Background: In the Phase 3 randomized, placebo-controlled VIALE-A study, venetoclax (Ven) + azacitidine (Aza) improved overall survival (OS) and complete response (CR) rates vs Aza + placebo (Pbo; DiNardo et al. N Engl J Med. 2020. In Press). Neutropenia and related infections are common in acute myeloid leukemia (AML) and exacerbated by Ven combinations. Antimicrobial prophylaxis is used variably in patients (pts) with AML receiving low-intensity therapies. Ven dose modifications are required for concomitant use of commonly used antimicrobials that are strong or moderate cytochrome P450 3A inhibitors (CYP3Ai). A previous report's findings showed that Ven dose reductions with CYP3Ai do not affect Ven exposure (Wei et al. Blood. 2020;135:2137). This analysis evaluated the use of prophylactic anti-infective CYP3Ai and these agents' effects on infections and efficacy outcomes in the VIALE-A study (NCT02993523).
Methods: Pts with newly diagnosed AML, aged ≥75 y or 18-75 yand ineligible for induction therapy were enrolled. Pts were randomized 2:1 to receive Ven (daily, orally) + Aza (75 mg/m2 d 1-7, subcutaneously/intravenously) or Pbo + Aza in 28-d cycles. Anti-infective prophylaxis was required for pts with absolute neutrophil count <500/µL. For concomitant use of CYP3Ai, Ven dose was reduced from the intended target dose of 400 mg to 200 mg (Cycle 1 ramp-up: d 1, 50 mg; d 2, 100 mg; d 3, 200 mg) for moderate CYP3Ai and 50 mg (Cycle 1 ramp-up: d 1, 10 mg; d 2, 20 mg; d 3, 50 mg) for strong CYP3Ai. Use of anti-infective CYP3Ai agents reported as being given for prophylaxis during Cycles 1 and 2 was assessed; there was a possibility that CYP3Ai agents were used for nonprophylactic purposes not documented as such. The effects of concomitant use of these agents with the required Ven dose reductions on infection and efficacy outcomes were evaluated.
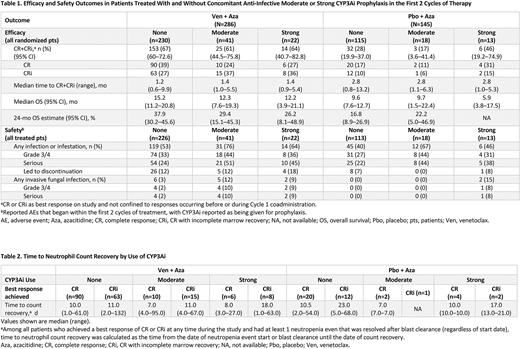
Results: In total, 286 pts were randomized to Ven + Aza and 145 to Pbo + Aza. Within the first 2 cycles of therapy, concomitant anti-infective prophylaxis agents considered moderate CYP3Ai were received by 41/286 pts (14%) in the Ven + Aza arm and 18/145 (12%) in the Pbo + Aza arm. Concomitant anti-infective prophylaxis agents considered strong CYP3Ai were received in the first 2 cycles by 22/286 pts (8%) treated with Ven + Aza and 13/145 pts (9%) treated with Pbo + Aza. The median duration of each prophylactic CYP3Ai agent use was 12.5 d (range, 1-614) in the Ven + Aza arm and 15 d (range, 1-731) in the Pbo + Aza arm.
The rates of CR+CR with incomplete marrow recovery (CRi) as a best response during the study were similar with concomitant use of moderate (61%; CR, 24%; CRi, 37%) or strong (64%; CR, 27%; CRi, 36%) CYP3Ai with adjusted-dose Ven vs no use of CYP3Ai (67%; CR, 39%; CRi, 27%; Table 1). The relatively lower CR and higher CRi rates appear to be due to delay in recovery of peripheral blood counts in pts who received strong CYP3Ai during the first 2 cycles (Table 2). The median time to first CR+CRi was 1.2 mo (range, 0.6-9.9), 1.4 mo (range 1.0-5.5), and 1.4 mo (range, 0.9-5.4) in those receiving no, moderate, and strong CYP3Ai agents, respectively, in the Ven + Aza arm. Median OS was not statistically different regardless of use of moderate or strong CYP3Ai in both arms, as noted by overlapping CIs (Table 1). The 24-month estimated OS was 26.2% (95% CI, 8.1-48.9) for pts receiving strong CYP3Ai vs 37.9% (95% CI, 30.2-45.6) for those receiving no CYP3Ai. Rates of any-grade or Grade 3/4 infections within the first 2 cycles varied between those with or without use of moderate CYP3Ai reported as being given as prophylaxis in both treatment arms (Table 1). Rates of invasive fungal infections were 3%, 12%, and 9% with Ven + Aza and 0%, 0%, and 15% with Pbo + Aza in pts receiving no, moderate, and strong CYP3Ai agents, respectively. Rates of discontinuation from infections were similar in both arms regardless of CYP3Ai use.
Conclusions:Antimicrobial prophylaxis with moderate or strong CYP3Ai was used in neutropenic pts in the VIALE-A study, with overall similar composite remission rates with Ven dose reductions. The use of CYP3Ai did not increase rates of discontinuation from infections. Analysis of differences in baseline characteristics, rates of infections, and efficacy outcomes for pts who began anti-infective prophylaxis at the initiation of therapy is ongoing.
Jonas:Amgen: Consultancy, Other: TRAVEL, ACCOMMODATIONS, EXPENSES (paid by any for-profit health care company), Research Funding; GlycoMimetics: Consultancy, Other: TRAVEL, ACCOMMODATIONS, EXPENSES (paid by any for-profit health care company), Research Funding; Celgene: Consultancy, Research Funding; Jazz: Consultancy, Research Funding; Takeda: Consultancy; Tolero: Consultancy; Treadwell: Consultancy; Forty Seven: Research Funding; Accelerated Medical Diagnostics: Research Funding; AROG: Research Funding; Daiichi Sankyo: Research Funding; F. Hoffmann-La Roche: Research Funding; Forma: Research Funding; AbbVie: Consultancy, Other: TRAVEL, ACCOMMODATIONS, EXPENSES (paid by any for-profit health care company), Research Funding; Genentech/Roche: Research Funding; Sigma Tau: Research Funding; Hanmi: Research Funding; Pfizer: Research Funding; LP Therapeutics: Research Funding; Incyte: Research Funding; Pharmacyclics: Research Funding. Dinardo:Takeda: Honoraria; Notable Labs: Membership on an entity's Board of Directors or advisory committees; Novartis: Consultancy; Daiichi Sankyo: Consultancy, Research Funding; Calithera: Research Funding; Celgene: Research Funding; Agios: Consultancy, Research Funding; AbbVie: Consultancy, Research Funding; ImmuneOnc: Honoraria. Fracchiolla:AbbVie: Honoraria, Membership on an entity's Board of Directors or advisory committees, Other: TRAVEL, ACCOMMODATIONS, EXPENSES (paid by any for-profit health care company); Gilead: Honoraria, Membership on an entity's Board of Directors or advisory committees, Other: TRAVEL, ACCOMMODATIONS, EXPENSES (paid by any for-profit health care company), Speakers Bureau; Pfizer: Honoraria, Membership on an entity's Board of Directors or advisory committees, Other: TRAVEL, ACCOMMODATIONS, EXPENSES (paid by any for-profit health care company), Speakers Bureau; Amgen: Honoraria, Membership on an entity's Board of Directors or advisory committees, Other: TRAVEL, ACCOMMODATIONS, EXPENSES (paid by any for-profit health care company), Speakers Bureau. Pristupa:Beigene: Honoraria, Research Funding; State Institution of Health of the Ryazan Regional Clinical Hospital: Current Employment; Pfizer: Honoraria, Research Funding, Speakers Bureau; Janssen: Honoraria, Research Funding; AbbVie: Honoraria, Research Funding; Takeda: Honoraria, Research Funding, Speakers Bureau; Daiichi Sankyo: Honoraria, Research Funding; Paraxel: Honoraria, Research Funding; Acerta: Honoraria, Research Funding; Pharmacyclics: Honoraria, Research Funding; Geron: Honoraria, Research Funding. Ishizawa:Takeda: Honoraria; Ono: Honoraria; Chugai: Honoraria; Eizai: Honoraria; Novartis: Honoraria, Research Funding; Celgene: Honoraria; SymBio: Research Funding; Bayer: Research Funding; AbbVie: Research Funding. Jin:The First Affiliated Hospital of Zhejiang University: Current Employment. Konopleva:Rafael Pharmaceutical: Research Funding; F. Hoffmann La-Roche: Consultancy, Research Funding; Genentech: Consultancy, Research Funding; Eli Lilly: Research Funding; Sanofi: Research Funding; Calithera: Research Funding; Kisoji: Consultancy; AstraZeneca: Research Funding; AbbVie: Consultancy, Research Funding; Amgen: Consultancy; Ascentage: Research Funding; Reata Pharmaceutical Inc.;: Patents & Royalties: patents and royalties with patent US 7,795,305 B2 on CDDO-compounds and combination therapies, licensed to Reata Pharmaceutical; Ablynx: Research Funding; Forty-Seven: Consultancy, Research Funding; Stemline Therapeutics: Consultancy, Research Funding; Cellectis: Research Funding; Agios: Research Funding. Ofran:AbbVie: Consultancy, Membership on an entity's Board of Directors or advisory committees; Novartis: Consultancy, Membership on an entity's Board of Directors or advisory committees; Astellas: Consultancy; Rambam Health Care: Current Employment; Pfizer: Consultancy, Membership on an entity's Board of Directors or advisory committees; Roche: Consultancy. Kovacsovics:Pfizer: Research Funding; Novartis: Research Funding; AbbVie: Research Funding; Jazz: Honoraria; Astella: Honoraria; Agios: Honoraria. Kantarjian:Daiichi-Sankyo: Honoraria, Research Funding; BMS: Research Funding; Actinium: Honoraria, Membership on an entity's Board of Directors or advisory committees; Oxford Biomedical: Honoraria; Janssen: Honoraria; Delta Fly: Honoraria; BioAscend: Honoraria; Jazz: Research Funding; Abbvie: Honoraria, Research Funding; Sanofi: Research Funding; Aptitute Health: Honoraria; Adaptive biotechnologies: Honoraria; Pfizer: Honoraria, Research Funding; Novartis: Honoraria, Research Funding; Amgen: Honoraria, Research Funding; Immunogen: Research Funding; Ascentage: Research Funding. Hong:Genentech, Inc.: Current Employment; F. Hoffmann-La Roche: Current equity holder in publicly-traded company. Duan:AbbVie: Current Employment, Other: may hold stock or options. Ainsworth:AbbVie: Current Employment, Current equity holder in publicly-traded company. Potluri:AbbVie: Current Employment, Other: may hold stock or stock options. Werner:AbbVie: Current Employment, Current equity holder in publicly-traded company. Svensson:AbbVie: Current Employment, Current equity holder in publicly-traded company. Pratz:Millennium: Research Funding; Daiichi Sankyo: Research Funding; Agios: Other: Scientific Advisory Board, Research Funding; Jazz Pharmaceutical: Consultancy; Celgene: Other: Scientific Advisory Board; Boston BioMedical: Consultancy; AbbVie: Other: Scientific Advisory Board, Research Funding; Astellas: Other: Scientific Advisory Board, Research Funding.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal