Novel therapies targeting BTK (ibrutinib), PI3Kδ (idelalisib) and BCL2 (venetoclax) are active in poor-risk chronic lymphocytic leukemia (CLL) and are widely administered to patients with relapsed/refractory (R/R)-CLL. Given the activity of ibrutinib in high-risk CLL patients, including those with del17p/TP53 mutation or germline IGHV genes, we assumed that this drug could diminish the prognostic utility of the CLL-IPI, because the outcome of patients with high- and very high-risk CLL-IPI scores may improve. Recently, Soumerai et al (Lancet Hematology, 2019) proposed a new risk score for overall survival (OS) based on four accessible markers, called BALL (β2-microglobulin, anemia, LDH, last therapy), in the setting of R/R-CLL patients receiving chemo-immunotherapy or targeted therapies in clinical trials. This model segregates CLL patients into three groups with significantly different OS. This multicenter, observational retrospective study aimed at validating the proposed BALL score for R/R-CLL real-world patients treated with ibrutinib. The primary objectives were to determine whether: i) the BALL score is of prognostic value for ibrutinib R/R-CLL patients, ii) the BALL score is predictive of progression-free survival (PFS); and iii) the CLL-IPI retains its prognostic power also in R/R patients treated with ibrutinib.
This study, from an institutional Italian multicenter working group on CLL ('Campus CLL'), included CLL patients collected from 18 Italian centers and 1 Israeli center, who received ibrutinib 420 mg/day outside of clinical trials as salvage therapy with available data for the calculation of the BALL and CLL-IPI scores at the time of the start of treatment. OS was estimated for all subgroups according to both scores. Additionally, risk-specific PFS was assessed. Kaplan-Meier curve, log-rank test and Cox regression analyses were performed. The prognostic accuracy of the predictive model was assessed by the Harrell's C-index.
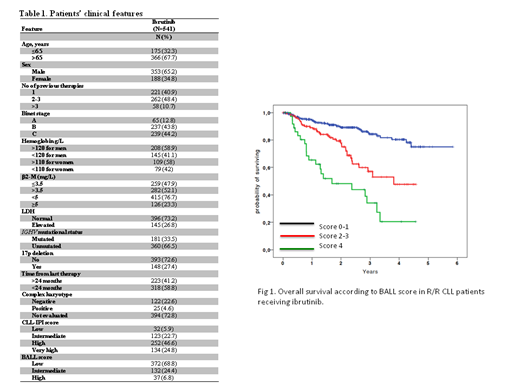
Overall, 541 CLL patients were included in this analysis. The majority of patients were Binet stages B and C (87.2%). The median age was 70.4 years (range 37 - 88) and 353 cases (65.2%) were male. The median number of previous therapies was 2 (range 1-9). The baseline patients' features are listed in Table 1. After a median follow-up of 1.8 years (range 1 month to 5.8 years), 101 patients had died and 206 experienced an event (death or progression). According to the BALL score, 372 patients (68.8%) were classified as low-risk, 132 (24.4%) as intermediate-risk and 37 (6.8%) as high-risk. Stratification of patients according to the BALL score predicted significant differences in terms of OS. Thus, low-risk patients had a 2-year OS probability of 89.2% (HR=1), intermediate-risk of 79.9% (HR=2.8, 95%CI 1.8-4.4, P<0.0001) and high-risk of 48.2% (HR=6.6, 95%CI 3.9-11, P<0.0001) (Figure 1). The C-statistic was 0.66 (P<0.001) for OS prediction. The CLL-IPI score indicated that 32 patients (5.9%) were classified as low-risk, 123 (22.7%) as intermediate-risk, 252 (46.6%) as high-risk and 134 (24.8%) as very high-risk. Stratification of patients according to the CLL-IPI score did not allow to predict significant differences in OS. Indeed, low-risk patients had a 2-year OS probability of 92.7% (HR=1), intermediate-risk patients of 88.4% (HR=2.3, 95%CI 0.5-9.9, P=0.26), high-risk of 84% (HR=3.3, 95%CI 0.8-13.7, P=0.1) and very high-risk of 76.4% (HR=4.3, 95%CI 1.038-17, P=0.046). Both the BALL and CLL-IPI scores failed to stratify patients significantly in terms of PFS.
Our multicenter retrospective study confirms the prognostic power of the BALL score in this real-world series of R/R-CLL patients treated with ibrutinib, as previously reported in the setting of clinical trials (Soumerai et al, Lancet Hematology 2019). Furthermore, the CLL-IPI did not retain a discriminative power in the current retrospective study of ibrutinib-treated patients, possibly due to i) the well-known activity of ibrutinib in high-risk CLL patients, i.e. those with TP53 disruption and/or germline IGHV genes, which represent the most heavily weighted adverse risk factors contributing to the CLL-IPI, and to ii) the fact that the CLL-IPI was designed for patients receiving front-line chemo-immunotherapy. Conversely, the BALL score may identify higher risk R/R-CLL patients potentially requiring alternative and more effective therapeutic strategies.
Mauro:Jannsen: Consultancy, Research Funding; Gilead: Consultancy, Research Funding; Abbvie: Consultancy, Research Funding; Shire: Consultancy, Research Funding; Roche: Consultancy, Research Funding. Coscia:Gilead: Membership on an entity's Board of Directors or advisory committees; Karyopharm Therapeutics: Research Funding; Janssen: Membership on an entity's Board of Directors or advisory committees, Research Funding; Abbvie: Membership on an entity's Board of Directors or advisory committees. Herishanu:Roche: Honoraria; Janssen: Honoraria; AbbVie: Honoraria. Varettoni:ABBVIE: Other: travel expenses; Roche: Consultancy; Gilead: Other: travel expenses; Janssen: Consultancy. Rigolin:AbbVie: Speakers Bureau; Gilead: Speakers Bureau; Gilead: Research Funding. Rossi:Gilead: Honoraria, Membership on an entity's Board of Directors or advisory committees, Research Funding; Abbvie: Honoraria, Other: Scientific advisory board; Janseen: Honoraria, Other: Scientific advisory board; Roche: Honoraria, Other: Scientific advisory board; Astra Zeneca: Honoraria, Other: Scientific advisory board. Gaidano:AbbVie: Membership on an entity's Board of Directors or advisory committees, Speakers Bureau; Janssen: Membership on an entity's Board of Directors or advisory committees, Speakers Bureau; Sunesys: Consultancy, Honoraria; Astra-Zeneca: Consultancy, Honoraria. Cuneo:Amgen: Honoraria; Abbvie: Honoraria, Speakers Bureau; Gilead: Honoraria, Speakers Bureau; Janssen: Honoraria, Speakers Bureau; Roche: Honoraria, Speakers Bureau. Foà:Roche: Consultancy, Speakers Bureau; Celltrion: Membership on an entity's Board of Directors or advisory committees; Abbvie: Consultancy, Speakers Bureau; Amgen Inc.: Consultancy, Membership on an entity's Board of Directors or advisory committees, Speakers Bureau; Amgen Inc.: Consultancy, Membership on an entity's Board of Directors or advisory committees, Speakers Bureau; Novartis: Consultancy, Membership on an entity's Board of Directors or advisory committees, Speakers Bureau; Janssen: Consultancy, Membership on an entity's Board of Directors or advisory committees, Speakers Bureau; Incyte: Consultancy, Membership on an entity's Board of Directors or advisory committees, Speakers Bureau; Shire: Consultancy, Membership on an entity's Board of Directors or advisory committees, Speakers Bureau; Incyte: Consultancy, Membership on an entity's Board of Directors or advisory committees, Speakers Bureau; Roche: Consultancy, Speakers Bureau; Novartis: Consultancy, Membership on an entity's Board of Directors or advisory committees, Speakers Bureau; Pfizer: Membership on an entity's Board of Directors or advisory committees, Speakers Bureau; Shire: Consultancy, Membership on an entity's Board of Directors or advisory committees, Speakers Bureau; Pfizer: Membership on an entity's Board of Directors or advisory committees, Speakers Bureau; Abbvie: Consultancy, Speakers Bureau; Janssen: Consultancy, Membership on an entity's Board of Directors or advisory committees, Speakers Bureau; Celltrion: Membership on an entity's Board of Directors or advisory committees.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal