Abstract
Background: CLL patients (pts) treated with novel targeted therapies continue to experience disease progression. Approximately 7-15% of relapsed CLL treated with ibrutinib or venetoclax developed Richter's syndrome (RS), an aggressive high grade lymphoma transformation. Standard chemotherapy has limited efficacy in RS. We hypothesized that checkpoint blockade would re-establish anti-tumor immune response in progressive CLL/RS and here we aim to update the clinical data and correlative analysis of MC1485: a phase 2 study of PD-1 blocking antibody pembrolizumab in relapsed/refractory CLL and RS.
Methods: Relapsed/refractory CLL including RS enrolled in the CLL arm of MC1485 trial was reported here. The primary end point of this study is overall response rate (ORR). Pts with prior allogeneic stem cell transplant were excluded. Pembrolizumab 200 mg was administered intravenously every 3 weeks. NCI CTCAE v4.0 and IWCLL 2008 criteria were used for non-hematological adverse events (AE) and for CLL grade hematological AE. Tumor expression of PD-1 and PD-L1 were assessed with standard immunohistochemical (IHC) staining. Percentage expressions of individual antigens were analyzed with whole-slide scanning followed by image analysis with Image-Pro software (Media Cybernetics). Fluorescence in situ hybridization (FISH) was performed on available CLL/RS tissue sections to assess copy number of chromosome 9p region that contains PD-L1 and PD-L2.
Results: 25 pts including 16 CLL and 9 RS (biopsy-proven large cell lymphoma) were enrolled in CLL arm. The median age was 69 years (46-81). Twelve (48%) pts had del(17p) or monosomy 17 or TP53 mutation. The median number of prior therapies was 4 (1-10). 96%, 72% or 48% of pts had received alkylator and anti-CD20 antibody, purine analog or anthracycline. Fifteen (60%) pts had prior ibrutinib and 12 (48%) progressed clinically while receiving ibrutinib, including 6 who progressed to RS and 6 who developed progressive CLL. The median number of pembrolizumab doses that pts received was 3 (1 to 21), administered over a median treatment duration of 11 weeks (1 to 56). Drug-related AE occurred in 21 pts (88%) with the most common ones being neutropenia in 9 (37%), cough in 7 (29%) and dyspnea in 6 (25%) pts. Drug-related grade 3 or above AE occurred in 9 (38%) pts with most common ones being thrombocytopenia (5, 21%), dyspnea (2, 8%), and fatigue (2, 8%). The most common immune-related AE was liver enzyme elevation (12%, 8% G3) and was reversible with therapy interruption or steroid therapy.
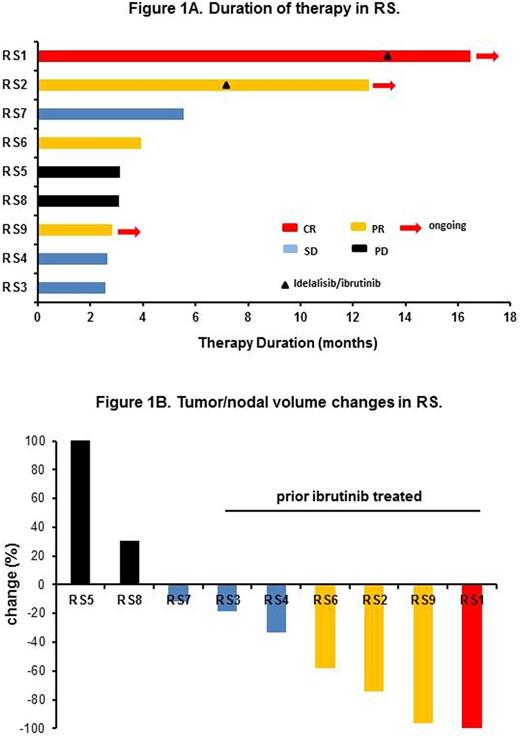
Based on investigator assessment using the Revised Response Criteria for Lymphomas and IWCLL 2008 criteria, one RS pt had complete response (CR, 4%), 3 RS had partial responses (PR, 12%). Among 9 RS pts, 1 CR (11%), 3 PR (33%), 3 SD (33%) and 2 PD (22%) were observed. Thus the ORR in RS was 44%. No CLL pts achieved a CR/PR, 3 had SD and 9 had progressive disease (PD). The ORR of all pts was 16%. Of the 4 RS who had CR/PR, responses occurred early after 2 cycles of therapy and median duration of therapy for CR/PR pts was 8.3 months with data cut-off by June 30th 2016 (Figure 1A). The causes for therapy discontinuation in RS were PD (3, 33%), alternate therapy (2, 22%), and thrombocytopenia due to increased marrow CLL (1, 11%). Increased marrow CLL was observed in 2 RS pts with PR to RS phase of disease assessed by PET, thus the protocol was amended to allow addition of idelalisib/ibrutinib to control the underlying marrow CLL. For RS pts who had a CR/PR or SD, variable nodal responses were observed (Figure 1B). In particular, 6 RS who had prior ibrutinib experienced CR or PR or SD with nodal responses.
After a median follow-up of 10.2 (1.9 to 16.1) months, the median overall survival (OS) for all pts was 10.7 months (95% confidence interval [CI], 5.4 to not reached). The 6 month OS rate for RS and CLL pts were 73% and 59%, respectively. Biomarker assessment using IHC analysis on 10 pts with available tumor/nodal tissues (6 RS and 4 CLL) showed an increased expression of PD-L1 (p = 0.02) and a trend for an increased expression of PD-1 (p = 0.1) in the group of pts with CR/ PR versus pts in the group with no clinical response. One out of 10 tested showed polysomic for chromosome 9p.
Conclusion: Pembrolizumab had an acceptable safety profile in CLL and RS patients. We confirmed that pembrolizumab has substantial therapeutic activity in RS. Single-agent pembrolizumab does not appear to have clear activity in CLL.
Ding:Merck: Research Funding. Parikh:Pharmacyclics: Honoraria, Research Funding. Shanafelt:GlaxoSmithKline: Research Funding; Genentech: Research Funding; Janssen: Research Funding; Pharmacyclics: Research Funding; Celgene: Research Funding; Cephalon: Research Funding; Hospira: Research Funding.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal