Visual Abstract
Measurable residual disease (MRD) evaluation by multiparameter flow cytometry (MFC) or quantitative polymerase chain reaction methods is an established standard of care for assessing risk of relapse before or after hematopoietic cell transplantation (HCT) for acute lymphoblastic leukemia (ALL). Next-generation sequencing (NGS)–MRD has emerged as a highly effective approach that allows for the detection of lymphoblasts at a level of <1 in 106 nucleated cells, increasing sensitivity of ALL detection by 2 to 3 logs. Early studies have shown superior results compared with MFC and suggest that NGS-MRD may allow for the determination of patients in whom reduced toxicity transplant preparative approaches could be deployed without sacrificing outcomes. Many centers/study groups have implemented immune modulation approaches based on MRD measurements that have resulted in improved outcomes. Challenges remain with NGS-MRD, because it is not commercially available in many countries, and interpretation of results can be complex. Through patient case review, discussion of relevant studies, and detailed expert opinion, we share our approach to NGS-MRD testing before and after HCT in pediatric and adult ALL. Improved pre-HCT risk classification and post-HCT monitoring for relapse in bone marrow and less invasive peripheral blood monitoring by NGS-MRD may lead to alternative approaches to prevent relapse in patients undergoing this challenging procedure.
Introduction
The use of next-generation sequencing (NGS) of B-cell receptor (BCR) and T-cell receptor (TCR) gene rearrangements to quantitate measurable residual disease (MRD) represents a major advancement in the clinical management of children and adults with acute lymphoblastic leukemia (ALL). ClonoSEQ (Adaptive Biotechnologies, Seattle, WA) is an NGS–based in vitro diagnostic assay that has received US Food and Drug Administration (FDA) clearance for MRD testing in B-cell ALL (B-ALL) and is currently in clinical use for this indication.1 Relative to multiparameter flow cytometry (MFC) or quantitative polymerase chain reaction (PCR), NGS-MRD achieves a greater depth of leukemia detection of at least 10–6 or 0.0001% in adequately cellular bone marrow (BM) specimens.2 Increased MRD sensitivity improves prognostication and provides greater lead time for interventions to ward off clinical relapse.3-8 Furthermore, NGS-MRD allows for clonal tracking, and the strong correlation between blood and BM results poses an opportunity to monitor MRD by less invasive means.5,9
Allogeneic hematopoietic cell transplantation (HCT) is presently reserved for children and adults with ALL at high risk for disease relapse with nontransplant therapies, namely, patients with persistent MRD after initial induction/consolidation or patients in first or later relapse. A burgeoning literature exists supporting the use of NGS-MRD in the HCT setting.3,5,6,9 However, clarity on the optimal timing of NGS-MRD testing in the context of transplant, nuances in interpretation, and options for therapeutic intervention based on peri-HCT results complicate the use of this technology. In this article, we use an exemplar case of a patient with B-ALL to illustrate clinical pearls and expert guidance associated with use of the clonoSEQ NGS-MRD assay in allogeneic HCT.
Case
A 20-year-old female was diagnosed with Philadelphia chromosome-like (Ph-like) P2RY8-CRLF2 rearranged B-ALL. At diagnosis, a BM aspirate sample was sent for clonoSEQ identification and demonstrated 2 dominant clonal sequences: sequence A, an immunoglobulin heavy chain (IgH) clonal sequence present in 650 000 out of 100 000 nucleated cells; and sequence B, an Igκ clonal sequence present in 580 000 out of 1 000 000 nucleated cells. After a pediatric multiagent chemotherapy induction, she achieved morphologic complete remission (CR) with 0.8% residual leukemic blasts by flow cytometry, which increased to 6% leukemic blasts after consolidation. She then received CD19–directed chimeric antigen receptor (CAR) T-cell therapy and achieved a CR by morphology and flow cytometry; however, clonoSEQ returned positive at 3 months after CAR T-cell infusion for residual disease with 2 clonal cells per million nucleated cells detected for sequence A (IgH) and 5 clonal cells per million nucleated cells detected for sequence B (Igκ). Laboratory evaluation of the patient was notable for a CD19 count of 80 cells per μL. Consolidative allogeneic HCT was recommended.
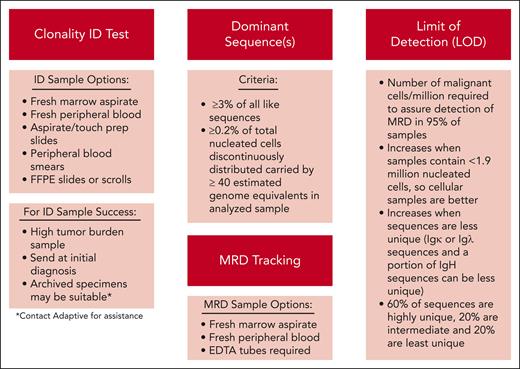
Identification of dominant sequence(s) for NGS-MRD tracking
This case demonstrates several important points for the practicing physician related to the use of NGS-MRD in ALL. First, unlike flow cytometry, in which a diagnostic sample is typically not necessary for subsequent MRD evaluation, the use of clonoSEQ requires initial sequencing of diagnostic leukemia material to identify the dominant clonal sequences for MRD tracking. Thus, during initial leukemia workup, it is important to send either fresh BM aspirate or peripheral blood for clonality assessment (Figure 1). Unstained BM aspirate slides, touch prep slides, or peripheral blood smears may also be used for sequence identification; however, decalcified BM core biopsy samples are not usable for this purpose. For patients with predominant lymphomatous presentation of ALL, unfixed frozen tissue or formalin-fixed paraffin-embedded slides or scrolls may be suitable. Samples with high tumor load that clearly represent the cytomorphologically defined disease are preferred. Some programs send clonoSEQ clonality identification testing on all patients with newly diagnosed ALL. When this is not feasible, we recommend sending clonality identification testing on patients who will likely receive HCT. In such cases, if clonoSEQ identification testing was not sent at diagnosis, at the time of HCT evaluation, we recommend sending samples or slides that were obtained or banked at diagnosis. Many centers now have procedures for banking samples obtained at diagnosis for future testing, and Adaptive has capabilities of using initial specimen samples or slides for determining tracking sequences.
Stepwise approach to identification of clonal dominant sequence(s) and subsequent MRD tracking.
Stepwise approach to identification of clonal dominant sequence(s) and subsequent MRD tracking.
Identification of dominant sequence(s) is performed by analysis of unique V(D)J (variable diversity joining) and/or TCR gene rearrangements present in the diagnostic material. For a sequence to be deemed “dominant,” it must represent ≥3% of sequences from the same locus (ie, IgH, Igκ, or Igλ) and ≥0.2% of the total nucleated cells in the sample known to contain a high percentage of blasts. Prior studies have shown that ∼90% of ALL samples tested will contain dominant sequence(s).6,7 The most common reasons for failure to identify a dominant sequence are immature blasts that have not yet undergone rearrangement (eg, reported in approximately one-third of early T-cell precursor ALL10), poor quality DNA, or low tumor burden in the evaluated sample. Approximately half of patients with B-ALL who do not have a dominant sequence within the BCRs will have a TCRβ or TCRγ rearrangement that could be used for MRD tracking; however, there is a paucity of data on whether TCR monitoring holds the same level of clinical utility in this setting.
Once identified, each dominant sequence is then assigned a “uniqueness” metric based on survey of a database of hundreds of individuals and computational modeling of locus-specific V(D)J rearrangements. This uniqueness metric is then used to determine sensitivity and limit of detection (LOD; defined as the number of malignant cells per million required to ensure detection of MRD in 95% of samples, hence, lower numbers indicate higher sensitivity; see Figure 1).11 After the identification of dominant sequence(s), MRD evaluation in future BM aspirates or blood samples is enabled. Of note, if identification of informative sequences is not successful with initial samples, it is worthwhile to send samples again at subsequent relapse in case an informative dominant sequence has emerged, or if the initial sample had poor DNA quality or low tumor burden.
Clinical considerations regarding pre-HCT NGS-MRD
As described in the example case above, residual disease detected by NGS-MRD after CAR T-cell therapy has been associated with a significantly increased risk of clinical relapse.3,12 In the setting of NGS-MRD detected >1 month after tisagenlecleucel CAR T-cell infusion, allogeneic HCT should be considered in transplant-naïve individuals, because these patients are highly likely to relapse regardless of persistent B-cell aplasia.13 The role of HCT as consolidation after CAR T-cell therapy in children who achieve NGS-MRD negative remission has recently been extensively reviewed, and there are data suggesting that HCT can be avoided in some patients with persistently negative NGS-MRD.13 When patients after CAR T-cell therapy have positive NGS-MRD, we do not know whether their blasts continue to express CD19 or CD22, making therapeutic choices difficult. Consolidation of CAR T-cell response with HCT in adults with ALL remains an area of debate; however, our current practice is to consider consolidative HCT in transplant-naïve adults with ALL who achieve NGS-MRD negative CR after commercial CAR T-cell therapies that do not persist long-term. The CAR-CURE trial (NCT05621291) follows children and young adults with serial NGS-MRD after CAR T-cell therapies that can persist long-term (tisagenlecleucel) to define which patients can avoid HCT. We would advocate for similar research in the adult ALL research community.
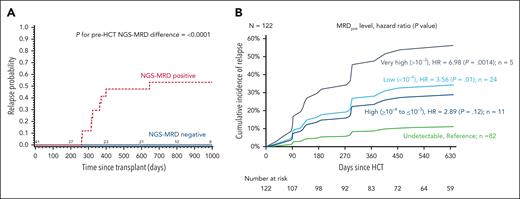
In general, clearance of NGS-MRD before HCT is preferred whenever feasible. Several studies have demonstrated that detectable MRD by NGS at any level before HCT predicts for increased risk of post-HCT relapse and inferior survival. This was shown in an analysis of children and adolescents enrolled on the Children’s Oncology Group/Pediatric Transplantation and Cell Therapy Consortium ASCT0431 trial, in which the 2-year probability of relapse among patients with detectable pre-HCT NGS-MRD was 53% vs 0% among patients who were NGS-MRD–positive and –negative, respectively (Figure 2A).3 Relapse risk was high even among patients at the lowest level of MRD detection (<10–6). Pre-HCT NGS-MRD improved sensitivity for determining risk of post-HCT relapse and poor overall survival compared with pre-HCT MFC. Similar findings have been reported among adult patients with ALL, in which any level of detectable pre-HCT NGS-MRD has been shown to associate with inferior transplant outcomes (Figure 2B).5,6
Associations between pre-HCT NGS-MRD and post-HCT relapse. In children and young adults with ALL (any level of NGS-MRD is considered positive)3 (A) and adult patients with ALL6 (B).
Blinatumomab, a bispecific T-cell engager targeting CD19, is FDA approved for use in both children and adults with B-ALL in first or second CR with MRD ≥10–3. Full approval of blinatumomab for this indication was based on results of the phase 2 BLAST trial14 in adults and the results of 2 phase 3 trials in children (NCT02101853 in the United States15 and NCT02101853 in Europe16). Although these trials did not evaluate the efficacy of blinatumomab at NGS-MRD levels <10–3, studies suggest that this approach is feasible and results in NGS-MRD clearance in a proportion of patients with very low detectable residual disease with 13 of 19 and 14 of 26 NGS-MRD-positive patients clearing NGS-MRD after blinatumomab in 2 small studies.17,18 Additionally, although CAR T cells were initially tested only in patients with relapsed or refractory morphologic B-ALL, real world evidence studies suggest that CAR T-cell therapies, including both tisagenleucleucel in children and brexucabtagene autoleucel in adults, are often successfully used in patients in CR to clear detectable MRD; however, this requires additional prospective study.19,20
It is important to note that the desire to obtain deep MRD clearance before HCT must be weighed against other practical considerations such as transplant and donor coordination. Therapy aimed at reduction of NGS-MRD may not always be feasible because transplant timing can vary based on donor selection and availability. Furthermore, it is possible that administering additional lines of therapy aimed at driving NGS-MRD to zero before transplant will not always be effective and may result in added toxicity and delay. If clearance of NGS-MRD cannot be obtained and the patient is an HCT candidate otherwise, we recommend proceeding with HCT because studies have shown that at least half of children and young adults with pre-HCT MRD that is negative by MFC and positive by NGS will still experience long-term disease-free survival.21
Case (continued)
The patient underwent myeloablative conditioning with total body irradiation (TBI) and fludarabine followed by HCT using an HLA-matched unrelated donor. Graft-versus-host disease (GVHD) prophylaxis included posttransplant cyclophosphamide, tacrolimus, and mycophenolate mofetil. Repeat BM examination 28 days after HCT showed CR with negative residual disease by MFC and clonoSEQ NGS-MRD.
Modifications of the HCT design and post-HCT approach based on NGS-MRD
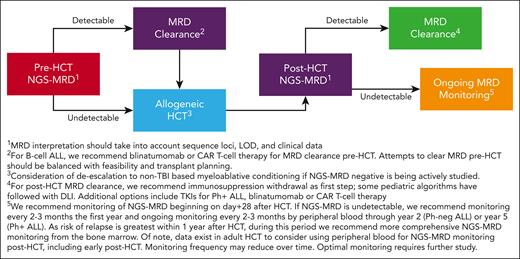
Emerging data suggest that the approach to transplant conditioning, GVHD prophylaxis, and post-HCT maintenance may be guided in part by peri-HCT NGS-MRD results (Figure 3). Myeloablative TBI, long considered a standard component of conditioning in ALL, results in increased late effects in children and second primary malignancies in adult recipients of HCT.22-24 Intriguing retrospective data showed that pre-HCT NGS-MRD may distinguish patients requiring myeloablative dosing of TBI from those for whom TBI can be safely omitted. In one study, children and young adults with detectable NGS-MRD pre-HCT had significantly reduced relapse rates and improved event-free survival after receiving myeloablative TBI-based conditioning, as opposed to non-TBI myeloablative chemotherapy conditioning regimens.25 However, patients with undetectable pre-HCT NGS-MRD had equally excellent outcomes after TBI or non-TBI conditioning. The PTCTC ONC1701 EndRAD Study (NCTN 03509961) is a phase 2 prospective clinical trial in children and young adults aged up to 25 years designed to use NGS-MRD to escalate or de-escalate transplant conditioning. Early results from a large observational cohort that is part of the study suggest that myeloablative TBI may potentially be omitted in patients with undetectable NGS-MRD before HCT,21 although reporting of the larger EndRAD phase 2 cohort is pending, and conclusive randomized trials have not been conducted to date. Data addressing this issue are lacking in adult ALL, but interestingly, findings in acute myeloid leukemia and myelodysplastic syndrome suggest that pre-HCT NGS-MRD may discriminate adults who require myeloablative transplant preparative regimens from those in whom reduced intensity conditioning would suffice.26
Approach to HCT for ALL in children and adults based on pre- and post-HCT NGS-MRD.
Approach to HCT for ALL in children and adults based on pre- and post-HCT NGS-MRD.
Persistence or emergence of detectable NGS-MRD after HCT suggests impending clinical relapse and portends a poor prognosis.3,5,6 Interventions to prevent emergence of NGS-MRD or to treat low-level MRD in this setting include reduction of post-HCT immunosuppression to induce graft-versus-leukemia and/or the introduction of additional agents intended to consolidate or maintain remission. Several studies have shown that an intervention “window” of ∼3 to 6 months exists between the first instance of detectable NGS-MRD and the development of clinical relapse after HCT.5,9,27 Early reduction or withdrawal of immunosuppression after HCT is an appealing strategy due to the inverse relationship between acute GVHD development and B- or T-ALL relapse.28-30 A secondary analysis of the pediatric ASCT0431 trial in fact demonstrated that among patients with detectable MRD after HCT, patients who developed acute GVHD had lower risk of relapse and higher event-free survival relative to patients who did not develop GVHD.27 However, only pediatric patients who experienced grades 1 to 3 acute GVHD received clinical benefit because the reduction in relapse risk was negated by an increased risk of transplant-related mortality in patients who developed grade 4 acute GVHD. This association was also seen in a large retrospective multicenter cohort of children from North America, Europe, and Australia; relapse rates among patients with detectable post-HCT MRD who developed acute GVHD were similar to those who were MRD-negative and did not develop acute GVHD.31 Immunosuppression withdrawal resulting in resolution of post-HCT NGS-MRD has been reported9; however, this approach must be applied on an individualized basis after weighing the associated risks and benefits, because reported literature addressing this approach is limited and mostly retrospective (see Table 1).
NGS-MRD in the context of MFC and qPCR MRD: approaches and interventions
| NGS-MRD pediatric and adult studies in HCT . | Other MRD approaches and comparative studies . | Interventions based upon MRD pre-/post-HCT . |
|---|---|---|
| Pulsipher et al3(n = 56): in children pre-HCT NGS-MRD negativity vs positivity led to a 2-y relapse rate of 0% vs 53% and 2-y OS 96% vs 48% in pre-HCT MRD-negative vs MRD-positive. Post-HCT NGS-MRD was highly sensitive in predicting relapse. Abdel-Azim et al21(n = 87): NGS-MRD–negative children pre-HCT had EFS/OS >90% whether treated with TBI or non-TBI. NGS MRD-positive: 2-year EFS of 50%. Liang et al6(n = 158): adult multicenter, n = 158. Pre-HCT NGS-MRD was undetectable (0), low (<10–4), high (≥10–4 and ≤10–3), and very high (>10–3) in 82 (67%), 24 (20%), 11 (9%), and 5 (4%), respectively. Any level of NGS-MRD detected pre-HCT increased relapse (HR, 6.31; P < .001). Muffly et al9(n = 62): strong correlation between PB and BM NGS-MRD. Sensitivity and specificity of NGS-MRD was 87% and 90% in PB. Median time from detection of PB MRD to relapse was 90 d after HCT and 60 d after CART. Short et al8(n = 44, in retrospective cohort; n = 65, in prospective cohort): NGS-MRD clarifies multilineage BCR::ABL ALL in adults; 32% discordance between BCR::ABL and NGS-MRD; and 11 of 65 patients achieved NGS-MRD negativity and did not relapse despite detection of BCR::ABL by PCR. | MFC (n = 105, 146): multiparameter flow cytometry (MFC) MRD highly predictive of relapse, EFS, and OS before HCT; poor sensitivity early after HCT but increased by 8-12 mo after HCT. Acute GVHD improves EFS/OS due to lower relapse, chronic GVHD does not due to higher treatment-related mortality.27,32 IgH/TCR PCR/qPCR MRD (n = 64, 71, 17, 91, 113): highly predictive of relapse, EFS, and OS before HCT; high risk >10–3, intermediate risk 10–3 to 10–4, low risk <10–4. High sensitivity for relapse prediction both early and late after HCT.33-37 Comparative studies: MFC vs PCRpediatric(N = 616): PCR has improved sensitivity compared to MFC-MRD, doubling the number of patients detected at risk in similar populations.31 MFC vs NGS-MRD pediatric(n = 184): NGS-MRD improved predictive power for relapse, EFS, and OS before HCT compared with MFC. NGS-MRD highly predictive of relapse at any time after HCT.3 MFC vs NGS-MRD adult (n = 25): in 17% of cases, MRD detected by NGS but not MFC.38 There was >80% relapse-free survival in NGS-MRD-negative patients.38 | Balduzzi et al39(n = 82): pre-HCT MRD >10–4 associated with 9.2 hazard of relapse. Additional chemotherapy to reduce MRD associated with fivefold reduction in risk of relapse. Pre-HCT MRD >10–4 + no GVHD post-HCT: IS tapered at month 2, relapse rate 46%. Post-HCT MRD associated with 2.5-fold risk of relapse in the first 100 d; rose to 7.8-fold risk if detected after 6 mo. All patients with MRD >10–3 after HCT relapsed. Rettinger et al40(n = 58): withdrawal of IS ± DLI given to some patients with mixed chimerism (MC) and/or MRD+ early after. Those with MC/MRD+ not getting intervention relapsed, but those with MC/MRD+ getting intervention had similar 3-y EFS to those who were MRD-negative and did not have MC. Lancaster et al41(n = 48): pediatric patients with >10−4 MRD by PCR pre-HCT had early withdrawal of IS ± DLI. EFS in the group remained low at 19%, but relapses were delayed up to 3 y. |
| NGS-MRD pediatric and adult studies in HCT . | Other MRD approaches and comparative studies . | Interventions based upon MRD pre-/post-HCT . |
|---|---|---|
| Pulsipher et al3(n = 56): in children pre-HCT NGS-MRD negativity vs positivity led to a 2-y relapse rate of 0% vs 53% and 2-y OS 96% vs 48% in pre-HCT MRD-negative vs MRD-positive. Post-HCT NGS-MRD was highly sensitive in predicting relapse. Abdel-Azim et al21(n = 87): NGS-MRD–negative children pre-HCT had EFS/OS >90% whether treated with TBI or non-TBI. NGS MRD-positive: 2-year EFS of 50%. Liang et al6(n = 158): adult multicenter, n = 158. Pre-HCT NGS-MRD was undetectable (0), low (<10–4), high (≥10–4 and ≤10–3), and very high (>10–3) in 82 (67%), 24 (20%), 11 (9%), and 5 (4%), respectively. Any level of NGS-MRD detected pre-HCT increased relapse (HR, 6.31; P < .001). Muffly et al9(n = 62): strong correlation between PB and BM NGS-MRD. Sensitivity and specificity of NGS-MRD was 87% and 90% in PB. Median time from detection of PB MRD to relapse was 90 d after HCT and 60 d after CART. Short et al8(n = 44, in retrospective cohort; n = 65, in prospective cohort): NGS-MRD clarifies multilineage BCR::ABL ALL in adults; 32% discordance between BCR::ABL and NGS-MRD; and 11 of 65 patients achieved NGS-MRD negativity and did not relapse despite detection of BCR::ABL by PCR. | MFC (n = 105, 146): multiparameter flow cytometry (MFC) MRD highly predictive of relapse, EFS, and OS before HCT; poor sensitivity early after HCT but increased by 8-12 mo after HCT. Acute GVHD improves EFS/OS due to lower relapse, chronic GVHD does not due to higher treatment-related mortality.27,32 IgH/TCR PCR/qPCR MRD (n = 64, 71, 17, 91, 113): highly predictive of relapse, EFS, and OS before HCT; high risk >10–3, intermediate risk 10–3 to 10–4, low risk <10–4. High sensitivity for relapse prediction both early and late after HCT.33-37 Comparative studies: MFC vs PCRpediatric(N = 616): PCR has improved sensitivity compared to MFC-MRD, doubling the number of patients detected at risk in similar populations.31 MFC vs NGS-MRD pediatric(n = 184): NGS-MRD improved predictive power for relapse, EFS, and OS before HCT compared with MFC. NGS-MRD highly predictive of relapse at any time after HCT.3 MFC vs NGS-MRD adult (n = 25): in 17% of cases, MRD detected by NGS but not MFC.38 There was >80% relapse-free survival in NGS-MRD-negative patients.38 | Balduzzi et al39(n = 82): pre-HCT MRD >10–4 associated with 9.2 hazard of relapse. Additional chemotherapy to reduce MRD associated with fivefold reduction in risk of relapse. Pre-HCT MRD >10–4 + no GVHD post-HCT: IS tapered at month 2, relapse rate 46%. Post-HCT MRD associated with 2.5-fold risk of relapse in the first 100 d; rose to 7.8-fold risk if detected after 6 mo. All patients with MRD >10–3 after HCT relapsed. Rettinger et al40(n = 58): withdrawal of IS ± DLI given to some patients with mixed chimerism (MC) and/or MRD+ early after. Those with MC/MRD+ not getting intervention relapsed, but those with MC/MRD+ getting intervention had similar 3-y EFS to those who were MRD-negative and did not have MC. Lancaster et al41(n = 48): pediatric patients with >10−4 MRD by PCR pre-HCT had early withdrawal of IS ± DLI. EFS in the group remained low at 19%, but relapses were delayed up to 3 y. |
DLI, donor lymphocyte infusion; EFS, event-free survival; IS, immunosuppression; OS, overall survival; PB, peripheral blood; qPCR, quantitative PCR.
In patients with Ph-positive (Ph+) B-ALL, the use of tyrosine kinase inhibitors to reduce MRD or as maintenance therapy after HCT is well established.42,43 Although the ideal intervention to eliminate detectable NGS-MRD after HCT remains undefined, the excellent data demonstrated with both blinatumomab and CAR T-cell therapies in MRD-positive patients suggest that either is a reasonable approach to a patient with emerging or persistent NGS-MRD after HCT. Donor-derived allogeneic CAR T-cell approaches for MRD clearance after HCT are also under study.44
Case (continued)
At 60 days after HCT, the patient developed stage 2 acute GVHD of the skin, which was treated with topical steroids with resolution. Repeat BM aspirate at 90 days after HCT showed ongoing CR; clonoSEQ NGS-MRD resulted as undetectable for sequence A (IgH) (LOD <1 per million nucleated cells) but detected sequence B (Igκ) as <1 clonal cells per million nucleated cells (LOD, 47 per million nucleated cells). Repeat clonoSEQ NGS-MRD 1 month later was negative. The patient was tapered off tacrolimus by 5 months after HCT without further GVHD. ClonoSEQ was sent from the BM at 6, 9, and 12 months after HCT and was negative for residual disease. Peripheral blood clonoSEQ was monitored every 2 to 3 months during the second year after HCT and remained negative.
Interpretation of NGS-MRD results and post-HCT monitoring
Proper interpretation of the results of the clonoSEQ report is imperative for clinicians using this test for clinical decision-making (Table 2). Although some patients with B-ALL may have one dominant sequence, others may have numerous dominant sequences in different loci. When using this assay for B-ALL MRD monitoring, one should take note of the LOD for the dominant sequence(s) described within the report because sequences with higher LOD pose an increased risk of generating a nonspecific signal. Relative to IgH (heavy chain) sequences, Igκ and Igλ (light chain) sequences often have higher LOD due to greater diversity and uniqueness of the IgH loci relative to light chain loci. This is a result of biology because heavy chains rearrange V, D, and J, whereas light chains only rearrange V and J, thus having less opportunities for diversity. Approximately 60% of identified IgH clonal sequences have very low LODs due to their highly unique nature, whereas 20% of IgH sequences are of intermediate uniqueness, and 20% are less unique (more likely to occur as background clonal presence). Another factor influencing LOD is the numbers of nucleated cells in the sample. The optimal LOD for detecting MRD at a sensitivity of 1 per million total cells is achieved when at least 1.9 million nucleated cells are present in the tested sample. Although MRD can still be detected when samples with lower numbers of nucleated cells are evaluated, the LOD per million will increase, and the 95% confidence intervals surrounding the detected MRD as an integer value per million will be wider. If patients are being tracked with TCRβ or TCRγ sequences, uniqueness may be lower (see “T-ALL and B-ALL without IgH sequences”). It is important to keep in mind that although an isolated less unique sequence may represent a nondisease-containing clonal expansion, when less unique sequences track with more specific (low LOD) sequences, such less unique sequences likely represent true disease and should not be ignored.
Tips for using and interpreting the clonoSEQ NGS-MRD assay
| Frequently asked questions . | Expert recommendation . |
|---|---|
| Can I use NGS-MRD for my patient if I did not send a sample for ID at diagnosis? | Identification of a dominant sequence from diagnostic material is required for subsequent MRD tracking. If not sent at diagnosis, dominant sequence identification can be performed using archived unstained marrow aspirate or peripheral smear slides. Relapse samples can also be used to define dominant sequences if not done previously. |
| Can I use NGS-MRD for a patient with a lymphoblastic lymphoma presentation of ALL? | Yes, dominant sequence identification can be performed on diagnostic tissue by sending FFPE slides or scrolls. |
| The ID report resulted as “polyclonal.” What should I do? | A polyclonal result indicates that dominant clonal sequences were not identified on the specimen sent for analysis. Etiologies include (1) immature leukemia that has not undergone rearrangement; (2) sample did not contain adequate malignant cells; and (3) degraded DNA. If an alternative sample is available, it can be sent for a second attempt. TCR sequences can also be explored. |
| What does it mean when the MRD report is positive but below LOD? | This means that dominant sequence(s) were detected below the limit of sequence detection. Each dominant sequence has an individual LOD. It is important to assess the result in context with the type of sequence detected (eg, IgH vs Igκ vs Igλ), the sequence LOD, and the clinical data. Positive IgH sequences below the LOD are often true positives and should not be ignored, as they have been shown to be highly predictive of relapse after HCT and CAR T-cell therapy. Isolated positive sequences (only 1 tracking dominant sequence from the baseline sample) in which LOD is high and more specific sequences are not present warrant repeat observations over time, because they may be false positives. |
| What if a new dominant sequence emerges as I follow my patient? | During the course of therapy, expansion pressure may lead to a hematopoietic clone rising to levels that meet the definition of dominant sequence. Such clonal sequences would then be reported out as new dominant sequences. If established tracking sequences associated with disease are not present, the new dominant sequence likely does not represent disease, and may decrease or disappear with time. |
| Can I use peripheral blood to track NGS-MRD? | Although not FDA cleared for ALL, the use of peripheral blood for NGS-MRD tracking is clinically available. Studies show strong correlation between blood and marrow for NGS-MRD assessment, although marrow generally has a higher level of sensitivity. |
| I do not understand the MRD report. What should I do? | Several resources are available to help with report interpretation. It is highly advised to reach out to an expert if there are questions, particularly if clinical intervention is under consideration. Adaptive offers services for their assay to answer these types of questions. |
| Frequently asked questions . | Expert recommendation . |
|---|---|
| Can I use NGS-MRD for my patient if I did not send a sample for ID at diagnosis? | Identification of a dominant sequence from diagnostic material is required for subsequent MRD tracking. If not sent at diagnosis, dominant sequence identification can be performed using archived unstained marrow aspirate or peripheral smear slides. Relapse samples can also be used to define dominant sequences if not done previously. |
| Can I use NGS-MRD for a patient with a lymphoblastic lymphoma presentation of ALL? | Yes, dominant sequence identification can be performed on diagnostic tissue by sending FFPE slides or scrolls. |
| The ID report resulted as “polyclonal.” What should I do? | A polyclonal result indicates that dominant clonal sequences were not identified on the specimen sent for analysis. Etiologies include (1) immature leukemia that has not undergone rearrangement; (2) sample did not contain adequate malignant cells; and (3) degraded DNA. If an alternative sample is available, it can be sent for a second attempt. TCR sequences can also be explored. |
| What does it mean when the MRD report is positive but below LOD? | This means that dominant sequence(s) were detected below the limit of sequence detection. Each dominant sequence has an individual LOD. It is important to assess the result in context with the type of sequence detected (eg, IgH vs Igκ vs Igλ), the sequence LOD, and the clinical data. Positive IgH sequences below the LOD are often true positives and should not be ignored, as they have been shown to be highly predictive of relapse after HCT and CAR T-cell therapy. Isolated positive sequences (only 1 tracking dominant sequence from the baseline sample) in which LOD is high and more specific sequences are not present warrant repeat observations over time, because they may be false positives. |
| What if a new dominant sequence emerges as I follow my patient? | During the course of therapy, expansion pressure may lead to a hematopoietic clone rising to levels that meet the definition of dominant sequence. Such clonal sequences would then be reported out as new dominant sequences. If established tracking sequences associated with disease are not present, the new dominant sequence likely does not represent disease, and may decrease or disappear with time. |
| Can I use peripheral blood to track NGS-MRD? | Although not FDA cleared for ALL, the use of peripheral blood for NGS-MRD tracking is clinically available. Studies show strong correlation between blood and marrow for NGS-MRD assessment, although marrow generally has a higher level of sensitivity. |
| I do not understand the MRD report. What should I do? | Several resources are available to help with report interpretation. It is highly advised to reach out to an expert if there are questions, particularly if clinical intervention is under consideration. Adaptive offers services for their assay to answer these types of questions. |
FFPE, formalin-fixed paraffin-embedded.
In the case described, sequence A, which was an IgH-dominant clonal sequence with an LOD of <1 clonal cell per million nucleated cells, was not detected; however, sequence B, an Igκ sequence with an LOD of 47 per million nucleated cells, was detectable at <1 clonal cell per million nucleated cells. In this situation, although it may initially appear concerning that MRD is “detectable,” lack of detection of the more unique IgH clonal sequence is reassuring. In this respect, in an analysis of adult patients undergoing HCT, it was shown that MRD detection of less sensitive Igκ and Igλ clonal sequences in the absence of detectable IgH sequences was not associated with clinical relapse.6 Emergence of new dominant clonal sequences (sequences that meet the criteria for dominant sequence mentioned above and shown in Figure 1) occurs occasionally, most commonly after HCT. These sequences should also be interpreted in the context of the patient’s clinical course; because they have not been strongly associated with relapse in studies to date, they likely represent a clonal expansion not associated with disease, and most tend to recede into the background repertoire with time (Figure 2). Thus, when using the clonoSEQ assay in clinical practice, it is important to incorporate the totality of clinical data: the dominant sequence loci and sequence LOD, the tracking behavior of sequences over time, as well as sequence abundance in the sample to better inform MRD results. For complex cases, consultation is available from Adaptive Biotechnologies, as well as from experts in the field.
After HCT, monitoring of NGS-MRD provides reassurance in patients with ongoing undetectable MRD and conversely sounds the early alarm to consider intervention in patients with emergence of detectable MRD. The ideal frequency and duration of monitoring in patients with undetectable post-HCT MRD is debatable; however, we recommend post-HCT assessment for ∼2 years in Ph– B-ALL and for up to 5 years in Ph+ B-ALL (Figure 3). Late relapses are reported more frequently in Ph+ B-ALL after tyrosine kinase inhibitor discontinuation, necessitating longer MRD follow-up.45 Reassuringly, patients entering HCT with undetectable NGS-MRD who remain NGS-MRD negative throughout the first year after HCT experience excellent outcomes with very low rates of subsequent relapse.3,6
The use of peripheral blood for NGS-MRD monitoring as a substitute for BM aspirations has been supported by recent literature (Table 1).5,9,38 In a prospective, observational study of adults with ALL undergoing HCT or CAR T-cell therapy (n = 62), paired blood and BM samples demonstrated excellent concordance of NGS-MRD, with discordant results noted in a small minority of paired samples: among 126 total paired samples analyzed, 14 pairs were discordant (7 pairs MRD-positive in PB/MRD-negative in BM; 7 pairs MRD-negative in PB/MRD-positive in BM).9 Other studies have shown that NGS-MRD testing using BM is ∼1 to 2 logs more sensitive than in the blood.5,38 Even so, NGS-MRD assessment of the blood is at least as sensitive (if not more) than MFC of the BM and represents a patient-centric approach to MRD monitoring by reducing the number of BM procedures. Some experts have proceeded with substituting peripheral blood for BM aspirate as a source for NGS-MRD monitoring in the post-HCT setting, particularly after the first year after HCT, although it is acknowledged that routine use of blood in this setting should be further examined through prospective study. Cost-analysis studies of peripheral blood NGS-MRD monitoring vs standard BM testing could help determine the cost effectiveness of this strategy and support the optimal frequency of surveillance.
NGS-MRD in ALL subgroups of interest: Ph+ ALL and T-ALL
Ph+ ALL
The International Consensus Classification recently updated the definition of BCR::ABL1 Ph+ B-ALL to include 2 subtypes: BCR::ABL1+ B-ALL-L (B-ALL with lymphoid-only involvement), which is analogous to B-ALL; and BCR::ABL1+ B-ALL-M (B-ALL with multilineage involvement), which is more akin to chronic myeloid leukemia in lymphoid blast phase.46 Multiple studies have now demonstrated discordance between NGS-MRD and quantitative PCR for BCR::ABL1 in patients with Ph+ B-ALL.8,47 Persistent detection of BCR::ABL1 by PCR in the absence of detectable MRD by NGS suggests BCR::ABL1+ B-ALL-M subtype and has not been shown to associate with inferior outcomes.8,48 In contrast, NGS-MRD appears prognostic in Ph+ B-ALL regardless of BCR::ABL1 PCR results (Table 1). Understanding this distinction is critical in pre- and post-HCT decision-making, because clinicians can be tempted to intervene when patients have persistent BCR::ABL1 PCR positivity. We recommend confirmation of positive BCR::ABL1 PCR positivity findings pre- and post-HCT with NGS or PCR-based TCR/IgH MRD measurements before intervention is considered.
T-ALL and B-ALL without IgH sequences
ClonoSEQ is available for use in T-ALL and B-ALL without IgH, Igκ, or Igλ sequences as a Clinical Laboratory Improvement Amendments–validated laboratory developed test service but has not been cleared by the FDA for this indication. For these indications, TCRβ and TCRγ loci are examined for dominant sequence(s). Currently, LOD is not provided for TCR-dominant sequences. Given that TCR sequences may be less unique (particularly TCRγ), caution in interpreting low-level detectable TCR clonal sequences in MRD reports is necessary. One study of NGS-MRD that included patients with T-ALL (n = 14) suggested better sensitivity at relapse prediction when loci at >1 TCRβ and TCRγ sequence was detected after HCT.49 More studies of TCRβ and TCRγ-based NGS-MRD monitoring pre- and post-HCT are needed to better understand the predictive power of this approach.
Conclusion
NGS of BCR and TCR loci to uncover deep MRD has had a significant impact on management of patients with ALL. In the context of allogeneic HCT, NGS-MRD pre- and post-HCT provide actionable prognostic information that can be used to guide clinical decisions. Considerable investigation is underway to identify patients who benefit the most from this technology and clarify its optimal use in a variety of settings. Although this article is focused on NGS-MRD in the context of HCT, early emergence of NGS-MRD in patients with ALL receiving non-HCT therapies is also of great importance to treating physicians. Consensus definitions in pediatrics have lowered the threshold for defining relapse to 1% disease detected by multiple modalities,50 but whether patients with very low levels of disease (10–5-10–7) should be treated early and whether more intensive therapies such as HCT could be avoided by using novel therapies is a key topic for future studies. Similar consensus definitions have not yet been applied in adult ALL. Once patients meet established definitions for transplant eligibility, the recommendations laid out in this article apply.
As with all new technology, it is important that clinicians firmly understand the results, which requires familiarity. For additional assistance with interpreting clonoSEQ results, we strongly recommend outreach to an HCT provider with expertise in this area or personnel at Adaptive Biotechnologies. It is also important to recognize a key limitation of NGS-MRD; namely, that it provides no information on leukemia immunophenotype and thus may not offer guidance when selecting immune-targeted therapy. Further investigation is also required to determine its usefulness in detecting and monitoring extramedullary disease. Finally, NGS-MRD is currently not generally available and/or affordable throughout the world outside of the United States (although recent reimbursement and availability has been achieved in Australia). Efforts should be made to broaden access to this transformative technology.
Acknowledgments
The authors thank Lanny Kirsh from Adaptive Biotechnologies, who read the manuscript to comment upon the technical accuracy of statements related to the clonoSEQ assay.
M.A.P. was supported by the NIH, National Cancer Institute (R01 CA181050 and P30CA040214), National Heart, Lung, and Blood Institute (NHLBI) (UG1HL069254), and the St. Baldrick’s Johnny Chrisstopher Fund. E.C.L. was supported by the NHLBI (5T32HL007093).
The content of this publication does not necessarily reflect the views or opinion of Adaptive Biotechnologies. Adaptive had no role in the final approval or submission of the manuscript.
Authorship
Contribution: All authors formulated the idea, planned and designed approach, wrote and edited the manuscript, and read and approved the final work.
Conflict-of-interest disclosure: L.M. reports advisory board fees from Kite, Autolus, Vor, and Cargo; research support from Jasper, Kite, Adaptive Biotechnologies, Bristol Myers Squibb, Astellas, Wugen, and Vor; honoraria from Kite; and consultancy from Astellas. M.A.P. reports advisory board fees from Pfizer, Cargo, Novartis, GentiBio, Bluebird, and Vertex; steering committee membership for Novartis and Autolus; and research support from Miltenyi and Adaptive Biotechnologies. The remaining authors declare no competing financial interests.
Correspondence: Michael A. Pulsipher, Pediatric Hematology and Oncology, Intermountain Primary Children’s Hospital, Huntsman Cancer Institute, Spencer Fox Eccles School of Medicine at the University of Utah, 2000 Circle of Hope Dr, Rm 3515, Salt Lake City, UT 84112; email: michael.pulsipher@hci.utah.edu.
References
Author notes
L.M., E.C.L., and J.G.D. contributed equally to this study.


This feature is available to Subscribers Only
Sign In or Create an Account Close Modal