Key Points
CB-derived adoptive cell therapy is a feasible and efficacious therapeutic approach for patients with refractory AML.
Treatment response depends on the development of CBU-derived chimerism.
Abstract
We conducted a prospective evaluation of cord blood (CB)–derived adoptive cell therapy, after salvage chemotherapy, for patients with advanced myeloid malignancies and poor prognosis. Previously, we reported safety, feasibility, and preliminary efficacy of this approach. We present updated results in 31 patients who received intensive chemotherapy followed by CB infusion and identify predictors of response. To enhance the antileukemic effect, we selected CB units (CBU) with shared inherited paternal antigens and/or noninherited maternal antigens with the recipients. Twenty-eight patients with acute myeloid leukemia (AML), 2 with myelodysplastic syndrome, and 1 in chronic myeloid leukemia myeloid blast crisis were enrolled; 9 had relapsed after allogeneic transplant. Response was defined as <5% blasts in hypocellular bone marrow at 2 weeks after treatment. Thirteen patients (42%) responded; a rate higher than historical data with chemotherapy only. Twelve had CBU-derived chimerism detected; chimerism was a powerful predictor of response (P < .001). CBU lymphocyte content and a prior transplant were associated with chimerism (P < .01). Safety was acceptable: 3 patients developed mild cytokine release syndrome, 2 had grade 1 and 2 had grade 4 graft-versus-host disease. Seven responders and 6 nonresponders (after additional therapy) received subsequent transplant; 5 are alive (follow-up, 5-47 months). The most common cause of death for nonresponders was disease progression, whereas for responders it was infection. CB-derived adoptive cell therapy is feasible and efficacious for refractory AML. Banked CBU are readily available for treatment. Response depends on chimerism, highlighting the graft-versus-leukemia effect of CB cell therapy. This trial was registered at www.clinicaltrials.gov as #NCT02508324.
Introduction
Patients with relapsed/refractory myeloid leukemia have a dismal prognosis with chemotherapy.1 Novel treatment approaches are necessary to improve outcomes. One such approach, adoptive immunotherapy with HLA haploidentical donor cells, has shown encouraging response rates in patients with acute myeloid leukemia (AML) and survival that exceeds that with chemotherapy alone.2,3 However, the logistics of identification and clearance of haploidentical donors, followed by stem cell mobilization and collection, have limited its use. We reasoned that unrelated cord blood (CB) cells would have similar or superior graft-versus-leukemia (GVL) effects and, because they are banked, would be readily available to treat patients. We conducted a prospective evaluation of CB-based adoptive cell therapy following salvage chemotherapy in patients with refractory AML/myelodysplastic syndrome (MDS) to control their disease and make them eligible for allogeneic transplant. We allowed enrollment of patients with very advanced disease, regardless of the number of prior treatments, and whether they had undergone allogeneic stem cell transplantation. In our initial report, we described feasibility, safety, and transient chimerism with a low incidence of acute graft-versus-host disease (GVHD) and responses in some refractory patients.4
In this article, we present updated outcomes on 31 patients who had measurable refractory disease and received intensive salvage chemotherapy followed by CB infusion. We report responses in 13 (42%) of them, in marked contrast with the published low response rates with chemotherapy only, and we identify patient and CB cell therapy characteristics that were associated with response.
Materials and methods
We briefly summarize eligibility, cord blood unit (CBU) selection for cell therapy, and chemotherapy. For details, refer to our previous publication.4 This study was performed under US Food and Drug Administration application 16423 and is registered at www.clinicaltrials.gov as #NCT02508324. All patients were treated at New York Presbyterian Hospital–Weill Cornell Medicine, and all CBU were obtained from the National Cord Blood Program (NCBP). The study was approved by the Institutional Review Boards of both institutions. Patient eligibility included a confirmed diagnosis of AML (primary induction failure or relapsed refractory)5-7 or myelodysplastic syndrome (MDS; refractory anemia with excess blasts 1 or 2) after failure of ≥2 cycles of chemotherapy, including relapse after allogeneic transplant.
CBU searches were performed using the NCBP standard search algorithm (NCBP online access - Websearch). Selection criteria included a precryopreservation total nucleated cell (TNC) dose of 0.5 to 2.5 × 107 per kilogram, CBU HLA match of 4 to 6 of 6 (HLA-A, HLA-B intermediate resolution, HLA-DRB1 high resolution) with patient, and CBU with shared inherited paternal antigen (IPA) targets and/or noninherited maternal antigen (NIMA) match with patient (supplemental Table 1). CBU with higher HLA allele match were preferred. Donors targeted by donor-specific anti-HLA antibodies (DSAs) were avoided. Patient HLA typing and CBU confirmatory HLA typing were performed at high resolution for HLA-A, HLA-B, HLA-C, and HLA-DRB1 loci (G-level typing) by Histogenetics (Ossining, NY; Clinical Laboratory Improvement Amendments #33D0985173, American Society for Histocompatibility and Immunogenetics #03-1-NY-26-2). Maternal samples of the CBU were also HLA typed to assign maternal and paternal antigens. Patient and CBU KIR genotyping was performed by Histogenetics.
Chemotherapy treatment and CBU infusion
Chemotherapy regimens were selected based on the patient’s prior treatments and ability to tolerate intensive therapy. In the current analysis, we include only patients who received intensive chemotherapy. The unrelated CBU was infused 24 to 72 hours from the completion of chemotherapy. The only exception was a patient who received liposomal adriamycin/cytarabine salvage; cell infusion was delayed for 7 days because of the prolonged half-life of the liposomal formulation. Patients did not receive any GVHD prophylaxis or additional immunosuppression. Granulocyte colony-stimulating factor was not used routinely.
The primary study objective was to examine the safety of treatment with salvage chemotherapy followed by CBU infusion. Adverse events were graded according to the Common Terminology Criteria for Adverse Events (version 4.3) from initiation of study treatment until an event took place; events were death, hematopoietic stem cell transplantation (HSCT), or relapse, whichever occurred first. Serious adverse events were grade 3-4 acute GVHD, unexplained prolonged myelosuppression, and grade 3-4 cytokine release syndrome (CRS).
Other objectives included assessment of response rates and duration of response, incidence and severity of GVHD, and CB cell therapy–derived chimerism. Because patients with very advanced leukemia were included, marrow response was followed by an immediate attempt at curative allotransplant, in a manner akin to the bridge approaches that we have previously studied.8,9 Hence, it was not possible to assess count recovery, a requisite for formal assessment of complete remission (CR).
Response to treatment was instead defined as effective cytoreduction (ie, <5% residual blasts in a hypocellular bone marrow [BM] or no blasts in an acellular BM [aplasia] obtained ∼14 days after infusion of the CB cells).8 Less than 5% blasts in day-14 BM has been shown to be predictive of excellent response in chemotherapy studies.10 To render our definition of response more stringent and consistent, we added the following criteria: (1) if the percentage of blasts was assessed by multiple methods (aspirate, morphology, immunohistochemistry, immunophenotyping), the assay that yielded the highest percentage was taken into account and (2) if overt disease progression occurred within 2 weeks after a bone marrow test that showed <5% blasts, this was not considered a response. As a result, 3 patients who had been classified as responses in our previous publication were reclassified as nonresponders, because of findings of >10% blasts by 1 of 3 assays in 2 recipients and overt disease progression within 1 week of biopsy in the third patient.
Chimerism studies were performed in peripheral blood (PB) or BM samples, as previously described.11 PB samples were obtained weekly after the CBU infusion for 8 consecutive weeks or until allogeneic transplantation. PB chimerism assessment was usually performed on isolated CD3+ and CD33+ cell fractions. When the white blood cell count was <4 × 109 per liter, unfractionated samples were analyzed. BM samples were evaluated for CBU chimerism when available.
A final objective was the feasibility of obtaining the CBU within the time frame needed for patient treatment. The number of CBU identified for each patient, their characteristics, and the time of completion of the CBU evaluation were recorded throughout the study.
Statistical analysis
CBU characteristics and patient outcomes are described using descriptive statistics.
The relationship between patient or treatment characteristics and response or CBU-derived chimerism was assessed by 2-sided χ2 tests for categorical variables and analysis of variance for continuous variables. P < .05 was considered significant. Multiple regression was used for multivariate analysis of the predictors of chimerism. Initially, all factors that were significant in univariate analyses were included. A stepwise model selection approach was used to identify all significant risk factors. Survival and progression-free survival were calculated using the Kaplan-Meier method and log-rank testing. Cumulative incidence of GVHD was calculated using relapse, death, or subsequent transplant as competing risks.
Results
Patients, disease, and treatment
From June 2015 to December 2019, 38 patients were treated with chemotherapy, followed by infusion of CBU cell therapy; 21 of whom were included in our previous publication.4 For the purposes of this article, we excluded 3 patients who had measurable disease but no excess blasts, because they were not readily evaluable for response, as defined above. We also excluded 4 additional patients who were deemed unfit to receive intensive chemotherapy and were treated with decitabine-containing regimens. We describe the outcomes of 31 patients, 14 women and 17 men, with a median age of 49 years (range, 22-77) and a median weight of 63 kg (range, 44-122) (Table 1). They had received a median of 3 prior treatment regimens, and 9 had relapsed following allogeneic HSCT. Sixteen had an Eastern Cooperative Oncology Group score ≥2. Seven patients had suffered prior fungal infections, and 1 patient, with prior allotransplant, had adenoviremia diagnosed within days of starting treatment. Twenty-eight patients had AML, 1 had myeloid blast crisis chronic myeloid leukemia, and 2 had high-risk MDS. All had chemotherapy-refractory disease. Twenty-seven patients had adverse European LeukemiaNet (ELN) risk.
Combination salvage chemotherapy with cladribine, cytarabine, and mitoxantrone was used in 21 patients.12 Other intensive salvage regimens,13,14 including standard- or intermediate-dose cytarabine, liposomal adriamycin/cytarabine (VYXEOS), clofarabine, fludarabine, high dose cytarabine, filgrastim, and mitoxantrone, etoposide, and cytarabine were used in 10 patients (Table 1).
CBU cell therapy characteristics
The median precryopreservation CBU TNC dose was 91.0 × 107 per kilogram (range, 63-179 × 107), and the median TNC dose was 1.4 × 107 per kilogram (range, 0.7-2.4 × 107). The median postthaw CD34+ cell dose was 0.3 × 105 per kilogram (range, 0.10-0.99 × 105). CBU had a median lymphocyte cell dose of 5.26 × 106 per kilogram (range, 1.4-10.21 × 106) precryopreservation and a median CD3+ cell dose of 3.5 × 106 per kilogram (range: 1.3-6.6 × 106) postthaw. Overall, CBU with much lower cell doses than those currently selected for transplantation were used. Furthermore, CBU were “older” (median storage time, 11 years; range, 6.0-18.0). CBU met all criteria for clinical use and had segment quality evaluation before release (n = 26), as per current practice. Median segment CD34+ cell viability was 96.1%.
HLA match was 6/6 for 1 patient, 5/6 for 19 patients, and 4/6 for 11 patients. Overall, 30 patients received CBU with shared IPA targets; of those, 8 had shared IPAs and NIMA match, and 1 had a CBU with NIMA match only. KIR genotyping was performed in 30 units and showed A/A haplotype in 15 units and B/x haplotype in 15 units.15
We were able to identify these units within the time interval needed for patient treatment. During the last 3 years of the study (n = 26 patients), selection was expedited, and CBU were ready for shipment within an average of 8 days after initiation of a search; in 50% of the requests, a CBU was ready to ship within 7 days (supplemental Table 2).
Chimerism and engraftment
Postinfusion chimerism was assessed in all patients. CBU-derived chimerism was detected in 12 (39%) patients. Five patients had full CBU chimerism. In 2 of those, the CBU was fully engrafted with CBU-derived PB count recovery. Two others died in aplasia, and the fifth, after an initial response, developed disease progression. Four of the 5 patients with full CBU chimerism had relapsed after allogeneic transplant. One patient had 95% CBU chimerism but died in aplasia from worsening fungal infection. Another patient, who also had relapsed after transplant, had 72% CBU-derived chimerism associated with skin GVHD. Chimerism was transient and was followed by the patient’s own count recovery with morphologic CR.
Transient low-level CBU-derived chimerism (1%-5%) was detected in 5 additional recipients. Lastly, 1 patient did not have detectable CBU chimerism on repeated testing in the first months after CBU infusion and, therefore, was considered nonchimeric (all host). However, low-level CBU chimerism was transiently detected 6 months later and after a subsequent stem cell transplant.
CRS and GVHD
No immediate CBU infusion reactions were observed. CRS, manifested as prolonged fever of unknown origin occurring in the week after CBU cell therapy infusion, was observed in 3 patients (2 were reported previously).4 In 2 patients, CRS was treated with systemic steroids; in a more recent patient, it was treated with tocilizumab and, later, with steroids. There were no cases of grade 3-4 CRS.
Acute GVHD was observed in 4 patients. Two patients, who had full CBU chimerism and PB count recovery, experienced grade 4 acute GVHD; 1 patient required steroids, and the other required steroids, tacrolimus, and ruxolitinib. Another patient had grade 2 skin-only GVHD. In a more recent patient with 72% CBU chimerism and skin GVHD, tacrolimus was initiated promptly, GVHD resolved, CBU-derived chimerism was transient, and CR resulted. The cumulative incidence of GVHD in the 12 patients with chimerism was 33% by day 30. No chronic GVHD was observed.
Other adverse events
Opportunistic infections were frequent in these heavily pretreated patients with prolonged preceding periods of immunosuppression and neutropenia (supplemental Table 3).
There were 4 nonrelapse deaths in patients who had responded. A 47-year-old who had relapsed after allotransplant developed posttransplant lymphoproliferative disease of the brain and pulmonary veno-occlusive disease with respiratory failure. A 35-year-old, also relapsed after prior allotransplant, developed disseminated adenovirus. A 70-year-old died from fungal sinusitis, and a 77-year-old died from sepsis.
Leukemia response and further treatment
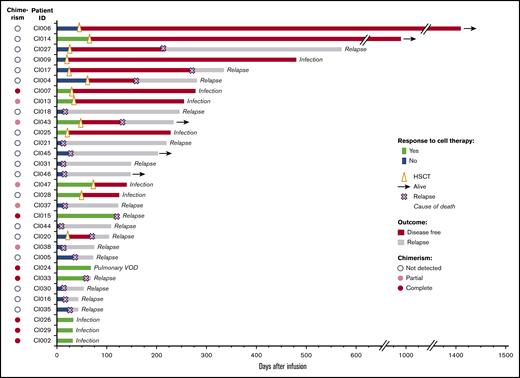
Thirteen (42%) patients responded (Figure 1). Twelve had hypocellular or empty marrow without residual blasts; 1 had 4% residual blasts but recovered with a CR. Of the 13 responders, 7 subsequently underwent HSCT; unfortunately 5 died from posttransplant infections. Additionally, 6 patients without response received further salvage and proceeded to HSCT16 ; 4 of them relapsed again.
Overall survival, progression-free survival, and causes of death
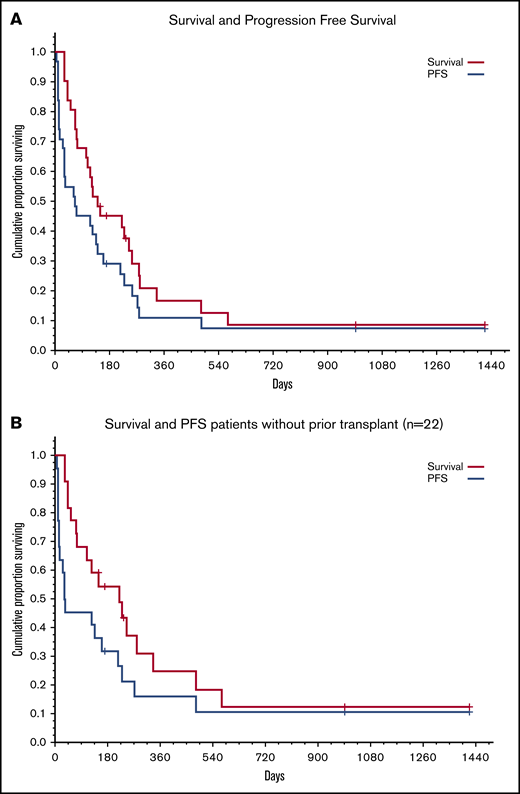
Five of the 31 patients are alive at a median follow-up of 234 days (range, 148-1418), 1 patient is in CR, 3 relapsed, and 1 developed HSCT donor-derived MDS. Estimated 1-year survival is ∼17% for the entire group and 25% for the 22 patients who had not undergone an allogeneic transplant (Figure 2). Twenty-six patients died at a median time of 120 days (range, 32-571) after CB cell therapy. Leukemia relapse was the cause of death in 16 patients. Eight patients died from infection (3 early deaths; 5 after HSCT), 1 patient died from multiorgan failure, and 1 patient died from GVHD/multiorgan failure. Among the 13 responders, only 2 relapsed, 4 died of early complications, and 5 died from posttransplant complications, primarily infections. In contrast, of the 18 patients who did not respond, 14 died from disease progression (Figure 1).
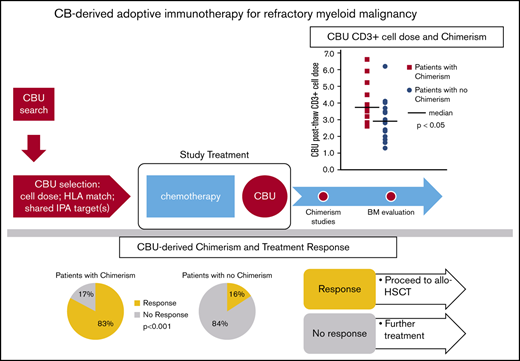
Response to CB adoptive cell therapy and patient outcomes. Of the 31 patients treated with intensive salvage chemotherapy, followed by CB cell therapy infusion, 13 (42%) responded, whereas the remaining 18 did not respond. Overall, 12 patients (39%) had chimerism detected. Thirteen patients received HSCT at a median of 44 days after the study treatment. Arrows indicate patients alive (as of May 2020, median follow-up: 234 days); for the remaining patients, the cause of death is indicated. VOD, veno-occlusive disease.
Response to CB adoptive cell therapy and patient outcomes. Of the 31 patients treated with intensive salvage chemotherapy, followed by CB cell therapy infusion, 13 (42%) responded, whereas the remaining 18 did not respond. Overall, 12 patients (39%) had chimerism detected. Thirteen patients received HSCT at a median of 44 days after the study treatment. Arrows indicate patients alive (as of May 2020, median follow-up: 234 days); for the remaining patients, the cause of death is indicated. VOD, veno-occlusive disease.
Survival and progression-free survival (PFS). (A) All patients (N = 31). (B) Patients with no prior allogeneic BM transplantation (n = 22).
Survival and progression-free survival (PFS). (A) All patients (N = 31). (B) Patients with no prior allogeneic BM transplantation (n = 22).
Predictors of response and chimerism
We evaluated the effect of patient, disease, treatment, and CBU characteristics on response. The only pretreatment variable that predicted response was a prior allotransplant. Seven of the 9 patients with prior transplant responded vs only 6 of 22 patients without a transplant (P = .01; Table 3).
Chimerism detected within the first 2 weeks after infusion was a strong predictor of response. Responses occurred in 10 of 12 patients with detected CBU chimerism compared with only 3 of 19 with no chimerism (P = .0002; Table 4). Therefore, the ability to induce CBU chimerism of any degree appeared to be the best predictor of response. The 2 variables that correlated strongly with CBU-derived chimerism were prior allotransplant and the lymphocyte content of the CB cell therapy.
Of the 9 patients who had relapsed after a prior allogeneic transplant, 8 had detectable chimerism compared with 4 of 22 without a prior transplant (P = .0002; Table 5).
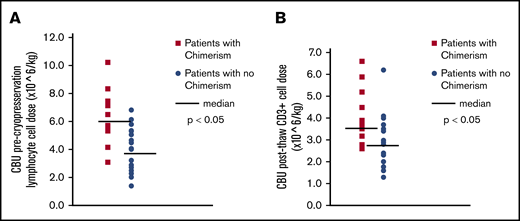
There was a very strong association between the lymphocyte content of the CBU and chimerism. This was true whether the lymphocyte content was assessed as percentage of lymphocytes at initial CBU evaluation, percentage of lymphocytes and total lymphocyte cell dose (total lymphocytes per kilogram) precryopreservation, percentage of lymphocytes postthaw, or CD3+ cell dose at infusion (CD3+ cells per kilogram) (all P values were significant; Table 2; Figure 3). Notably, the lymphocyte cell dose did not correlate with the degree of chimerism. Lymphocyte cell doses were similar between patients who had chimerism levels <10% and those who had high levels or full chimerism. We did not observe any effect of TNC or CD34+ cell doses on chimerism.
CBU lymphocyte content and chimerism. (A) CBU precryopreservation lymphocyte cell dose (×106 per kilogram) and chimerism CBU complete blood count with differential were obtained before cryopreservation, as per current practice, and the total lymphocyte content of the CBU, as well as the lymphocyte cell dose (per kilogram of patient weight) were calculated. The orange symbols indicate the precryopreservation lymphocyte doses of patients who had CBU-derived chimerism detected after treatment, whereas the blue symbols indicate the cell doses of the patients who did not have detectable chimerism. The black horizontal lines indicate the medians for each group. (B) CBU postthaw CD3 cell dose (×106 per kilogram) and chimerism CBU evaluation by flow cytometry was performed after thawing, following standard practice, and the CD3+ cell doses were calculated. The orange symbols indicate the postthaw CD3+ cell doses of patients who had CBU-derived chimerism detected after treatment, whereas the blue symbols indicate the cell doses of the patients who did not have detectable chimerism. The horizontal black lines indicate the medians for each group.
CBU lymphocyte content and chimerism. (A) CBU precryopreservation lymphocyte cell dose (×106 per kilogram) and chimerism CBU complete blood count with differential were obtained before cryopreservation, as per current practice, and the total lymphocyte content of the CBU, as well as the lymphocyte cell dose (per kilogram of patient weight) were calculated. The orange symbols indicate the precryopreservation lymphocyte doses of patients who had CBU-derived chimerism detected after treatment, whereas the blue symbols indicate the cell doses of the patients who did not have detectable chimerism. The black horizontal lines indicate the medians for each group. (B) CBU postthaw CD3 cell dose (×106 per kilogram) and chimerism CBU evaluation by flow cytometry was performed after thawing, following standard practice, and the CD3+ cell doses were calculated. The orange symbols indicate the postthaw CD3+ cell doses of patients who had CBU-derived chimerism detected after treatment, whereas the blue symbols indicate the cell doses of the patients who did not have detectable chimerism. The horizontal black lines indicate the medians for each group.
Lastly, the degree of HLA match between recipient and CBU correlated with response: 11 of 20 patients with CB units matched at 5/6 or 6/6 loci responded compared with 9 of 16 patients with 4/6 matched cell therapy units (P = .047; Table 2). The association of HLA match and chimerism was not significant. Further, we did not find a relationship with allele level HLA match or NIMA matching. Additionally, there was no difference in the chimerism results based on A/A or B/x CBU KIR haplotype (33% and 43%, respectively).
The relationships between prior BM transplantation and chimerism, as well as between lymphocyte dose and chimerism, were independent of each other.
Discussion
Despite the introduction of numerous novel chemotherapy drugs, challenges remain in the treatment of relapsed/refractory AML. Targeted cell therapy with chimeric antigen receptor (CAR) T cells is still at an early stage.17 Adoptive cell therapy using haploidentical cells has shown promise but has found little implementation in the United States.2,3,18 We adapted the concept and prospectively studied the combination of salvage chemotherapy and CB-based adoptive immunotherapy for treatment of patients with refractory AML/MDS. We reported safety, feasibility, and preliminary efficacy previously.4 Other investigators have also recently shown encouraging response and duration of response when treating older AML patients with hypomethylating agents, followed by CB infusion.19 In this article, we report updated results for 31 patients and new insights derived from our analysis.
Eligibility for this study was restricted to patients with relapsed/refractory disease and adverse prognosis. Patients had failed an average of 3, and a maximum of 7, prior treatments, and 9 had relapsed after allogeneic transplants. Such patients are prone to toxicities and have low responses and very poor long-term survival.
Treatment safety was acceptable, with only 4 early deaths; none of them attributed to the CB infusion, but rather to worsening of prior infections or related to chemotherapy. Adverse events included 3 cases of mild CRS and 4 cases of GVHD. Two of the latter were severe, but occurred early in our experience. In a more recent case, earlier intervention with immunosuppression averted deterioration.
There was a powerful association between CBU-derived chimerism and response (Table 4). Ten of 12 (83%) patients with any degree of chimerism responded. Overall, 13 of the 31 patients (43%) responded. These rates are much higher than any data reported previously in patients with advanced leukemia. For example, investigators at MD Anderson Cancer Center recently reported outcomes in 674 patients undergoing second salvage.1 The rate of CR (CR+CRp [CR with low platelets]) was only 13%, and the overall rate of response (CR+CRp+PR [partial response]+bone marrow CR) was 22%. These salvage results are consistent with the response rates that we observed among our 19 patients without detectable chimerism, 3 of whom (16%) had a response. Further, the CLAGM (cladribine, cytarabine, mitoxanthrone) regimen has remission rates (CR+CRi [CR with incomplete hematologic recovery]) of 40% to 65% in several trials, but these typically enrolled younger patients or had restrictions based on estimated risk for treatment-related mortality, such as exclusion of patients with prior allotransplant and those at high risk for treatment-related mortality.12,20-22 The association of chimerism with response, as well as the high response rate in chimeric patients compared with historical data, strongly suggest that, indeed, the CB cell therapy and its GVL effect were the major determinants of response. The use of intensive salvage chemotherapy may have facilitated a response. Such an effect has been described with other cell therapies, most notably CAR T cells, where dose intensity of cyclophosphamide has been associated with higher T-cell expansion and higher response rates,23-25 as well as with CB-derived CAR natural killer cells.26 It is not clear whether the chemotherapy creates a favorable cytokine environment or lymphodepletion or exerts a direct antileukemic effect.
Moreover, the response rates among patients who had relapsed after allotransplant are particularly impressive compared with responses (CR+PR) of, at best, 35% with donor lymphocyte infusion(s) (DLI) in similar patients.27,28 Even recent studies implementing DLI with chemotherapy had modest success: the German Cooperative Group recently reported combination of azacitidine and DLI (median CD3+ dose of 31 × 106 per kilogram), and the Italian group reported DLI with posttransplant cyclophosphamide for AML patients who relapsed after transplant.29,30 In both studies, CR+PR rates were in the range of 33% for patients with high number of blasts. In contrast, we observed chimerism in 8 patients and responses in 7 of the 9 patients treated with a median CB CD3+ dose of only 3.5 × 106 per kilogram.
The most disappointing finding is the poor long-term outcome. A total of 13 patients (7 responders and 6 nonresponders who received additional salvage chemotherapy) went on to transplant or to a second transplant, but many have relapsed again or died from posttransplant complications. Only 5 of 31 patients remain alive; however, this low long-term survival should be viewed in the context of the very advanced disease and multiple prior treatments. Indeed, using the criteria proposed by Kantarjian et al1 for prediction of response and survival after second salvage for refractory AML, all but 1 of our patients were in the highest risk category, with an estimated probability of CR/CRp of 8%, median survival < 2 months, and 0% probability of 1-year survival.1 Although our patients were younger than the group described by Kantarjian et al (median age, 49 years vs 60 years), they were more heavily treated: they had received an average of 3 (up to 7) prior regimens, as opposed to only 2 in the study by Kantarjian et al, and several had failed allogeneic transplant, whereas such patients were excluded from the Kantarjian et al study.1 In that context, 1-year survival of 17% in our patients and 25% in those who had not undergone a transplant is encouraging.
Because chimerism was the best predictor of response, we were interested in identifying determinants of chimerism. The most immunosuppressed hosts, those with a prior allotransplant, readily allowed engraftment of the CBU; in some cases, this CBU-derived chimerism was complete and durable. Further, a cell therapy CBU with higher T-cell content was associated with an increased likelihood of chimerism. Conversely, a higher HLA match between CBU and patient correlated with response but had a weak association with chimerism. Further, NIMA matching in 8 of the 31 patients was not associated with more frequent chimerism.31,32 We have previously provided evidence that sharing IPAs favorably affected outcomes in CB transplantation, probably because of maternal microchimerism.33 We have also detected long-term maternal microchimerism in some transplant recipients.34 For the current study, we selected CBU that shared IPA targets with the recipients and were able to find such units for all but 1 patient. This CBU selection may have further contributed to the high response rates. Lastly, donor KIR haplotype influences GVL effects in HLA-matched or haploidentical transplants; however, we were not able to detect such effects in this small series.15
Although microchimerism was well tolerated, full or dominant CBU chimerism was associated with GVHD and toxicity. In the future, we plan to implement GVHD prophylaxis whenever a high level of chimerism is observed. High-level chimerism was more common in patients with prior allotransplants. On the other hand, the lymphocyte dose was not predictive of high-level vs low-level chimerism.
The limitations of this study are several. The assessment of response was based on day-14 BM evaluation, which has excellent positive predictive value for remission but lacks specificity.10 Second, our chimerism assay cannot reliably detect levels <1%.11 This may explain the interesting case of CB chimerism 6 months after transplant that was not detected on previous occasions. It is likely that this patient, and possibly others, had microchimerism below the limit of detection early after treatment. More sensitive detection methods may need to be used in future trials. Finally, the advanced disease and multiple prior treatments contribute to toxicity; although several patients achieved disease control, most succumbed to infections and died.
Our analysis provides insights on how to further optimize this strategy, which ultimately may result in an effective, reasonably affordable, and widely available treatment for AML making use of previously stored relatively small CBU that are currently underutilized. Such a treatment approach is feasible: as our experience increased, appropriate CBU could be identified within 8 days, on average, from search request (supplemental Table 2). The large NCBP inventory of previously HLA typed and fully tested CBU enabled us to evaluate many units quickly (average, 59 per patient) and identify those meeting study criteria promptly. Furthermore, increasing the number of subjects with chimerism is likely to improve response rates and may be achieved by modifying the selection criteria for the CBU (eg, by choosing units with higher lymphocyte content for cell therapy); patients who received CBU with CD3+ cell doses above the median had a higher probability of chimerism (P = .04). Although CD3+ cell counts are not routinely performed before cryopreservation, complete blood counts are, and the percentage of lymphocytes is helpful in estimating the CD3+ cell dose. Changes in chemotherapy regimens, to increase immunosuppression, may also increase chimerism.35
In conclusion, use of CBU as adoptive immunotherapy, in combination with salvage chemotherapy for patients with refractory myeloid malignancies, represents a novel, safe, and effective approach to induce disease control. Response rates closely correlate with detectable chimerism, suggesting a GVL effect. The CBU lymphocyte content is a major determinant of chimerism and, thus, of response. CBU for cell therapy can be obtained quickly so treatment can be administered promptly.
Data-sharing requests should be sent to Koen van Besien (kov9001@med.cornell.edu).
Acknowledgments
The authors thank their patients for courage and willingness to participate in investigational studies. They also express gratitude to the clinical and administrative staff of their transplant program for their unwavering dedication to the care of patients, most recently under very difficult circumstances. They are grateful for the NCBP staff for efforts to ensure high-quality CB units, as well as to the obstetricians and obstetrical nursing staff at all NCBP collection sites. They are indebted to the thousands of mothers who have generously donated their infants’ CB for the common good.
Authorship
Contribution: O.C. analyzed data and wrote and reviewed the manuscript; E.M.F. performed CBU evaluation, collected and analyzed data. and reviewed the manuscript; G.J.R. designed the study, reviewed the manuscript, and cared for patients; U.G. cared for patients, collected and analyzed data, and reviewed the manuscript; A.S. and K.v.B. designed the study, analyzed and interpreted the data, and wrote the manuscript; A.S. performed CBU Eselection; K.v.B. was responsible for patient care; M.C., J.-M.H., S.L., S.A.M., A.A.P., N.O., E.K.R., T.S., and M.S.S. cared for patients and reviewed the manuscript; L.D., M.S.A., and C.R. participated in CBU preparation and reviewed the manuscript; D.G. participated in data collection and management and reviewed the manuscript; and P.D. reviewed the manuscript.
Conflict-of-interest disclosure: A.S. was the Medical Director of the NCBP until January 2020. The remaining authors declare no competing financial interests.
Correspondence: Koen van Besien, Weill Cornell Medical College/NYP, 520 East 70th St, New York, NY 10021; e-mail: kov9001@med.cornell.edu.
References
Author notes
The full-text version of this article contains a data supplement.