
Background
Azacitidine (AZA) is one of the hypomethylating agents (HMAs) proven to significantly prolong overall survival (OS) for patients with higher-risk myelodysplastic syndromes (h-MDS) via a prospective phase 3 trial. However, the optimal treatment schedule of AZA for h-MDS has not yet been prospectively determined. As a 7-day treatment schedule includes weekends, a 5-day administration has also been applied with h-MDS, though solid evidence has not been established. Based on this background, we planned to compare AZA treatment on 7-day and 5-day schedules.
Patients
Patients diagnosed with de novo or treatment-related MDS (FAB-defined RAEB and RAEB-t), aged 16 years or older were eligible for this study, if they showed adequate performance status (ECOG PS 0-2) and no history of HMA treatment or chemotherapy. Candidates for allogeneic stem-cell transplantation were excluded.
Study design
This was a multicenter, randomized, open-label, phase 3 trial to compare the efficacy of AZA treatment for 7-days (AZA-7) to 5-days (AZA-5). The primary endpoint was 2-year OS rate (2y OS). AZA was given subcutaneously at 75 mg/m² every 28 days. To test the non-inferiority of AZA-5 to AZA-7, 2y OS of AZA-7 was estimated at 30%, and the delta was defined as 11%, for which 410 patients were needed to complete the trial. However, because of the poor recruitment, this study closed prematurely, and the protocol-planned interim analysis (when 200 patients were registered) was performed. This protocol was approved by the IRB at each trial center. All patients provided written informed consent, and the trial was conducted in accordance with the Declaration of Helsinki. The efficacy of AZA was evaluated using IWG2006 criteria, and treatment was continued until study completion, or relapse, unacceptable toxicity, or disease progression was observed. Efficacy was analyzed by intention-to-treat. Secondary endpoints were the hematological response rate, 2-year leukemia-free survival (2y LFS) rate, the cytogenetic response, and occurrence of adverse events.
Results
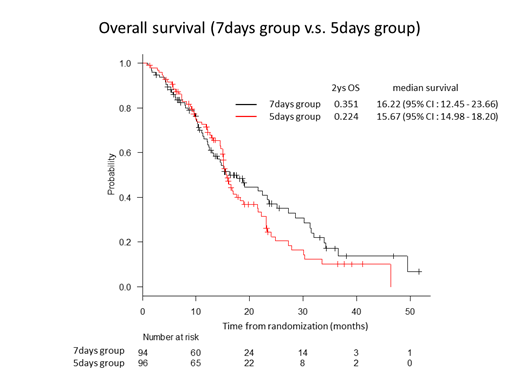
Between January 2013 and Jun 2018, 201 patients were randomly assigned to AZA-7 (n=99) or AZA-5 group (n=102). The median age of all patients at the enrollment was 73.5 years old (range, 48 to 91). Between the two groups, there was no significant difference in the baseline characteristics, such as sex, type of MDS (de novo or treatment-related), percent of BM blasts, FAB and WHO classification, and IPSS and IPSS-R risk. After excluding 5 and 6 patients from AZA-7 and AZA-5, respectively (poor data entry, change in diagnosis), at the time of last follow-up, 59 and 67 patients died in AZA-7 and AZA-5, respectively. The Kaplan-Meier estimates of the 2y OS and 2y LFS were 35.1% (95% CI, 23.8 to 46.7) and 27.3% (95% CI, 17.3 to 38.3) for AZA-7, and 22.4% (95% CI, 13.1 to 33.3) and 20.5% (95% CI, 11.6 to 31.2) for AZA-5, respectively. There was no significant difference in the frequency of erythroid, platelet or neutrophil improvement, or hematological response (complete or partial response, or any hematological improvement) between the two groups. The most common non-hematological adverse event of grade 3 to 4 (in ≥20% of the patients in either group) was febrile neutropenia (33.7% for AZA-7, and 30.5% for AZA-5).
Conclusion and Discussion
Because of the premature termination of this trial, statistical analysis for the primary endpoint (2y OS of 35.1% for AZA-7, and 22.4% for AZA-5) could not provide any solid evidence. However, considering the difference of more than 10% in 2y OS, and no major differences in the safety profiles, we favor AZA-7 to AZA-5 for MDS patients with RAEB and RAEB-t.
Kiguchi:Tejin Co., Ltd.: Research Funding; Taiho Pharmaceutical Co., Ltd.: Research Funding; SymBio Pharmaceutical Co., Ltd.: Research Funding; Celgene Co., Ltd.: Research Funding; Janssen Pharmaceutical Co., Ltd.: Research Funding; Sumitomo Dainippon Pharmaceutical Co., Ltd.: Research Funding; Novartis Pharmaceutical Co., Ltd.: Research Funding; Nippon Shinyaku Co., Ltd.: Research Funding; Astellas Pharmaceutical Co., Ltd.: Research Funding; MSD CO., Ltd.: Research Funding; Kyowa Hakko Kirin Co., Ltd.: Research Funding; Otsuka Pharmaceutical Co., Ltd.: Research Funding; Bristol-Myeres Squibb Co., Ltd.: Research Funding; Sanofi K.K., Ltd.: Research Funding; Celltrion, Inc.: Research Funding. Usuki:Daiichi Sankyo Co., Ltd.: Research Funding, Speakers Bureau; Astellas Pharma Inc: Research Funding, Speakers Bureau. Suzuki:kyowa Hakko Kirin: Consultancy, Honoraria; MSD k.k.: Research Funding; Nippon Shinyaku: Honoraria; Novartis: Honoraria; takeda: Research Funding; Incyte and Pfizer: Research Funding; astellas: Research Funding; Celgene: Honoraria; Chugai: Honoraria, Research Funding; daiichi sankyo: Research Funding; eisai: Research Funding. Tomita:Chugai Pharmaceutical Co., Ltd.: Honoraria, Research Funding; Kyowa Kirin: Research Funding; Taiho Pharma: Research Funding. Handa:Ono: Research Funding. Maeda:Janssen Pharmaceutical K.K..: Honoraria; Nippon Shinyaku Co., Ltd.: Honoraria. Iyama:Otsuka Pharmaceutical Co., Ltd.: Honoraria; Otsuka Pharmaceutical Factory: Honoraria; Astellas Pharma: Honoraria; Daiichi Sankyo: Honoraria; Allexion Pharma: Honoraria; CSL Behring: Honoraria. Yamauchi:Astellas, AbbVie: Consultancy; Pfizer, Chugai, Teijin, Solasia: Research Funding. Kiyoi:Daiichi Sankyo Co., Ltd: Research Funding; Bristol-Myers Squibb: Research Funding; Pfizer Japan Inc.: Honoraria; Nippon Shinyaku Co., Ltd.: Research Funding; Otsuka Pharmaceutical Co.,Ltd.: Research Funding; Perseus Proteomics Inc.: Research Funding; FUJIFILM Corporation: Research Funding; Astellas Pharma Inc.: Honoraria, Research Funding; Zenyaku Kogyo Co., Ltd.: Research Funding; Chugai Pharmaceutical Co., Ltd.: Research Funding; Sumitomo Dainippon Pharma Co., Ltd.: Research Funding; Eisai Co., Ltd.: Research Funding; Kyowa Hakko Kirin Co., Ltd.: Research Funding; Takeda Pharmaceutical Co., Ltd.: Research Funding. Miyazaki:Chugai: Research Funding; Otsuka: Honoraria; Novartis: Honoraria; Nippon-Shinyaku: Honoraria; Dainippon-Sumitomo: Honoraria; Kyowa-Kirin: Honoraria.
Author notes
Asterisk with author names denotes non-ASH members.

This icon denotes a clinically relevant abstract

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal