INTRODUCTION
The introduction of 20 years ago of imatinib, the first tyrosine kinase inhibitor (TKI), has drastically improved the survival of patients (pts) with Chronic Phase (CP) Chronic Myeloid Leukemia (CML), with a life expectancy similar to that of the general population. First used in patients after interferon (IFN) failure, imatinib and other TKIs have become standard frontline therapy. This analysis was done to investigate a 20-year outcome of Imatinib in pts Interferon-alpha failure.
METHODS
The long term outcome of 154 patients treated at our Institution with imatinib 400 mg after interferon failure from 1999-2000 was analyzed.
RESULTS
The median age was 44 (16 to 79yrs). 16 pts (11%) had prior malignancies 6 (38%) breast cancer, 3 (19%) skin cancer, 2 (12%) each prostate cancer and lymphoma, and 1 patient (6%) each lung cancer, colon cancer, and hairy cell leukemia. At the time of this analysis, 26 pts (17%) remained on imatinib of whom 22 pts (85%) had decreased their dosage. The median dose at last follow-up (FU) was 400mg (100-400mg). 128 pts (83%) discontinued imatinib permanently. Among them, 47 pts (37 %) changed to second generation TKIs, including 27 pts (58%) to dasatinib, 13 pts (28%) to nilotinib, and 7 (15%) to bosutinib.28 (60 %) received therapy with only 2 TKIs, and 19 (41%) received more than two TKIs. 81 pts (64%) did not receive a second TKI. Of these, 29 (36%) died, 23 (29%) were lost to follow up, 4 pts (12%) transformed to blast phase, 6 (8%) had stem cell transplant, 6 (8%) received farnesyltransferase inhibitor, 2 (3%) hyper-CVAD, 2 (3%) gemtuzumab ozogamicin, 1 (2%) homoharringtonine, 1(2%) with decitabine, and 7 (9%) had elective treatment discontinuation. All these 7 pts have remained treatment-free with a median follow up after discontinuation of 11.8 months. At baseline 13.6% had cardiac/vascular comorbidities and none had renal comorbidities. At 5-years, 8.4% had cardiac/vascular adverse events (AEs) and 1.2% had renal AEs. At last follow up, 13.4% had developed cardiac/vascular AEs and 3.8% renal AEs. 12 pts (8%) acquired second cancers after the start of imatinib. Of those, 2 pts each (16%) had esophageal, prostate, breast, and pancreatic cancers, and one each glioblastoma, basal cell cancer, lung cancer, and melanoma each had 1 patient (6%).
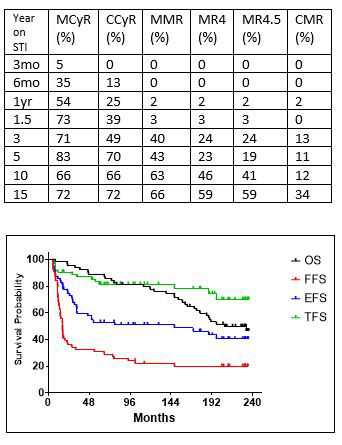
At the time of this report, intention to treat (ITT) responses are available for 63 patients. The median FU for these pts was 232 months, with a median overall survival (OS) of 51%. At three months, the ITT responses were: 35% for MCyR, 13% for CCyR. At 6 months they were 54% MCyR, 25% CCyR, and 2% each for MR4, MR4.5, and CMR (undetectable transcripts with ≥100,000 copies ABL). At one year they were 73% MCyR, 39% CCyR, and 3% for MMR, MR4, and MR4.5, respectively. At 18 months, responses were 67% MCyR, 38% CCyR, 5% for MMR, MR4, and MR4.5, respectively, and 2% for CMR. At three years, best responses included 71% MCyR, 49% CCyR, 40% MMR, 24% MR4 and MR4.5, and 13% CMR. At five years best responses were 83% MCyR, 70% CCyR, 43% MMR, 23% MR4, 19% MR4.5, and 11% CMR. At ten years, best responses were 66% for MCyR and CCyR, 63% for MMR, 46% for MR4, 41% for MR4.5, and 12% for CMR. Lastly, at fifteen years, best responses were 72% for MCyR and CCyR, 66% for MMR, and 59% for MR4 and MR4.5, and 34% for CMR. The overall best response rates (at any time) for these 63 pts was 87% for MCyR, 79% for CCyR, 62% for MMR, 51% for MR4 and MR4.5, and 33% for CMR (Table). The median duration of sustained MR4.5 for 21 pts was 139 months, and only 3 pts (15%) lost it while on therapy. The one year, five years, and nineteen years survival outcomes are: Overall Survival (OS) 98%, 89%, and 50%, respectively; Failure free survival (FFS) 68%, 31%, and 20%, respectively; Event Free Survival (EFS) 86%, 53%, and 40%, respectively. Treatment Free Survival (TFS), 90%, 81%, and 70%, respectively.
CONCLUSION
These results represent the longest follow-up of imatinib therapy available to date and demonstrate the long-term benefit of TKIs even in the setting of IFN failure.
Kantarjian:Ariad: Research Funding; Pfizer: Honoraria, Research Funding; Daiichi-Sankyo: Research Funding; Takeda: Honoraria; Amgen: Honoraria, Research Funding; Agios: Honoraria, Research Funding; Astex: Research Funding; BMS: Research Funding; Cyclacel: Research Funding; AbbVie: Honoraria, Research Funding; Immunogen: Research Funding; Actinium: Honoraria, Membership on an entity's Board of Directors or advisory committees; Novartis: Research Funding; Jazz Pharma: Research Funding. Borthakur:Incyte: Research Funding; Oncoceutics, Inc.: Research Funding; Bayer Healthcare AG: Research Funding; NKarta: Consultancy; AbbVie: Research Funding; AstraZeneca: Research Funding; BioTheryX: Membership on an entity's Board of Directors or advisory committees; BioLine Rx: Consultancy, Membership on an entity's Board of Directors or advisory committees, Research Funding; Cyclacel: Research Funding; Argenx: Membership on an entity's Board of Directors or advisory committees; Eli Lilly and Co.: Research Funding; Agensys: Research Funding; Merck: Research Funding; Cantargia AB: Research Funding; Cantargia AB: Research Funding; Merck: Research Funding; Oncoceutics: Research Funding; Novartis: Research Funding; Janssen: Research Funding; BMS: Research Funding; Tetralogic Pharmaceuticals: Research Funding; Argenx: Membership on an entity's Board of Directors or advisory committees; BioTheryX: Membership on an entity's Board of Directors or advisory committees; FTC Therapeutics: Membership on an entity's Board of Directors or advisory committees; Arvinas: Research Funding; GSK: Research Funding; Eisai: Research Funding; FTC Therapeutics: Membership on an entity's Board of Directors or advisory committees; Xbiotech USA: Research Funding; Polaris: Research Funding; Strategia Therapeutics: Research Funding; Arvinas: Research Funding; PTC Therapeutics: Consultancy. Verstovsek:Genetech: Research Funding; CTI BioPharma Corp: Research Funding; Promedior: Research Funding; Sierra Oncology: Research Funding; Ital Pharma: Research Funding; Pharma Essentia: Research Funding; Astrazeneca: Research Funding; Pragmatist: Consultancy; Protaganist Therapeutics: Research Funding; Incyte: Research Funding; Roche: Research Funding; NS Pharma: Research Funding; Celgene: Consultancy, Research Funding; Gilead: Research Funding; Constellation: Consultancy; Novartis: Consultancy, Research Funding; Blueprint Medicines Corp: Research Funding. Ravandi:Amgen: Honoraria, Membership on an entity's Board of Directors or advisory committees, Research Funding; Selvita: Research Funding; Menarini Ricerche: Research Funding; Cyclacel LTD: Research Funding; Macrogenix: Consultancy, Research Funding; Xencor: Consultancy, Research Funding. Cortes:Pfizer: Consultancy, Honoraria, Research Funding; Bristol-Myers Squibb: Consultancy, Research Funding; Jazz Pharmaceuticals: Consultancy, Research Funding; Takeda: Consultancy, Research Funding; Astellas Pharma: Consultancy, Honoraria, Research Funding; BiolineRx: Consultancy; Sun Pharma: Research Funding; Novartis: Consultancy, Honoraria, Research Funding; Daiichi Sankyo: Consultancy, Honoraria, Research Funding; Immunogen: Consultancy, Honoraria, Research Funding; Merus: Consultancy, Honoraria, Research Funding; Forma Therapeutics: Consultancy, Honoraria, Research Funding; Biopath Holdings: Consultancy, Honoraria. Kadia:Amgen: Membership on an entity's Board of Directors or advisory committees, Research Funding; Bioline RX: Research Funding; Celgene: Research Funding; Jazz: Membership on an entity's Board of Directors or advisory committees, Research Funding; Pfizer: Membership on an entity's Board of Directors or advisory committees, Research Funding; Genentech: Membership on an entity's Board of Directors or advisory committees; Pharmacyclics: Membership on an entity's Board of Directors or advisory committees; Takeda: Membership on an entity's Board of Directors or advisory committees; BMS: Research Funding; AbbVie: Consultancy, Research Funding. Wierda:Cyclcel: Research Funding; Sunesis: Research Funding; Xencor: Research Funding; Janssen: Research Funding; Oncternal Therapeutics Inc.: Research Funding; KITE pharma: Research Funding; Juno Therapeutics: Research Funding; Miragen: Research Funding; Loxo Oncology Inc.: Research Funding; GSK/Novartis: Research Funding; Gilead Sciences: Research Funding; Acerta Pharma Inc: Research Funding; Pharmacyclics LLC: Research Funding; Genentech: Research Funding; AbbVie: Research Funding. Garcia-Manero:Amphivena: Consultancy, Research Funding; Helsinn: Research Funding; Novartis: Research Funding; AbbVie: Research Funding; Celgene: Consultancy, Research Funding; Astex: Consultancy, Research Funding; Onconova: Research Funding; H3 Biomedicine: Research Funding; Merck: Research Funding. Jabbour:Takeda: Consultancy, Research Funding; BMS: Consultancy, Research Funding; Adaptive: Consultancy, Research Funding; Amgen: Consultancy, Research Funding; AbbVie: Consultancy, Research Funding; Pfizer: Consultancy, Research Funding; Cyclacel LTD: Research Funding.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal