Key Points
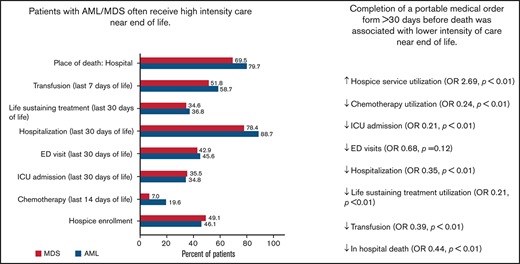
Patients with AML or MDS often receive high-intensity care near EOL, including admission to the hospital/ICU and life-sustaining treatments.
Completion of a portable medical order form >30 days before death was associated with lower intensity care near the end of life.
Abstract
Patients with acute myeloid leukemia (AML) or a myelodysplastic syndrome (MDS) experience high rates of hospitalization, intensive care unit (ICU) admission, and in-hospital death at the end of life. Early goals-of-care (GOC) discussions may reduce the intensity of end-of-life (EOL) care. Portable Medical Order forms, known as Medical Orders for Life-Sustaining Treatment (MOLST) forms in New York state, assist patients in translating GOC discussions into specific medical orders that communicate their wishes during a medical emergency. To determine whether the timing of completion of a MOLST form is associated with EOL care in patients with AML or MDS, we conducted a retrospective study of 358 adult patients with AML or MDS treated at a single academic center and its affiliated sites, who died during a 5-year period. One-third of patients completed at least 1 MOLST form >30 days before death. Compared with patients who completed a MOLST form within 30 days of death or never, those who completed a MOLST form >30 days before death were less likely to receive transfusion (adjusted odds ratio [AOR], 0.39; P < .01), chemotherapy (AOR, 0.24; P < .01), or life-sustaining treatments (AOR, 0.21; P < .01) or to be admitted to the ICU (AOR, 0.21; P < .01) at EOL. They were also more likely to use hospice services (AOR, 2.72; P < .01). Earlier MOLST form completion was associated with lower intensity of care near EOL in patients with MDS or AML.
Introduction
Acute myeloid leukemia (AML) and myelodysplastic syndromes (MDS) are life-threatening hematologic malignancies that often require intensive supportive therapy. Despite many advances in treatment, these diseases prove fatal for most patients. Patients with hematologic malignancies are more likely to receive high-intensity end-of-life (EOL) care than patients with solid tumors.1-3 Studies focused on EOL care in AML have demonstrated high rates of hospitalization within the last 30 days of life, intensive care unit (ICU) admission within the last 30 days of life, and in-hospital death, as well as low use of hospice services.1-5 In a Surveillance, Epidemiology, and End Results–Medicare database study of patients with MDS, 28% of patients were admitted to the ICU within the last 30 days of life, and 49% were enrolled in a hospice.5 Although high-intensity care leads to remissions and cure in some cases, it often does not. High-intensity care is associated with worse quality of life near the end of life.6,7 In patients with incurable hematologic malignancies, high-intensity EOL care occurs for a variety of reasons, including patients’ desire for all available medical interventions, patients’ and family members’ difficulty in understanding the limitations of life-sustaining treatments,8-10 lack of provider training for conducting goals-of-care (GOC) conversations,10 provider concern about taking away hope,11 clinicians’ unrealistic expectations,11 and providers’ uncertainty about prognosis.8-12 These issues may contribute to circumstances in which patients receive futile or unwanted medical care that ultimately does not improve their outcomes or EOL experience.
Early palliative care referrals and/or early GOC discussions may limit the intensity of EOL care for patients with hematologic malignancies.12 In a study that included 366 patients with advanced cancer (29 of whom had a hematologic malignancy), palliative care referrals made >3 months before death were associated with decreased emergency department (ED) visits or hospitalizations within the last 30 days of life and decreased likelihood of death during a hospital admission.12 In a separate study focused on patients with hematologic malignancies using the Surveillance, Epidemiology, and End Results–Medicare registry, the use of “early” billed palliative care services (initiated >30 days before death) was associated with a lower likelihood of ED visits, hospitalizations, or ICU admissions in the last 30 days of life and a lower likelihood of dying while in the hospital.13 Another study of 383 patients with hematologic malignancies showed that having the first GOC discussion >30 days before death and having a hematologic oncologist present were associated with lower odds of ICU admission within the last 30 days of life and earlier hospice use.14 More studies are needed to determine the impact of early GOC discussions on EOL measures in specific subsets of hematologic malignancies, such as AML and MDS, which generally have poorer prognoses than other hematologic malignancies such as lymphoma and multiple myeloma.
Portable medical order (POLST; www.polst.org) forms are designed for patients who are seriously ill, to translate GOC discussions into specific medical orders that communicate the patient’s wishes during a medical emergency. These orders usually include the patient’s preferences for several medical interventions including cardiopulmonary resuscitation. In studies conducted primarily in patients with solid tumors, POLST forms were completed in a minority (22% to 35%) and were associated with a decreased likelihood of in-hospital death and increased use of hospice care.15,16 Little is known about the use of POLST forms in specific subsets of hematologic malignancies and their association with EOL measures.
The goal of this study was to better understand the use of POLST forms in patients with AML or MDS and to assess its association with the use of EOL measures.
Methods
Study design, setting, and sample
We conducted a retrospective study of consecutive patients aged ≥18 years who were treated for AML or MDS at a single academic cancer center and its affiliated practices and died from 1 January 2014 to 31 December 2019. This research was approved by the University of Rochester Research Subjects Review Board.
Data collection
A trained investigator (M.L.) extracted demographic and clinical information from the electronic medical record, which contains data from the 2 largest health care systems in Rochester, New York, as well as scanned notes from other health care systems. Demographic data included age at diagnosis, sex, race and ethnicity, and ZIP code (used to categorize residence as urban vs rural based on the 2010 Rural-Urban Commuting Areas Codes (RUCAs) Categorization C.17,18 Clinical data included all comorbidities (without the use of standard comorbidity indices), Eastern Cooperative Oncology Group performance status (ECOG-PS), diagnosis (AML and/or MDS), and intent of care as stated in the treating oncologist’s note (curative, palliative, or not stated). We also collected dates of palliative care referral and visit. Any ambiguous cases were discussed with senior authors (K.P.L. and/or J.H.M.).
POLST form
MOLST form (Medical Orders for Life-Sustaining Treatment, the term used for the POLST form in New York State) data were extracted from the electronic medical record (only data from scanned copies of forms were used). For each patient, the number of MOLST forms completed, with the date of each form, was recorded. Data collected from each MOLST form included the patient’s preferences for cardiac resuscitation, intubation, future hospitalization/transfer, artificially administered fluids, and nutrition (ie, feeding tube and intravenous fluids), and antibiotics. In addition, we also recorded which individual completed the MOLST form (ie, oncologist vs other). Throughout the study period, there were no changes in how POLSTs were completed, entered into the medical record, or displayed in the medical record during the study period.
EOL measures
We collected EOL measures that have been established in published studies and are endorsed by the American Society of Clinical Oncology (ASCO)1-4 : (1) place of death (hospital, home, or other), (2) transfusion within the last 7 days of life, (3) use of life-sustaining treatment (eg, mechanical ventilation, vasopressors, tracheostomy, dialysis for acute kidney injury, percutaneous endoscopic gastrostomy, and cardiopulmonary resuscitation)19 ; (3) hospital admission, ED visit, and ICU admission within the last 30 days of life; (4) chemotherapy within the last 14 days of life; and (5) hospice enrollment.
Statistical analyses
Descriptive statistics including medians and interquartile ranges (IQRs) for continuous variables and counts and proportions of categorical variables were used to describe the study population as a whole and within AML and MDS cohorts. We summarized time from first hematology visit, MOLST form completion (including who completed the MOLST), palliative care referral, palliative care visit, and hospice enrollment, to date of death in the AML and MDS cohorts to understand the timing of these interventions. Among the patients who completed a MOLST form, we compared time from completion of the form to death according to specialty of the provider who completed the form, using the nonparametric Kruskal-Wallis test.
To better understand the timing of MOLST form completion and palliative care referral/visit relative to first hematology visit, we used cumulative incidence functions to estimate the probability of MOLST form completion and palliative care referral/visit within 12 weeks of the first hematology visit, accounting for the competing risk of death. We chose 12 weeks, as ASCO recommends palliative care referral within 8 weeks of the first hematology visit.20 Because many patients in this study did not have their diagnosis at the time of the first visit, we chose to extend the period to 12 weeks to account for the time of diagnosis.
To evaluate the association of MOLST form completion with EOL measures, we divided the patients into those who completed the first MOLST form >30 days before death (early) vs those who never completed or completed the first MOLST form within 30 days of death (delayed). We chose 30 days based on the ASCO Quality Oncology Practice Initiatives and the results of a prior study.21 In addition, given that many of our EOL measures were defined within 30 days of death, using a cutoff of 30 days rather than yes/no responses for MOLST form completion eliminated cases in which the MOLST form was completed after the outcome (eg, hospice enrollment). We did not exclude patients who survived <30 days from their AML or MDS diagnosis. Fisher’s exact test was used to identify the characteristics of patients that were associated with early MOLST form completion. We used multivariate logistic regression to model the association of early form completion on EOL measures, adjusted for age, race, number of comorbidities, diagnosis, disease status at first hematology visit, and geographical location (rural vs urban). We did not include ECOG-PS and intent of care in the models because of the large amount of missing data. We explored interactions between early MOLST form completion and each adjustment variable; significant interactions are highlighted in “Results.”
For sensitivity analyses, we excluded patients who died within the first 30 days of diagnosis and repeated the multivariate logistic regression. All reported P-values are 2-sided, with P < .05 indicating statistical significance. All analyses were performed with SAS 9.4 (SAS Institute, Inc, Cary, NC).
Results
Demographics
We included 358 patients: 238 had AML and 120 had MDS (Table 1). Among the patients with AML, the median age was 67 years (range 20-95) and 84% were white. The median age of patients with MDS was 75 years (range, 26-93). Median times from first hematology visit to death in patients with AML and MDS were 9.8 (IQR, 18.3) and 17.9 (IQR, 28.1) months, respectively.
MOLST forms
Of 358 patients, 12 had transferred to our institution, and data on their completion of a MOLST form were unavailable. A large proportion (85.8%; 297of 346) of the patients had completed a MOLST form before death. One-third of the patients (118 of 346) completed their first MOLST form >30 days of death, 51.7% (179 of 346) completed the form within 30 days of death, and 14.2% (49 of 346) never completed the form.
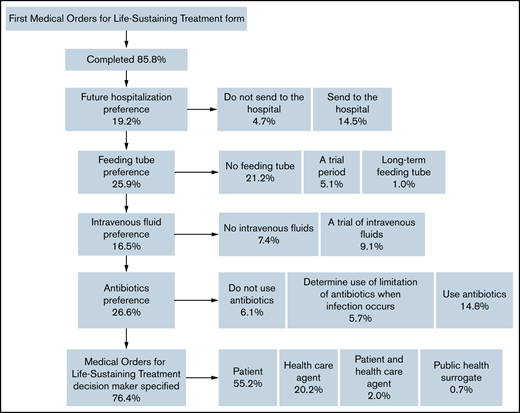
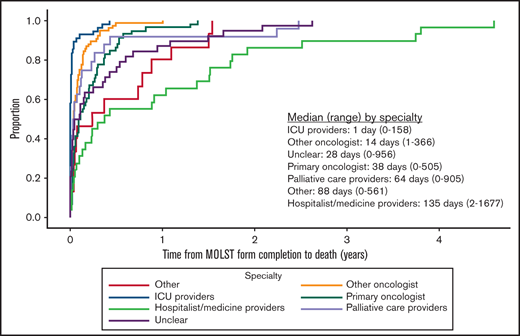
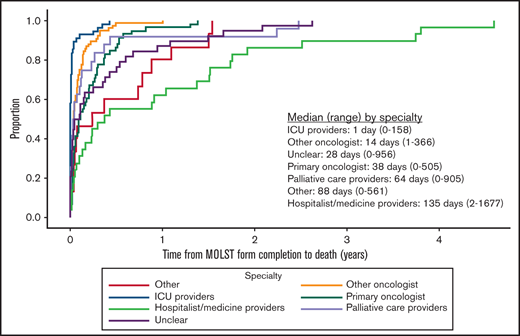
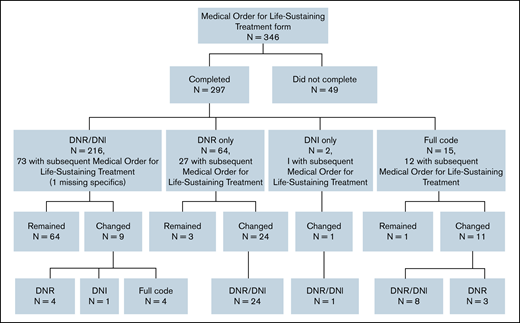
Among those who had a MOLST form completed before death, 4.0% (12 of 297) completed the form before the initial hematology visit, and 96.0% (285 of 297) completed it on the date of or after the initial hematology visit. Approximately two-thirds (184 of 297; 61.6%) had completed 1 MOLST form, 29.6% (88 of 297) had completed 2, and 8.4% (25 of 297) had completed ≥3; 5.1% (15 of 297) of those completing their first MOLST forms requested full code. Do-not-resuscitate (DNR) and do-not-intubate (DNI) orders were present in 72.7% (216 of 297), DNR-only orders in 21.6% (64 of 297), and DNI-only orders in 0.7% (2 of 297). Preferences for future hospitalization/transfer (19.2%; 57 of 297), feeding tube (25.9%; 77 of 297), IV fluids (16.5%; 49 of 297), and antibiotics (26.6%; 79 of 297) were indicated (Figure 1). The primary oncologist completed the first MOLST form in 19.5% (58 of 297) of cases; the remaining MOLST forms were completed by other oncologists (25.6%; 76 of 297), hospitalist or other medicine providers (9.8%; 29 of 297), ICU providers (18.5%; 55 of 297), palliative care providers (8.1%; 24 of 297), or other (5.1%; 15 of 297). We were not able to determine the clinician subspecialty (unclear) in 13.5% (40 of 297), usually because the signature on the MOLST form was illegible. Time from initial MOLST form completion to death, as a function of clinician subspecialty, is shown in Figure 2; there was a significant difference among the various specialties (P < .0001). Among patients who had DNR and/or DNI orders (n = 282), 4 changed to full code in a subsequent MOLST completion (Figure 3). Among patients who did not have DNR/DNI orders (15 of 297) in the first MOLST, 11 changed to DNR and/or DNI in a subsequent MOLST.
Flowchart describing patient preferences on the first MOLST form. IV, intravenous fluids; MOLST, Medical Orders for Life-Sustaining Treatment.
Flowchart describing patient preferences on the first MOLST form. IV, intravenous fluids; MOLST, Medical Orders for Life-Sustaining Treatment.
Time from initial MOLST form completion to death as a function of clinician subspecialty. “Unclear” subspecialty refers to providers who could not be identified because the signature on the MOLST form was illegible.
Time from initial MOLST form completion to death as a function of clinician subspecialty. “Unclear” subspecialty refers to providers who could not be identified because the signature on the MOLST form was illegible.
Flowchart depicting changes in code status as stated on the MOLST forms. MOLST, Medical Order for Life-Sustaining Treatment.
Flowchart depicting changes in code status as stated on the MOLST forms. MOLST, Medical Order for Life-Sustaining Treatment.
Compared with those who completed a MOLST form within 30 days of death or never completed a MOLST form, those who completed a MOLST form >30 days before death were more likely to be older (median, 73.5 vs 67.0 years; P < .0001), have more comorbidities (median, 4 vs 3; P = .01), and have palliative intent documented in their medical record (52.5 vs 38.6%; P = .03; Table 2).
EOL measures
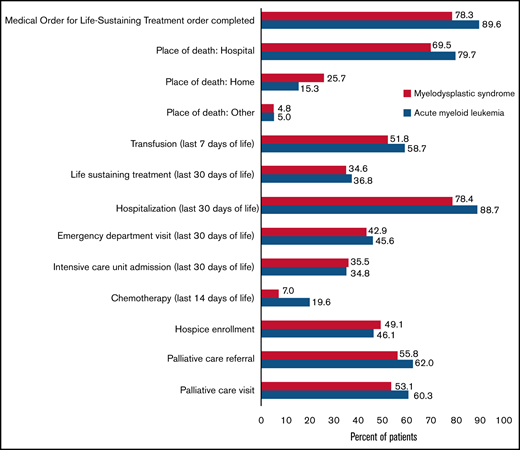
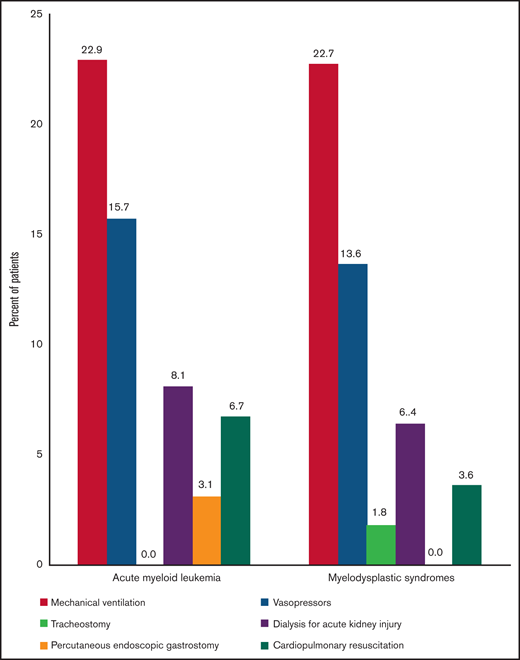
Figure 4 shows the EOL measures for AML and MDS. Life-sustaining treatment was used in 36.8% (82 of 223) and 34.6% (38 of 110) of patients with AML and MDS, respectively, within the last 30 days of life. Figure 5 demonstrates the use of various life-sustaining treatments in patients with AML and MDS. Among patients with AML (n = 223), 14.8%, 12.1%, and 9.9% used 1, 2, and ≥3 types of life-sustaining treatment, respectively. Among patients with MDS (n = 110), 17.3%, 7.3%, and 10% used 1, 2, and ≥3 types of life-sustaining treatment, respectively. In terms of hospice, 46.1% of patients with AML (n = 105) and 49.1% of patients with MDS (n = 55) were enrolled; 27.2% (n = 62) and 28.6% (n = 32) of patients with AML and MDS were enrolled for >3 days, respectively. For those who were in hospice (n = 160), median length of time in hospice was 4.5 days (IQR, 8 days).
EOL measures in patients with AML and MDS. AML, acute myeloid leukemia; ED, emergency department; ICU, intensive care unit; MDS, myelodysplastic syndrome; MOLST; Medical Order for Life-Sustaining Treatment.
EOL measures in patients with AML and MDS. AML, acute myeloid leukemia; ED, emergency department; ICU, intensive care unit; MDS, myelodysplastic syndrome; MOLST; Medical Order for Life-Sustaining Treatment.
Table 3 demonstrates the timing between completion of the MOLST form, referral to palliative care, visit from palliative care, and hospice enrollment and date of death. The probability of palliative care referral, palliative care visit, and completing a MOLST form within 12 weeks of diagnosis was 16.7%, 16.2%, and 23.7%, respectively for AML. The probability of palliative care referral, palliative care visit, and completing a MOLST form within 12 weeks of diagnosis was 7.1%, 6.3%, and 12.0%, respectively for MDS.
Multivariate analyses
On multivariate analyses, early completion of a MOLST form (vs delayed completion or form never completed) was associated with lower use of transfusion (adjusted odds ratio [AOR] 0.39; 95% confidence interval [CI], 0.24-0.64; P = .0002), life-sustaining treatment (AOR, 0.21; 95% CI, 0.11-0.38; P < .0001), ICU admission (AOR, 0.21; 95% CI, 0.11-0.39; P < .0001), and chemotherapy (AOR, 0.24; 95% CI, 0.10-0.59; P = .002) near the end of life, as well as higher use of hospice services (AOR, 2.69; 95% CI, 1.66-4.38; P < .0001) as well as hospice care for >3 days (AOR, 2.50; 95% CI, 1.49-4.17; P = .001; Tables 4 and 5). Early MOLST form completion and diagnosis showed a significant interaction with in-hospital death (P = .01) and hospitalization (P = .005). Among patients with AML, early completion of a MOLST form was associated with lower odds of in-hospital death (AOR, 0.25; 95% CI, 0.12-0.51; P = .0001) and EOL hospitalization (AOR, 0.15; 95% CI, 0.06-0.37; P < .0001). Among patients with MDS, early MOLST form completion was not significantly associated with in-hospital death (AOR, 1.09; 95% CI, 0.44-2.69; P = .85) or hospitalization (AOR, 0.99; 95% CI 0.37-2.67; P = .99) near the end of life.
In our sensitivity analyses, after excluding 29 patients who died within the first 30 days of diagnosis, multivariate logistic regression demonstrated similar results.
Discussion
It is critical to understand the EOL experience of patients with AML and MDS, to ensure that patient preferences are accounted for in decision making. We found that most patients died in the hospital and that more than one-third were in the ICU and receiving life-sustaining treatment near the end of life. Completion of the MOLST form, palliative care referrals and visits, and hospice enrollment generally occurred very close to the time of death. Patients who completed a MOLST form early (vs late or never) were less likely to receive high-intensity care near EOL.
Relatively few studies have explored EOL care in patients with MDS or AML.3,22 Our findings confirm those of others, demonstrating that most patients with AML are admitted to the hospital near EOL,23 die while in the hospital,23 receive initial palliative care services within the last 2 weeks of life,23 and receive hospice services <7 days before death.3,4 Our finding that approximately one-third of patients with AML and MDS are admitted to the ICU within the last 30 days of life is also consistent with results in other studies.3-5 To our knowledge, our study is the first to investigate how MOLST forms are used in patients with MDS or AML and their association with EOL measures. In prior studies of solid tumors, POLST forms were completed in 22% to 35% of patients, compared with >80% in our study.15,16 The higher rate of completion of MOLST forms in our study may be the result of a higher frequency of health care contacts in AML and MDS and a high proportion of patients being hospitalized near the end of life.3 However, it is important to note that only one-third of patients completed these forms >30 days before death. Those who completed a MOLST form early were less likely to be admitted to or die in the hospital (patients with AML only); to be admitted to the ICU; and to receive blood transfusions, chemotherapy, and life-sustaining treatments near the end of life and were more likely to enroll in hospice compared with those who completed MOLST forms late or never. Our findings suggest that early MOLST completion, reflecting early GOC discussions, may result in lower intensity of care near EOL in patients with AML or MDS. In this regard, our data confirm and extend those of Odejide and colleagues who found that early GOC discussions were associated with decreased ICU admissions within the last 30 days of life14 in patients with hematologic malignancies.
Our study raises the question of why GOC discussions and completion of MOLST forms do not occur earlier in patients with AML or MDS. Barriers may include unrealistic patient/family member expectations,11,24,25 patients’/family members’ difficulty in accepting a poor prognosis,8-10,24 patients’ desire for aggressive treatment,10,24 patients’ incapacity to make GOC decisions,8,9 patients’/family members’ difficulty in understanding the limitations of life-sustaining treatments,8-10 lack of family agreement about GOC,8-10 lack of provider training for conducting GOC conversations,10 provider concern about taking away hope,11 clinicians’ unrealistic expectations,11 and providers’ uncertainty about prognosis.11 In patients with hematologic malignancies, health care providers have difficulty in identifying the EOL phase as a result of the continuing potential for cure of advanced disease and the often-rapid pace of decline near death. It is often challenging for patients and physicians to avoid the use of “aggressive” treatments when there is still a potential for cure. In this regard, “signposts” have been developed to help identify the EOL phase in patients with hematologic malignancies.11 Importantly, most studies addressing barriers to discussing EOL care are from the perspective of health care providers. Studies from patients’ perspectives are needed to determine their willingness to discuss preferences for EOL care earlier in the disease course.
According to the national POLST program, completion of the POLST form is appropriate for patients with serious illness or frailty whose health care provider would not be surprised if they died within a year. We argue that this definition applies to a large percentage of patients with AML or MDS, particularly older patients or younger patients with relapsed/refractory or high-risk disease (eg, those with complex cytogenetics or TP53 mutations). It is our belief that chemotherapy (even if given with curative intent) and GOC discussions/POLST form completion could occur concurrently in those patients so that they have a realistic view of treatment outcomes and a chance to set limits on the intensiveness of EOL care should they so choose. As a prior study suggested,14 hematologic oncologist participation in GOC discussions/POLST completion may be important in guiding the use of intensive treatments and hospice care.
Our study has limitations. First, it included patients who died over a time period (retrospective) rather than patients who were dying currently (prospective).26,27 Because it is impossible to accurately predict death, question is raised of the utility of using a cutoff point for MOLST completion before death. Nonetheless, our study informs the need to investigate the use of POLST forms in a population of patients who are likely to die (eg, poor-risk AML or high-risk MDS and relapsed AML) and to prospectively evaluate the association of MOLST form completion with EOL measures. Second, we acknowledge that patients may have engaged in early GOC discussions but did not complete a MOLST form because their preference was for no limitations to EOL care. MOLST forms may also have been completed and not scanned into the electronic medical record. Third, although our electronic medical record contains data from the 2 largest health care systems in Rochester, New York, and scanned notes from other health care systems, data from patients who received some of their care outside of our system may be missing. Fourth, although our study demonstrates an association between early completion of a MOLST form and EOL measures, it does not establish causation. Completion of a MOLST form may be a proxy for disease progression, illness severity, or transition from disease- to comfort-oriented care, and these patients may have been less likely to use aggressive care because of perceived futility. That said, our findings are consistent with those of other studies that have demonstrated a benefit of early GOC discussions on EOL measures in patients with cancer.6,14,28,29 Moreover, our findings support National Comprehensive Cancer Network palliative care guidelines recommending that GOC discussions begin while patients have a life expectancy of years to months and that decisions be documented in the medical record, including MOLST/POLST.30 Fifth, MOLST/POLST forms are specific to the United States, and the content of these forms varies across states. Thus, the generalizability of our findings to other countries and states must be determined. Sixth, the single-center design and lack of racial and ethnic diversity may also limit the generalizability of our findings. Seventh, although we adjusted our analysis for patient comorbidities, we did not adjust for disease-related characteristics that may affect EOL measures. Despite these limitations, this study provides important data that justify the development of intervention studies to prospectively evaluate the effects of early completion POLST/MOLST forms on EOL care for patients with AML and MDS.
In summary, we found that patients with AML or MDS often receive high-intensity care near EOL and that early MOLST completion is associated with lower intensity care. Future studies should be aimed at better understanding patients’ wishes regarding EOL care, their perceptions of what constitutes patient-centered EOL care, their willingness to engage in earlier GOC conversations, and the impact of early GOC discussions and completion of a MOLST form on EOL measures.
Acknowledgment
The authors thank Susan Rosenthal for editorial assistance.
This research study was supported by LoGerfo Medical Student Fellowship through the University of Rochester Medical Center (M.L.); and National Institutes of Health, National Cancer Institute grant K99CA237744 and the Wilmot Research Fellowship Award (K.P.L.).
Authorship
Contribution: M.L., K.P.L., and J.H.M. conceived and designed the study; A.M.B., performed the analyses; M.L. acquired the data; M.L., K.P.L., and J.H.M. wrote the manuscript; and all authors interpreted the data and critically revised the manuscript.
Conflict-of-interest disclosure: K.P.L. has served as a consultant to Pfizer and Seattle Genetics. The remaining authors declare no competing financial interests.
Correspondence: Jason H. Mendler, James P. Wilmot Cancer Center, University of Rochester School of Medicine and Dentistry, 601 Elmwood Ave, Box 704, Rochester, NY 14624; e-mail: jason_mendler@urmc.rochester.edu.
References
Author notes
K.P.L. and J.H.M. contributed equally to this study.
Requests for data sharing may be submitted to Jason H. Mendler (jason_mendler@urmc.rochester.edu).