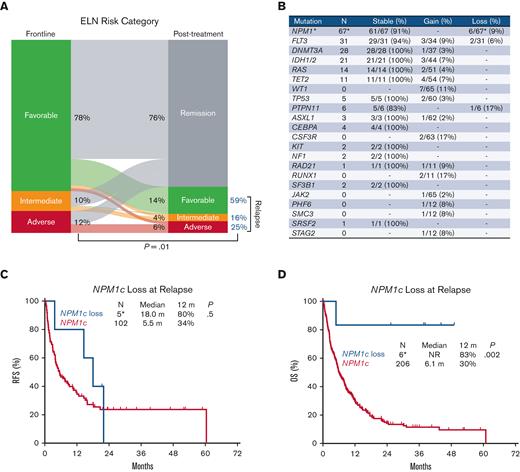
Consistent with the favorable prognosis associated with NPM1c at AML diagnosis, 76% of the evaluable patients showed remission following first-line therapy (Figure 1A). Among the remaining patients with relapsed disease (24%), there was an overall increase in ELN risk, reflecting cytogenetic and mutational changes at relapse. There was an increase in the ELN intermediate risk proportion from 10% at diagnosis to 16% at relapse, and an increase in the ELN adverse risk proportion from 12% to 25% (P = .01) (Figure 1A). Mutations in NPM1, DNMT3A, and FLT3 were stable at relapse (detected both at baseline and relapse), reflecting the frequent persistence at relapse of leukemia clones and subclones detected at diagnosis (Figure 1B). However, we identified NPM1c loss at relapse in 6 of 212 evaluable patients (3%) and FLT3 mutation loss in 2 of 31 patients (6%) with FLT3 mutation at diagnosis (Figure 1B). Notably, mutations in the WT1 gene were gained in 7 of 65 evaluable patients (11%), a pattern previously identified in FLT3 mutated AML relapse.16,17 In addition, 4 patients acquired mutations in TET2 at relapse, 3 acquired mutations in IDH1 or 2, and 2 acquired mutations in TP53. A full list of mutations gained or lost at relapse is provided in the supplement (supplemental Tables 2 and 3).
Clonal architecture of AML with mutated NPM1 at relapse. (A) Change in the ELN risk classification at relapse. (B) Mutational evolution, including stability, gain, or loss of mutations at relapse. N is the number of patients with the corresponding mutation at diagnosis among those evaluable by mutational analysis performed at diagnosis and at the time of relapse. The percentages for stability and loss were calculated as the number of patients with mutations that persisted or were lost at relapse divided by patients with mutations in the corresponding gene present at diagnosis. Percentage gain was calculated as the number of patients with mutations acquired at relapse divided by the number of patients without mutations in the corresponding gene at diagnosis. (C-D) Impact of NPM1c loss at relapse on relapse-free survival and overall survival. ∗Two of the 6 patients who lost NPM1c at relapse underwent mutational analysis at diagnosis before referral to our center.
Clonal architecture of AML with mutated NPM1 at relapse. (A) Change in the ELN risk classification at relapse. (B) Mutational evolution, including stability, gain, or loss of mutations at relapse. N is the number of patients with the corresponding mutation at diagnosis among those evaluable by mutational analysis performed at diagnosis and at the time of relapse. The percentages for stability and loss were calculated as the number of patients with mutations that persisted or were lost at relapse divided by patients with mutations in the corresponding gene present at diagnosis. Percentage gain was calculated as the number of patients with mutations acquired at relapse divided by the number of patients without mutations in the corresponding gene at diagnosis. (C-D) Impact of NPM1c loss at relapse on relapse-free survival and overall survival. ∗Two of the 6 patients who lost NPM1c at relapse underwent mutational analysis at diagnosis before referral to our center.
Mutational co-occurrence patterns at relapse of AML with NPM1c were mostly similar to patterns described in the frontline setting with co-occurrence of NPM1c with DNMT3A, FLT3, IDH1, and IDH2 mutations.1 In this R/R cohort, NPM1c more commonly co-occurred with DNMT3A (50% vs 17%; P < .0001), FLT3-ITD (49% vs 11%; P < .0001), IDH1/2 (20% vs 9%; P < .0001), and WT1 (19% vs 10%; P = .009) mutations compared with NPM1wt (Table 1). Most FLT3-ITD cases (71%) were detected at a high allelic ratio (AR) (AR > 0.5). In contrast, mutations in ASXL1, RUNX1, TET2, and TP53 co-occurred less commonly with NPM1c than with NPM1wt patients (5% to 31% vs 22% to 65%; P < .0001).
Impact of comutations on outcomes in NPM1c R/R AML
Given that the favorable prognosis associated with NPM1c is context dependent and particularly influenced by the co-occurrence of FLT3-ITD mutations, we examined the responses corresponding to these specific comutations following salvage 1 (supplemental Table 4). We observed that response rates were similar in patients with NPM1c with or without co-occurring FLT3-ITD mutations (CR/CRi, 55% vs 58% respectively; P = .9), with a trend for an improved OS in patients with NPM1c and wild-type FLT3 compared with NPM1c and FLT3-ITD comutations (median OS, 8.6 vs 5.8 months; P = .05) (supplemental Figure 1).
Similarly, NPM1c with co-occurring mutations in DNMT3A, IDH1, TET2, or RAS had similar response rates compared with NPM1c and the corresponding wild-type genes. However, NPM1c and IDH2 comutations was associated with a higher CR rate compared with NPM1c and IDH2 wild-type (CR rates, 50% vs 27%; P = .03) in addition to an improved OS (median OS, 14.5 vs 5.8 months, respectively; P = .04) (supplemental Table 4, supplemental Figure 1). TET2 comutations with NPM1c was associated with worse OS compared with NPM1c and wild-type TET2 (median OS, 5.1 vs 8.3 months, respectively; P = .01) (supplemental Table 4, supplemental Figure 1).
NPM1c loss at relapse
NPM1c is considered a founding leukemia event that is stable throughout the disease process and therefore has been used as a surrogate for MRD.18 However, in a small fraction of cases, NPM1c is lost at relapse.11,12 Among patients with newly diagnosed AML with NPM1c, 3% (6 of 212 patients) relapsed without NPM1c. NPM1c loss at relapse was associated with improved RFS and OS compared with persistence of NPM1c, although the numbers were small for this comparison (n = 6 vs n = 206) (1-year RFS, 80% vs 34%; P = .5; median OS, NR vs 6.1 months; 1-year OS, 83% vs 30%; P = .002) (Figure 1C,D). This highlights the possibility that NPM1c loss at relapse may represent a de novo leukemia.11
Outcomes by line of therapy in NPM1c R/R AML
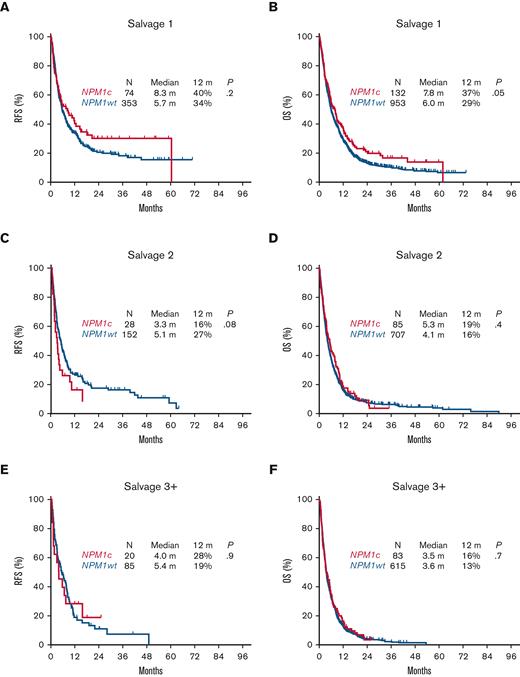
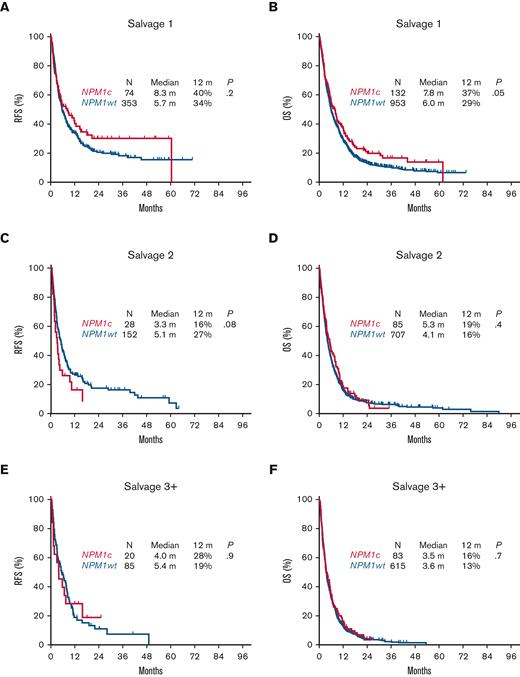
As expected, the response rates decreased sequentially with each line of salvage therapy for all patients with R/R AML. Patients with NPM1c had higher response rates compared with those with NPM1wt following S1 with a CR/CRi rate of 56% vs 37%, respectively (P < .0001), and a significant but less pronounced difference with subsequent lines of therapy (S2, 33% vs 22%; P = .02; ≥S3, 24% vs 14%; P = .02) (Table 2). There was no significant difference in 30-day mortality between NPM1c and NPM1wt, regardless of salvage regimen (9% each in S1, 17% vs 13% in S2, and 13 vs 14% in ≥S3).
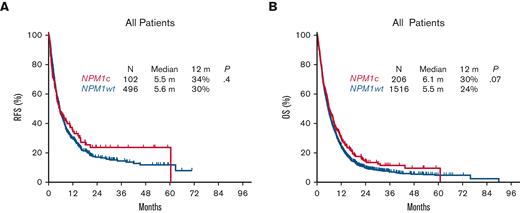
However, despite the relatively higher response rates associated with NPM1c AML, there was no significant difference in RFS or OS compared with NPM1wt in the aggregate population, with a median RFS of 5.5 vs 5.6 months (P = .4) and a median OS of 6.1 vs 5.5 months respectively (P = .07) (Figure 2A,B). Albeit a trend for an improved RFS and OS associated with NPM1c following salvage 1 (median RFS, 8.3 vs 5.7 months; P = .2; median OS, 7.8 vs 6.0 months; P = .05), survival outcomes were similar with subsequent salvage lines of therapy (median RFS, 3.3 vs 5.1 months; P = .08; median OS, 5.3 vs 4.1 months; P = .4) in salvage 2, and (median RFS, 4.0 vs 5.4 months; P = .9; median OS, 3.5 vs 3.6 months; P = .7) in salvage 3 (Figure 3).
Survival associated with NPM1 mutational status at AML relapse.
Survival associated with NPM1 mutational status at AML relapse.
Relapse-free survival and overall survival for patients with relapsed or refractory AML with NPM1c by line of therapy. (A) Relapse-free survival following S1. (B) Overall survival following S1. (C) Relapse-free survival following S2. (D) Overall survival following S2. (E) Relapse-free survival following S3+. (F) Overall survival following S3+.
Relapse-free survival and overall survival for patients with relapsed or refractory AML with NPM1c by line of therapy. (A) Relapse-free survival following S1. (B) Overall survival following S1. (C) Relapse-free survival following S2. (D) Overall survival following S2. (E) Relapse-free survival following S3+. (F) Overall survival following S3+.
When restricting the analysis to patients with R/R AML and a diploid karyotype only, there was no difference in RFS or OS according to NPM1 mutational status. The median OS for diploid R/R NPM1c AML was 8.0 months vs 7.9 months in those with diploid R/R NPM1wt AML (P = .2) (supplemental Figure 2). Similarly, there was no difference in RFS or OS according to NPM1 mutational status in the subgroup of patients with R/R AML below the age of 60 years or in the subgroup above this age cut-off (supplemental Figure 3).
Outcomes by type of therapy in NPM1c R/R AML
Combinations with Venetoclax
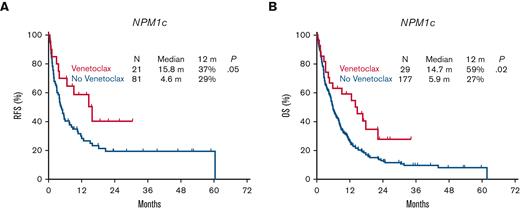
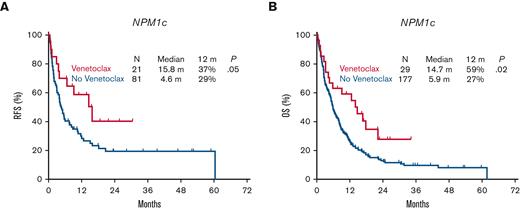
Patients with R/R NPM1c AML treated with HI regimens had higher response rates than those with NPM1wt, with a CR/CRi rate of 63% vs 37%, respectively (P < .0001) (Table 2). Conversely, there was no impact of the NPM1 mutational status on response rates when LI regimens were used (CR/CRi, 34% with NPM1c vs 26% with NPM1wt; P = .1) (Table 2). However, the addition of venetolax to LI regimens used in salvage therapy led to an improved response rate in patients with NPM1c, with a CR/CRi rate of 71% vs 32% in those with NPM1wt (P = .02). This in turn led to improved RFS with a median of 15.8 months for NPM1c patients who received venetoclax vs 4.6 months for NPM1wt (P = .05), and an improved OS with a median of 14.7 months vs 5.9 months, respectively (P = .02) (Figure 4). These outcomes in patients with NPM1c associated with LI and venetoclax matched those associated with standard HI regimens with a median OS of 14.5 months vs 8.1 months respectively (P = .4) (supplemental Figure 4). Only 5 patients received HI regimens with the addition of venetoclax for salvage at the time of this analysis, thereby limiting the comparison of outcomes.
Impact of the addition of venetoclax on survival in relapsed or refractory AML with NPM1c.
Impact of the addition of venetoclax on survival in relapsed or refractory AML with NPM1c.
Outcomes with other targeted therapies
The advent of therapies targeting specific mutations has increased the treatment arsenal for AML, particularly in patients with NPM1c, where mutations in FLT3 or IDH frequently co-occur. The use of an FLT3 inhibitor, either alone or in combination, was associated with a CR/CRi rate of 57% (29/56 patients) in patients with NPM1c and FLT3 comutations (CR/CRi of 43% with FLT3 inhibitor alone and 59% with FLT3 inhibitor combinations) (supplemental Tables 5 and 6). In contrast, IDH inhibitor-based therapies had an associated CR/CRi rate of 33% (2/6 patients) in patients with NPM1c and IDH comutations. Only 2 patients with NPM1c received gemtuzumab ozogamicin, 1 of them achieved CRi (supplemental Tables 5 and 6).
Impact of HSCT in NPM1c R/R AML
Among patients evaluated in this analysis, 211 (12%), including 197 with CR or CRi and 14 with other responses, received an allogeneic hematopoietic stem cell transplant (HSCT). Among them, 149 (70%) underwent transplantation after achieving remission following their first relapse. The proportion of patients who received an allo-HSCT was similar in the NPM1c and NPM1wt groups (all salvage, 16% vs 12%; P = .1; S1, 19% vs 13%, P = .06; S ≥ 2, 11% vs 9%, P = .6). The median time from the start of therapy to HSCT was 2.8 months (range, 0.5 to 14.3).
In a landmark analysis, HSCT was associated with improved RFS and OS in patients with R/R AML with NPM1c, regardless of the salvage line status. The median RFS for patients with NPM1c and HSCT was 20.7 months compared with 4.0 months for those with NPM1c who did not have HSCT (P < .0001), whereas the median OS was 22.2 months vs 8.6 months, respectively (P < .0001) (supplemental Figure 5).
Multivariate analysis
To assess the independent prognostic effect of various factors in this group of patients with R/R AML (both NPM1c and NPM1wt), we performed univariate and multivariate analyses, including baseline characteristics, mutational and cytogenetic status, duration of first remission, and type of therapy received. The NPM1 mutational status had no effect on OS in this analysis (supplemental Table 7). The only independent factors identified that predicted worse OS in this group of patients with R/R AML included older age, mutated TP53, and duration of first remission of less than 6 months (supplemental Table 7).
Discussion
In this study, we found that AML with NPM1c at relapse had similar survival outcomes to those with the wild-type gene. Despite a marginal increase in response rates following salvage therapy, NPM1c was not associated with an improved RFS or OS when all therapies were considered. In a limited analysis, we found significantly improved response rates when venetolcax was added to therapy, leading to a decreased risk of relapse and an improved overall survival. However, a longer follow-up with larger cohorts of patients is needed to validate this finding. It is unclear whether this susceptibility is related to comutations with IDH, an established vulnerability to BCL2 inhibition, or is broadly applicable to all patients with mutated NPM1.19
The proportion of comutations in AML with NPM1c at relapse seemed mostly similar to the previously described genomic composition at diagnosis, albeit a relatively higher frequency of FLT3-ITD (49% vs 39% in Papaemmanuil et al).1 Previous analyses have shown an increase in high risk copy number alterations at relapse of AML with NPM1c when using methods with an improved resolution compared with conventional cytogenetics available in our analysis.11 In addition, gain of distinct FLT3-ITD clones at relapse, despite relatively preserved mutational proportions compared with what is seen at diagnosis, or selection of inherently resistant leukemia cells following induction therapy (NPM1c forms 30% to 35% of cases at diagnosis vs 12% at relapse), could explain the observed resistance and poor outcomes in this setting.10 This pattern of mutations at relapse differs from what is expected when all patients with FLT3 mutations receive a frontline FLT3 inhibitor, which leads to loss of these mutations at relapse in some. Notably, this cohort also included FLT3 wild-type and older or unfit patients with AML who received frontline LI therapies without the addition of a FLT3 inhibitor.17,20
Interestingly, we found that NPM1c loss at relapse (seen in about 3% of patients) was associated with relatively improved outcomes similar to what has been previously described, further justifying the concept that these leukemias could be arising from a de novo clone rather than persistence and evolution of the original founding clone.10-12 This could affect use of NPM1c for MRD monitoring, therefore addition of phenotypic MRD assays such as multicolor flow cytometry would be complimentary. It remains unclear if this rare occurrence justifies the need to confirm NPM1 mutational status for trials investigating NPM1c directed therapies in the R/R setting but could be justified for registrational studies.
The study is limited by the retrospective nature of the analysis, genomic and biological heterogeneity of the subsets included, and the use of various types of therapy in a single center. Therefore, these results must be interpreted within the context of these limitations.
There is relatively no data on the outcomes of relapsed AML with NPM1c. To the best of our knowledge, this is the first study to examine the outcomes of these patients with each line of therapy. The median OS of AML with NPM1c following S1 was 7.8 months, with a decrease to 5.3 months following S2, and 3.5 months following S3 and beyond. These dismal outcomes indicate the unmet need for novel therapeutic strategies. Among salvage therapies for AML with NPM1c, HI treatment regimens and the addition of venetoclax to LI regimens appeared to be the most advantageous. However, comparison across these therapy options is limited by considerations such as age and fitness. Nevertheless, HI regimens or venetoclax combinations are likely to be the preferred backbone for the addition of novel agents to this entity. Notable examples include menin or spleen tyrosine kinase (SYK) inhibitors.21,22 Early results from the ongoing phase I trials investigating menin inhibition in this population are encouraging (NCT04065399; NCT04067336; NCT04752163; NCT04811560; NCT05153330; NCT04811560).23,24
In conclusion, AML with NPM1c is associated with poor outcomes at relapse. The use of HI regimens and/or the addition of venetoclax to salvage therapy was associated with improved outcomes in patients with NPM1c in this setting. Combination strategies incorporating emerging novel therapies should be rapidly evaluated to further improve outcomes and long-term survival.
Acknowledgment
G.C.I. received funding through the K12 Paul Calabresi Clinical Scholarship Award (NIH/NCI K12 CA088084).
Authorship
Contribution: G.C.I., A.B., S.V., and N.D. designed the study and wrote the manuscript; S.V. and A.B. analyzed the data; M.K., C.D.D., T.M.K., G.B., E.J., N.P., M.Y., N.J.S., A.M., K.S., L.M., S.P., K.T., G.T., S.L., K.P., M.A., K.B., G.G.M., F.R., and H.K. provided suggestions and revised the manuscript; G.C.I. and N.D. supervised the analysis; and all authors read and approved the final version of the manuscript.
Conflict-of-interest disclosure: G.C.I. reports research funding from Celgene, Kura Oncology, Syndax, and Novartis, and received consultancy fees from Novartis and Kura Oncology. M.K. reports grant support from Agios, Rafael Pharmaceuticals, Cellectis, Ascentage, AstraZeneca, Sanofi, Ablynx, Genentech, Forty Seven, AbbVie, F Hoffmann-La Roche, and Calithera; research funding from Agios, Rafael Pharmaceuticals, KisoJi, Eli Lilly, Ascentage, AstraZeneca, Sanofi, Ablynx, Genentech, Forty Seven, AbbVie, Stemline Therapeutics, and Calithera; intellectual property rights from Eli Lilly, Novartis, and Reata Pharmaceuticals; current stock options in Reata Pharmaceuticals; consultancy for Genentech, F Hoffmann-La Roche, and AbbVie; and honoraria from Genentech, F Hoffmann-La Roche, and AbbVie. C.D.D. reports membership on an entity's Board of Directors or advisory committees from GlaxoSmithKline and Notable Labs; current stock options in a privately-held company in Notable Labs; honoraria from Immune-Onc, Bristol Myers Squibb, Agios/Servier, Takeda, Novartis, Foghorn, Celgene, a Bristol Myers Squibb company, and Forma; research funding from Immune-Onc, Bristol Myers Squibb, AbbVie, Agios/Servier, Foghorn, Celgene, a Bristol Myers Squibb company, and Forma; and consultancy from AbbVie and Agios/Servier. T.M.K. reports consultancy from AbbVie, Sanofi-Aventis, Liberum, Jazz, Genentech, Daiichi Sankyo, Novartis, Pfizer, and Aglos; grant or research support from AbbVie, Genentech, Bristol Myers Squibb, and Amgen; speaker's bureau fees from Cure; and research support from Genfleet, Cellonkos, Astellas, AstraZeneca, Ascentage, and Pulmotech. G.B. reports consultancy fees from Protagonist Therapeutics, Novartis, and GlaxoSmithKline (GSK); research funding from Ryvu and Astex; and membership to an entity’s Board of Directors or advisory committees from Takeda, and ArgenX. E.J. reports research grants from AbbVie, Adaptive Biotechnologies, Amgen, Pfizer, and Takeda, and consultancy fees from AbbVie, Adaptive Biotechnologies, Amgen, Bristol Myers Squibb, Genentech, Incyte, Novartis, Pfizer, and Takeda. N.P. reports consultancy from DAVA Oncology, LFB Biotechnologies, MustangBio, Stemline Therapeutics, Affymetrix, Roche Diagnostics, Blueprint Medicines, Clearview Healthcare Partners, Novartis Pharmaceuticals, Celgene Corporation, Incyte, Protagonist Therapeutics, AbbVie Pharmaceuticals, Aptitude Health, Bristol Myers Squibb, ImmunoGen, Pacylex Pharmaceuticals, CareDx, Daiichi Sankyo, Springer Science + Business Media, Samus, Plexxicon, and Cellectis. N.J.S. reports consultancy for Takeda Oncology, Jazz Pharmaceuticals, NGMBio, AstraZeneca, and Amgen; research funding from Takeda Oncology and Astellas; honoraria from Amgen and Novartis. K.S. reports consultancy and research funding from Novartis, and membership on the Board of Directors or advisory committees of Pfizer and Daiichi-Sankyo. K.T. reports consultancy for Novartis, Celgene/Bristol Myers Squibb, GSK, and Symbio Pharmaceuticals; and membership on Symbio Pharmaceuticals, Board of Directors or advisory committee. M.A. reports consultancy from Daiichi Sankyo, Inc., Amgen, and AstraZeneca; patents licensed, royalty bearing and research funding from Jazz Pharmaceuticals; equity ownership with Aptose, Eutropics, Senti Bio, Oncolyze, BrooklynITX, SAB, Chimerix, and Reata; membership with SAB; research funding from the Breast Cancer Research Foundation; and membership with BiolineRx on an entity's Board of Directors or advisory committees. F.R. reports honoraria from Astex, Taiho, Bristol Myers Squibb, Xencor, Agios, AstraZeneca, Novartis, AbbVie, Celgene, Jazz, Syros Pharmaceuticals, and Amgen; research funding from Astex, Taiho, Bristol Myers Squibb, Xencor, Agios, Prelude, AbbVie, Celgene, Jazz, Syros Pharmaceuticals, and Amgen; membership on the Board of Directors or advisory committees of Bristol Myers Squibb and Celgene; and consultancy for Syros Pharmaceuticals. H.K. reports research funding from Ariad, Astex, Bristol Myers Squibb, Cyclacel, Daiichi-Sankyo, Pfizer, Immunogen, Jazz, Novartis, and honoraria from Pfizer, Immunogen, Actinium, and Takeda. N.D. reports consultancy for Novartis, Trovagene, AbbVie, Genentech, Amgen, Pfizer, Bristol Myers Squibb, Astellas, Daiichi Sankyo, Sevier, ImmunoGen, Gilead Sciences, Trillium, Dava Oncology (Arog), Celgene, Syndax, Shattuck Labs, Agios, Kite Pharmaceuticals, SOBI, STAR Therapeutics, and Jazz Pharmaceuticals; is a member of the Data Monitoring Committee for Jazz Pharmaceuticals; and research funding from Trovagene, Genentech, FATE Therapeutics, Hanmi, Amgen, Pfizer, Bristol Myers Squibb, Novimmune, Astellas, Daiichi Sankyo, Sevier, ImmunoGen, Glycomimetics, Gilead Sciences, Trillium, Karyopharm, Newave, and AbbVie. The remaining authors declare no competing financial interests.
Correspondence: Ghayas C. Issa, Department of Leukemia, The University of Texas MD Anderson Cancer Center, 1400 Holcombe Blvd, Unit 428, Houston, TX 77030; e-mail: gcissa@mdanderson.org; and Naval Daver, Department of Leukemia, The University of Texas MD Anderson Cancer Center, 1400 Holcombe Blvd, Unit 428, Houston, TX 77030; e-mail: ndaver@mdanderson.org.
References
Author notes
Data are available on request from the corresponding authors, Ghayas Issa (gcissa@mdanderson.org) or Naval Daver (ndaver@mdanderson.org).
The full-text version of this article contains a data supplement.
G.C.I., A.B., and S.V. contributed equally to this work.