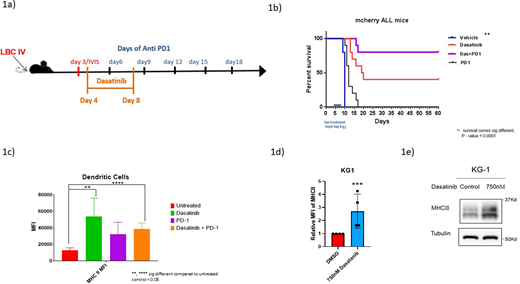
Anti-PD1 therapy in hematologic malignancies has shown clinically inferior effects when compared to solid tumors.1 However, immunotherapy has been part of the standard of care in the treatment of acute leukemia for over 40 years. Recently, loss of MHCII was shown to be a mechanism of Immune escape in patients with acute myeloid leukemia after stem cell transplantation.2 Here we demonstrate, in pre-clinical models, that responses to anti-PD1 can be enhanced by increased antigen presentation through induction of MHCII. We used a Philadelphia chromosome positive (Ph+) Acute Lymphoblastic Leukemia (ALL) syngeneic mouse model and treated 4 cohorts of mice with the following regimens: 1) vehicle 2) single agent Dasatinib; 3) single agent anti-PD1; 4) combination anti-PD1 + Dasatinib (Figure 1a). Single agent Dasatinib, was highly active against this model compared to vehicle. Although anti-PD1 therapy showed little or no activity as a single agent, when combined with sub-therapeutic doses of Dasatinib, we observed significantly enhanced survival of mice compared to single agent anti-PD1 or to single agent Dasatinib. (Figure 1b) In our mouse model, Dasatinib increased the MHCII on the surface of the antigen presenting cells in the tumor microenvironment, most notably in dendritic cells (CD11c+ cells) from mouse tumor-infiltrating lymphocytes collected from the mice bone marrow after treatment (Fig 1c). To prove whether Dasatinib-induced MHCII expression can occur independently of a tumor microenvironment, we treated antigen presenting cell lines (KG1) with Dasatinib and were able to confirm an increased MHCII expression by flow cytometry and by western blotting. (Fig 1d) Since Dasatinib is approved for the treatment of Philadelphia chromosome positive ALL, CML, and has been used in a variety of other settings in the treatment of both malignant hematology and solid tumors, this data has immediate translational potential. In fact, Dasatinib in combination with anti-PD1 therapy is currently being tested in a variety of phase I/II trials in various kinds of malignancies (NCT04284202, NCT03516279). Here we show that induction of MHCII by Dasatinib may serve as a biomarker and could predict the potential benefit from the combination treatment. Additionally, targeting anti-PD1 following Dasatinib administration may increase response rates of patients treated with Dasatinib, providing a rationale for sequential treatment design in patients with persistent minimal residual disease.
Figure 1. (a) C57BL/6J mice were injected via tail vein injection with a transplantable, immunocompetent BCR-ABL+ B-ALL model and treated with vehicle/Dasatinib/or anti-PD1 as described in the schema. (b) Overall survival of the mice treated in the previous experiment. (c) MHCII cell surface expression on dendritic cells (CD11+ cells) from the bone marrow of treated mice. (d) relative Cell surface expression of MHCII in the KG1 cell line (APC cell line) treated with Dasatinib vs. vehicle. (e) protein expression of MHCII in the KG1 cell line (APC cell line) treated with Dasatinib vs. vehicle.
1. Masarova L, Kantarjian H, Ravandi F, Sharma P, Garcia-Manero G, Daver N. Update on Immunotherapy in AML and MDS: Monoclonal Antibodies and Checkpoint Inhibitors Paving the Road for Clinical Practice. Adv Exp Med Biol 2018;995:97-116.
W2. Christopher MJ, Petti AA, Rettig MP, et al. Immune Escape of Relapsed AML Cells after Allogeneic Transplantation. New England Journal of Medicine 2018;379:2330-41.
Koller:Jazz Pharmaceuticals, Inc.: Consultancy. Konopleva:Stemline Therapeutics: Consultancy, Research Funding; F. Hoffmann La-Roche: Consultancy, Research Funding; Forty-Seven: Consultancy, Research Funding; Sanofi: Research Funding; Genentech: Consultancy, Research Funding; Ablynx: Research Funding; Cellectis: Research Funding; Agios: Research Funding; AbbVie: Consultancy, Research Funding; Ascentage: Research Funding; Kisoji: Consultancy; Reata Pharmaceutical Inc.;: Patents & Royalties: patents and royalties with patent US 7,795,305 B2 on CDDO-compounds and combination therapies, licensed to Reata Pharmaceutical; AstraZeneca: Research Funding; Amgen: Consultancy; Calithera: Research Funding; Eli Lilly: Research Funding; Rafael Pharmaceutical: Research Funding.
anti-pd1 therapy in acute lymphoid leukemia
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal