Visual Abstract
Experts from the European Leukemia Net (ELN) working group for adult acute lymphoblastic leukemia have identified an unmet need for guidance regarding management of adult acute lymphoblastic leukemia (ALL) from diagnosis to aftercare. The group has previously summarized their recommendations regarding diagnostic approaches, prognostic factors, and assessment of ALL. The current recommendation summarizes clinical management. It covers treatment approaches, including the use of new immunotherapies, application of minimal residual disease for treatment decisions, management of specific subgroups, and challenging treatment situations as well as late effects and supportive care. The recommendation provides guidance for physicians caring for adult patients with ALL which has to be complemented by regional expertise preferably provided by national academic study groups.
Introduction
The European Working Group for Adult is part of the European Leukemia Net (ELN) and was founded by representatives of national academic multicenter study groups for adult acute lymphoblastic leukemia (ALL) in Europe. The group has identified an unmet need for guidance regarding management of adult ALL from diagnosis to aftercare. A previous publication covered diagnostic approaches, prognostic factors and assessment of adult ALL.1 With increasing complexity of therapeutic options there is also a need for guidance regarding clinical management. Standard therapy with pediatric-based regimens is successful but requires optimization. Also, treatment decisions based on minimal residual disease (MRD) require standards. Immunotherapy is integrated to an increasing extent into first-line therapies, whereas management of relapse is still a considerable challenge. Treatment approaches must be adapted to specific subgroups, such as older patients or Ph/BCR::ABL1-positive ALL and practical management is challenging in specific situations, such as secondary leukemia. Finally, the increasing number of long-term survivors highlights the need for optimized aftercare and surveillance for late effects. Therefore, the group decided to develop an ELN recommendation for management of ALL as published for other entities.2,3
Methods
The panel includes 17 members representing national study groups. Members met in person and defined topics, tables, and responsibilities of coauthors (supplemental Table 1, available on the Blood website). Coauthors performed literature searches of PubMed database and considered relevant abstracts. The manuscript was reviewed by all coauthors. Formal corrections were performed by the corresponding author. Disagreements were summarized and discussed in the whole group. The group agreed on the final version of the manuscript. Owing to rapid innovation and availability of new data, together with a lack of randomized trials for many essential questions, most of the statements have an evidence level of “expert recommendation” for clinical practice.
Induction and consolidation therapy in Ph-negative ALL
General principles
The aim of intensive induction therapy is to obtain a complete remission (CR) in as many patients and as early, safely, and deep as possible. CR rate in adults aged between 15 to18 years and 55 to 65 years with Ph/BCR::ABL1-negative (Ph-) ALL is ∼90%.4 Approximately 5% display primary resistance after 2 cycles, and ∼5% die early of disease- or therapy-related complications. Treatment is usually risk-adapted using prognostic factors (PF) and course of MRD for treatment decisions regarding intensity of chemotherapy, use of immunotherapies, or indication for stem cell transplantation (SCT). A comprehensive diagnostic characterization is the basis for optimal management as described previously.1
Pediatric-based chemotherapy in adult ALL
In a meta-analysis 25 out of 27 reports clearly favored the pediatric-based approach as standard of care (SoC).5 In patients up to 45 to 55 years overall survival (OS) improved to an average of 60% (supplemental Table 2). Comparison of pediatric-based regimens vs standard Hyper-CVAD led to similar conclusions in a monocentric trial.5 Pediatric-based therapy was particularly effective in standard risk ALL6-9 and patients with negative MRD6,10-12 ensuring OS of ∼70%.13-16 The typical compounds are corticosteroids (especially dexamethasone [Dexa]), vincristine, antimetabolites (6-thiopurines, cytarabine, and methotrexate) and asparaginase (ASP; pegylated or not) plus intensive supportive care, avoidance of inappropriate dose reductions/delays and a risk-adapted SCT.
Induction
For prephase, corticosteroids are usually administered for 5 to 7 days; other drugs are occasionally added, for example, cyclophosphamide (CP) and intrathecal (IT) prophylaxis after sampling of cerebrospinal fluid (CSF). First induction lasts ∼4 weeks and carries the highest risk of complications, mandating for intensive support, including granulocyte colony stimulating factor (G-CSF), transfusions and optimal prophylaxis and management of infections. The induction backbone consists of vincristine, steroids, an anthracycline, and ASP. Dexa is highly active, including activity in the central nervous system (CNS), but requires careful adaption of dose and schedule. If Dexa is administed over a prolonged period, an increasd risk of severe infections has been observed. Therefore, shorter term application with interruptions has been implemented by several groups during induction therapy. Early high-dose (HD) anthracyclines worsen myelotoxicity and mucositis.17 A randomized trial failed to demonstrate an advantage from increasing the CP dose upfront, however fractionated doses were of benefit to patients aged >55 years receiving less intensive consolidation.13 Pegylated asparaginase (PEG-ASP) provides a longer asparagine depletion (serum enzymatic activity ≥0.1 IU/mL detectable for 14 to 30 days depending on dose) compared with the native compound.5,14,15,18,19 Compared with native ASP, the drug causes rarely severe allergic reactions in adults. ASP in general can cause coagulopathy, thrombosis, hyperglycemia, pancreatitis, and liver toxicity being more frequent and of greater concern in adults than children.20,21 It is crucial to establish a specific schedule for toxicity monitoring and management20,21; PEG-ASP schedule and dosing should be adapted to age, body mass index (higher toxicity if body mass index is >30 kg/m2) and hepatosteatosis (higher toxicity if positive ultrasound scan)14,15 and to the intended duration of activity. Any other potentially hepatotoxic drugs should not be administered during expected ASP activity duration, with the exception of strict clinical indication.18,22 More recently premedication before use of PEG-ASP has been discussed to reduce the risk of infusion reactions which are sometimes difficult to differentiate from real allergic reactions. Any premedication should only be administered if drug monitoring can be offered. Otherwise, there is the risk to overlook inactivation of ASP.
The second induction or first consolidation consists usually of CP, cytarabine (AC) and mercaptopurine (MP) or HD-methotrexate (MTX)/HD-AC.23 HD-AC/idarubicin can be used as salvage.13 The few patients failing to enter CR after 2 induction courses have highly resistant ALL and are candidates to alternative immunotherapies depending on the protocol.
Consolidation
Consolidation is administered to patients in CR. Outcome is best with rotational multidrug cycles consisting of HD-MTX and HD-AC (also useful as CNS-penetrating agent), PEG-ASP and other drugs (supplemental Table 2). BFM (Berlin-Frankfurt-Munster group)–based regimens include a delayed reinduction phase. The average duration of consolidation is ≥6 months, for 6 to 8 total courses. HD-AC, HD-MTX, etoposide, and CP were essential to improve outcome of high-risk subsets and T-ALL. HD-AC (4-8 doses at 1-3 g/m2), PEG-ASP and HD-MTX (1-1.5 g/m2, followed by folinic acid rescue) are administered in blocks. Higher MTX dosages of 3 to 5 g/m2 are used especially for patients whio are at high risk and T-ALL. A randomized trial demonstrated an improved outcome for patients receiving consolidation MTX at 3 vs 0.5 g/m2, however the lower dose is unusual.12 A phase 2 trial in adolescents and young adults (AYA) used a lower MTX dose with weekly dose adaptions (Capizzi style).6 Capizzi MTX showed in pediatric trials (up to age 30) outcome superior to MTX 5 g/m2 in T-ALL but not in B-ALL.24,25
Given the heterogeneous consolidation protocols, the comparable results after adjustment for patient age and risk class, and the lack of randomized comparisons, at present no single regimen can be recommended as SoC for Ph-negative ALL. Pediatric-based regimens are favored. It is strongly recommended to participate in (or adopt) prospective clinical trials. Experience and established guidelines at the sites are important for adoption of distinct protocols.
Frontline targeted therapy of Ph-negative B-LIN ALL
Because of the many opportunities offered by immunotherapies,26 modified regimens with targeting agents are tested to increase response, OS, and if possible, to reduce the high toxicity burden because of intensive chemotherapy and SCT. The identification of surface markers as target for immunotherapies is therefore essential part of initial diagnosis.1 The compounds and mechanisms of action are discussed in “Relapsed/refractory ALL.”
Evidence is accumulating rapidly that immunotherapy can improve antileukemic efficacy (Table 1). Anti-CD20 antibodies, the bispecific CD19/CD3 antibody blinatumomab (Blina) and the CD22 antibody-drug conjugate inotuzumab ozogamicin (InO) have been integrated into frontline and salvage regimens in clinical trials.
Trials with upfront targeted or subset-specific therapy in adult Ph− ALL
| Study (reference) . | ALL subset . | Combination treatment . | Age (y) . | No. . | CR (%) . | CRD/ RFS (%) . | OS (%) . | EFS (%) . | FUP . | Annotations . |
|---|---|---|---|---|---|---|---|---|---|---|
| Rituximab/ofatumumab (anti-CD20 antibody) | ||||||||||
| MDACC27 | CD20+ B-ALL | Hyper-CVAD + R ×8 in cycles 1-4, ×4 in maintenance cycles 6, 18 | 68 | 100 | 70 | 75 | — | 3 y | Better outcome vs no R patients (P < .001 for CRD, P = .003 for OS); outcome not improved by R in patients aged 60+ | |
| GMALL28 | CD20+ B-ALL | GMALL 07/2003 + R ×8 (SR), ×3 (HR) in induction/consolidation | 15-55 | 117 | 94 | 64 | 75 | — | 3 y | Better outcome vs no R patients (P < .009 for CRD), with better/faster MRD clearing (MRD < 0.01% 60% at day 21) |
| GRAALL-200329 | CD20+ B-ALL | A: SOC B: SOC + R ×16-18 during induction/consolidation | 40 (24-53) | 105 104 | 94 92 | — | 50 61 | 43 55 | 4 y | Better outcome vs no R patients (P < .04 for EFS, P =. 02 for relapse incidence) |
| UKALL 1430 | CD20+/− B-ALL (29% Ph+) | A: SOC B: SOC + R ×4 in induction | 25-65 | 288 289 | 92 94 | — | — | 42 48 | 3 y | No overall benefit from R (P = .28), better outcome with R after MAC SCT (EFS 72% vs 50%, P = .03) |
| MDACC31 | CD20+/− B-ALL | Hyper-CVAD + ofatumumab ×8 in induction/consolidation | 41 (18-71) | 46 CD20+ 23 CD20− | 93 | — | 66 70 | 61 65 | 4 y | CD20+: EFS and OS with R 43% (P = .119) and 48% (P = .123); CD20-: EFS and OS with R 50% (P = .89) and 62% (P = .61) |
| Blinatumomab | ||||||||||
| GIMEMA LAL 231732 | CD19+ B-ALL | SOC + blinatumomab ×2 in consolidation | 18-65 | 146 | 90 | 56-87 | 65-91 | 48-85 | 1.5 y | 95% MRD negative after blinatumomab I (P = .001); OS by age (P = .0009) and DFS/EFS by Ph-like (P = .006) and MRD (P = .0002) |
| GRAALL-2014- QUEST33 | CD19+ B-ALL (high risk) | SOC + blinatumomab ×5 in consolidation/ maintenance | 35 (18-60) | 95 | NR | 69-90 | 92 | — | 1.5 y | 74% MRD negative after blinatumomab; DFS by high or very high risk class (P = .018) |
| MDACC34 | B-ALL | Hyper-CVAD + blinatumomomab ×2 after induction and/or x2-4 after consolidation ± R/ofatumumab (CD20+) ± InO ×4 after consolidation | 34 (17-59) | 58 (20 with InO) | 100 | 84 | 85 | — | 3 y | 76% and 95% MRD negative after cycle 1 and overall, resp.; no relapse/death in InO-treated group (OS 100%) |
| MDACC35 | B-ALL | Hyper-CVAD 4 cycles (50% dose reduction) + blinatumomab ×4 after induction and ×3 after consolidation + R/ofatumumab (CD20+) in induction/consolidation | 37 (29-45) | 38 | 100 | 73 | 81 | — | 3 y | Amended to Hyper-CVAD 2 cycles (75% dose reduction) in HR patients; overall MRD negativity 97%; OS 88% no HR feature vs 76% any HR feature |
| HOVON36 | CD19+ B-ALL (37% Ph+) | SOC + blinatumomab ×2 (prephase and after consolidation I) | 53 (18-70) | 71 | 77 | — | 53 (Ph−) | 68 (Ph−) | 2 y | 53% and 91% MRD negative after blinatumomab I and II, resp.; EFS and OS 71% and 73% in patients ≤60 years |
| GMALL0837 | CD19+ B-ALL (MRD positive) | Blinatumomab 1 cycle in MRD positive patients after consolidation I | 18-35 | 63 | all CR/MRD+ | — | — | 71 | 3 y | 55% molecular CR after blinatumomab I; subsequent SCT indicated in all patients |
| ECOG-ACRIN (phase 3)38 | CD19+ B-ALL (MRD negative) | A: SOC consolidation B: SOC consolidation + blinatumomab x4 | 51 (30-70) | 112 112 | 81 | — | NR 71.4 | — | 5 y | CR rate on all study patients (n = 488); median OS arm B not reached (NR, >70% at 5 years) vs 71.4 months (P = .003) |
| Nelarabine (T-specific nucleoside analog) | ||||||||||
| MDACC39 | T-ALL/LBL | Hyper-CVAD + nelarabine after course 8 (×2) or 5 and 7 (×1 each) | 30 (13-78) | 81 | NR | — | 57 | 52 | 5 y | OS non-ETP 63% vs ETP 32% (P < .001); non-ETP: OS nelarabine 83% vs no nelarabine 38% (P = .003) |
| UKALL 14 (Phase 3)40 | T-ALL | A: SOC B: SOC + nelarabine ×1 after induction II | 25-65 | 75 69 | 90 87 | — | 61 65 | 57 61 | 3 y | No benefit from single nelarabine course (3 doses), all P values not significant |
| Study (reference) . | ALL subset . | Combination treatment . | Age (y) . | No. . | CR (%) . | CRD/ RFS (%) . | OS (%) . | EFS (%) . | FUP . | Annotations . |
|---|---|---|---|---|---|---|---|---|---|---|
| Rituximab/ofatumumab (anti-CD20 antibody) | ||||||||||
| MDACC27 | CD20+ B-ALL | Hyper-CVAD + R ×8 in cycles 1-4, ×4 in maintenance cycles 6, 18 | 68 | 100 | 70 | 75 | — | 3 y | Better outcome vs no R patients (P < .001 for CRD, P = .003 for OS); outcome not improved by R in patients aged 60+ | |
| GMALL28 | CD20+ B-ALL | GMALL 07/2003 + R ×8 (SR), ×3 (HR) in induction/consolidation | 15-55 | 117 | 94 | 64 | 75 | — | 3 y | Better outcome vs no R patients (P < .009 for CRD), with better/faster MRD clearing (MRD < 0.01% 60% at day 21) |
| GRAALL-200329 | CD20+ B-ALL | A: SOC B: SOC + R ×16-18 during induction/consolidation | 40 (24-53) | 105 104 | 94 92 | — | 50 61 | 43 55 | 4 y | Better outcome vs no R patients (P < .04 for EFS, P =. 02 for relapse incidence) |
| UKALL 1430 | CD20+/− B-ALL (29% Ph+) | A: SOC B: SOC + R ×4 in induction | 25-65 | 288 289 | 92 94 | — | — | 42 48 | 3 y | No overall benefit from R (P = .28), better outcome with R after MAC SCT (EFS 72% vs 50%, P = .03) |
| MDACC31 | CD20+/− B-ALL | Hyper-CVAD + ofatumumab ×8 in induction/consolidation | 41 (18-71) | 46 CD20+ 23 CD20− | 93 | — | 66 70 | 61 65 | 4 y | CD20+: EFS and OS with R 43% (P = .119) and 48% (P = .123); CD20-: EFS and OS with R 50% (P = .89) and 62% (P = .61) |
| Blinatumomab | ||||||||||
| GIMEMA LAL 231732 | CD19+ B-ALL | SOC + blinatumomab ×2 in consolidation | 18-65 | 146 | 90 | 56-87 | 65-91 | 48-85 | 1.5 y | 95% MRD negative after blinatumomab I (P = .001); OS by age (P = .0009) and DFS/EFS by Ph-like (P = .006) and MRD (P = .0002) |
| GRAALL-2014- QUEST33 | CD19+ B-ALL (high risk) | SOC + blinatumomab ×5 in consolidation/ maintenance | 35 (18-60) | 95 | NR | 69-90 | 92 | — | 1.5 y | 74% MRD negative after blinatumomab; DFS by high or very high risk class (P = .018) |
| MDACC34 | B-ALL | Hyper-CVAD + blinatumomomab ×2 after induction and/or x2-4 after consolidation ± R/ofatumumab (CD20+) ± InO ×4 after consolidation | 34 (17-59) | 58 (20 with InO) | 100 | 84 | 85 | — | 3 y | 76% and 95% MRD negative after cycle 1 and overall, resp.; no relapse/death in InO-treated group (OS 100%) |
| MDACC35 | B-ALL | Hyper-CVAD 4 cycles (50% dose reduction) + blinatumomab ×4 after induction and ×3 after consolidation + R/ofatumumab (CD20+) in induction/consolidation | 37 (29-45) | 38 | 100 | 73 | 81 | — | 3 y | Amended to Hyper-CVAD 2 cycles (75% dose reduction) in HR patients; overall MRD negativity 97%; OS 88% no HR feature vs 76% any HR feature |
| HOVON36 | CD19+ B-ALL (37% Ph+) | SOC + blinatumomab ×2 (prephase and after consolidation I) | 53 (18-70) | 71 | 77 | — | 53 (Ph−) | 68 (Ph−) | 2 y | 53% and 91% MRD negative after blinatumomab I and II, resp.; EFS and OS 71% and 73% in patients ≤60 years |
| GMALL0837 | CD19+ B-ALL (MRD positive) | Blinatumomab 1 cycle in MRD positive patients after consolidation I | 18-35 | 63 | all CR/MRD+ | — | — | 71 | 3 y | 55% molecular CR after blinatumomab I; subsequent SCT indicated in all patients |
| ECOG-ACRIN (phase 3)38 | CD19+ B-ALL (MRD negative) | A: SOC consolidation B: SOC consolidation + blinatumomab x4 | 51 (30-70) | 112 112 | 81 | — | NR 71.4 | — | 5 y | CR rate on all study patients (n = 488); median OS arm B not reached (NR, >70% at 5 years) vs 71.4 months (P = .003) |
| Nelarabine (T-specific nucleoside analog) | ||||||||||
| MDACC39 | T-ALL/LBL | Hyper-CVAD + nelarabine after course 8 (×2) or 5 and 7 (×1 each) | 30 (13-78) | 81 | NR | — | 57 | 52 | 5 y | OS non-ETP 63% vs ETP 32% (P < .001); non-ETP: OS nelarabine 83% vs no nelarabine 38% (P = .003) |
| UKALL 14 (Phase 3)40 | T-ALL | A: SOC B: SOC + nelarabine ×1 after induction II | 25-65 | 75 69 | 90 87 | — | 61 65 | 57 61 | 3 y | No benefit from single nelarabine course (3 doses), all P values not significant |
A, B: study arms in randomized trials; CR, complete remission; CRD/RFS, CR duration/relapse-free survival; FUP, follow-up; GIMEMA, Gruppo Italiano Malattie Ematologiche dell’Adulto; GRAALL, Group for Research on Adult ALL; GMALL, German Multicenter Group for Adult ALL; HOVON, Hemato-Oncology Foundation for Adults in The Netherlands; InO, inotuzumab ozogamicin; LBL, lymphoblastic lymphoma; MAC SCT, myeloablative conditioned SCT; MDACC, MD Anderson Cancer Center; NR, not reported; R, rituximab; SOC, standard of care (chemotherapy).
Approximately 40% of adult B-lineage (B-LIN) ALL express the CD20 antigen in >10% to 20% ALL blasts. Anti-CD20 antibodies were succesfully tested in de novo ALL.27,29 In a randomized study, rituximab (Ritux) significantly decreased the relapse risk (RR), contributing to increase in the rate of SCT realization and improved OS.29 Interestingly, patients treated with Ritux developed fewer allergic reactions to ASP; however, early MRD response was not improved. A randomized trial administering Ritux irrespective of CD20 expression failed to demonstrate a benefit30; however, in this study only 4 doses were prescribed (vs at least 8 doses in other trials).
Preliminary data with Blina, partly in association with InO are encouraging and confirm high rates of conversion to MRD negativity, with low RR and an improved OS at limited follow-up between 1 and 4 years.32,33,35,37 A randomized trial with Blina consolidation added to a standard backbone for patients with MRD negative ALL aged between 30 and 70 years indicates an advantage for patients treated with Blina in terms of OS according to a preliminary analysis.38 Thus, there is increasing evidence that Blina likely benefits in consolidation of patients with negative and positive MRD.
Overall, the use of Ritux is recommended for the management of CD20+ ALL, for at least 8 doses. Similarly, because of the favorable early results and high expectations with upfront InO and Blina in CD22+ and CD19+ B-ALL, participation into InO and Blina trials for untreated patients is recommended until results from ongoing studies are available. The concurrent use of an anti-CD20 antibody in CD20+ ALL represents SoC as well as the use of Blina in case of molecular failure.
Maintenance therapy
Maintenance therapy is strongly recommended in all patients. Maintenance therapy was not tested in randomized trials but any attempts to omit maintenance have resulted in inferior outcomes.41,42 Insufficient maintenance therapy significantly worsens OS.7,41 Long-term drug exposure is probably needed to eradicate MRD.42
MP and MTX are the main drugs in maintenance. Intermittent IT prophylaxis is part of most regimens. Some groups include other compounds, such as the POMP regimen (MP, MTX, prednisone, and vincristine),7,13,41 whereas the benefit is debated. In addition, data on a potential benefit of vincristine or steroid pulses in pediatric patients with ΔIKZF1 are controversial.43,44 The use of Dexa instead of prednisone in maintenance may lead to an increased incidence of infection-related deaths.45 Therefore, maintenance without vincristine or steroid pulses is preferred by most groups. A total treatment duration of 2 to 2.5 years including maintenance is recommended. Intervals of ∼3 months are suggested for MRD testing during maintenance.
CNS-directed prophylaxis
CNS infiltration occurs in 5% to 10% of adults at diagnosis. It can be classified as none (CNS-1), <5 white cells/μL (CNS-2), >5 white cells/μL or a cranial nerve palsy (CNS-3) but pragmatically, one should be suspicious if any blast cells are seen in the CSF.46 It must be routinely ruled out and subsequently monitored by analysis of cytospin preparations and/or multicolor flow-cytometry (MFC) analysis of CSF obtained at or soon after the time of diagnosis. Some protocols recommend waiting until peripheral blood (PB) blasts have been cleared or are reduced below a certain cutoff to prevent the risk of “seeding” the CNS from a traumatic procedure. Postponing of lumbar puncture bears on the other hand the risk to not detect initial CNS involvement which may be even more frequent in patients with high white blood cell count (WBC). Factors predicting a higher risk of initial CNS disease are T-ALL, high presenting WBC, and high-risk cytogenetics, such as Ph+ ALL and t(4;11).1,47 MRD detection in CSF has been attempted but is not part of standard management.
All patients require CNS prophylaxis. Cranial irradiation, IT chemotherapy, and components of systemic therapy which cross the blood-brain barrier have all been used. Currently, cranial irradiation is rarely administered as prophylaxis. There are insufficient data to clearly dissect the relative values of CNS penetrating systemic therapy vs IT prophylaxis; therefore, both are recommended although the critical component is most likely the IT prophylaxis.
IT chemotherapy
MTX, ARAC, and steroids can all be safely administered IT, and all have been used, either singly or in combination with no clear benefits to any specific IT regimen. IT medications should be administered in sufficient volume to distribute well throughout the CSF. An equivalent volume of CSF to the volume of drug is usually removed before administration and the CSF should always be sent for analysis. The patient should remain recumbent for at least 1 hour.48 It is a critical that conventional “cutting” needles carry a high risk of hygroma49 and subdural hemorrhage; therefore atraumatic needles are recommended.50 The ideal number of IT injections is undetermined, but most protocols recommend 8 to 15 total doses. Close attention must be paid to the supportive care, such as blood product support. In rare instances when lumbar punctures are difficult to perform an Ommaya reservoir can be considered as an individual alternative taking procedure related risks and uncertainties regarding adequate dosing into account. Practical aspects of IT treatment are reviewed by Olmos-Jimenez et al.51
Systemic chemotherapy
Systemic chemotherapy may also help prevent CNS relapse. A meta-analysis of 43 trials showed a benefit to adding HD-MTX in terms of reduction in RR and improved event-free survival (EFS), although only a small effect on CNS disease.52 A recent randomized trial showed a benefit in terms of disease-free survival (DFS) (but no impact on CNS relapse) for 3 g/m2 over 0.5 g/m2 with no excess toxicity for the higher dose.53 Dexa may be superior to prednisolone in reducing CNS RR.54
MRD-based treatment modification
Given the paramount prognostic impact of MRD response, most groups recommend therapy changes based on MRD. As described previously MRD based treatment decisions require adequate and standardized methodology for MRD detection.1 Important decision factors are time point and level of MRD, method, presence of other PFs, and suggested treatment modifications.37 Early good MRD response is associated with favorable outcome. In one series 12% of patients at standard risk showed no detectable MRD at day 11 of induction and had an excellent prognosis.55 A later time point after first consolidation was identified as most prognostically relevant for RR.56 Thus, earlier time points are suitable to identify patients for potential deescalation of therapy in clinical trials, whereas later time points are helpful to recognize candidates for treatment escalation. The most relevant time point depends on protocol. If it is the primary goal to identify patients with chemotherapy resistance, all relevant chemotherapy compounds should have been administered before changes are made keeping in mind that MRD testing does not represent the extramedullary compartment. In most protocols, the relevant time point will be ∼2 to 3 months from diagnosis.
It is also important to identify the most predictive level of MRD. A threshold of 0.01% (ie, 10−4) is often considered because this is in line with the sensitivity of MRD assays. Each log-level increase of MRD is associated with shorter time to subsequent hematologic relapse.57 The median time to hematologic relapse was 7.6 months vs 4.9 months for patients with MRD >0.01% vs 0.1%, respectively.56 Patients with low-level MRD that is <0.01% may have an intermediate prognosis58,59 and should be considered for MRD-based treatment interventions in the future.
In addition, the method of MRD detection influences treatment decisions. Thus, quantitative polymerase chain reaction (PCR) measurement of clonal immunoglobulin/T-cell receptor rearrangements (IG/TR) requires 4 to 6 weeks to set-up the appropriate assay. MFC may deliver results more quickly but may yield uncertain results particularly in regenerating bone marrow (BM). Decisions based on the safe confirmation of negative MRD status require strict adherence to methodological prerequisites.1 This requires MRD assessment in an experienced reference laboratory and a sensitivity of at least 0.01% for the time point which is considered for the treatment decision.
Most trials on the prognostic impact of MRD were conducted in newly diagnosed patients. In adult ALL limited data demonstrate a correlation between persistent MRD after therapy with new targeted compounds in the relapsed/refractory (R/R) setting60 but correlation is less stringent than that in first-line therapy. Nevertheless, MRD response to salvage therapy indicates the antileukemic efficacy of a defined approach and is relevant for patients.
Very few attempts for reduction of standard therapy have been made so far. One randomized trial in pediatric patients who are at standard risk with good MRD response evaluated the outcome with a reduced reinduction regimen. This modification was associated with an inferior DFS, particularly in children aged >10 years.61 Further clinical trials are warranted to evaluate treatment reduction particulary in the era of immunotherapies.
Most groups build their indication for SCT on MRD. SCT provides a survival advantage for patients with poor MRD response.56,62 However, the realization of SCT requires time and many patients relapse during this period despite continued chemotherapy.56 In addition, a high MRD level before SCT is associated with a higher RR after SCT.63-66 The prognostic impact of MRD before SCT is correlated to the time-interval between MRD detection and transplant, the potential interim therapies, the conditioning regimens, and other transplant related factors.
It is recommended to change therapy in patients with persistent or recurrent MRD. Compounds with different mechanisms of action compared with chemotherapy are potentially most promising. To date, Blina is the only compound tested in a pivotal trial for MRD positive B-LIN ALL. Patients with an MRD >0.1% were included either in first or later remission. The primary end point, achievement of a complete molecular remission (CMR) after 1 cycle, was achieved in 78%. Furthermore, a favorable median survival of 36.5 months was obtained and patients achieving a CMR had a significant benefit. This treatment was a bridge to SCT in 67% of the patients. The RR after SCT was low. However, OS was impaired by a treatment-related mortality (TRM) >30% which is explained by the high median age (42 years), the high rate of full conditioning and mismatch donors.67,68 On the other hand, most patients without subsequent SCT relapsed and beyond complete molecular remission no factors predicting RR in patients without SCT were identified. In the future immunotherapies or other targeted drugs should be evaluated in the setting of MRD persistence or recurrence, not hematologic relapse only.
Overall, patients with MRD >0.01% after 3 blocks of standard therapy have an indication for SCT and for targeted therapies. To date, there is no evidence that treatment reduction, except for omission of SCT, can be recommended outside of clinical trials in patients with favorable course of MRD. Whether SCT can be omitted in all patients with molecular CR, including those in specifically unfavourable subgroups should be investigated.69 After conversion from MRD positive to MRD negative status using new compounds such as Blina, subsequent SCT remains the standard in younger patients with matched donor. In older patients with high risk of TRM either dose-reduced conditioning regimens or a consolidation or maintenance strategy should be followed.
SCT
SCT is a complex multistep, multifactorial, and highly individualized treatment concept. As such, it is considered an effective treatment for preventing relapse, combining myeloablative doses of chemotherapy and/or radiotherapy with potential beneficial graft-versus-leukemia reaction. Unfortunately, it is also associated with significant incidence of TRM, reaching 13% after SCT from HLA-matched sibling donors (MSD) and 21% for SCT from unrelated donors (URD).70 Haploidentical SCT with posttransplant CP is increasingly implemented in many countries. Overall, indication for SCT is weighed against the reduction of the RR and the risk of TRM. Despite attempts to elaborate prognostic scores, the potential benefit in many individual cases is uncertain.71 The role of autologous (auto) SCT appears questionable and mainly affected by the MRD status.72 Results of recent SCT trials are summarized in Table 2.
Results of SCT for adults with ALL (selected studies)
| Study . | Population . | Donor . | N . | OS (%) . | LFS (%) . | RI (%) . | TRM (%) . |
|---|---|---|---|---|---|---|---|
| Nishiwaki et al, 201373 | Ph− in CR1 | MSD | 388 | 65 (4 y) | 62 (3 y) | 25 (3 y) | 13 (3 y) |
| URD | 434 | 64 (4 y) | 61 (3 y) | 17 (3 y) | 22 (3 y) | ||
| Cord blood | 95 | 57 (4 y) | 51 (3 y) | 22 (3 y) | 27 (3 y) | ||
| Ph− in CR > 1 | MSD | 89 | 47 (4 y) | 48 (3 y) | 31 (3 y) | 21 (3 y) | |
| URD | 158 | 39 (4 y) | 38 (3 y) | 26 (3 y) | 36 (3 y) | ||
| Cord blood | 53 | 48 (4 y) | 44 (3 y) | 29 (3 y) | 27 (3 y) | ||
| Giebel et al, 201670 | Ph+ and Ph−, CR1 | MSD | 252 | 71 (2 y) | 61 (2 y) | 26 (2 y) | 13 (2 y) |
| URD | 310 | 67 (2 y) | 60 (2 y) | 19 (2 y) | 21 (2 y) | ||
| Cahu et al, 201674 | T-ALL in CR1 | MSD+URD | 414 | 54 (5 y) | 51 (5 y) | — | — |
| T-ALL in CR2 | 93 | 37 (5 y) | 33 (5 y) | — | — | ||
| T-ALL in CR > 2 or active disease | 94 | 12 (5 y) | 9 (5 y) | — | — | ||
| Brissot et al, 201575 | Ph+ in CR1 | MSD | 234 | 45 (5 y) | 36 (5 y) | 43 (5 y) | 20 (5 y) |
| URD | 249 | 47 (5 y) | 40 (5 y) | 30 (5 y) | 30 (5 y) | ||
| Giebel et al, 201876 | Ph+ in molecular CR1 | MSD | 255 | 70 (2 y) | 55 (2 y) | 28 (2 y) | 18 (2 y) |
| URD | 247 | 69 (2 y) | 60 (2 y) | 19 (2 y) | 22 (2 y) | ||
| Autologous | 67 | 70 (2 y) | 52 (2 y) | 47 (2 y) | 2 (2 y) | ||
| Pavlu et al, 201777 | Ph+ and Ph−, primary induction failure∗ | Various | 86 | 23 (5 y) | 17 (5 y) | 54 (5 y) | 29 (5 y) |
| Santoro et al, 202078 | Ph+ and Ph− in CR1 | Matched URD | 809 | 62 (3 y) | 53 (3 y) | 28 (3 y) | 19 (3 y) |
| Mismatched URD | 289 | 62 (3 y) | 55 (3 y) | 25 (3 y) | 20 (3 y) | ||
| Haploidentical† | 136 | 54 (3 y) | 49 (3 y) | 28 (3 y) | 23 (3 y) | ||
| Nagler et al, 202179 | Ph+ and Ph− in CR1 or CR2 | MSD | 1891 | 67 (2 y) | 55 (2 y) | 32 (2 y) | 13 (2 y) |
| Haploidentical† | 413 | 59 (2 y) | 51 (2 y) | 26 (2 y) | 23 (2 y) | ||
| Beelen et al, 202280 | Ph−, HR, CR1, prospective trial | MSD | 176 | 59 (5 y) | 56 (5 y) | 23 (5 y) | 21 (5 y) |
| URD | 366 | 58 (5 y) | 55 (5 y) | 25(5 y) | 20 (5 y) | ||
| Marks et al, 202281 | Unfit for myeloablative conditioning, CR1 | MSD/URD | 249 | 55 (4 y) | 47 (4 y) | 34 (4 y) | 20 (4 y) |
| Study . | Population . | Donor . | N . | OS (%) . | LFS (%) . | RI (%) . | TRM (%) . |
|---|---|---|---|---|---|---|---|
| Nishiwaki et al, 201373 | Ph− in CR1 | MSD | 388 | 65 (4 y) | 62 (3 y) | 25 (3 y) | 13 (3 y) |
| URD | 434 | 64 (4 y) | 61 (3 y) | 17 (3 y) | 22 (3 y) | ||
| Cord blood | 95 | 57 (4 y) | 51 (3 y) | 22 (3 y) | 27 (3 y) | ||
| Ph− in CR > 1 | MSD | 89 | 47 (4 y) | 48 (3 y) | 31 (3 y) | 21 (3 y) | |
| URD | 158 | 39 (4 y) | 38 (3 y) | 26 (3 y) | 36 (3 y) | ||
| Cord blood | 53 | 48 (4 y) | 44 (3 y) | 29 (3 y) | 27 (3 y) | ||
| Giebel et al, 201670 | Ph+ and Ph−, CR1 | MSD | 252 | 71 (2 y) | 61 (2 y) | 26 (2 y) | 13 (2 y) |
| URD | 310 | 67 (2 y) | 60 (2 y) | 19 (2 y) | 21 (2 y) | ||
| Cahu et al, 201674 | T-ALL in CR1 | MSD+URD | 414 | 54 (5 y) | 51 (5 y) | — | — |
| T-ALL in CR2 | 93 | 37 (5 y) | 33 (5 y) | — | — | ||
| T-ALL in CR > 2 or active disease | 94 | 12 (5 y) | 9 (5 y) | — | — | ||
| Brissot et al, 201575 | Ph+ in CR1 | MSD | 234 | 45 (5 y) | 36 (5 y) | 43 (5 y) | 20 (5 y) |
| URD | 249 | 47 (5 y) | 40 (5 y) | 30 (5 y) | 30 (5 y) | ||
| Giebel et al, 201876 | Ph+ in molecular CR1 | MSD | 255 | 70 (2 y) | 55 (2 y) | 28 (2 y) | 18 (2 y) |
| URD | 247 | 69 (2 y) | 60 (2 y) | 19 (2 y) | 22 (2 y) | ||
| Autologous | 67 | 70 (2 y) | 52 (2 y) | 47 (2 y) | 2 (2 y) | ||
| Pavlu et al, 201777 | Ph+ and Ph−, primary induction failure∗ | Various | 86 | 23 (5 y) | 17 (5 y) | 54 (5 y) | 29 (5 y) |
| Santoro et al, 202078 | Ph+ and Ph− in CR1 | Matched URD | 809 | 62 (3 y) | 53 (3 y) | 28 (3 y) | 19 (3 y) |
| Mismatched URD | 289 | 62 (3 y) | 55 (3 y) | 25 (3 y) | 20 (3 y) | ||
| Haploidentical† | 136 | 54 (3 y) | 49 (3 y) | 28 (3 y) | 23 (3 y) | ||
| Nagler et al, 202179 | Ph+ and Ph− in CR1 or CR2 | MSD | 1891 | 67 (2 y) | 55 (2 y) | 32 (2 y) | 13 (2 y) |
| Haploidentical† | 413 | 59 (2 y) | 51 (2 y) | 26 (2 y) | 23 (2 y) | ||
| Beelen et al, 202280 | Ph−, HR, CR1, prospective trial | MSD | 176 | 59 (5 y) | 56 (5 y) | 23 (5 y) | 21 (5 y) |
| URD | 366 | 58 (5 y) | 55 (5 y) | 25(5 y) | 20 (5 y) | ||
| Marks et al, 202281 | Unfit for myeloablative conditioning, CR1 | MSD/URD | 249 | 55 (4 y) | 47 (4 y) | 34 (4 y) | 20 (4 y) |
MSD, matched sibling donor; RI, relapse incidence; URD, unrelated donor.
No CR after at least 2 cycles of induction.
Unmanipulated graft.
Ph-negative ALL
Most prospective studies evaluating the role of SCT have been conducted considering the availability of MSD. Patients in CR1 with MSD were offered SCT, whereas those lacking MSD were treated either with auto SCT or chemotherapy. Most of the trials demonstrated a beneficial effect of MSD-SCT (reviewed by Bassan et al4 and Hoelzer et al82). However, these studies were performed before the era of routine assessment of MRD and with non–pediatric-based comparator arms. Currently, MRD status is considered the most important PF, driving SCT indication. In 2 subsequent trials 522 Ph-negative patients who are at high risk up to the age of 55 years were intended for SCT.62 Among these, 54% received a transplant in CR1 (MSD or URD). SCT was associated with longer leukemia-free survival (LFS) in patients with postinduction MRD ≥10−3 but not in good MRD responders. This observation, however, may not necessarily apply to less intensive chemotherapy protocols and a randomized trial assessing SCT indication in MRD-negative patients who are high risk is ongoing.37
Indications for SCT refer mainly to younger adults aged up to 55-60 years and vary among study groups.1,83 There is no consensus regarding older patients. High variability in clinical practice between study groups and individual centers has been reported.83 In MRD-negative older patients, SCT should not be used outside prospective clinical trials. It is crucial to continue MRD monitoring after SCT to identify upcoming relapses.
Ph-positive ALL
SCT was associated with significant OS benefit in patients treated with imatinib and chemotherapy84 and therefore remains SoC. Indications may be restricted with the introduction of third generation tyrosine-kinase inhibitors (TKI) or with consideration of specific PFs. This, however, requires verification in prospective trials with sufficiently long follow-up. Clinical trials with third generation TKI and immunotherapy in older patients may provide relevant data on long-term survival without SCT.
Strict MRD monitoring should be performed after SCT.85 Selection of TKI or decision on TKI change in MRD-positive patients should be based on ABL-kinase domain (KD) mutation status, level, and time of MRD reoccurrence. Patients with early MRD-negative status may either be treated prophylactically or preemptively for at least 1 year of continuous molecular CR. Both strategies have been documented feasible in a prospective randomized trial.86
SCT procedure aspects
Donor type choice does not differ from other leukemias. All ALL MSD, URD, or haploidentical donors may be considered. However, the choice of conditioning regimen may influence outcome. A randomized trial in pediatric ALL showed a clear benefit of myeloablative total body irradiation (TBI) (12 Gy)–based conditioning over chemotherapy-based regimens mainly because of reduced RR.87 In case of limited access to TBI or for patients who cannot tolerate this procedure, conditioning based on IV busulfan or HD thiotepa may be an alternative.88,89 Results of prospective study in adults indicate noninferiority of busulfan/ CP compared with TBI at a total dose of 9 Gy plus CP.90 For older patients reduced-intensity conditioning (RIC) should be considered preferably within clinical trials. The broadest experience comes from a trial which offered conditioning with fludarabine, melphalan, and alemtuzumab.81 Nevertheless, the role of RIC SCT in older patients remains uncertain, particularly with the availability of immunotherapies and third generation TKI.
Antithymocyte globulin (ATG) as part of conditioning prevents chronic graft-versus-host disease (GVHD) as confirmed by 2 recent retrospective studies by the EBMT but was associated with increased RR without significant impact on OS.91,92 G-CSF mobilized PB stem cells is the most frequently source of stem cells.93
Treatment of specific subgroups
Adolescents and young adults
AYA with cancer have been recognized as a vulnerable population in transition between childhood and adulthood aged between 15 and 20 years. A definition of 15 to 39 years old was used defining therapeutic strategies based on unmodified pediatric protocols, whereas it is also recognized that patients aged between 15 to 25 years are more exposed to psychosocial issues.94 Currently, intensive pediatric-based protocols are used up to the age of 55 to 65 years. The outcome of AYA is lower than in children even within pediatric trials. Contributing factors include differences in disease biology, historically different therapeutic approaches, and lack of inclusion in clinical trials. Psychosocial issues may decrease adherence95 to long-lasting and complex protocols. Multidisciplinary teams aware of these complex situations should be involved in treating AYA with ALL.
Biology in AYA
The incidence of ALL is lower in AYA than in children or older adults. T-ALL and LBL are more frequent. Some molecular entities are identified more frequently in AYA, such as iAMP21, IGH@ rearrangements, MEF2D rearrangements, or Ph-like ALL.
Ph-negative ALL
Many historical comparisons addressed the outcome of AYA concomitantly treated in pediatric studies or historic trials designed for adult ALL (reviewed by Boissel and Baruchel96) and confirmed the advantage of pediatric strategies. These observations have led to different strategies in variable age groups including (1) the expansion of pediatric-based protocols in adult patients,14,37,97-100 (2) the setting of specific AYA trials,101 or (3) the extension of upper age limit in pediatric trials.10 Thereby outcome of AYA has improved significantly (supplemental Table 2). Currently, there is no evidence that AYA have poorer results with modern pediatric-based regimens compared with unmodified pediatric protocols. Any age cutoffs for defined protocols should be based on a clear rationale.
Ph-positive ALL
Although most adult groups use dose-reduced chemotherapy plus TKI but recommend SCT, pediatricians have maintained intensive chemotherapy and reduced SCT indications mostly based on early response including MRD.102 Owing to the rarity of the disease in AYA, patients should be included in prospective trials.
Older patients (>55-65 years)
Many groups set an upper age limit for unmodified pediatric-based protocols at the age of 55 years. This is supported by a recent publication indicating a mortality in CR of ∼35% in patients aged between 55 and 59 years and treated according to an intensive pediatric-based protocol.13
Biologic and clinical features
Older patients usually suffer from B-LIN ALL.103,104 The incidence of poor PFs, such as pro-B-ALL, including KMT2A-rearranged ALL, and early T-ALL increases with age.104,105 There is a high incidence of Ph+ALL (24%-51%) or complex aberrations.103,106 Older patients’ performance status frequently deteriorates quickly with the onset of disease and comorbidities are frequent. In one study the incidence of any comorbidity assessed by the HCT-CI (Hematopoietic Cell Transplantation Comorbidity Score) was 76%. Early death (ED) was significantly associated with comorbidity.107 Furthermore, secondary ALL is more frequent in the older population.108
PFs
Potential PFs for ED risk included comorbidity, age, and performance status before onset of leukemia.103,104 PFs for RR are similar to those in younger patients. In older patients with less intensive therapy, a higher rate of MRD persistence can be expected.104 Therefore, prospective evaluation of MRD is essential to identify those who could benefit from alternative, experimental treatments.
Management
A prephase therapy can be essential particularly for stabilization of the general condition. Induction therapy is the most critical phase for management. ED has a wide range (0%-42%) (Table 3). The most frequent cause is infection (reviewed by Gokbuget103). Even with an age adjusted chemotherapy >95% of the patients experience grade 3 to 4 hematologic toxicity during induction. The Swedish registry reported intensive care unit admission in 17% of the patients aged <55 years113 and ED was similar with intensive or so called palliative approaches.119
Results of chemotherapy and antibody therapy in older patients
| Study . | Year . | Age . | Ph+ . | Patients (N) . | CR/CRi rate (%) . | Early death (%) . | Failure (%) . | CCR (%)∗ . | DFS/EFS (%)∗ . | OS (%)† . |
|---|---|---|---|---|---|---|---|---|---|---|
| Chemotherapy | ||||||||||
| Hunault-Berger et al109 | 2010 | |||||||||
| Arm 1 | 68 (55-77) | No | 31 | 90 | 7 | 3 | 32 (2 y) | n.r. | 35 (2 y) | |
| Arm 2 | 66 (60-80) | 29 | 72 | 10 | 17 | 52 (2 y) | 24 (2 y) | |||
| Goekbuget et al110 | 2012 | 57 (55-85) | No | 268 | 76 | 14 | 10 | 32 (5 y) | n.r. | 23 (5 y) |
| Fathi et al111 | 2016 | 58 (51-72) | Yes | 30 | 67 | 3 | 30 | n.r. | 52 (2 y) | 52 (2 y) |
| Ribera et al112 | 2016 | 66 (56-79) | No | 54 | 74 | 14 | 14 | n.r. | 24 (2 y)‡ | 30 (2 y)‡ |
| Kozlowski et al113,§ | 2017 | 69 (62-82) | Yes | 35 | 71 | 20 | 9 | n.r. | n.r. | 20 (3 y) |
| Kozlowski et al113,|| | 2017 | 63 (55-79) | Yes | 79 | 89 | 13 | n.r. | n.r. | n.r. | 39 (3 y) |
| Goekbuget et al104 | 2022 | 68 (56-86) | No | 841 | 73 | 14 | 13 | 37 (3 y) | n.r. | 28 (5 y) |
| Cohort 1 | 593 | 72 | 15 | 13 | 32 | |||||
| Cohort 2 | 248 | 75 | 9 | 16 | 50 | |||||
| Chemo-Immunotherapy | ||||||||||
| Stelljes et al114 InO chemo | 2022 | 64 (56-80) | No | 43 | 100 | 0 | 0 | n.r. | 73 (2 y) | 81 (2 y) |
| Chevallier et al115 InO chemo | 2022 | 68 (55-84) | No | 131 | 90 | n.r. | n.r. | n.r. | 50 (2 y) | 54 (2 y) |
| Goekbuget et al116 Chemo Blina | 2021 | 65 (56-76) | No | 34 | 83 | 7 | 10 | n.r. | 89 (1 y) | 84 (1 y) |
| Advani et al117 Blina mono | 2022 | 75 (66-84) | No | 29 | 66 | n.r. | n.r. | n.r. | 37 (3 y) | 37 (3 y) |
| Nasnas et al118 InO Blina chemo | 2022 | 68 (60-87) | No | 80 | 99 | 0 | 0 | n.r. | n.r. | 46 (5 y) |
| Study . | Year . | Age . | Ph+ . | Patients (N) . | CR/CRi rate (%) . | Early death (%) . | Failure (%) . | CCR (%)∗ . | DFS/EFS (%)∗ . | OS (%)† . |
|---|---|---|---|---|---|---|---|---|---|---|
| Chemotherapy | ||||||||||
| Hunault-Berger et al109 | 2010 | |||||||||
| Arm 1 | 68 (55-77) | No | 31 | 90 | 7 | 3 | 32 (2 y) | n.r. | 35 (2 y) | |
| Arm 2 | 66 (60-80) | 29 | 72 | 10 | 17 | 52 (2 y) | 24 (2 y) | |||
| Goekbuget et al110 | 2012 | 57 (55-85) | No | 268 | 76 | 14 | 10 | 32 (5 y) | n.r. | 23 (5 y) |
| Fathi et al111 | 2016 | 58 (51-72) | Yes | 30 | 67 | 3 | 30 | n.r. | 52 (2 y) | 52 (2 y) |
| Ribera et al112 | 2016 | 66 (56-79) | No | 54 | 74 | 14 | 14 | n.r. | 24 (2 y)‡ | 30 (2 y)‡ |
| Kozlowski et al113,§ | 2017 | 69 (62-82) | Yes | 35 | 71 | 20 | 9 | n.r. | n.r. | 20 (3 y) |
| Kozlowski et al113,|| | 2017 | 63 (55-79) | Yes | 79 | 89 | 13 | n.r. | n.r. | n.r. | 39 (3 y) |
| Goekbuget et al104 | 2022 | 68 (56-86) | No | 841 | 73 | 14 | 13 | 37 (3 y) | n.r. | 28 (5 y) |
| Cohort 1 | 593 | 72 | 15 | 13 | 32 | |||||
| Cohort 2 | 248 | 75 | 9 | 16 | 50 | |||||
| Chemo-Immunotherapy | ||||||||||
| Stelljes et al114 InO chemo | 2022 | 64 (56-80) | No | 43 | 100 | 0 | 0 | n.r. | 73 (2 y) | 81 (2 y) |
| Chevallier et al115 InO chemo | 2022 | 68 (55-84) | No | 131 | 90 | n.r. | n.r. | n.r. | 50 (2 y) | 54 (2 y) |
| Goekbuget et al116 Chemo Blina | 2021 | 65 (56-76) | No | 34 | 83 | 7 | 10 | n.r. | 89 (1 y) | 84 (1 y) |
| Advani et al117 Blina mono | 2022 | 75 (66-84) | No | 29 | 66 | n.r. | n.r. | n.r. | 37 (3 y) | 37 (3 y) |
| Nasnas et al118 InO Blina chemo | 2022 | 68 (60-87) | No | 80 | 99 | 0 | 0 | n.r. | n.r. | 46 (5 y) |
Arm 1, continuous infusion doxorubicin; Arm 2, pegylated doxorubicin; Cohort 1, original version of protocol; Cohort 2, more dose density, PEG-ASP consolidation, MRD-based Blina; CCR, continuous complete remission; CRi, complete remission with incomplete recovery; InO, inotuzumab ozogamicin; n.r., not reported; Ph+, Ph/BCR::ABL1 positive ALL included yes or no.
Median months or probability.
Probability.
Estimated from Kaplan-Meier curve.
EWALL protocol.
ABCVD protocol.
Outcome
Population-based studies reported CR rates of 40% to 70% and OS of 6% to 30%.106,113,119,120 With protocols specifically designed for older patients with ALL CR rates of 43% to 90% and OS rates <30% to 40% after 5 years can be achieved (Table 3). As in protocols for younger patients, steroids and vincristine are the most important drugs in induction therapy. One central question is whether anthracyclines must be included, which type of anthracycline and which dose-intensity. Anthracyclines contribute considerably to BM toxicity. One approach is the use of idarubicin in induction based on a potentially lower cardiac and hepatic toxicity. The results of liposomal anthracyclines in older adults with ALL are not convincing. Tolerability of ASP is reduced in induction in older patients (reviewed by Gokbuget103). One group reported an induction mortality of 34% in patients aged 60 to 65 years treated with an intensive induction including Dexa, high doses of daunorubicin and 2 doses of PEG-ASP.22 Overall, it is advisable to start ASP in older patients later during consolidation.
Although options for intensification of induction therapy are limited, there is still space for intensification of consolidation therapy. To date the largest prospective trial was conducted by the GMALL used a pediatric-based 2-phase induction followed by alternating consolidation cycles and maintenance up to 2.5 years. In CD20 positive ALL 8 doses of Ritux were added. The median age was 67 (55-85) years. The CR rate was 76% in 268 patients; ED rate was 14% and mortality in CR 6%. OS at 5 years was 23%.110 Based on this protocol a consensus treatment approach for older patients with ALL was defined by the EWALL. The Hyper-CVAD protocol was associated with a CR rate of 88%, death in CR of 31%, and OS at 5 years of 21%.27 With the most recent version of the GMALL protocol including MRD-based immunotherapy a CR rate of 75%, ED of 9%, and OS of 50% at 3 years was reported104 (Table 3).
Overall, these standard regimens are the basis for further improvement by moderate time and dose intensification in consolidation, consideration of SCT with RIC and decision on targeted therapies based on MRD. Immunotherapies such as Blina should be considered in older patients with B-LIN ALL showing persistent MRD67; also patients with PR or failure after induction can benefit from treatement with InO or Blina. Older patients may also benefit from the addition of Ritux to the chemotherapy backbone.
Further improvement can be expected from complementing or replacing chemotherapy with immunotherapy independent of MRD. Older patients show a similar tolerability of Blina and InO as younger patients.121,122 In the first experience with a combination of InO with dose reduced chemotherapy and Ritux in patients aged >63 years a CR rate of 98% was reported in 48 evaluable patients. Seventy-eight percent of the patients achieved negative MRD status. However, despite the use of dose-reduced chemotherapy, 93% of the patients developed grade 3 to 5 infections, 17% increases of bilirubin or transaminases and 81% had prolonged thrombocytopenias of >6 weeks with grade 3 to 4 hemorrhage in 15%. Veno-occlusive disease/sinusoidal obstruction syndrome (VOD/SOS) was observed in 8% of the cases. The mortality in CR was however 22% to 25% and the 3-year OS was 56%.123 Importantly, the dose of InO was adapted during the trial in a total of 5 steps and in a further modification Blina was added in the latest cohort after 4 cycles of InO-based chemotherapy.
In older patients trials with different combinations of chemotherapy and sequential trials with Blina and chemotherapy are ongoing and yielded promising interim results.114-118 Longer follow-up will be essential, but it is of interest that despite reduced chemotherapy, the risk of TRM was quite high in some trials.115,118 Particularly in patients aged >70 years with poor risk cytogenetics, outcome was very poor.118
Although these combined approaches are of great interest and bear promise for older patients, it will be a challenge to define new standard regimens and demonstrate their benefit compared with historical data which would be necessary to obtain not only marketing authorization of immunotherapies for first line but also reimbursement in different health care systems.
Ph/BCR::ABL1-positive (Ph+) ALL
Diagnosis and molecular features
Clinical presentation and diagnosis are comparable to Ph-negative ALL. Cardiovascular assessment is particularly important because of the safety profile of TKIs and attention to the echocardiography is relevant as some TKI may cause QT prolongation. It is essential to identify Ph+ ALL as early as possible.
Confirmation of Ph+ ALL relies on karyotyping and/or molecular genetics.1 Additional chromosomal aberrations have been linked to inferior outcome.124-126 PCR analysis of BCR::ABL1 should not be the sole diagnostic test as atypical BCR::ABL1 transcripts may be missed, but determining the BCR::ABL1 isoform is important for subsequent MRD analysis. There is no unequivocal data on the prognostic relevance of the more frequent p190 isoform vs the p210 breakpoint present in one-third of patients.
Principles of treatment
TKIs induce CR in 90% to 95% of patients, display low toxicity, and enable a greater proportion of patients to undergo SCT. TKI should be initiated as early as possible, because delays during induction reduce their efficacy. During induction it is possible to rely on a TKI only to achieve CR, but in practice TKI are usually combined with steroids and often vincristine. Simultaneous administration with most chemotherapeutic agents is feasible. CNS-prophylaxis is mandatory.
Induction and postremission chemotherapy
The combination of imatinib with nonintensive induction was prospectively studied, demonstrating a higher CR rate and lower induction mortality without compromising OS.84 Chemotherapy de-escalation during induction is applicable to all age groups but has not been universally adopted.127-129 Because a minority of patients achieve a deep molecular response after TKI-only induction, additional antileukemic modalities are required as postremission therapy130 (reviewed by Foa and Chiaretti131) (Table 3).
Postremission therapy is commonly initiated with 1 or 2 consolidation cycles combining a TKI with more intensive chemotherapy, analogous to treatment of Ph-negative patients; use of ASP is discouraged by some groups.132 Depending on comorbidities, transplant risk, age, patient preference, and MRD level, further postremission therapy will then consist either of SCT, continued consolidation and maintenance therapy, or switching to an alternative treatment if response is deemed unsatisfactory. Consolidation is followed by maintenance with the initial or an alternative TKI, depending on tolerability and efficacy.133-135 Stopping the TKI in patients who did not undergo transplant is discouraged, even in patients with prolonged MRD-negativity.
MRD
MRD monitoring for Ph+ ALL should rely on BCR::ABL1 measurement but complemented by 1 additional method.1
Kinase domain (KD) mutations
Point mutations in the KD of BCR::ABL1 contribute to most relapses on TKI. Increasing BCR::ABL1 transcripts should prompt mutational analysis136 as the type of mutation may inform which TKI to switch to. In most cases ponatinib will be the TKI of choice. Switching even to the most appropriate TKI will rarely induce a prolonged response in case of overt hematologic relapse. Next-generation sequencing with a sensitivity of 1% to 5% is the method of choice to detect evolving mutated clones in the MRD setting.136 The clinical relevance of low-level mutations at diagnosis remains unclear.135,137
Selecting TKI
Published studies with long-term data indicate that RD and OS with regimens combining chemotherapy and either imatinib or second-generation TKI is comparable138 which may in part be attributable to SCT as a confounding factor. In contrast, a randomized study in pediatric patients showed superiority of dasatinib over imatinib when combined with intensive chemotherapy.139 One caveat of this trial is however, the imatinib dosing, which was lower than typically prescribed for pediatric ALL. Treatment strategies that do not rely on SCT or combination with intensive chemotherapy may benefit from second and third generation TKI. Thus, first-line ponatinib combined with hyper-CVAD resulted in high CR and molecular CR rates, and the 5-year OS (71%) was superior to historical controls.140 In a phase 2 trial of ponatinib plus standard induction and consolidation chemotherapy in patients with newly diagnosed Ph+ ALL, the 3-year EFS and OS rates were 70% and 96%, respectively141 (Table 4). Preliminary data of a randomized trial with ponatinib vs imatinib in combination with dose-reduced chemotherapy in 245 patients showed that ponatinib yielded a significantly higher MRD response rate (34% vs 17%), whereas OS data were not different.146
Prospective trials of 2nd and 3rd generation TKI as front-line therapy for Ph+ ALL
| TKI . | Reference . | N . | Age . | Treatment . | CR rate (%) . | Molecular response (%) . | Allogeneic SCT rate (%) . | RFS/EFS (%) . | Survival (%) . |
|---|---|---|---|---|---|---|---|---|---|
| Dasatinib | |||||||||
| 70 mg BID | Foa et al, 2011130 | 53 | 54 (24-77) | Prednisone | 15 (d 85) | 42 | 22 (20 mo) | 31 (20 mo) | |
| 50 mg BID/100 mg QD 100 mg QD-70 mg QD | Ravandi et al, 2015134 | 72 | 55 (21-80) | Hyper-CVAD | 96 | 65 | 17 | 44 (5 y) | 46 (5 y) |
| 50 mg BID/100 mg QD 70 mg QD | Ravandi et al, 2016129 | 97 | 44 (20-60) | Hyper-CVAD | 88 | — | 42 | 62 (3 y) | 69 (3 y) |
| 140 mg QD; >70 y: 100 mg QD | Rousselot et al, 2016135 | 71 | 69 (55-83) | EWALL backbone | 24 | 10 | 28 (5 y) | 36 (5 y) | |
| 140 mg QD | Foà et al, 2020142 | 63 | 54 (24-82) | Corticosteroids | 98 | 60 (2 cycles of blinatumomab | 38 | 88 (18 mo med. FU) | 95 (18 mo med. FU) |
| Nilotinib | |||||||||
| 400 mg BID | Kim et al, 2015143 | 90 | 47 (17-71) | Intensive chemotherapy | 91 | 77 (3 mo) | 63 | 72 (2 y) | 72 (2 y) |
| 400 mg BID | Ottmann et al, 2018144 | 72 | 66 (55-85) | EWALL backbone | 94 | 58 <0.01 | 33 | 42 (4 y) | 47 (4 y) |
| Ponatinib | |||||||||
| 45-30-15 mg QD | Jabbour et al, 2018127 | 76 | 47 (39-61) | Hyper-CVAD | 98 | 83 | 20 | 67 (5 y) | 71 (5 y) |
| 30-15 mg QD | Ribera et al 2022141 | 30 | 49 (19-59) | Intensive chemotherapy alone | 100 | 71 at week 16 | 87 | 70 (3 y) | 96 (3 y) |
| 45-30-15 QD | Martinelli et al, 2022145 | 44 | 66.5 | 90.9 at week 6, 86.4 at month 6 | 47.7 at week 6 40.9 at month 6 | 11 (transplant unplanned) | 48 (3 y) | 58 (3 y) |
| TKI . | Reference . | N . | Age . | Treatment . | CR rate (%) . | Molecular response (%) . | Allogeneic SCT rate (%) . | RFS/EFS (%) . | Survival (%) . |
|---|---|---|---|---|---|---|---|---|---|
| Dasatinib | |||||||||
| 70 mg BID | Foa et al, 2011130 | 53 | 54 (24-77) | Prednisone | 15 (d 85) | 42 | 22 (20 mo) | 31 (20 mo) | |
| 50 mg BID/100 mg QD 100 mg QD-70 mg QD | Ravandi et al, 2015134 | 72 | 55 (21-80) | Hyper-CVAD | 96 | 65 | 17 | 44 (5 y) | 46 (5 y) |
| 50 mg BID/100 mg QD 70 mg QD | Ravandi et al, 2016129 | 97 | 44 (20-60) | Hyper-CVAD | 88 | — | 42 | 62 (3 y) | 69 (3 y) |
| 140 mg QD; >70 y: 100 mg QD | Rousselot et al, 2016135 | 71 | 69 (55-83) | EWALL backbone | 24 | 10 | 28 (5 y) | 36 (5 y) | |
| 140 mg QD | Foà et al, 2020142 | 63 | 54 (24-82) | Corticosteroids | 98 | 60 (2 cycles of blinatumomab | 38 | 88 (18 mo med. FU) | 95 (18 mo med. FU) |
| Nilotinib | |||||||||
| 400 mg BID | Kim et al, 2015143 | 90 | 47 (17-71) | Intensive chemotherapy | 91 | 77 (3 mo) | 63 | 72 (2 y) | 72 (2 y) |
| 400 mg BID | Ottmann et al, 2018144 | 72 | 66 (55-85) | EWALL backbone | 94 | 58 <0.01 | 33 | 42 (4 y) | 47 (4 y) |
| Ponatinib | |||||||||
| 45-30-15 mg QD | Jabbour et al, 2018127 | 76 | 47 (39-61) | Hyper-CVAD | 98 | 83 | 20 | 67 (5 y) | 71 (5 y) |
| 30-15 mg QD | Ribera et al 2022141 | 30 | 49 (19-59) | Intensive chemotherapy alone | 100 | 71 at week 16 | 87 | 70 (3 y) | 96 (3 y) |
| 45-30-15 QD | Martinelli et al, 2022145 | 44 | 66.5 | 90.9 at week 6, 86.4 at month 6 | 47.7 at week 6 40.9 at month 6 | 11 (transplant unplanned) | 48 (3 y) | 58 (3 y) |
BID, twice a day; FU, follow-up; med., median; QD, once a day.
Dosing considerations apply to all TKI; for imatinib maintaining a high initial dose (800 mg per day for 6-8 weeks) has been associated with better outcome.128 Ponatinib (45 mg per day) and nilotinib have been linked to cardiovascular adverse events, lowering the ponatinib dose when CR is reached to 30 mg per day reduces the risk of arterial occlusive events without compromising efficacy.127 Doses <15 mg per day ponatinib are discouraged by pharmacokinetic data.127,147 Rigorous attention to normalization of blood pressure, blood glucose, and lipids is essential. Other comorbidities to be considered include pulmonary disease (dasatinib) and diabetes (nilotinib). Nilotinib and ponatinib may cause clinically symptomatic pancreatitis, particularly in patients with a history of pancreatitis. Selecting TKI for first-line therapy depends on treatment regimen, comorbidities, and drug approval in different health care systems.
Immunotherapy
Combining TKI with immunotherapy may enhance antileukemic activity. Whether addition of Ritux enhances efficacy as in Ph-negative ALL has not been established. Blina was shown to be highly effective in postremission therapy combined with dasatinib.142 In this trial, all but 1 patient (98%) achieved a CR after dasatinib, and up to 60% obtained molecular response measured by BCR::ABL1 MRD after 2 cycles of Blina. At 18 months median follow-up, DFS was 88%. Observation of several CNS relapses emphasizes the need for intensive CNS-directed prophylaxis. The role of SCT in this setting remains to be established, because ∼50% of the patients received SCT based on investigators’ choice. Several ongoing trials are evaluating the combination of TKI with Blina as first-line therapy for older patients with Ph+ ALL. Also, the role of SCT may be defined in the context of ongoing randomized trials (NCT06061094).
CNS prophylaxis
Dasatinib crosses the blood-brain barrier,148 but the clinical relevance of CSF penetration of TKI is not well-described. Therefore, prophylaxis of CNS relapses is an essential part of all TKI based regimens and usually relies on intensive and prolonged application of IT prophylaxis.
Salvage therapy
Other than TKI, the modalities are the same as for other B-LIN ALL; most patients will have developed KD mutations. Second- and third-generation TKI have response rates of 16% to 46%149,150 with OS of 6 to 9 months.147 OS is poor even in patients referred for SCT.151 With InO in R/R Ph+ ALL higher response rates, longer PFS, and a higher rate of SCT compared with SoC were observed but no benefit in OS.122,152 Single-agent Blina induced CR/CRh in 36% of R/R Ph+ ALL, including patients with the T315I mutation.153 Median relapse-free survival (RFS) and OS were 6.7 months and 7.1 months, respectively. Concurrent use of Blina and ponatinib in R/R Ph+ ALL resulted in a remarkably high CR rate (96%) and encouraging OS (median 20 months), with acceptable tolerability.154 Optimal positioning of chimeric antigen receptor (CAR) T-cell therapy in the treatment of Ph+ ALL remains to be determined. Avoiding relapse is paramount because salvage therapies remain unsatisfactory. In addition to immunotherapies, BH3-inhibitors, and the novel allosteric kinase inhibitor ABL001 (asciminib) are of interest; both have a favorable toxicity profile and act synergistically with available therapeutic modalities used in Ph+ ALL.
Ph-like ALL
Approximately 10% to 30% of cases with B-LIN ALL are characterized by a gene expression profile similar to that of Ph+ ALL, and they were therefore named BCR::ABL1-like or Ph-like ALL.
Diagnosis
Ph-like ALL was first described in adult ALL.155,156 One signature in pediatric ALL, named BCR::ABL1-like, was based on hierarchical clustering of 110 gene probe sets.157 Another signature with 257 gene probe sets, named Ph-like, was based on the prediction analysis of microarrays (PAM) classifier.158 A comparison of both classifiers found a relatively small degree of overlapping, with only 18% of patients consistently classified. However, both classifiers identified patients with a poor outcome and tyrosine kinase fusion genes.159 Only 9 probe sets overlapped between the 2 lists, most likely because of different approaches, algorithms, and patient demographics.
The identification of Ph-like ALL remains challenging and is not SoC. A 15-gene LFS (LDA) gene expression card can be used in clinical diagnostics using quantitative RT-PCR coupled with a mathematical algorithm. The LDA card includes gene probes for the most frequent tyrosine kinase genomic lesions in Ph-like ALL. The concordance rate between the LDA card and the PAM method is 87%.160 Another approach is the “BCR::ABL1-like predictor,” based on the real-time quantitative–PCR quantification of 9 specifically overexpressed genes.161 For comparability of clinical trials the method for identification of Ph-like ALL should be clearly described. There is increasing evidence that BCR::ABL-like ALL should be identified as a subgroup in all patients.
Molecular characterization
Genomic screens and next-generation sequencing have revealed a highly diverse range of aberrations; these include many new fusion genes, including ABL1-class and JAK-class fusion genes.162 Ph-like ALL aberrations can be grouped in 5 major subclasses (supplemental Table 3). Because most studies were carried out in children and young adults, the type of genomic lesions in older adults are less well-understood and might differ.
Incidence
The incidence of Ph-like ALL differs by geographic region, age, reference group, and the methods used for identification. With the PAM-classifier the incidence of Ph-like cases in US cohorts was 20% to 24% in adolescents and older adults (>39 years old), with a higher incidence of 28% in younger adults (aged 21-39 years)161 (Table 5). It is essential to note whether incidences refer to B-LIN as a whole or to a group named B-other, that is, those without distinct cyctogenetic or molecular aberrations.
Results in adult Ph-like ALL
| Study . | Age group (y) . | Total patients and frequency of Ph-like ALL in B-other∗ . | 5-year survival (EFS or OS) . |
|---|---|---|---|
| Roberts et al, 2014162 | 16-20 | 77 (21%) | EFS 41%, OS 66% |
| 21-39 | 46 (27%) | EFS 24%, OS 26% | |
| Herold et al, 2014163 | 16-20 | 5 (19%) | |
| 21-39 | 12 (18%) | DFS (all ages) 19% | |
| 40-55 | 4 (9%) | OS (all ages) 22% | |
| 55-84 | 5 (7%) | ||
| Boer et al, 2015159 | 16-20 | 6 (25%) | EFS (all ages) 24% |
| 21-39 | 9 (19%) | OS (all ages) 30% | |
| 40-71 | 6 (11%) | ||
| Jain et al, 2017164 | 15-39 | 33 (42%) | |
| 40-84 | 16 (24%) | OS (all ages) 23% | |
| Roberts et al, 2017165 | 21-39 | 96 (28%) | EFS 24% |
| 40-59 | 62 (20%) | EFS 21% | |
| 60-86 | 36 (24%) | EFS 8% | |
| Chiaretti et al, 2018161 | 0-15 | 2 (9%) | EFS (excluding children) 22% |
| 15-35 | 29 (29%) | OS (excluding children) 37% | |
| 35 | 23 (31%) | ||
| Stock et al, 20196 | 17-39 | 41 (31%) | OS (3 y), 63% |
| Chiaretti et al, 2021166 | 18-65 | 28 (32%) | EFS (2 y), 34% |
| OS (2 y), 40% |
| Study . | Age group (y) . | Total patients and frequency of Ph-like ALL in B-other∗ . | 5-year survival (EFS or OS) . |
|---|---|---|---|
| Roberts et al, 2014162 | 16-20 | 77 (21%) | EFS 41%, OS 66% |
| 21-39 | 46 (27%) | EFS 24%, OS 26% | |
| Herold et al, 2014163 | 16-20 | 5 (19%) | |
| 21-39 | 12 (18%) | DFS (all ages) 19% | |
| 40-55 | 4 (9%) | OS (all ages) 22% | |
| 55-84 | 5 (7%) | ||
| Boer et al, 2015159 | 16-20 | 6 (25%) | EFS (all ages) 24% |
| 21-39 | 9 (19%) | OS (all ages) 30% | |
| 40-71 | 6 (11%) | ||
| Jain et al, 2017164 | 15-39 | 33 (42%) | |
| 40-84 | 16 (24%) | OS (all ages) 23% | |
| Roberts et al, 2017165 | 21-39 | 96 (28%) | EFS 24% |
| 40-59 | 62 (20%) | EFS 21% | |
| 60-86 | 36 (24%) | EFS 8% | |
| Chiaretti et al, 2018161 | 0-15 | 2 (9%) | EFS (excluding children) 22% |
| 15-35 | 29 (29%) | OS (excluding children) 37% | |
| 35 | 23 (31%) | ||
| Stock et al, 20196 | 17-39 | 41 (31%) | OS (3 y), 63% |
| Chiaretti et al, 2021166 | 18-65 | 28 (32%) | EFS (2 y), 34% |
| OS (2 y), 40% |
B-other: Ph negative without other defining molecular abnormalities (eg, KMT2A, low hypodiploid, hyperdiploid).
MRD response and prognostic significance
Adults with Ph-like ALL had higher rates of MRD positivity after induction. This was associated with an inferior EFS and OS. Seventy percent of patients with Ph-like ALL remained MRD positive.165 This was confirmed in other adult populations.166,167 Data on the impact of MRD response in Ph-like ALL are conflicting. The high RR does not always correlate with MRD response.32 On the other hand, pediatric patients with Ph-like ALL and negative MRD status after standard treatment had no inferior outcome.168 Therefore, aiming at MRD negativity retains prognostic importance also in this subgroup.
Treatment
Novel immunotherapies, such as Blina and InO are investigated32,140,169 and appear to be effective according to anectodal reports of successful treatment. Most groups rely on an MRD-based approach and would offer SCT and/or targeted therapy because of poor MRD response. There are no studies confirming that SCT overcomes the higher RR in this subgroup.
Given the genetic heterogeneity in Ph-like ALL, translation of this subtype to a useful treatment algorithm is a challenge. The ABL class fusions, usually identified by fluorescence in situ hybridization were sensitive to SRC/ABL TKIs, such as imatinib and dasatinib, and there are several anecdotal clinical reports.170 JAK2 fusions, EPOR rearrangements, and activating JAK-STAT mutations were highly sensitive to the JAK2 inhibitor, ruxolitinib in preclinical studies. Another approach could be the use of ponatinib.161,170 For clinical practice, identification of ABL-fusions and MRD-based treatment modifications are essential and several groups recommend identifying Ph-like ALL as a whole subgroup. Diagnostic methods require standardization, but the upfront identification offers the opportunity to further analyze outcomes of this subgroup.
T-ALL
T-ALL comprises 25% of adult ALL171 and treatment is similar to that for B-LIN ALL. After standard induction, an intensification phase containing CP and AC is usual. Intensive use of ASP was highly effective in pediatric T-ALL.172 It is not clear whether nelarabine as consolidation improves outcome in adults. When combined with the hyper-CVAD protocol in newly diagnosed adults with T-ALL/LBL, there was no OS benefit with the addition of nelarabine compared with the historical data.173 A randomized pediatric trial has evaluated nelarabine in newly diagnosed T-ALL/LBL.174 In this study, the 5-year DFS was 88% in the nelarabine group vs 82% in the no-nelarabine group, with an acceptable toxicity. The benefit of nelarabine was not observed in OS. Of note, the rate of isolated and combined CNS relapses was reduced in patients treated with nelarabine. In adult ALL a recent trial did not show a benefit of one course of nelarabine instead of induction phase 2.40 The GMALL 08/2013 trial added 2 cycles of nelarabine in standard-risk ALL and reported excellent outcome; the exact role of nelarabine in this intensive pediatric-based regimen remains to be defined. On the other hand, nelarabine had limited efficacy in MRD positive immature T-ALL175 (Table 1). The MDACC reported improved OS for patients treated with nelarabine compared with retrospective controls, with the notable exception of ETP ALL which did poorly.39 All trials in adults did not reveal specific safety concerns. It is recommended to participate in nelarabine-based trials whenever possible and until more robust data are available.
As for B-LIN ALL MRD testing is of utmost importance and provides guidance for SCT indication. ETP ALL appears to have a poor prognosis with chemotherapy and unfavourable oncogenetics should also lead to consideration of SCT indication. Older patients with high-risk T-ALL may benefit from a RIC if they have a well-matched donor.81
LBL
Lymphoblastic lymphoma (LBL) is a rare entity of 1% to 3% NHL. Eighty percent to 90% are T-cell (T-LBL). LBL and ALL are separated by an arbitrary cut-point of 25% BM infiltration. Gene expression profile and single nucleotid polymorphism profiling revealed differences between T-ALL and T-LBL.176 T-LBL shows a prevalence of 70% of thymic subtype with a favourable outcome compared with 18% early T and 9% mature T-type.
Patient characteristics and PF
T-LBL has a male predominance, presents most often with advanced stage III to IV, and 90% of patients with T-LBL have a mediastinal bulky mass, sometimes with concomitant pleural and pericardial effusions. CNS involvement is seen in 5% to 10%, lymph node or other organ involvement in 70%, and mostly LDH values are increased. In contrast to T-ALL, PB values are most often normal. There are no accepted adverse PFs. They differ in various trials, such as increased LDH, CNS involvement, and in one study a gene classifier was prognostic.177
Therapy
Rarely, tumor lysis syndrome and, in some cases, thoracic compression require immediate therapeutic intervention by corticosteroids. Treatment should be based on ALL regimens.177,178 The CR rate in most studies is 70% to 90%, the EFS is 60% to 70% at 5 years, and the OS range is also between 60% and 70%. CNS prophylaxis is administered as in ALL.
In earlier studies despite mediastinal irradiation of 24-36Gy for residual tumors, the mediastinal relapse rate was high.178 This is now compensated by more intensive systemic chemotherapy. Positron emission tomography may theoretically guide therapy of residual disease179; however, the exact timing, the predictive role, and treatment consequences after positive positron emission tomography are not established. MRD in the BM or PB may help to evaluate the response similar to ALL. SCT has obtained equal results of an OS of 70% compared with chemotherapy alone; the known selection bias for SCT must be considered, however. SCT is therefore reserved for patients with T-LBL in second CR or refractory cases.175,177
Relapsed/refractory ALL
Conventional chemotherapy salvage
Depending on protocol and subtype, 5% to 10% of patients will be primary refractory, and an additional 30% to 60% of patients will relapse. Although BM is the most frequent site of relapse, extramedullary relapses can occur, and incidence may even increase with more widespread use of immunotherapies. Adult patients with R/R ALL have a poor prognosis with salvage chemotherapies. A new CR is attained in 20% to 40% of patients depending on treatment line, but these remissions are in general not durable despite subsequent SCT. Prolonged DFS and cure are observed in ∼10% to 15%113,180-185 (supplemental Table 4). An international study of 1706 patients with R/R Ph− B-LIN ALL reported 3-year OS of only 10%.185 Predictors for outcomes include age, duration of first remission, response to initial salvage therapy, and ability to undergo SCT. For patients who are candidates for salvage therapy, the choice of regimens will be based on the patient age and comorbidities, disease characteristics (eg, immunophenotype, genetic characteristics, and extramedullary involvement among others), type of prior therapy (including SCT), and duration of prior remission. R/R ALL is an emergency and patients should be referred to experienced centers to establish a comprehensive management plan since sequencing and timing of salvage therapies are essential for outcome. SCT indication should be considered in all patients with R/R ALL.
Treatment of extramedullary relapses
CNS is the most frequent site of extramedullary relapse. CNS-directed treatment followed by combined systemic therapy is warranted.186 Recurrences within the CNS usually coincide with or predict systemic relapse; immediate MRD testing is therefore recommended. Although rapid improvement in neurological symptoms and signs can often be achieved with intravenous or oral Dexa, IT therapy should be started promptly twice per week until the CSF is negative. Systemic reinduction therapy should include drugs able to cross the blood-brain barrier, such as HD-MTX or -AC. Patients with isolated or combined CNS relapse are candidates for SCT. Although there are no data to support any conditioning regimen in these circumstances, TBI regimens are preferred. A cranial boost of 6 Gy has been shown to be tolerable in adults receiving TBI-based conditioning.187 There are no randomized data on the potential benefit or harms of giving additional IT therapy posttransplant, and the practice varies by institution. MRD testing in the CSF may help to guide management. Regarding immunotherapies, elimination of CNS leukemia has been observed in patients treated with CD19 CAR T cells. Treatment of other extramedullary relapses is rather individualized, depending on location. Usually, chemotherapy or local therapies, for example, irradiation are attempted because the efficacy of immunotherapies is not clear in this stetting.
New chemotherapeutic and targeted drugs
Liposomal vincristine was developed with the goal of increasing drug exposure of vincristine to leukemic cells while minimizing dose-limiting neurotoxicity. The overall response rate was (CR, CRi, and PR) was 32% with a median RD of 23 weeks and median OS of 4.6 months.188 Grades 3 to 4 neuropathy occurred in 25% of patients.
Clofarabine, a nucleoside analogue approved for use in children with R/R ALL, has as single agent a response rate of only 12% in adults. Response to combination regimens (etoposide/mitoxantrone, cyclophosphamide, or cytarabine) have ranged from 17% to 36%189 but the toxicity of these regimens is high, precluding the use of subsequent SCT in most patients.
Nelarabine was approved for R/R T-ALL/LBL. The first study showed a CR rate of 23% in 26 adult patients with R/R disease. A larger phase 2 study including 126 heavily pretreated R/R adult patients (ages 18-81 years) showed a CR rate of 36% and a PR rate of 10%, with 80% of CR patients bridged to SCT.190 Significant neurologic toxicities are observed in ∼10%. It is not clear whether combination therapies yield higher responses and with the increasing use of nelarabine in first-line therapy, the options in R/R ALL are more limited.
Immunotherapies
Antigens expressed on the surface of ALL cells (CD19, CD20, CD22, and CD52) have proven to be appropriate targets for monoclonal antibodies (MoAb), used either alone or in combination with chemotherapy. MoAb and CAR T cells have thus been developed, demonstrating high antileukemic activity essentially when targeting the B-lineage CD19 and CD22 surface antigens (Table 6). Data for first line have been already discussed. Antigen targeting is more difficult to develop in T-ALL patients, even if the potential of targeting CD38, CD3, CD5, CD7, CD1a, and CCR9 is currently investigated.
Immunotherapy studies in R/R BCP-ALL
| Reference . | Study . | Eligibility . | Patients, N . | Median age (y) . | Prior alloHSCT . | ORR . | Molecular response . | Bridge to alloHSCT . | OS estimate or median . |
|---|---|---|---|---|---|---|---|---|---|
| Multicenter antibody-based studies | |||||||||
| Kantarjian et al, 2016122 | INO phase 3 (INO-VATE) | ≥18 years R/R CD22+ ALL | 109 (67% salvage 1) | 47 | 16% | 81% | 63% | 40% | 7.7 mo |
| Topp et al, 2015191 | Blinatumomab phase 2 | ≥18 years Ph-neg R/R ALL CR1 <12 months | 189 (61% salvage 1) | 36 | 34% | 43% | 35% | 40% | 6.1 months |
| Kantarjian et al, 2017192 | Blinatomomab phase 3 (TOWER) | ≥18 years Ph-neg R/R ALL CR1 <12 months | 271 (42% salvage 1) | 37 | 35% | 44% | 36% | 14% | 7.7 months |
| Gokbuget et al, 201867 | Blinatumomab phase 2 (BLAST) | ≥18 years ALL in HCR MRD ≥ 0.1% | 116 (65% CR1) | 45 | NR | NR | 80% complete MRD response | 67% | 36.5 months |
| Brown et al, 2021193 | Blinatumomab COG AALL1331 | 1-30 years HR/IR ALL in first relapse | 105 (all salvage 1) | 9 | NR | NR | 79% | 73% | 79% at 24 months |
| Reference . | Study . | Eligibility . | Patients, N . | Median age (y) . | Prior alloHSCT . | ORR . | Molecular response . | Bridge to alloHSCT . | OS estimate or median . |
|---|---|---|---|---|---|---|---|---|---|
| Multicenter antibody-based studies | |||||||||
| Kantarjian et al, 2016122 | INO phase 3 (INO-VATE) | ≥18 years R/R CD22+ ALL | 109 (67% salvage 1) | 47 | 16% | 81% | 63% | 40% | 7.7 mo |
| Topp et al, 2015191 | Blinatumomab phase 2 | ≥18 years Ph-neg R/R ALL CR1 <12 months | 189 (61% salvage 1) | 36 | 34% | 43% | 35% | 40% | 6.1 months |
| Kantarjian et al, 2017192 | Blinatomomab phase 3 (TOWER) | ≥18 years Ph-neg R/R ALL CR1 <12 months | 271 (42% salvage 1) | 37 | 35% | 44% | 36% | 14% | 7.7 months |
| Gokbuget et al, 201867 | Blinatumomab phase 2 (BLAST) | ≥18 years ALL in HCR MRD ≥ 0.1% | 116 (65% CR1) | 45 | NR | NR | 80% complete MRD response | 67% | 36.5 months |
| Brown et al, 2021193 | Blinatumomab COG AALL1331 | 1-30 years HR/IR ALL in first relapse | 105 (all salvage 1) | 9 | NR | NR | 79% | 73% | 79% at 24 months |
| . | . | . | Patients . | . | . | . | . | ||
|---|---|---|---|---|---|---|---|---|---|
| Reference . | Institution . | Coactivation domain . | Screened, N . | Median age, y (range) . | Infused, N . | ORR % . | CRS incidence, % . | Neurotoxicity incidence, % . | OS estimate or median . |
| CART19 studies | |||||||||
| Maude et al, 2014195 | Penn | 4-1BB | NR | 14 (5-60) | 30 | 90% | 100% (severe, 27%) | 43% | 78% at 6 months |
| Davila et al, 2014196 | MSKCC | CD28 | NR | 50 (NA) | 16 | 88% | 44% (severe) | 25% (Gr 3/4) | NR |
| Lee et al, 2015197 | NCI | CD28 | NR | 15 (5-27) | 21 | 67% | 76% (Gr 3/4, 29%) | 19% (Gr 3/4, 5%) | 52% at 12 months |
| Turtle et al, 2016198 | FHCRC | 4-1BB | 32 | 40 (20-73) | 30 | 97% | 83% | 50% (Gr 3/4, 50%) | NR |
| Gardner et al, 2017199 | SCRI | 4-1BB | 45 | 12 (1-25) | 43 | 93% | 93% (severe, 23%) | 44% (severe, 21%) | 69.5% at 12 months |
| Maude et al, 2018200 | Multicentre | 4-1BB | 92 | 11 (3-23) | 75 | 81% | 77% | 40% (Gr 3/4, 13%) | 76% at 12 months |
| Park et al, 2018201 | MSKCC | CD28 | 83 | 44 (23-74) | 53 | 83% | 85% (Gr 3/4, 26%) | 48% (Gr 3/4, 42%) | Median, 12.5 months |
| Hay et al, 2019202 | FHCRC | 4-1BB | 59 | 39 (20-76) | 53 | 85% | NR | NR | Median, 20 months in MRD-neg pts |
| Shah et al, 2021203 | Multicentre | CD28 | 71 | 44 (30–59) | 55 | 71% | 89% (Gr3/4 24%) | 60% (Gr3/4/5 26%) | Median 18.2 months |
| . | . | . | Patients . | . | . | . | . | ||
|---|---|---|---|---|---|---|---|---|---|
| Reference . | Institution . | Coactivation domain . | Screened, N . | Median age, y (range) . | Infused, N . | ORR % . | CRS incidence, % . | Neurotoxicity incidence, % . | OS estimate or median . |
| CART19 studies | |||||||||
| Maude et al, 2014195 | Penn | 4-1BB | NR | 14 (5-60) | 30 | 90% | 100% (severe, 27%) | 43% | 78% at 6 months |
| Davila et al, 2014196 | MSKCC | CD28 | NR | 50 (NA) | 16 | 88% | 44% (severe) | 25% (Gr 3/4) | NR |
| Lee et al, 2015197 | NCI | CD28 | NR | 15 (5-27) | 21 | 67% | 76% (Gr 3/4, 29%) | 19% (Gr 3/4, 5%) | 52% at 12 months |
| Turtle et al, 2016198 | FHCRC | 4-1BB | 32 | 40 (20-73) | 30 | 97% | 83% | 50% (Gr 3/4, 50%) | NR |
| Gardner et al, 2017199 | SCRI | 4-1BB | 45 | 12 (1-25) | 43 | 93% | 93% (severe, 23%) | 44% (severe, 21%) | 69.5% at 12 months |
| Maude et al, 2018200 | Multicentre | 4-1BB | 92 | 11 (3-23) | 75 | 81% | 77% | 40% (Gr 3/4, 13%) | 76% at 12 months |
| Park et al, 2018201 | MSKCC | CD28 | 83 | 44 (23-74) | 53 | 83% | 85% (Gr 3/4, 26%) | 48% (Gr 3/4, 42%) | Median, 12.5 months |
| Hay et al, 2019202 | FHCRC | 4-1BB | 59 | 39 (20-76) | 53 | 85% | NR | NR | Median, 20 months in MRD-neg pts |
| Shah et al, 2021203 | Multicentre | CD28 | 71 | 44 (30–59) | 55 | 71% | 89% (Gr3/4 24%) | 60% (Gr3/4/5 26%) | Median 18.2 months |
FHCRC, Fred Hutchinson Cancer Research Center; HCR, hematologic CR; HR, high risk; IR, intermediate risk; MSKCC, Memorial Sloan Kettering Cancer Center; NCI, National Cancer Institute; NR, not reported; ORR, overall response rate; SCRI, Seattle Children’s Research Institute.
Inotuzumab
InO is an anti-CD22 antibody conjugated to calicheamicin, a cytotoxic antibiotic agent. InO has been approved for R/R CD22-positive ALL based on a randomized study, the rate of CR/CRi was 81% after InO vs 33% after standard salvage chemotherapy122 Among responders, the rate of MRD negativity was 78% in the InO group vs 28% in the control group. Median OS was 7.7 vs 6.7 months, respectively. Liver-related adverse events (AE) were more common in the InO group, with an 11% incidence of VOD vs 1%, respectively. Results were confirmed in long-term follow-up.204 It is open whether combination of InO and chemotherapy can further improve outcome of R/R ALL. To reduce the VOD risk, it is recommended to administer only up to 2 cycles InO before SCT, to avoid double alkylators in conditioning and to strinctly monitor patients for VOD signs after SCT. InO retains effectiveness also in patient with high proliferative relapse205 and shows some efficacy in extramedullary relapse.206 A close surveillance of liver function tests is advised.
Blinatumomab
Blina is a bispecific T-cell engager antibody construct that enables autologous CD3-positive T cells to target and eliminate CD19-positive B cells including B-LIN blasts. Blina has been approved for R/R ALL based on a randomized study. The CR/CRi rate was 44% after Blina vs 25% after standard salvage chemotherapy.192 MRD negativity was 76% in the Blina group vs 48% in the control group and median OS was 7.7 vs 4.4 months, respectively. The gain was more marked in patients receiving Blina as first salvage therapy and in patients with BM infiltration <50%.207 AE of interest were manageable cytokine release syndrome (CRS) and transient neurologic events. More favorable results with Blina were observed in MRD-positive ALL as discussed above. Two recent pediatric randomized studies have also investigated the use of Blina as postreinduction therapy in first relapse in an MRD setting and as bridge to transplant.193,194 These studies demonstrated fewer and less severe toxicities, higher rates of MRD response, greater likelihood of proceeding SCT and improved DFS and OS in the Blina arm.
Chimeric antigen receptor T-cells (CAR-T)
CAR-T targeting the CD19 antigen have generated promising results in children and adults with R/R ALL. Characteristics and results of largest noncomparative CAR-T studies published to date are detailed in Table 6.195-203 Median ages were relatively low. Overall response rates ranged from 67% to 97% in patients who were infused, and complete MRD response by MFC was achieved in most responders. Most patients had very advanced disease with a substantial proportion of patients relapsing after prior SCT. In order to make results comparable to other approaches in R/R ALL the evaluation of intent-to-treat populations is essential, because not all patients receive an intended CAR-T-cell therapy owing to relapse or death during bridging or toxicities. Overall applicability of the procedure has been evaluated in the mainly pediatric ELIANA trial, in which 75 of the 92 patients (81%) screened were infused.199 The rationale for an upper age-limit of 25 years in this trial is unclear. It led to the approval of tisagenlecleucel for patients aged between 1 and 25 years old with R/R ALL.
Data in adult ALL are still limited and data from clinical trials and real-world indicate that the treatment principle is less effective in full cytologic relapse. The Zuma-3 trial yielded promising results in 55 patients with heavily pretreated adult R/R ALL and a median age of 40 (28-52) years. Overall, 71 patients were enrolled. 62% of the treated patients had BM infiltration >25% at time of infusion. The CR/CRi rate was 71% in the patients who received infucion and 54% of all patients. The median OS of patients who received treatment was 18 months and the median RFS 11.6 months. This trial led to marketing authorization of brexucabtagene autoleucel.203
CRS and neurotoxicity are more common and more severe with CAR-T as compared with Blina, likely because of massive, induced CAR-T expansion/activation and endothelial activation.208 This makes CAR-T a therapy that should be administered in specialized centers with a trained intensive care unit. Many issues still need to be elucidated, including the impact of CAR-T subsets and persistence and the role of prior alloSCT and disease burden at infusion time on CAR-T efficacy and the need for subsequent SCT. Genetically engineered “off-the-shelf” allogeneic CAR-T, aiming to increase the applicability and rapidity of the procedure, are under clinical development. Owing to a relatively high incidence of CD19-negative ALL recurrence, strategies combining CD19 and CD22 targeting, using dual or bispecific CAR-T, are also investigated.
In countries with all options available, the selection and sequencing of Blina, InO, and CAR-T in R/R BLIN ALL is debated. There is no clear recommendation, because no comparative studies have been conducted to date; furthermore, the availability of CAR T cells is limited. One might propose favoring Blina in patients with relatively low disease burden and preserved T-cell functions, whereas InO might be used to reduce high disease burden though trials were limited to patients with peripheral blasts <10 000/μL. A sequence of both compounds, InO followed by Blina is also of interest. CAR-T might be indicated to treat more advanced disease, particularly recurrence after SCT. Even if CAR-T have been used as a bridge-to-transplant in most patients treated to date, the question of whether CAR-T could replace SCT in the next future is of great clinical interest. The potential negative impact of Blina209 or InO failure before CAR-T infusion requires larger evaluation. It remains unclear whether prior failure is a selection of unfavorable disease biology or whether there is a specific underlying mechanism.
Other investigational drugs
The most promising targeted agents are those targeting major molecular pathways controlling cell proliferation and apoptotic response (eg, multiple kinases and members of BCL-2, TP53, RAS, mTOR/PI3K, pre-B/B-cell receptor, and NOTCH networks).26 A variety of precision medicine approaches are under investigation though often based on considerations of disease biology and anecdotal cases only.210 Further investigation in prospective clinical trials remains a challenge. Some anecdotal remissions have been reported using FLT3 inhibitors, JAK inhibitors in activated IL7R pathway cases, imatinib or dasatinib in NUP214-ABL1 mutated cases. More recently, promising data for menin inhibitors have been reported for KMT2a rearranged acute leukemias. Targeted therapy is less developed in T-ALL.211 The CD38 antigen has been considered as a potential target in T-ALL. Combining targeting of CD38 with conventional relapse therapy has been explored.212,213 Targeting apoptotic pathways with BCL-2 inhibitors are currently tested in clinical trials. Thus a selective BCL-2 inhibitor, with low-dose navitoclax, a BCL-XL/BCL-2 inhibitor was evaluated in 47 patients with relapsed T- and B-cell disease and achieved a remarkable 60% CR rate with 28% proceeding to potentially curative therapy.214 Frequent activation of the NOTCH pathway in T-ALL/LBL, through NOTCH1 or FBXW7 gene mutations, led to developing NOTCH targeting approaches, such as γ-secretase inhibitors (GSIs) or NOTCH inhibiting antibodies.215 Several groups are attempting to develop CAR-T cells for relapsed T-cell disease that avoid the problem of fratricide and prolonged T-cell aplasia.216 A first-in-man trial of donor-derived CAR T cells from China in 20 patients has made major progress with high efficacy and acceptable toxicity.217
Late effects
With improving cure rates in adult ALL, research in late effects, quality of life, and prognostic factors for late AE becomes increasingly important. Survivors of childhood cancer have a higher mortality, more chronic medical conditions, impaired general and mental health, functional impairment, and poorer social parameters compared to healthy relatives and the general population.218,219 The GMALL group has recently summarized medical conditions in 538 long-term survivors of ALL therapy. Sixty-six percent of the patients had no comorbidity. The most frequent diagnoses were infections (12%), fatigue (13%), and GVHD (15%), whereas the most frequently affected organs classes were endocrine (17% men and 24% women), neurologic (27%), and skin (18%).220 Late effects can include general sexual hormone deficiency, thyroid disorders, premature menopause, infertility, osteonecrosis and osteoporosis, cardiotoxicities, neuropsychological disorders, fatigue, and second malignancies. The incidence of late effects is higher in patients with SCT compared with those without and additional late effects, such as skin disorders in the context of chronic GVHD, sicca syndrome, restrictive and obstructive pulmonary disorders and cataract may occur.220,221 Although patients can maintain their fertility after chemotherapy,222 infertility is observed in almost all transplanted patients.223,224
Avascular osteonecrosis is a clinically relevant problem which is particularly observed in AYA. Intensified treatment with steroids is a risk factor. Effects may be enhanced by asparaginase or HD-MTX. In one pediatric study with postinduction Dexa or prednisone pulses the incidence of skeletal toxicity was overall 13% (11% fractures and 4% osteonecrosis). It was higher (25% vs 11%) in older children (>10 years vs <10 years) treated with Dexa pulses.225,226 In contrast the UKALL/ECOG2993 study for adult ALL found an overall incidence of 4% osteonecrosis. The incidence was higher in younger (<20 years) vs older patients.227 Approximately half of pediatric patients with osteonecrosis underwent surgical procedures.228 There might be a correlation between coagulation disturbances and osteonecrosis. A PAI-1 gene polymorphism, which has been previously associated with risk of thrombosis was also associated with osteonecrosis in childhood ALL.229 Recently a potential correlation between hypertriglyceridemia—a side effect of ASP—and osteonecrosis has been reported.230 Most cases of osteonecrosis in patients with ALL occur within the first 3 years of treatment and hip and knees are the most frequent locations. The most sensitive diagnostic method is magnetic resonance imaging (MRI). Some authors suggest performing at least 1 regular MRI of hip and knees in high-risk populations at the end of therapy. Early detection, prevention, and management of osteonecrosis is a major joint challenge for pediatric and adult hematologists.231
Cardiotoxicity, particularly cardiomyopathy, pericarditis, or congestive heart failure, is often attributed to anthracyclines.232 The risk seems to be correlated with the overall drug dose, although it may also be idiosyncratic and occur after few applications. Factors, such as mediastinal irradiation, infections, or combination of different cardiotoxic drugs may enhance the risk.232 Cardioprotection with dexroxazone may be an option, for example, in patients with higher cumulative doses of anthracyclines.233 Cardiotoxicity, for example manifested as a reduced ejection fraction may become manifest years after treatment. Cardiovascular check-up should, therefore, be part of aftercare.
Unspecific neuropsychological disorders are often described in adult patients with ALL.220 This includes complaints about cognitive disturbances. For this phenomenon, known as “chemo-brain,” a physiological correlate is unknown.234 A relevant number of cured patients with ALL suffer from fatigue.220,235 Fatigue is probably underdiagnosed in aftercare236; its detection, for example, by specific questionnaires should be attempted.
Survivors of ALL have an additional risk to develop cancer. PFs for secondary malignancies may be alkylating agents, epipodophyllotoxins, CNS irradiation, TBI conditioning before SCT, and moreover different gene aberrations.237 In adult ALL survivors the most frequent second malignancies are hematopoietic neoplasias which mostly occur within the first 5 to 10 years after end of chemotherapy.238 In addition, a variety of solid tumors is observed such as breast, thyroid, gastrointestinal, lung, skin, urogenital, brain, or sarcoma.182
It is very important to provide all physicians involved in aftercare with sufficient information about treatment and the relevant aspects of after-care. Long-term observation of cured patients with ALL is an essential part of ongoing and future clinical trials and a major challenge in an environment of limited financial ressources for academic groups. Depending on the different health care systems joint forces in dedicated late effect clinics can combine pediatric and adult hematology teams.
Management of specific situations
ALL in pregnancy
Acute leukemia is estimated to affect 2 in 100 000 pregnancies. Current treatments are typically chosen based on case reports and retrospective analyses and depend heavily on personal and societal considerations as well as the medical considerations of gestational age, disease biology and the patient’s clinical status. A comparison of 15 women treated during pregnancy to 330 nonpregnant controls provided evidence that pregnancy does not necessarily affect the overall outcome of ALL.239 Pregnant women with ALL should always be managed in experienced centers with close cooperation with the obstetric and foetal medicine teams. The management depends on the trimester of pregnancy. During the first trimester, termination of pregnancy is a strong consideration because foetal safety precludes delivering of standard ALL therapy. Subsequently, many chemotherapy agents can be safely administered to achieve maternal CR although systemic antifolates should be avoided at any time during pregnancy. The potential use of ASP later during pregnancy is debated, particularly because of its effects on coagulation and depending on risk-benefit ratio it should rather be postponed to treatment cycles after delivery. The use of specific steroids for fetal lung maturation should be considered. Delivery should be timed for the date consistent with foetal survival and be performed in the setting of adequate maternal hematopoiesis.
MPAL
Leukemias of ambiguous lineage origin present a problem for both diagnosis and management (reviewed by Gökbuget et al,1 Gokbuget et al,220 and Khan et al.240 In pediatric mixed phenotype acute leukemias (MPAL) initial treatment with ALL-oriented regimens is recommended.241,242 Lack of response should prompt a switch to AML-orientated regimens. A similar strategy for adults is reasonable. It is debated whether the diagnosis of MPAL represents an indication for SCT in CR1 in general or only in cases of persistent MRD.
Secondary ALL
ALL arising as a second malignancy (with or without cytotoxic therapy for a prior malignancy) is increasingly recognized (reviewed by Aldoss et al243). Secondary ALL occurs after a variety of antecedent diagnosis including breast cancer, lymphomas, myeloma, sarcomas, and neuroblastoma. Cases in which there has been no prior cytotoxic therapy, inherited germ line mutations in tumor suppressor genes or oncogenes such as TP53 (Li-Fraumeni syndrome) or mismatched repair genes (MSH2, MLH1, MSH6, and PMS2) should be suspected.244 For ALL after prior cytotoxic therapy, US SEER registry data showed a median latency of 60 months and a significantly inferior median 5-year survival compared with de novo ALL.245 Development of ALL after therapy with lenalidomide may represent a specific biologic correlation.246 Generally, treatment strategy is similar to newly diagnosed ALL although a higher risk of toxicity might be expected.
ALL and Down syndrome
Individuals with Down syndrome (DS) have a 10- to 20-fold increased risk of ALL, which usually occurs during childhood and is associated with a genetic susceptibility. ALL correlated to DS is well-described for pediatric patients. Recently 3 distinct molecular subtypes with higher incidence in DS-ALL have been described (CRLF2 rearranged, C/EBP alteration, and IGH::IGF2BP1 rearrangement).247 Pediatric patients with DS-ALL have an increased RR and TRM, mainly because of infections. Although standard therapies are usually offered to patients with DS-ALL, some modifications are recommended, such as reduction of anthracyclines and high-dose methotrexate. Intensive prophylaxis and monitoring for infections is essential. Replacement of toxic chemotherapies by immunotherapies is tested in clinical trials. DS-ALL in adults should be managed accordingly.248
Supportive care
Supportive care recommendations of the EWALL have been published.249 In the following paragraphs, only selected specific aspects for management of ALL are reviewed.
High WBC
When patients present with very high WBC, leukostasis with resultant tissue damage can occur in vulnerable capillary regions, causing organ compromise. Leukostasis is diagnosed on the presence of unexplained hypoxia, neurological symptoms, renal failure, cardiac ischemia, priapism, or severe retinopathy. Management of “symptomatic” leukostasis is a medical emergency. It may be treated with leukapheresis, but this should not be seen as a substitute for prompt initiation of ALL therapy, such as steroid prephase.
TLS
Patients with a high risk of developing tumor lysis syndrome (TLS) (WBC >100 × 109/L, high tumor burden)250 should receive increased hydration (3 L/m2 per day), unless there is evidence of renal insufficiency and oliguria and rasburicase prophylaxis. Urinary alkalinization is no longer recommended.250 Intractable fluid overload, hyperkalemia, hyperuricemia, hyperphosphatemia, or hypocalcemia are indications for renal dialysis.
Antiinfection prophylaxis
Antibacterial, antifungal, and antiviral prophylaxis and active management of infections are of utmost importance, given the fact that infections are still the major cause of death in CR. Management should be performed according to respective guidelines.
Growth factors
The prophylactic administration of G-CSF in ALL shortened neutropenia duration,251-254 improved adherence to chemotherapy schedule and in one study reduced the incidence of infections.252 Based on these data the majority of European study groups have implemented prophylactic use of G-CSF in their pediatric-based protocols since decades. A joint analysis of long-term follow-up data from 5 European clinical trials revealed survival advantages in the G-CSF group, particularly in DFS (5-y DFS 38% vs 24%) and OS in T-ALL (5-y 51% vs 29%), probably as the result of improved dose-dense administration of chemotherapy courses without significant decrease of the 8 weeks mortality rate.255
Menstruation prophylaxis
To suppress menorrhagia during thrombocytopenic periods and avoid undesired pregnancy during chemotherapy, continuous administration of progestational agents is preferred over combined oral contraception, especially during ASP treatment period and its prothrombotic state. For norethisterone a nonsignificant association between exposure to higher doses and risk of thromboembolism has been described; medroxyprogesterone acetate was discussed as preferable option in patients with high risk of venous embolism.256 Progestational agents should not be used >6 months to prevent meningioma occurrence that has been reported lately.
Preservation of fertility
As part of informed consent before ALL therapy, hematologists should address the possibility of infertility in patients treated during their reproductive years, discuss fertility preservation options, and refer all patients to appropriate reproductive specialists. Sperm cryopreservation should be proposed in all men before start of chemotherapy. In women, ovarian tissue cryopreservation is an option to be considered before SCT. To limit the risk for disease reintroduction after ovarian tissue transplantation, quantification of ovarian MRD is being explored to decide for ovarian reimplantation.257 So far there is no clear recommendation for the use of gonadotropin-releasing hormone (GnRH) agonists (GnRHa) for fertility preservation, particularly because of conflicting data on their effect on the risk of ovarial insufficiency and likelihood of later pregnancy. Patients should have the opportunity for counseling on the different available options and GnRHa may be used based on individual considerations.258 It is recommended to reevaluate ovarian function after completion of chemotherapy (AMH, antral follicular count).
Summary and outlook
The basis for adequate management of ALL is the application of comprehensive diagnostic tools, the identification of prognostic factors including the continous monitoring of MRD.1 The standard treatment of ALL is often defined by national study groups and international consortia259 as part of prospective treatment optimization trial. Furthermore, the clinical data bases of study groups often include comprehensive data on biologic markers and are sources of reliable real-world data.
Currently, excellent results are achieved with current standard pediatric-based chemotherapy particularly in younger patients. To reduce the burden of morbidity and mortality, less toxic regiments are an important goal. The gap between health care systems is increasing. Although many new compounds can be used in first-line based on integration into NCCN guidelines in the United States,260 specific marketing authorization and reimbursement decisions are required in most European countries.
Immunotherapy with antibodies is standard for R/R ALL. Availability of CAR-T as well as data in adult ALL are still limited and the sequencing of these therapies will be a challenge. The indication for CAR-T in relapse after SCT is generally accepted, if possible, already in the setting of MRD relapse. In R/R ALL the definition of adequate bridging therapies and the criteria to decide on the need of subsequent (second) SCT are essential.
For Ph+ ALL, the selection of TKIs, the efficacy of chemotherapy-free regimens, indications for change of TKI, the impact of MRD measured with different methods, and the role of immunotherapies and HSCT will be important research questions.
Less progress has been made regarding prognostic classification of T-ALL, and there are few promising new compounds.261 Although overall prognosis of T-ALL is quite favorable, new approaches are urgently required for poor prognostic subgroups because survival after relapse is rare.
Late effects of treatment, such as osteonecrosis are increasingly relevant with improved ALL survival, requiring joint efforts of pediatric and adult study groups to better understand biology, improve surveillance and define potential treatment modifications.
Many new treatment approaches are applicable only in small subcohorts of ALL and represent a challenge for successful clinical trials. This applies also for the need of more individualized relapse therapies integrating molecular and immune targets as well as in vitro response data. These strategies require international collaboration including joint forces of pediatric and adult study groups.
Regulatory prerequisites are expanding regarding marketing authorization and reimbursement for new drugs in ALL, which is treated with complex combination therapies. The evaluation of new compounds in rare molecular subgroups of ALL will require European-level collaboration with the European Medical Agency and intergroup data sharing, as in the Harmony IMI initiative (https://www.harmony-alliance.eu). New strategies for clinical trial design must be developed in concertation with patient representatives, and there is an urgent need to support and maintain the successful infrastructure of independent academic study groups through funding programs.259
In the next years, these challenges will become evident for the integration of new monoclonal antibodies and cell therapies into first line. Several clinical trials are ongoing in Europe and worldwide requiring harmonization to enable future meta-analyses.57,185,262 First-line therapies will not address the impact of a single compound but on new combination strategies, risk stratifications and approaches, such as SCT. One major research question will certainly focus on the future role of SCT in adult and pediatric ALL.
Future ALL treatment will be based on very successful standards with increasing implementation of immunotherapies. The goal is to improve the prognosis of patients who are at high risk but also significantly reduce treatment-related morbidity and mortality for patients of all ages.
Authorship
Contribution: All coauthors reviewed literature, wrote parts of the manuscript, reviewed the manuscript, and approved the final manuscript.
Conflict-of-interest disclosure: N.G. has received institutional research funding from Amgen, Clinigen, Incyte, Jazz, Novartis, Pfizer and Servier and received speaker honoraria or fees for advisory board participation from Amgen, AstraZeneca, Autolus, Celgene, Clinigen, Gilead, Incyte Jazz, Novartis, Pfizer, and Servier. N.B. received honoraria from Amgen, Incyte, Jazz Pharma, Novartis, Pfizer, Servier and research grants from Amgen, Novartis, Incyte, Jazz Pharma, and Sanofi. S.C. served on the advisory boards of Incyte, Pfizer, and AbbVie and as a consultant for Gilead and Amgen. H.D. received research funding from Amgen, Astellas, Celgene, Incyte, Jazz, Pfizer, and Servier and honoraria from Daiichi Sankyo, Incyte, Jazz, and Servier. M.D. received research support from AstraZeneca. R.F. received speaker honoraria from Amgen, Novartis, and AstraZeneca and served on the advisory board of Merck Sharp & Dohme. S.G. received speaker honoraria or served on the advisory boards of Amgen, Novartis, Pfizer, Gilead, and Zentiva; received speaker honoraria from Angelini; and received travel grants from Gilead. M.H. received research support from Novartis, travel support from AbbVie and Beigene, and speaker honoraria from Servier; joined the advisory board of Servier; and was part of advisory boards of Amgen and Clinigen. D.I.M. provided consulting for Pfizer, Novartis, and Kite. O.O. received speaker honoraria from, joined advisory boards of, and received research grants from Incyte; received research support from Amgen and Celgene; and received honoraria for advisory boards from Autolus. A.R. received research support from Servier. P.R. received research grants from Pfizer and Incyte. J.R. received speaker honoraria from and served on the advisory boards of Amgen, Pfiizer, Shire, Ariad, Incyte, and Takeda and received research support from Amgen. R.B. served on the advisory boards of Novartis and Kite/Gilead and received honoraria or travel support from Amgen, Incyte, Servier, Jazz, and Pfizer. The remaining authors declare no competing financial interests.
Correspondence: Nicola Gökbuget, University Hospital, Department of Medicine II, Theodor Stern Kai 7, 60590 Frankfurt, Germany; email: goekbuget@em.uni-frankfurt.de.
References
Author notes
The online version of this article contains a data supplement.

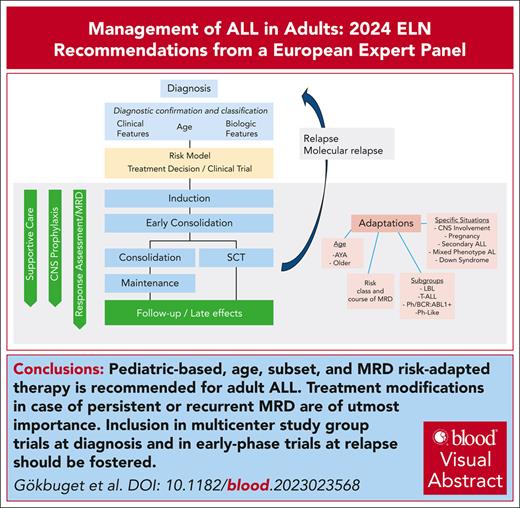
This feature is available to Subscribers Only
Sign In or Create an Account Close Modal