Myelodysplastic syndromes (MDS) are clonal hematopoietic stem cell (HSC) malignancies characterized by ineffective hematopoiesis. Genetic alterations do not fully explain the molecular pathogenesis of the disease, indicating that other types of lesions, such as transcriptional aberrations, may play a role in its development. Moreover, MDS prevalence is almost exclusive to older patients, suggesting that elderly-related alterations may predispose to the development of this clinical entity. Thus, study of the transcriptional lesions occurring in the aging-MDS axis could shed some light of the molecular bases of the disease.
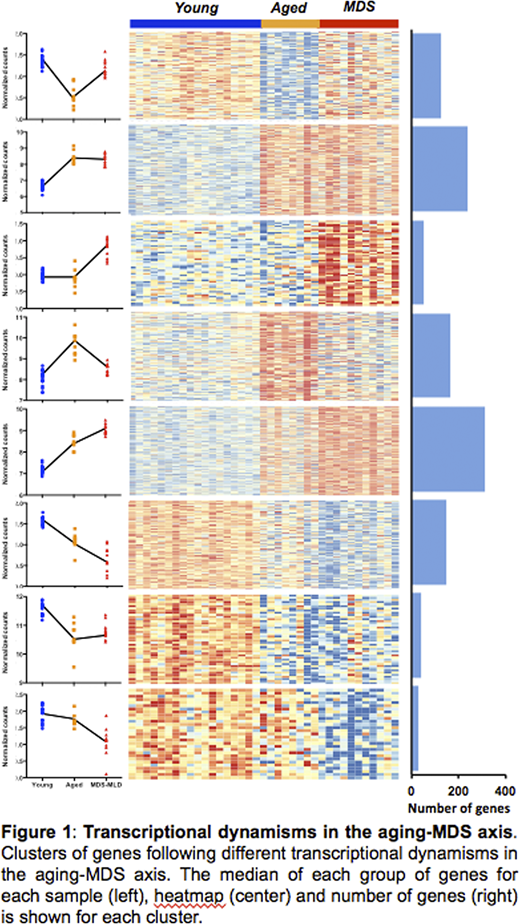
To characterize the transcriptional profile of HSCs in aging and MDS, we isolated CD34+, CD38-, CD90+, CD45RA- cells from 11 untreated MDS patients with unilineage and multilineage dysplasia (median of 75 y/o), as well as from 16 young and 8 elderly healthy donors (median of 21 and 70 y/o, respectively), and their expression profile was analyzed using MARS-seq. Unsupervised principal component analysis demonstrated that the three groups of HSCs clustered separately, indicating that different expression profiles characterize healthy young and elderly, and MDS-associated HSCs. To better understand the gene expression deregulation of HSCs, we analyzed the transcriptional dynamisms along the aging-MDS axis, detecting groups of genes following different patterns of expression. Some gene clusters showed exclusive alteration either in aging or in the progression from elderly HSCs to MDS-HSCs, other groups of genes presented a continuous alteration along the axis, and some displayed opposite regulation in aging and in the transition to MDS (Figure 1).
Genes showing specific downregulation in aging were involved in DNA damage sensing and repair, and in cell cycle regulation, whereas genes overexpressed in this process were enriched in apoptosis regulators and in cancer-associated genes, including AML-related factors. These findings indicate that transcriptional changes in aging may predispose for MDS and AML, and potentially other malignancies.
Interestingly, we detected a group of genes in which the age-mediated upregulation of gene expression was reversed to that of young HSCs in MDS, indicating a "rejuvenation" profile of malignant HSCs. These genes were involved in response to inflammation, to different types of stress conditions such as hypoxia or radiation, and to cytokines. Elderly HSCs may upregulate such genes in response to the known inflammatory microenvironment of elderly bone marrow. Intriguingly, the decrease in expression detected in MDS suggests that malignant HSCs lose the ability of reacting to such stimuli, possibly favoring their survival in a hostile microenvironment.
Finally, the analyses performed allowed for the identification of genes showing MDS-specific deregulation. Genes specifically overexpressed in MDS compared to normal (both young and elderly) HSCs, we enriched in transcriptional and epigenetic regulators, and among them, we detected the presence of DDIT3/CHOP, a member of the CCAAT/enhancer-binding protein (C/EBP) family of transcription factors. To determine its potential effects on hematopoietic deregulation, DDIT3 was exogenously overexpressed in healthy HSCs. Notably, its upregulation produced an erythroid bias in an ex-vivo differentiation system, with an increase in the percentage of erythroblasts and a decrease in granulocytes and monocytes compared to HSCs transduced with the empty vector. Transcriptomic analysis of transduced HSCs not subjected to differentiation demonstrated how DDIT3 overexpression produced an erythroid-prone state of HSCs, suggesting it may act as a pioneer factor in MDS-HSCs. Furthermore, gene set enrichment analysis showed that DDIT3 overexpression produced an MDS-like transcriptional profile, suggesting this factor may be key in the acquisition of the disease.
Altogether, our results demonstrate that HSCs undergo transcriptional changes in the aging-MDS axis that may alter their intrinsic functions as well as their response to the microenvironment, ultimately contributing to the acquisition of the disease. In particular, our data show that DDIT3 may be a potential driver of MDS transformation.
Paiva:Amgen, Bristol-Myers Squibb, Celgene, Janssen, Merck, Novartis, Roche, and Sanofi; unrestricted grants from Celgene, EngMab, Sanofi, and Takeda; and consultancy for Celgene, Janssen, and Sanofi: Consultancy, Honoraria, Research Funding, Speakers Bureau. Díez-Campelo:Celgene Corporation: Consultancy, Membership on an entity's Board of Directors or advisory committees, Research Funding; Novartis: Consultancy, Membership on an entity's Board of Directors or advisory committees, Research Funding.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal