Recent clinical trials have reported that in combination with hypomethylating agents, the BCL-2 inhibitor venetoclax can induce responses in over 70% of older previously untreated AML patients who are unfit for conventional chemotherapy. These findings led to the recent United States Food and Drug Administration approval of this regimen for this population, and it is now considered to be the standard care. However, a significant minority of patients do not achieve a remission and are refractory. In addition, the majority of patients who do achieve a remission ultimately relapse. It is therefore critical to identify AML patients who are likely to be resistant to venetoclax-based therapy.
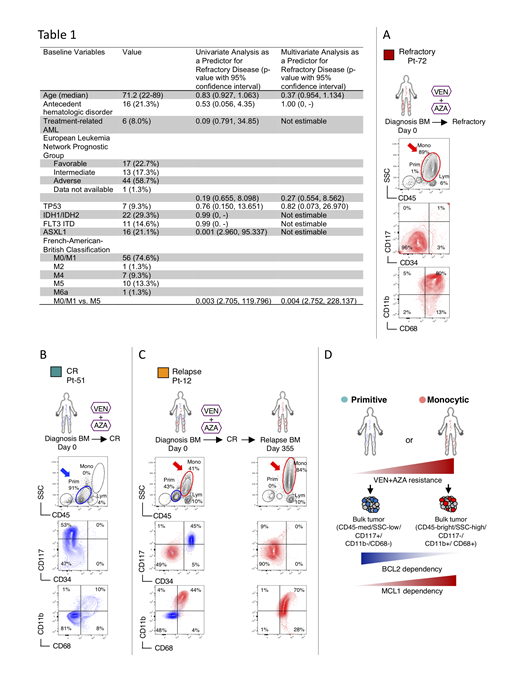
To initially address this question, we retrospectively reviewed 75 newly diagnosed AML patients who received venetoclax + azacitidine (VEN+AZA) at our institution and analyzed several baseline clinical features to determine the ability of each to predict disease that was refractory to treatment (defined as a lack of complete remission [CR], CR with incomplete recovery of peripheral blood counts [CRi], partial remission, or morphological leukemia free state [MLFS]). Both univariate and multivariate analyses revealed the presence of FAB-M5 to be associated with disease that was refractory to VEN+AZA (Table 1). Given that FAB-M5 represents AML with monocytic differentiation, these findings indicate a strong correlation between myeloid differentiation status and resistance to venetoclax.
Using multicolor flow cytometry, we show bone marrow specimens of typical FAB-M5 patients who were refractory to VEN+AZA presented dominant monocytic disease that has an immunophenotype of CD45-bright/SSC-high/CD117-/CD11b+/CD68+ (Figure A). In contrast, bone marrow specimens of typical FAB-M0/M1/M2 patients who achieved CR with VEN+AZA presented as a single dominant disease population that is CD45-med/SSC-low/CD117+/CD11b-/CD68- (Figure B). In a subset of AML patients, we observed the co-existence of both phenotypically primitive and monocytic populations, which we term "MPM" AML (for Mixed Primitive/Monocytic). We observe that after attaining CR with VEN+AZA treatment and subsequent relapse, MPM-AML showed almost complete loss of the primitive subpopulation, and evolved to a dominant monocytic disease (Figure C). These data indicate that VEN+AZA treatment induces strong selection of the monocytic phenotype. Importantly, when we compared the immunophenotype of six pairs of diagnostic/relapse specimens from AML patients treated with conventional intensive induction chemotherapy, we observed selection of a more primitive phenotype, suggesting the drive toward a monocytic phenotype observed at relapse appears to be a unique consequence of VEN+AZA therapy. To our knowledge, selection of a monocytic phenotype at relapse has never been previously observed in AML, suggesting the relapse after VEN+AZA may represent a new clinical entity.
Mechanistically, using RNA-seq we show the global transcriptome of monocytic AMLs are distinct from primitive AMLs, suggesting they represent two broad classes of AML with likely differential responses to therapy. Indeed, we demonstrate that AML with a primitive immunophenotype is dependent on BCL-2 activity as a means to drive oxidative phosphorylation, a critical requirement for survival of leukemia stem cells. Conversely, AML with a more differentiated monocytic phenotype is no longer dependent on BCL-2, but rather switches to MCL-1 as a mediator of oxidative phosphorylation. Using colony-forming and xenograft assays, we show the stem and progenitor potential of monocytic AMLs are selectively more sensitive to MCL-1 inhibition comparing to BCL-2 inhibition.
Together, our study suggests a significantly higher refractory/relapse risk for monocytic AML patients treated with VEN+AZA (Figure D). Further, for those AML patients who do respond to initial VEN+AZA treatment, the therapy drives a powerful selective process resulting in emergence of more differentiated monocytic disease in some patients. Based on these findings, we propose that AML exists on a developmental spectrum that is inherently fluid, where with appropriate selective pressure the disease can acquire characteristics of a more differentiated state. Further, our data indicate that optimal AML therapy will require strategies designed to target both primitive and myeloid phenotypes.
Pollyea:Forty-Seven: Consultancy, Membership on an entity's Board of Directors or advisory committees; Janssen: Consultancy, Membership on an entity's Board of Directors or advisory committees; Astellas: Consultancy, Membership on an entity's Board of Directors or advisory committees; Abbvie: Consultancy, Membership on an entity's Board of Directors or advisory committees, Research Funding; Celyad: Consultancy, Membership on an entity's Board of Directors or advisory committees; Agios: Consultancy, Membership on an entity's Board of Directors or advisory committees; Celgene: Consultancy, Membership on an entity's Board of Directors or advisory committees; Diachii Sankyo: Consultancy, Membership on an entity's Board of Directors or advisory committees; Gilead: Consultancy, Membership on an entity's Board of Directors or advisory committees; Takeda: Consultancy, Membership on an entity's Board of Directors or advisory committees; Pfizer: Consultancy, Membership on an entity's Board of Directors or advisory committees, Research Funding. Savona:TG Therapeutics: Membership on an entity's Board of Directors or advisory committees, Research Funding; Takeda: Membership on an entity's Board of Directors or advisory committees, Research Funding; Selvita: Membership on an entity's Board of Directors or advisory committees; Karyopharm Therapeutics: Consultancy, Equity Ownership, Membership on an entity's Board of Directors or advisory committees; Incyte Corporation: Membership on an entity's Board of Directors or advisory committees, Research Funding; Sunesis: Research Funding; Celgene Corporation: Membership on an entity's Board of Directors or advisory committees; Boehringer Ingelheim: Patents & Royalties; AbbVie: Membership on an entity's Board of Directors or advisory committees.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal