Key Points
Circulating heme sheds EPCR from the renovascular endothelium, leading to sickle CKD.
Plasma EPCR levels were elevated in patients with SCD and CKD.
Visual Abstract
Chronic kidney disease (CKD) is a major contributor to morbidity and mortality in sickle cell disease (SCD). Anemia, induced by chronic persistent hemolysis, is associated with the progressive deterioration of renal health, resulting in CKD. Moreover, patients with SCD experience acute kidney injury (AKI), a risk factor for CKD, often during vaso-occlusive crisis associated with acute intravascular hemolysis. However, the mechanisms of hemolysis-driven pathogenesis of the AKI-to-CKD transition in SCD remain elusive. Here, we investigated the role of increased renovascular rarefaction and the resulting substantial loss of the vascular endothelial protein C receptor (EPCR) in the progressive deterioration of renal function in transgenic SCD mice. Multiple hemolytic events raised circulating levels of soluble EPCR (sEPCR), indicating loss of EPCR from the cell surface. Using bone marrow transplantation and super-resolution ultrasound imaging, we demonstrated that SCD mice overexpressing EPCR were protective against heme-induced CKD development. In a cohort of patients with SCD, plasma sEPCR was significantly higher in individuals with CKD than in those without CKD. This study concludes that multiple hemolytic events may trigger CKD in SCD through the gradual loss of renovascular EPCR. Thus, the restoration of EPCR may be a therapeutic target, and plasma sEPCR can be developed as a prognostic marker for sickle CKD.
Introduction
Individuals with sickle cell disease (SCD), characterized by acute and chronic intravascular hemolysis, suffer from multiorgan disease that shortens the average life span. The damage to the kidneys is widespread and causes numerous complications, contributing to 16% to 18% of the overall mortality in this patient population.1 Chronic kidney disease (CKD) is the most common form of renal disease, affecting ∼58% of individuals with SCD.2 The development of CKD is age dependent and is primarily identified by increased albuminuria and reduced kidney function, represented as the estimated glomerular filtration rate (eGFR). Prospective and retrospective studies have confirmed that the risk of albuminuria and CKD increases with age in patients with SCD.3-5 Approximately 30% of adults with SCD experienced a rapid decline in eGFR, with a loss of >3.0 mL/min per 1.73 m2 per year.4 Sickle CKD stages were independently associated with hemoglobinuria and markers of intravascular hemolysis.2 Elevated albuminuria is also associated with low hemoglobin (Hb) level and intrinsic hemolytic stress, promoting endothelial dysfunction that impacts internal organs.6 Chronic and acute intravascular hemolytic events, pertinent to SCD, elevate circulating free heme, which is implicated in vaso-occlusion, oxidative stress, inflammation, endothelial toxicity, and end-organ damage.7-9 We have previously shown that vascular permeability deteriorates with age10 and that an acute elevation of heme triggers clinically relevant acute kidney injury (AKI), with a rapid decline in glomerular filtration rate (GFR) and albuminuria in SCD mice.11 Renal pathology in SCD includes extensive peritubular microvascular congestion, which can lead to CKD via microvascular rarefaction.12-14 Moreover, AKI is considered a prominent risk factor for CKD, and damage to the renal microvasculature after AKI potentially expedites the transition to CKD.15 Despite these clinical and experimental associations, mechanistic studies linking hemolysis to renal vascular endothelial impairment and progression to CKD in SCD have not been described.
The inflammatory milieu in SCD regulates the endothelial barrier function through adhesion- and secretion-dependent mechanisms.16 The endothelial protein C receptor (EPCR) is a transmembrane glycoprotein that augments activated protein C (APC) generation through the thrombin-thrombomodulin complex, and APC functions as an anticoagulant by inhibiting coagulation factors, factor VIII and factor V.17,18 In addition to its role in anticoagulation, EPCR plays a critical role in endothelial cytoprotection and stability, primarily by facilitating the induction of protease-activated receptor-1 (PAR1) signaling through APC.17,19,20 The presence of functional EPCR on the endothelial cell surface is essential for the cytoprotective effects of EPCR.21 Soluble EPCR (sEPCR) detectable in plasma is generated by the cleavage of the EPCR molecule near the transmembrane domain, resulting in decreased cell-surface expression of EPCR and increased plasma levels of sEPCR.22,23 Higher levels of sEPCR have been reported in patients with systemic inflammatory diseases and conditions associated with enhanced thrombin generation.24 Deficiency of cellular EPCR is attributed to endothelial dysfunction and renal injury.25
In this study, we aimed to determine the mechanism by which hemolysis drives renal endothelial impairment and promotes CKD in SCD. Here, we present novel data showing that progressive CKD in SCD is associated with a gradual loss of renal microvascular endothelial integrity and detachment of EPCR from the renal microvasculature. Extracellular heme triggers loss of EPCR from the renal endothelium, resulting in CKD. Furthermore, we identified plasma sEPCR as a potential biomarker for CKD in individuals with SCD.
Methods
For a full description of the materials and methods, refer to supplemental Materials, available on the Blood website.
Mice
The University of Pittsburgh Institutional Animal Care and Use Committee (protocol numbers 22101947 and 22061337) approved all mouse studies. Knockin Townes SCD mice (strain number 013071)26 expressing human-Hb βS (SS) and control mice expressing human-Hb βA (AA) were purchased from Jackson Laboratories and bred at the University of Pittsburgh vivarium. The EPCR overexpressor mice (EPCR-Ox) were generated as described previously.27 Overexpression of EPCR was achieved by placing the expression of the EPCR gene under the Tie2 promoter. The SS mice were IV infused with ferric heme (hemin; 14 μmol/kg body weight; 5 times on alternate days). Whole blood samples and spot urine samples were collected from the mice. Both female and male mice were used in the experiments.
Human plasma samples
Biochemical analysis
Urine creatinine and albumin were evaluated using a colorimetric assay. Enzyme-linked immunosorbent assays were used to assess plasma and urinary sEPCR, plasma cystatin C (Cys C), urinary kidney injury molecule-1 (KIM-1), plasma soluble thrombomodulin (sTM), and renal tissue thrombin, following the manufacturer’s instructions.
GFR
GFR was measured in mice using a noninvasive transcutaneous device that records fluorescence from infused fluorescein isothiocyanate (FITC)-sinistrin (7.5 mg/100 g body weight). FITC-sinistrin clearance was recorded, and the T1/2 for FITC-sinistrin clearance was determined using software provided by the manufacturer.11,29
SRU imaging
Super-resolution ultrasound (SRU) imaging was performed on paraspinal fur-shaved anesthetized mice using a programmable ultrasound (US) system equipped with a 15.6-MHz linear array US probe (L22-14v, Verasonics). Commercial lipid-shelled microbubbles (diameter, 1-4 mm; DEFINITY; Lantheus Medical Imaging) were used as the US contrast agents. The captured signals were postprocessed through clutter filtering, microbubble center localization, microbubble tracking, and frame summation and analyzed using MATLAB (MathWorks)30-32 (supplemental Figures 10-11).
Bone marrow transplantation
The EPCR overexpressor (EPCR-Ox) mice and C57BL/6 (EPCR-wild type [EPCR-WT]) mice were transplanted with bone marrow cells from homozygous sickle (SS) or control (AA) mice as we have described previously.11,33 Total Hb was measured using the AVOXimeter (ITCMED) and reticulocytes by flow cytometry. Hb gel electrophoresis was performed using a system from Helena Laboratories.
Histopathology and immunofluorescence staining
Paraffin-embedded kidney tissue sections were stained with hematoxylin and eosin and Prussian blue stain. For immunofluorescence staining, renal tissue sections were incubated with primary antibodies for EPCR, CD31, smooth muscle actin, or β-catenin. The sections were counterprobed with the appropriate secondary antibodies conjugated with fluorescent probes. Images were captured and processed using an Olympus AX70 microscope, an all-in-one APX100 microscope, and the cellSens software.
Renal vascular leakage
Vascular leakage was studied by IV injection of cell-impermeable Evans blue dye, as described elsewhere.10
Statistical analysis
Two-tailed paired or unpaired Student t test, 1-way analysis of variance, and Pearson or Spearman correlation were performed. Data obtained from the female and male mice were combined. GraphPad Prism 10 software was used for all the statistical comparisons.
Results
Age-dependent deterioration of renal health in SCD was associated with renal microvascular rarefaction
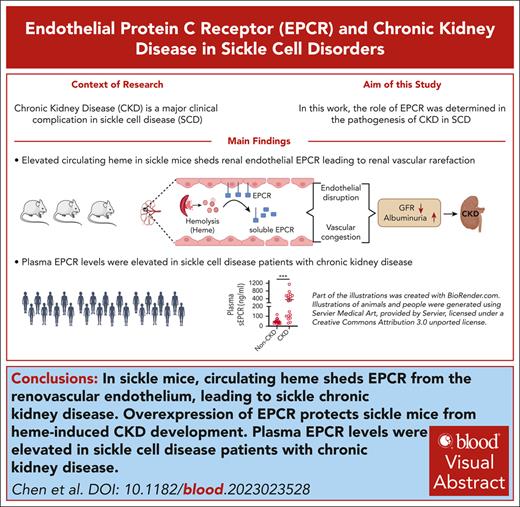
Because a progressive decline in renal function, diagnosed with overt albuminuria and loss of GFR, leads to CKD in individuals with SCD, we tested whether the Townes SCD mouse model recapitulates age-dependent CKD progression. We found that SS mice exhibited a significant decrease in GFR and an increase in albuminuria (urinary albumin to creatinine ratio [uACR]) from 1 to 10 months of age. Furthermore, 1-month-old SS mice had more pronounced albuminuria than 10-month-old control AA mice (Figure 1A-B). Because serum Cys C has recently been recommended as an early and accurate biomarker of CKD,34 we measured plasma Cys C in our cohort of differentially aged SS mice and older AA mice, noting a significant age-dependent elevation of Cys C in SS mice (Figure 1C). Previous studies have shown a longitudinal age-dependent increase in urinary KIM-1, a tubular epithelial injury marker, in SS mice.35 Although 1-month SS mice had comparable levels of urinary KIM-1 to 10-month-old AA mice, we observed significant elevation of KIM-1 in 3 to 10-month-old SS mice (Figure 1D). Histopathology of renal tissue from patients with SCD and CKD revealed substantial congestion and sickling within the renal vasculature.13 The SS mouse kidneys showed a progressive increase in congestion involving the renal corticomedullary peritubular area (Figure 1E). Previously, we have shown that renal vascular leakage in SS mice is correlated with age and a gradual drop in Hb.10 Although progressive CKD is evident in SCD, a significant drop in Hb has also been reported in patients aged >30 years.36 In this study, we assessed renal vascular leakage in a group of SS mice (1-, 3-, 6-, and 10-month-old) using Evan blue extravasation and correlated their level of renal vascular leakage with their intensity of anemia. An age-dependent increase in renal vascular leakage was significantly associated with the loss of total Hb, thus linking the lack of endothelial integrity with anemia (supplemental Figure 1A-B). SRU, which uses injectable microbubbles to noninvasively measure renal vascularity, was previously shown to accurately quantify the microvascular rarefaction that occurred during the AKI-to-CKD progression in mice exposed to ischemia-reperfusion injury.32 We used SRU imaging technology to determine renal microvascular changes in 1-month-old and 10-month-old SS mice. The density of the peritubular vasculature in the renal cortex and corticomedullary areas was significantly lower in 10-month-old SS mice than in 1-month-old mice, indicating age-dependent loss of renal microvasculature in SCD. In contrast, the vessel density in 10-month-old AA mice was comparable to that in 1-month-old SS mice (Figure 1F-H). Although older SS mice had a significantly lower renal blood volume than older AA mice, we did not observe a significant difference in renal blood volume between older and younger SS mice (Figure 1I).
Progressive renal damage and development of CKD in sickle mice. (A) Townes sickle (SS) mice from different age groups (1-10 months; n = 9; M, 5; F, 4) and older (10 months; n = 9; M, 3; F, 6) control (AA) mice were used to assess GFR noninvasively using a transcutaneous device. An age-dependent decline in GFR was evident with hyperfiltration in younger (1-month) SS mice. (B) Urinary albumin and creatinine were measured in the urine collected from the same cohort of mice as in (A) before GFR measurement, and the ratio (uACR) indicated a significant increase in albuminuria. (C) Plasma Cys C, measured using enzyme-linked immunosorbent assay in the same cohort of mice, was higher in SS mice of all ages than in AA mice. (D) Elevated urinary KIM-1 in the SS mice, with no difference between older AA and 1-month-old SS mice. (E) Representative hematoxylin and eosin images showing an age-dependent increase in renal microvascular congestion (arrows) in SS mice (scale bar = 20 μm). (F) SRU images of the kidneys in a separate cohort of AA (10-month; n = 3; M, 2; F, 1) and SS (1-month [n = 3; M, 2; F, 1] and 10-month [n = 3; M, 2; F, 1]) mice, showing representative overlaid B-mode images. Quantitation of vessel density in the (G) cortex and (H) corticomedullary regions of interest, as described in the supplemental Methods, showing a loss of renal microvasculature in older SS mice (n = 3). (I) Reduced renal blood volume (rBV) in older SS mice compared with age-matched AA mice (n = 3). ∗P < .05; ∗∗P < .01; ∗∗∗P < .001 (1-way analysis of variance (ANOVA) between differentially aged SS mice and unpaired Student t test between 10-month-old AA and SS mice). F, female mice; M, male mice; ns, nonsignificant.
Progressive renal damage and development of CKD in sickle mice. (A) Townes sickle (SS) mice from different age groups (1-10 months; n = 9; M, 5; F, 4) and older (10 months; n = 9; M, 3; F, 6) control (AA) mice were used to assess GFR noninvasively using a transcutaneous device. An age-dependent decline in GFR was evident with hyperfiltration in younger (1-month) SS mice. (B) Urinary albumin and creatinine were measured in the urine collected from the same cohort of mice as in (A) before GFR measurement, and the ratio (uACR) indicated a significant increase in albuminuria. (C) Plasma Cys C, measured using enzyme-linked immunosorbent assay in the same cohort of mice, was higher in SS mice of all ages than in AA mice. (D) Elevated urinary KIM-1 in the SS mice, with no difference between older AA and 1-month-old SS mice. (E) Representative hematoxylin and eosin images showing an age-dependent increase in renal microvascular congestion (arrows) in SS mice (scale bar = 20 μm). (F) SRU images of the kidneys in a separate cohort of AA (10-month; n = 3; M, 2; F, 1) and SS (1-month [n = 3; M, 2; F, 1] and 10-month [n = 3; M, 2; F, 1]) mice, showing representative overlaid B-mode images. Quantitation of vessel density in the (G) cortex and (H) corticomedullary regions of interest, as described in the supplemental Methods, showing a loss of renal microvasculature in older SS mice (n = 3). (I) Reduced renal blood volume (rBV) in older SS mice compared with age-matched AA mice (n = 3). ∗P < .05; ∗∗P < .01; ∗∗∗P < .001 (1-way analysis of variance (ANOVA) between differentially aged SS mice and unpaired Student t test between 10-month-old AA and SS mice). F, female mice; M, male mice; ns, nonsignificant.
Sickle mice experienced a progressive loss of EPCR from renal microvasculature
Sequestration of erythrocytes, causing vascular leakage and endothelial dysfunction, is associated with the loss of EPCR from the cerebral microvasculature in severe malaria.37 EPCR plays a crucial role in supporting APC–mediated endothelial cytoprotection through antiapoptotic, anti-inflammatory, and barrier stabilization mechanisms. Various proteases generated during inflammation and coagulation often cleave the EPCR molecule near the transmembrane domain, thus decreasing the cell surface expression of EPCR and increasing plasma levels of its soluble form, sEPCR.22,23 The lack of cellular EPCR is attributed to endothelial dysfunction and renal injury.25 Due to compromised endothelial barrier integrity, EPCR may detach from the membrane, raising sEPCR levels in the circulation. In the cohort of differentially aged SS mice, plasma, and urine sEPCR levels were elevated in an age-dependent manner, with younger SS mice having an sEPCR level equivalent to that in older AA mice (Figure 2A-B). Consequently, using immunofluorescence staining, we discovered that the renal endothelium of older SS mice lacked EPCR, whereas the renal endothelium of younger SS mice and older AA mice retained similar levels of EPCR expression (Figure 2C-D).
Age-dependent loss of EPCR from the renal vascular endothelium in SS mice. (A) Plasma and (B) urine collected from older (10-month) AA mice (n = 5-6; M, 2; F, 3-4) and differentially aged (1-10 months) SS mice (n = 5-6; M, 2; F, 3-4) showing a gradual age-dependent increase in plasma and urinary sEPCR in SS mice. (C) Representative immunofluorescence images showing the loss of EPCR from the renal microvascular endothelium in older SS mice (6 months or 10 months) compared with younger mice (1 or 3 months). CD31 is an endothelial marker (scale bar = 50 μm). (D) The ratio of the staining intensity of EPCR and CD31 was calculated from the older AA mice (10 months; n = 5; M, 2; F, 3) and SS mice of the indicated age (n = 5; M, 2; F, 3 in each age group) using ImageJ software. The EPCR staining intensity gradually diminished with age in the SS mice. ∗P < .05; ∗∗P < .01; ∗∗∗P < .001 (1-way ANOVA between differentially aged SS mice and unpaired Student t test between 10-month-old AA and SS mice).
Age-dependent loss of EPCR from the renal vascular endothelium in SS mice. (A) Plasma and (B) urine collected from older (10-month) AA mice (n = 5-6; M, 2; F, 3-4) and differentially aged (1-10 months) SS mice (n = 5-6; M, 2; F, 3-4) showing a gradual age-dependent increase in plasma and urinary sEPCR in SS mice. (C) Representative immunofluorescence images showing the loss of EPCR from the renal microvascular endothelium in older SS mice (6 months or 10 months) compared with younger mice (1 or 3 months). CD31 is an endothelial marker (scale bar = 50 μm). (D) The ratio of the staining intensity of EPCR and CD31 was calculated from the older AA mice (10 months; n = 5; M, 2; F, 3) and SS mice of the indicated age (n = 5; M, 2; F, 3 in each age group) using ImageJ software. The EPCR staining intensity gradually diminished with age in the SS mice. ∗P < .05; ∗∗P < .01; ∗∗∗P < .001 (1-way ANOVA between differentially aged SS mice and unpaired Student t test between 10-month-old AA and SS mice).
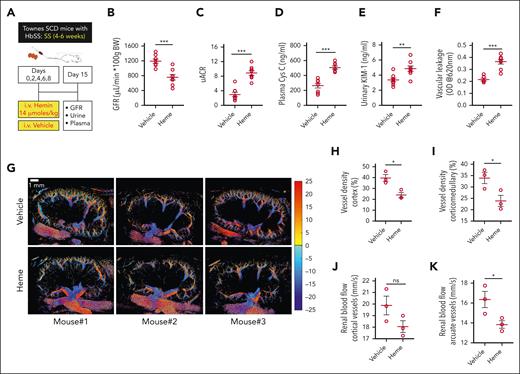
Repeated low-dose heme challenges in younger sickle mice triggered vascular rarefaction and CKD
We previously reported that age-dependent worsening of anemia in SS mice is associated with the extent of intravascular hemolysis and elevated heme in circulation.33 Moreover, we have reported that extracellular heme triggers severe congestion in the renal corticomedullary region during AKI in SS mice.11 In this study, we found that SS mice with IV injection of purified free heme had significantly greater renal endothelial permeability than age-matched control (AA) littermates (supplemental Figure 2). These results prompted us to postulate that a mild elevation in circulating heme might instigate endothelial impairment in the kidneys. To test whether repeated mild heme elevations in younger SS mice could result in renal complications similar to those observed in older SS mice unchallenged with heme, we injected a group of SS mice (3 months) either with a single dose (20 μmol/kg) or multiple doses (14 μmol/kg; 5 times on alternate days) of heme and monitored kidney function for 20 days. A single heme dose led to an initial drop in GFR and an increase in albuminuria, which was later restored, indicative of AKI that improved over time. However, the mice that were challenged with multiple doses of heme failed to recover their baseline GFR and uACR, suggesting the development of CKD after repeated hemolytic events (supplemental Figure 3A-B). In another longitudinal cohort of younger SS mice (4-6 weeks), we found significant albuminuria after repeated heme challenge that did not improve over 4 weeks after the final heme injection (supplemental Figure 3C). We hypothesized that multiple acute hemolytic events instigate persistent endothelial damage that leads to progressive CKD in SCD. To test this hypothesis, younger SS mice (4-6 weeks) were IV infused with vehicle or heme (14 μmol/kg; 5 times on alternate days). One week after the last heme challenge, the multiheme-challenged mice had significantly lower GFR and higher uACR than the vehicle-injected mice (Figure 3A-C). Kidney injury biomarkers, such as plasma Cys C and urinary KIM-1, were also significantly elevated in SS mice injected with multiple heme doses (Figure 3D-E). Furthermore, the multiheme challenge caused a substantial loss of vascular barrier integrity in the kidneys (Figure 3F). These data implied that the sustained CKD that developed in younger SS mice after multiple hemolytic events was comparable to that observed in older SS mice not challenged with heme (Figure 1A-D). Moreover, renal failure in sickle mice was associated with renal endothelial barrier disruption.
CKD development in sickle mice after repeated hemolytic events. (A) Experimental scheme showing the regimen of repeated low-dose hemin injections, mimicking multiple hemolytic events in SS mice. (B) Kidney function represented by GFR, 1 week after the last hemin challenge in the younger SS mice (4-6 weeks) compared with the vehicle-injected mice (n = 8; M, 4; F, 4). (C-E) Increased albuminuria (represented by uACR), plasma Cys C, and urinary KIM-1 in multiheme-challenged SS mice showing CKD development in younger SS mice. (F) Evans blue extravasation data showing elevated renal endothelial barrier disruption in younger SS mice injected with the multiheme regimen (n = 5-6; M, 2-3; F, 3). (G) Vehicle- and heme-injected SS mice were subjected to SRU imaging 1 week after the last hemin challenge. The reconstructed 2-dimensional, cross-sectional, SRU images of the long-axis mouse kidney showing vessel distribution and the velocity map for the vehicle and hemin-challenged mice (n = 3; M, 2; F, 1). The velocity values were separated into positive (displayed as red in the color map) and negative values (displayed as blue in the color map), representing the blood flow direction toward and away from the US probe to separate the arteries and veins, respectively. Quantitation of (H-I) vessel densities and (J-K) renal blood flow calculated from the SRU data showing loss of vessels and reduced blood flow in the SS mouse kidney after multiheme-induced CKD development. ∗P < .05; ∗∗∗P < .001 (unpaired Student t test).
CKD development in sickle mice after repeated hemolytic events. (A) Experimental scheme showing the regimen of repeated low-dose hemin injections, mimicking multiple hemolytic events in SS mice. (B) Kidney function represented by GFR, 1 week after the last hemin challenge in the younger SS mice (4-6 weeks) compared with the vehicle-injected mice (n = 8; M, 4; F, 4). (C-E) Increased albuminuria (represented by uACR), plasma Cys C, and urinary KIM-1 in multiheme-challenged SS mice showing CKD development in younger SS mice. (F) Evans blue extravasation data showing elevated renal endothelial barrier disruption in younger SS mice injected with the multiheme regimen (n = 5-6; M, 2-3; F, 3). (G) Vehicle- and heme-injected SS mice were subjected to SRU imaging 1 week after the last hemin challenge. The reconstructed 2-dimensional, cross-sectional, SRU images of the long-axis mouse kidney showing vessel distribution and the velocity map for the vehicle and hemin-challenged mice (n = 3; M, 2; F, 1). The velocity values were separated into positive (displayed as red in the color map) and negative values (displayed as blue in the color map), representing the blood flow direction toward and away from the US probe to separate the arteries and veins, respectively. Quantitation of (H-I) vessel densities and (J-K) renal blood flow calculated from the SRU data showing loss of vessels and reduced blood flow in the SS mouse kidney after multiheme-induced CKD development. ∗P < .05; ∗∗∗P < .001 (unpaired Student t test).
We then used SRU to assess the vascular structure and blood flow velocity in vehicle- and heme-induced SS mice. Obvious rarefaction of the renal vasculature was identified in the heme-induced SS mice compared with the vehicle-induced ones (Figure 3G). Analysis of the reconstructed 2-dimensional, cross-sectional SRU images of the long-axis mouse kidneys around the cortex and the corticomedullary junction areas revealed significantly lower vessel densities in SS mice after the heme challenge (Figure 3H-I). Accordingly, blood flow in the renal vessels decreased, with substantial attenuation in the arcuate blood vessels (Figure 3J-K). The hindrance of the flow through the renal vasculature of the heme-challenged sickle mouse kidney compared with that of the vehicle-injected mouse kidney was clearly visible in the imaging livestream of the injected microbubble (supplemental Video 1). Whole-kidney slices from heme-induced SS mice showed severe infarction and the corresponding renal tissue sections exhibited severe vascular congestion. Heme also causes renal tubular injury via the release of iron after its degradation in the tubular epithelial cells.38 Increased iron accumulation was evident in the renal tubular epithelium of heme-challenged SS mice (supplemental Figure 4A-B).
Immunofluorescence staining of the renal tissue sections (Figure 3) showed a reduced abundance of both EPCR and CD31 staining in the heme group compared with that in the vehicle group, indicating that multiheme insults led to the detachment of EPCR from the renal vascular endothelial surface (Figure 4A-B). Consequently, the concentrations of both plasma and urinary EPCR were significantly higher in heme-induced SS mice than in vehicle-treated SS mice (Figure 4C-D). Renal tubular epithelial cells also express EPCR.39 We found that EPCR expressed in the renal cortical tubular epithelial cells of the SS mice kidneys remained intact after the heme challenge. Additionally, no elevation of EPCR was evident in the cell supernatant of heme-challenged HK-2 cells, immortalized human renal tubular epithelial cells (supplemental Figure 5A-C). SCD represents a state of hypercoagulation, whereas EPCR is involved in the anticoagulation process. Moreover, elevated thrombin generation reportedly contributes to nephropathy and vascular congestion.40 Thus, we tested total thrombin generation in the kidney tissue and found higher thrombogenesis in heme-challenged SS mice than in vehicle-injected SS mice (Figure 4E).
Alterations in the renal microvascular EPCR distribution in SS mice after multiheme-induced CKD. (A) Representative images from immunofluorescence staining indicating a lack of EPCR in the renal endothelium marked with CD31-positive staining. (B) Quantitation of staining intensity (EPCR/CD31 ratio), indicating significant deterioration of renal endothelial EPCR in heme-challenged mice (n = 6; M, 3; F, 3). (C-D) Higher concentrations of EPCR in the plasma (C) and urine (D) detected in the heme-challenged SS mice compared with those challenged with vehicle (n = 8; M, 4; F, 4). (E) Quantitation of thrombin accumulation in SS mouse kidneys after a multiheme challenge (n = 6; M, 3; F, 3). ∗P < .05; ∗∗P < .01 (unpaired Student t test).
Alterations in the renal microvascular EPCR distribution in SS mice after multiheme-induced CKD. (A) Representative images from immunofluorescence staining indicating a lack of EPCR in the renal endothelium marked with CD31-positive staining. (B) Quantitation of staining intensity (EPCR/CD31 ratio), indicating significant deterioration of renal endothelial EPCR in heme-challenged mice (n = 6; M, 3; F, 3). (C-D) Higher concentrations of EPCR in the plasma (C) and urine (D) detected in the heme-challenged SS mice compared with those challenged with vehicle (n = 8; M, 4; F, 4). (E) Quantitation of thrombin accumulation in SS mouse kidneys after a multiheme challenge (n = 6; M, 3; F, 3). ∗P < .05; ∗∗P < .01 (unpaired Student t test).
Overexpression of EPCR protected sickle mice from heme-induced CKD development
Loss of EPCR from the renal endothelium potentially destabilizes the vasculature, leading to kidney damage under hemolytic conditions. We postulated that sustained expression of EPCR would maintain vascular endothelial integrity, thus impeding heme-induced CKD. To confirm our hypothesis, we studied a genetic mouse model with overexpression of EPCR in the endothelium (EPCR-Ox). These transgenic mice (originally termed as Tie2-EPCR) express abundant EPCR on all endothelial beds.27 We created bone marrow chimera mice with the SCD phenotype on the EPCR-Ox (SSEPCR-Ox) and EPCR-WT (B6; SSEPCR-WT) background (supplemental Figure 6A-B). Moreover, we generated control bone marrow chimera mice by transplanting bone marrow from AA mice to EPCR-WT and EPCR-Ox mice, generating AAEPCR-WT and AAEPCR-Ox mice, respectively (supplemental Figure 6B). After successful engraftment, the SSEPCR-Ox and SSEPCR-WT mice were anemic, similar to the donor SS mice, with low hematocrit and high reticulocyte counts (supplemental Table 1). Eight weeks after bone marrow transplantation, the renal function of the SSEPCR-Ox mice was analogous to that of younger SS mice (Figure 1A), with SSEPCR-Ox mice having higher GFR and lower albuminuria than SSEPCR-WT mice at baseline. Interestingly, SSEPCR-Ox mice had a significantly elevated uACR compared with AAEPCR-Ox mice. SSEPCR-Ox mice had lower total plasma heme (TPH) levels in circulation than SSEPCR-WT mice. Both AAEPCR-WT and AAEPCR-Ox mice had reduced comparable TPH much lower than their SS counterparts (Figure 5A-B; supplemental Figure 6C; supplemental Table 2).
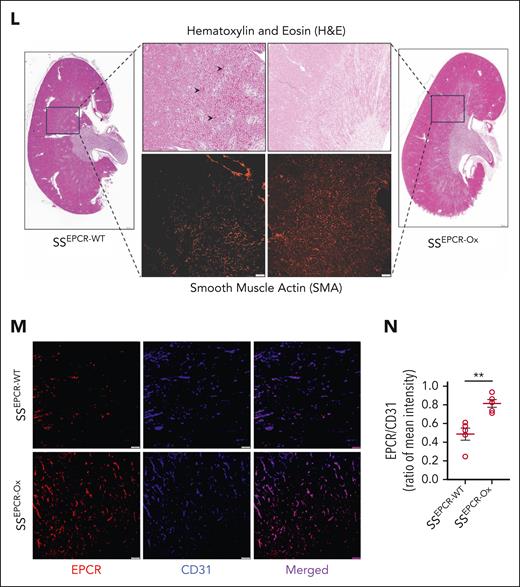
Preservation of renal health after heme-induced CKD in SCD mice with EPCR overexpression. (A-D) EPCR-WT and EPCR-Ox mice underwent transplantation with SS bone marrow to create sickle chimera mice with normal (SSEPCR-WT) and overexpression (SSEPCR-Ox) of EPCR on the vascular endothelium. The mice were then subjected to a multiheme challenge. GFR was measured, and plasma and urine samples were collected at baseline (BL) and 7 days after the last heme injection (H). Worsening renal function showing (A) reduced GFR and increased (B) uACR, (C) plasma Cys C, and (D) urinary KIM-1 in SSEPCR-WT compared with SSEPCR-Ox mice (n = 5-7; M, 2-3; F, 3-4). (E) Plasma sEPCR at BL and after the multiheme challenge in SSEPCR-WT and SSEPCR-Ox mice (n = 6; M, 3; F, 3). (F) Total thrombin in the kidney tissues harvested from SSEPCR-WT and SSEPCR-Ox mice (n = 5-6; M, 2-3; F, 3). (G) Representative restructured SRU images showing the scarcity of renal vasculature in SSEPCR-WT mice (n = 3; M, 1; F, 2). (H-I) Quantitation of vessel density in SSEPCR-WT and SSEPCR-Ox mice (n = 3). (J-K) Calculated renal blood flow in the indicated sickle bone marrow chimera mice after multiheme challenge (n = 3). (L) Representative stitched images (scale bar = 500 μm) of the whole kidney from the multiheme-challenged SSEPCR-WT and SSEPCR-Ox mice. The indicated areas are digitally enlarged to show corticomedullary congestion (arrowhead). Serial tissue sections stained for smooth muscle actin showing microvessel distribution in the indicated area (scale bar = 100 μm). (M-N) Representative immunofluorescence images (scale bar = 50 μm) and quantitation of the EPCR/CD31 intensity ratio in renal tissue sections from SSEPCR-WT and SSEPCR-Ox mice (n = 3) after heme challenge. ∗P < .05; ∗∗P < .001 (paired and unpaired Student t test).
Preservation of renal health after heme-induced CKD in SCD mice with EPCR overexpression. (A-D) EPCR-WT and EPCR-Ox mice underwent transplantation with SS bone marrow to create sickle chimera mice with normal (SSEPCR-WT) and overexpression (SSEPCR-Ox) of EPCR on the vascular endothelium. The mice were then subjected to a multiheme challenge. GFR was measured, and plasma and urine samples were collected at baseline (BL) and 7 days after the last heme injection (H). Worsening renal function showing (A) reduced GFR and increased (B) uACR, (C) plasma Cys C, and (D) urinary KIM-1 in SSEPCR-WT compared with SSEPCR-Ox mice (n = 5-7; M, 2-3; F, 3-4). (E) Plasma sEPCR at BL and after the multiheme challenge in SSEPCR-WT and SSEPCR-Ox mice (n = 6; M, 3; F, 3). (F) Total thrombin in the kidney tissues harvested from SSEPCR-WT and SSEPCR-Ox mice (n = 5-6; M, 2-3; F, 3). (G) Representative restructured SRU images showing the scarcity of renal vasculature in SSEPCR-WT mice (n = 3; M, 1; F, 2). (H-I) Quantitation of vessel density in SSEPCR-WT and SSEPCR-Ox mice (n = 3). (J-K) Calculated renal blood flow in the indicated sickle bone marrow chimera mice after multiheme challenge (n = 3). (L) Representative stitched images (scale bar = 500 μm) of the whole kidney from the multiheme-challenged SSEPCR-WT and SSEPCR-Ox mice. The indicated areas are digitally enlarged to show corticomedullary congestion (arrowhead). Serial tissue sections stained for smooth muscle actin showing microvessel distribution in the indicated area (scale bar = 100 μm). (M-N) Representative immunofluorescence images (scale bar = 50 μm) and quantitation of the EPCR/CD31 intensity ratio in renal tissue sections from SSEPCR-WT and SSEPCR-Ox mice (n = 3) after heme challenge. ∗P < .05; ∗∗P < .001 (paired and unpaired Student t test).
After the multiheme challenge, the SSEPCR-Ox mice showed resistance to CKD development, with unaltered GFR, uACR, and plasma Cys C, whereas the SSEPCR-WT mice had significantly reduced GFR and elevated uACR and plasma Cys C (Figure 5A-C). Multiheme challenge elevated urinary KIM-1 in both SSEPCR-WT and SSEPCR-Ox mice (Figure 5D). Importantly, the heme challenge did not increase the plasma sEPCR level in SSEPCR-Ox mice (Figure 5E). Moreover, SSEPCR-Ox mice exhibited less thrombogenesis than SSEPCR-WT mice (Figure 5F). These data indicate that the stronger presence of EPCR protected the renal vascular endothelium in SSEPCR-Ox mice.
The SRU images showed a distinct improvement in the cortical and corticomedullary vessel densities in SSEPCR-Ox mice (Figure 5G-I), along with enhanced renal blood flow (Figure 5J-K; supplemental Video 2). Hematoxylin and eosin–stained images of whole kidney (cut into longitudinal halves) were captured and stitched together. The renal blood vessels in the corticomedullary areas were severely congested in SSEPCR-WT, whereas those in SSEPCR-Ox mice were relatively empty. The congested corticomedullary area was abundant with vasculature, as evident by the ample smooth muscle actin staining of serial renal sections from SSEPCR-WT and SSEPCR-Ox (Figure 5L). Immunofluorescence imaging recapitulated the SRU findings, showing significantly augmented expression of EPCR in the renal endothelium of SSEPCR-Ox mice (Figure 5M-N). Overall, these data suggest that overexpression of EPCR attenuated the effect of persistent hemolytic stress, thus preventing CKD development in sickle mice.
Elevated sEPCR in plasma was associated with CKD in adult patients with SCD
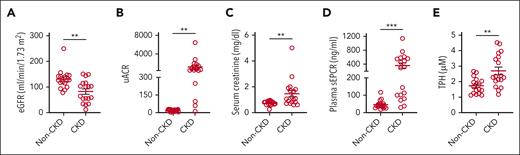
Chronic and acute hemolytic events are common in SCD, and elevated levels of plasma heme are associated with many severe clinical manifestations. We tested the clinical relevance of the increased level of plasma sEPCR observed in SCD mice in a group of SCD individuals with or without CKD. Plasma samples from a randomly selected group of 35 patients with SCD from the biorepository of the Walk-PHaSST study2,28 were separated into 2 groups, CKD (n = 18) and non-CKD (n = 17), based on the prognostic CKD criteria defined by the national kidney foundation (Kidney Disease Improving Global Outcomes [KDIGO], 2012).41 The CKD group had significantly greater albuminuria and lower eGFR than the non-CKD group (Table 1; Figure 6A-C). Importantly, we found that patients with SCD and CKD had significantly higher plasma sEPCR and TPH than those without CKD (Figure 6D-E). Moreover, sEPCR was significantly correlated with albuminuria and TPH, whereas TPH and albuminuria were positively associated (supplemental Figure 7A-C). These data strongly suggest that patients with SCD with high levels of plasma sEPCR may be vulnerable to progressive CKD, particularly under hemolytic conditions.
Clinical characterization of human participants
| . | Non-CKD n = 17 . | CKD n = 18 . |
|---|---|---|
| Female, n | 10/7 | 9/9 |
| Age (y) | 40 (26-51) | 45 (33-52) |
| Hb (g/dL) | 9.3 (7.0-10.0) | 8.1 (6.6-9.4) |
| White blood cells (×103/μL) | 11.0 (8.7-12.5) | 10.2 (6.6-12.3) |
| Neutrophils (×103/μL) | 5.3 (4.9-6.1) | 5.8 (3.2-7.7) |
| Platelets (×103/μL) | 371 (354-501) | 323 (223-410) |
| HbS (%) | 78 (40-90) | 75 (59-88) |
| HbF (%) | 5 (1-9) | 7 (3-17) |
| SS genotype, n | 17 | 17 |
| Hydroxyurea, n | 4 | 10 |
| . | Non-CKD n = 17 . | CKD n = 18 . |
|---|---|---|
| Female, n | 10/7 | 9/9 |
| Age (y) | 40 (26-51) | 45 (33-52) |
| Hb (g/dL) | 9.3 (7.0-10.0) | 8.1 (6.6-9.4) |
| White blood cells (×103/μL) | 11.0 (8.7-12.5) | 10.2 (6.6-12.3) |
| Neutrophils (×103/μL) | 5.3 (4.9-6.1) | 5.8 (3.2-7.7) |
| Platelets (×103/μL) | 371 (354-501) | 323 (223-410) |
| HbS (%) | 78 (40-90) | 75 (59-88) |
| HbF (%) | 5 (1-9) | 7 (3-17) |
| SS genotype, n | 17 | 17 |
| Hydroxyurea, n | 4 | 10 |
Cells contain median (interquartile range [IQR]) unless otherwise specified.
HbF, hb F (fetal); HbS, hb S (sickle).
Association between plasma sEPCR and CKD in patients with SCD. A cohort of patient samples (N = 35) from the Walk-PHaSST biorepository was subdivided into 2 groups: non-CKD and CKD. eGFR (A), uACR (B), and serum creatinine (C) in the indicated groups of patients with SCD. Level of plasma sEPCR (D) and TPH (E) showing elevated circulating EPCR and heme in the CKD group of patients with SCD. ∗∗P < .01; ∗∗∗P < .001 (unpaired Student t test).
Association between plasma sEPCR and CKD in patients with SCD. A cohort of patient samples (N = 35) from the Walk-PHaSST biorepository was subdivided into 2 groups: non-CKD and CKD. eGFR (A), uACR (B), and serum creatinine (C) in the indicated groups of patients with SCD. Level of plasma sEPCR (D) and TPH (E) showing elevated circulating EPCR and heme in the CKD group of patients with SCD. ∗∗P < .01; ∗∗∗P < .001 (unpaired Student t test).
Discussion
Kidney disease gradually develops in individuals with SCD. Microalbuminuria is evident in childhood, progressing to proteinuria and deteriorating GFR in early adulthood, with CKD prevalent in adulthood.5 Previous studies have reported age-dependent deterioration of renal health in sickle mice.35 In this study, we discovered the role of heme, a hemolytic byproduct, in the pathogenesis of the gradual worsening of renal health in Townes’ sickle transgenic mice, recapitulating renal health conditions in human patients with SCD. The pathogenesis of CKD in SCD is complex, but several studies have invariably demonstrated its association with low Hb and hemolysis.42-45 Severe anemia (Hb, 7-9 g/dl) is more common among patients with SCD.46 Moreover, exacerbation of anemia and AKI are predisposing factors for CKD in patients with SCD.47-49 We have previously shown that sickle mice exhibit an age-dependent increase in hemolytic anemia severity, as indicated by a gradual elevation in extracellular heme and other intravascular hemolytic markers.33 Heme triggers endothelial barrier disruption in various organs associated with SCD, including the kidneys.10,50-53 Endothelial injury in the renal peritubular microvessels is closely linked to rarefaction and interstitial fibrosis, leading to CKD progression.54-56 This study showed an association between aging, anemia, and loss of renal endothelial barrier integrity, which led to CKD pathologies including extensive microvascular congestion, rarefaction, and chronic thrombotic microangiopathy.
During the first decade of life, renal complications in SCD are primarily limited to glomerular hyperfiltration and urinary concentrating defects, followed by proteinuria, leading to CKD in late adolescence or adulthood.57 Accordingly, younger sickle mice had significantly higher GFR and slightly higher albuminuria than older AA mice (Figure 1A-B). Larger meta-analysis have shown that episodes of single and repeated AKI significantly raise the risk of CKD,58,59 whereas the true incidence of AKI, primarily diagnosed during acute hemolytic events such as vaso-occlusive crisis or acute chest syndrome in SCD, may be underestimated.48,49 Our current experimental strategy of using multiple heme challenges in younger sickle mice corroborates these hemolytic events.
Abnormal alterations, such as degradation and perfusion impairment of the renal microvasculature are associated with progressive kidney disease.60,61 SRU imaging is an emerging technology that can identify microvessels in vivo with unprecedented spatial resolution, beyond the standard acoustic diffraction limit. The technology has been successfully applied and validated to assess structural and flow changes of the renal microvasculature in animal models using a high-frequency small animal transducer.31,32,62 Using a sophisticated imaging sequence and signal processing algorithms (supplemental Methods; supplemental Figures 10 and 11), we performed noninvasive SRU imaging to quantify the structural and blood flow changes of the renal microvasculature in experimental sickle mice. Along with the physiological evidence of CKD, significantly reduced corticomedullary microvascular density revealed that chronic exposure of the microvascular endothelium to circulating extracellular heme promoted CKD progression. Using a clinical curved array transducer for a deeper imaging depth and larger imaging field of view, this sophisticated SRU technology has the potential to be easily translated into clinical applications in human subjects.
Although EPCR is primarily expressed in the larger blood vessels, multiple studies have shown that EPCR is expressed in the intestinal and renal microvascular endothelium, as well as in the renal tubular epithelium.25,39,63,64 An elevated plasma EPCR level has been reported in lupus erythematosus sepsis. Multiple factors, such as inflammation and increased metalloproteolytic activities, have been implicated in EPCR shedding from the endothelium, thereby raising its plasma level.22,65 The urinary level of EPCR has been shown to be significantly elevated in antibody-mediated acute renal allograft rejection patients.66 In our study, increased plasma and urinary EPCR was associated with heme-induced CKD in SCD mice, potentially due to inflammation and a loss of endothelial integrity caused by elevated circulating heme. We identified both membrane and truncated fractions of EPCR in the experimental mice plasma, indicating that circulating sEPCR may result from the loss of the renal endothelium as well as detached truncated EPCR fragments (supplemental Figure 8). Because we did not identify any loss of EPCR from tubular epithelium in SS mice challenged with heme, the circulating sEPCR that passed through the leaky glomerular filtration might contribute to urinary sEPCR. Moreover, activation of coagulation and insufficient protein C and protein S, hallmarks of SCD, are associated with increased thrombogenesis, which contributes to vascular congestion.67 Previous studies have reported enhanced thrombus formation in cerebral microvessels of mice expressing sickle Hb.68
In this study, we also found elevated thrombin generation in heme-challenged sickle mice kidneys. Earlier studies have shown that patients with SCD and sickle cell mice have lower levels of protein C (PC) and APC activity.69 Reduced APC activity leads to increased thrombin generation, which may trigger thrombin-induced, PAR1-dependent proinflammatory signaling. In addition to its anticoagulant function, APC has cytoprotective signaling functions.70 APC cytoprotective signaling functions depend on EPCR and PAR1.21 Recent studies show that APC-PAR1 biased signaling involving APC cleavage of PAR1 at R46 reduces thromboinflammation in sickle mice.71 EPCR plays a crucial role in determining PAR1 cleavage and signaling specificity of APC.72,73 Loss of EPCR in SCD, as observed in this study, may impair APC cytoprotective signaling. Endothelial cells lose TM upon exposure to cell-free Hb, and patients with SCD have elevated sTM associated with an increased risk of multiorgan failure.74 We found that sTM was induced significantly in the multiheme-challenged SS mice (supplemental Figure 9). Recent studies have shown that TM switches the PAR1 cleavage specificity of thrombin (from R41 to R46) and triggers the cytoprotective signaling of thrombin.75 Interestingly, the sTM level was significantly reduced in SSEPCR-Ox mice compared with SSEPCR-WT mice (supplemental Figure 9). At present, the mechanism for reduced sTM levels in SSEPCR-Ox mice and its relevance to the pathogenesis of SCD are unknown. Apart from thrombotic occlusion, heme-induced renal microvascular rarefaction may involve the induction of metalloproteinase signaling that can cleave EPCR and vascular endothelial cadherin (VE-cadherin), which constitutes a major endothelial junctional complex.25,76 Moreover, hemolysis limits nitric oxide bioavailability associated with increased angiotensin II synthesis, which contributes to severe vasoconstriction, potentially leading to vascular rarefaction.77,78 A significant reduction in vessel density may be responsible for the loss of EPCR, or vice versa, and we found that restoration of EPCR expression ameliorated heme-induced CKD. Expression of EPCR is essential for vascular cryoprotection. Novel therapeutic strategies inducing EPCR overexpression and vascular endothelial integration with synergistic PAR-1 activation may improve hemolysis-induced sickle CKD.
The interpretation of our data is associated with a few limitations. First, heme-induced detachment of EPCR from other vascular beds apart from the kidneys may contribute to the elevation of plasma sEPCR. Second, the level of urinary KIM-1 was elevated in both SSEPCR-WT and SSEPCR-Ox mice after a multiheme challenge. Although, the overexpression of EPCR preserved endothelial integrity improving renal function in sickle mice, the tubular epithelial injury remained unresolved. Third, congestion in the corticomedullary region may also be accompanied by debris from injured renal tubules. Fourth, we found a bimodal distribution of plasma sEPCR levels in patients with SCD, with 1 group showing an sEPCR level similar to that in the non-CKD group (Figure 6D) and the other group showing an approximately threefold elevation in plasma sEPCR. Age, sex, and severity of CKD may contribute to the level of plasma sEPCR. In this study, with limited number of CKD samples (n = 18), we did not find a significant correlation between sEPCR and age, sex, Hb fraction, albuminuria, or eGFR (supplemental Table 3). Moreover, a longitudinal assessment with a larger cohort of patients is required to assess the effect of hydroxyurea use on EPCR shedding. Furthermore, several single nucleotide polymorphisms (SNPs) in the PROCR gene have been associated with altered function of EPCR and disease outcome.79 For example, the presence of 1 such SNP (Ser219Gly; rs867186) resulted in increased plasma sEPCR levels. The rs867186-G variant in exon 4 of PROCR causes a serine-to-glycine substitution in the transmembrane region, rendering the EPCR more susceptible to cleavage.76,80,81 The differential distribution of the level of plasma sEPCR may be due to the presence of specific SNPs within the PROCR gene, which may inspire future studies identifying PROCR SNPs in larger and multiple cohorts of patients with SCD.
Nonetheless, our current findings introduce a new pathobiological mechanism by which circulating heme promotes the progressive development of CKD through EPCR shedding from the renal vascular bed. This study also highlights that overexpression of EPCR may be a therapeutic target for improving renal health, whereas circulating EPCR may be developed as a biomarker for an increased risk of CKD development among patients with SCD.
Acknowledgments
This work was supported by the National Institutes of Health (NIH), National Institute of Diabetes and Digestive and Kidney Diseases (grants R01DK124426 and R01DK132145 [S.G.], R01DK131991 [R.J.T.], P30DK079307, and U54DK137329); NIH, National Heart, Lung, and Blood Institute (grant R01HL157465-01A1 [K.K.]); and funds from the Hemophilia Center of Western Pennsylvania and Vitalant (S.G.).
Authorship
Contribution: Q.C. performed super-resolution ultrasound (SRU) studies and analyzed and interpreted data; R.H. conducted immunofluorescence staining and microscopy; D.C. conducted bone marrow transplantation (BMT) experiments, mouse genotyping, hemoglobin gel electrophoresis, and assisted with SRU experiments; D.L. performed enzyme-linked immunosorbent assays and analyzed the data; S.C.L. assisted with BMT experiments and microscopy; P.M. performed western blot experiments; Y.Z. provided the human samples; S.M.N. provided the clinical data and conducted statistical analysis; R.J.T. was involved in SRU studies; C.T.E. was responsible for generating the endothelial protein C receptor (EPCR) overexpressor mice; L.V.M.R. provided the EPCR overexpressor mice and critically reviewed the manuscript; K.K. developed SRU algorithms and supervised all SRU experiments; and S.G. conceived and designed the study, analyzed and interpreted the data, supervised the project, and wrote the manuscript with consultation and contributions from all authors.
Conflict-of-interest disclosure: S.G. receives research funding (not relevant to the present study) from Pfizer Inc as part of a sponsored research agreement. The remaining authors declare no competing financial interests.
Correspondence: Samit Ghosh, Department of Medicine, Pittsburgh Heart, Lung, and Blood Vascular Medicine Institute, BST-E1200-10A, 200 Lothrop St, Pittsburgh, PA 15261; email: sag130@pitt.edu.
References
Author notes
Presented in abstract form at the 63rd annual meeting of the American Society of Hematology, Atlanta, GA, 11 December 2021 (available at https://doi.org/10.1182/blood-2021-154027), and the 2023 IEEE (Institute of Electrical and Electronics Engineers) International Ultrasonics Symposium, Montreal, QC, Canada, 3-8 September 2023.
Original data are available on request from the corresponding author, Samit Ghosh (sag130@pitt.edu).
The online version of this article contains a data supplement.
There is a Blood Commentary on this article in this issue.
The publication costs of this article were defrayed in part by page charge payment. Therefore, and solely to indicate this fact, this article is hereby marked “advertisement” in accordance with 18 USC section 1734.

![Progressive renal damage and development of CKD in sickle mice. (A) Townes sickle (SS) mice from different age groups (1-10 months; n = 9; M, 5; F, 4) and older (10 months; n = 9; M, 3; F, 6) control (AA) mice were used to assess GFR noninvasively using a transcutaneous device. An age-dependent decline in GFR was evident with hyperfiltration in younger (1-month) SS mice. (B) Urinary albumin and creatinine were measured in the urine collected from the same cohort of mice as in (A) before GFR measurement, and the ratio (uACR) indicated a significant increase in albuminuria. (C) Plasma Cys C, measured using enzyme-linked immunosorbent assay in the same cohort of mice, was higher in SS mice of all ages than in AA mice. (D) Elevated urinary KIM-1 in the SS mice, with no difference between older AA and 1-month-old SS mice. (E) Representative hematoxylin and eosin images showing an age-dependent increase in renal microvascular congestion (arrows) in SS mice (scale bar = 20 μm). (F) SRU images of the kidneys in a separate cohort of AA (10-month; n = 3; M, 2; F, 1) and SS (1-month [n = 3; M, 2; F, 1] and 10-month [n = 3; M, 2; F, 1]) mice, showing representative overlaid B-mode images. Quantitation of vessel density in the (G) cortex and (H) corticomedullary regions of interest, as described in the supplemental Methods, showing a loss of renal microvasculature in older SS mice (n = 3). (I) Reduced renal blood volume (rBV) in older SS mice compared with age-matched AA mice (n = 3). ∗P < .05; ∗∗P < .01; ∗∗∗P < .001 (1-way analysis of variance (ANOVA) between differentially aged SS mice and unpaired Student t test between 10-month-old AA and SS mice). F, female mice; M, male mice; ns, nonsignificant.](https://ash.silverchair-cdn.com/ash/content_public/journal/blood/144/5/10.1182_blood.2023023528/2/m_blood_bld-2023-023528-gr1.jpeg?Expires=1766641897&Signature=YTB4SCSi5SpMLwimoouBOxDR2kg7vQjYls89I8ezJK7yoIAjWBnC12AMOgxRCAV4dCRu9V6Aum-vDlCrira~-tftXlXwY9adjeOABzPQphjZEiPpcCjdbD~9tAPaACRGcwjd-NzFtUNEUaTs3PgCWAQd3kpO-lUgc8Z0ltNL0Qml0SGPqEClAFxp1gLCzUKYqPI2LtlRzEkjL9V2F5aR3dWoaJX5ghSg96NLPAd1Wm8kN6rjAv6AhqbMWIdZ0ILkQvI1GYgzfe9jDKSIyUwKduHAKIlUFiST-wYyPrxz~0JjzicX4wLRDyDUunY3kME5eZSMAcSXpsnd7W0vn5foOA__&Key-Pair-Id=APKAIE5G5CRDK6RD3PGA)



This feature is available to Subscribers Only
Sign In or Create an Account Close Modal