Background: Somatic mutations in isocitrate dehydrogenase 1 (IDH1) are reported in 6-10% of patients (pts) with acute myeloid leukemia (AML). Ivosidenib (IVO; AG-120) is an oral, potent, targeted inhibitor of mutant IDH1 (mIDH1) approved for treatment of mIDH1 relapsed/refractory AML, and newly diagnosed (ND) AML ineligible for intensive chemotherapy (IC). Here we report the genetic mutation and multiparameter flow analyses on longitudinal samples collected from pts receiving IVO + azacitidine (AZA) in the phase 1b portion of the ongoing phase 1b/2 study of mIDH1/2 inhibitors + AZA in pts with IC-ineligible ND AML (NCT02677922).
Methods: Pts received oral IVO 500 mg daily continuously and subcutaneous AZA 75 mg/m2 on Days 1-7 in 28-day cycles. The secondary efficacy endpoint of complete remission (CR) plus CR with partial hematologic recovery (CR+CRh) rate was sponsor derived, and CRh was defined as CR with absolute neutrophil count >0.5 × 109/L and platelets >50 × 109/L. Genomic DNA from baseline and longitudinal bone marrow (BMMCs) and peripheral blood mononuclear cells (PBMCs) were used for molecular studies. Co-occurring mutation profiling of 20/23 (14 CR/CRh) pts by targeted next-generation sequencing (NGS) using the ACE Extended Cancer Panel was performed, with 500× average target coverage for the full coding region of 1400 genes (detection limit 2%). mIDH1 variant allele frequency (VAF) for 23/23 (16 CR/CRh) pts was also tested by a highly sensitive BEAMing digital PCR assay (detection limit 0.02-0.04%). Multiparameter flow cytometry was conducted centrally on bone marrow aspirate from 14/23 (12 CR/CRh) pts (detection limit 1%). Fisher's exact test (two-sided) was applied for statistical analysis.
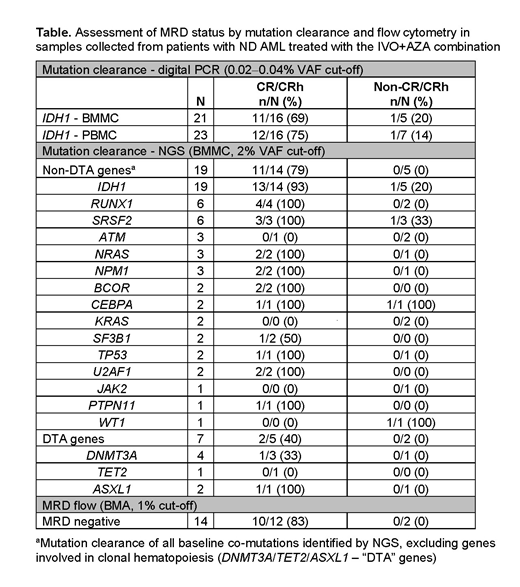
Results: As of February 19, 2019, 23 pts received IVO+AZA (11 male; median age 76 years [range 61-88]). Median duration of treatment was 15.1 months (range 0.3-32.2); 10 pts remained on treatment as of the data cutoff. Overall response rate was 78% (18/23 pts): CR 61% (14/23), CR with incomplete hematologic or platelet recovery 9% (2/23), and morphologic leukemia-free state 9% (2/23). CR+CRh rate was 70% (16/23). The top 3 most frequently co-mutated genes at study entry were RUNX1 (7/20 pts, 35%), SRSF2 (7/20, 35%), and DNMT3A (4/20, 20%). Given the small sample size, no gene or pathway identified at baseline was statistically associated with clinical response or resistance. Interestingly, CR/CRh was achieved in ND AML pt populations who typically have a poor prognosis, or did not achieve a CR/CRh response to single-agent IVO therapy: 3/3 pts with poor risk karyotypes (local cytogenetics), 1/2 harboring TP53 mutations, and 3/5 with RTK pathway (KRAS, NRAS, PTPN11) mutations. Longitudinal mutation clearance (MC) of mIDH1 and the most frequent baseline co-mutations in CR/CRh and non-CR/CRh pts are summarized in the Table. IDH1-MC in BMMCs was observed in 13/14 (93%) CR/CRh pts by NGS and in 11/16 (69%) by digital PCR. Utilizing the 2-log more sensitive digital PCR assay specific to mIDH1, there was strong concordance in the mIDH1 VAF observed in BMMCs and PBMCs (Pearson correlation coefficient [r]=0.919) with 12/16 (75%) CR/CRh pts achieving MC in PBMCs, and 11/12 (92%) achieving MC in both BMMCs and PBMCs. These IDH1-MC rates are higher than those previously observed in IC-ineligible ND AML pts treated with single agent IVO (Roboz et al. ASCO 2019; NCT02074839). Similarly, in CR/CRh pts with available baseline co-mutation data by NGS, all mutations were cleared in 11/14 (79%) pts, apart from mutations in the "DTA" (DNMT3A/TET2/ASXL1) genes typically associated with clonal hematopoiesis. In contrast, mutations in the "DTA" genes were cleared in 2/5 (40%) CR/CRh pts. Orthogonal evaluation of the depth of these remissions by flow cytometry found that 10/12 (83%) CR/CRh pts achieved measurable residual disease (MRD) negativity.
Conclusion: Combination of IVO+AZA in IC-ineligible ND AML leads to a high rate of clinical response with molecular remissions. The strong association between MC, clinical response, and flow cytometry MRD in this phase 1b study warrants further investigation of single gene mIDH1 VAF as a biomarker for monitoring response in pts with mIDH1 AML treated with IVO+AZA. Furthermore, the high concordance of mIDH1 VAF between BMMCs and PBMCs indicates that peripheral blood could be a surrogate tissue for monitoring mIDH1 VAF in these pts.
Daigle:Agios: Employment, Equity Ownership. Choe:Agios: Employment, Equity Ownership; Agios: Employment, Equity Ownership. Quek:Celgene: Research Funding, Speakers Bureau; Agios: Research Funding. DiNardo:jazz: Honoraria; syros: Honoraria; celgene: Consultancy, Honoraria; notable labs: Membership on an entity's Board of Directors or advisory committees; agios: Consultancy, Honoraria; medimmune: Honoraria; daiichi sankyo: Honoraria; abbvie: Consultancy, Honoraria. Stein:Stemline: Speakers Bureau; Amgen: Consultancy, Speakers Bureau; Celgene: Speakers Bureau. Stein:Daiichi Sankyo, Inc.: Membership on an entity's Board of Directors or advisory committees; Bioline: Membership on an entity's Board of Directors or advisory committees; Genentech: Membership on an entity's Board of Directors or advisory committees; Celgene Corporation: Membership on an entity's Board of Directors or advisory committees; Agios: Consultancy, Membership on an entity's Board of Directors or advisory committees; Astellas Pharma US, Inc: Membership on an entity's Board of Directors or advisory committees; Novartis: Membership on an entity's Board of Directors or advisory committees; PTC Therapeutics: Membership on an entity's Board of Directors or advisory committees; Syros: Membership on an entity's Board of Directors or advisory committees. Fathi:Agios, Astellas, Celgene, Daiichi Sankyo, Novartis, Takeda, Amphivena, Kite, Forty Seven,Trovagene, NewLink genetics, Jazz, Abbvie, and PTC Therapeutics: Consultancy; Amphivena, Kite, Jazz, NewLink Genetics,: Honoraria. Schuh:Jazz: Honoraria, Membership on an entity's Board of Directors or advisory committees; Pfizer: Honoraria, Membership on an entity's Board of Directors or advisory committees; Amgen: Honoraria, Membership on an entity's Board of Directors or advisory committees; Teva Canada Innovation: Honoraria, Membership on an entity's Board of Directors or advisory committees; Astellas: Honoraria, Membership on an entity's Board of Directors or advisory committees; Celgene: Honoraria, Membership on an entity's Board of Directors or advisory committees; Agios: Honoraria; AbbVie: Honoraria, Membership on an entity's Board of Directors or advisory committees. Kantarjian:Agios: Honoraria, Research Funding; Immunogen: Research Funding; Pfizer: Honoraria, Research Funding; Jazz Pharma: Research Funding; Daiichi-Sankyo: Research Funding; Cyclacel: Research Funding; BMS: Research Funding; Novartis: Research Funding; Amgen: Honoraria, Research Funding; AbbVie: Honoraria, Research Funding; Astex: Research Funding; Actinium: Honoraria, Membership on an entity's Board of Directors or advisory committees; Takeda: Honoraria; Ariad: Research Funding. Zhang:Agios: Employment, Equity Ownership; Agios: Employment, Equity Ownership. Winkler:Agios: Employment. Vyas:Celgene: Research Funding, Speakers Bureau; Novartis: Research Funding, Speakers Bureau; Pfizer: Speakers Bureau; Daiichi Sankyo: Speakers Bureau; Forty Seven, Inc.: Research Funding; Abbvie: Speakers Bureau; Astellas: Speakers Bureau. Wu:Agios: Employment, Equity Ownership.
Ivosidenib is an IDH1 inhibitor indicated for the treatment of AML with a susceptible IDH1 mutation as detected by an FDA-approved test in: 1) adult patients with newly-diagnosed AML who are more than 75 years old or who have comorbidities that preclude use of intensive induction chemotherapy; 2) Adult patients with relapsed or refractory AML. It is being investigated in clinical trials in combination with azacitidine in patients with IDH1-mutant newly diagnosed AML.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal