Abstract
Background: Allogeneic HSCT from either an HLA-identical sibling or an unrelated donor is a potentially curative treatment for patients with hemoglobinopathies and erythroid disorders (ED), such as Thalassemia Major (TM), Sickle Cell Disease (SCD) and Diamond-Blackfan Anemia (DBA). The limited historical experience with HLA-haploidentical HSCT in this setting has reported a disease-free survival probability lower than that reported using HLA-matched donors. In the last few years, we have developed a novel method of graft manipulation, based on the selective depletion of α/β+ T-cells and CD19+ B-cells (ClinicalTrial.gov identifier: NCT01810120), which was shown to be safe and effective in children with multiple types of non-malignant disorders (Bertaina el al, Blood 2014). To further optimize this approach through the acceleration of the recovery of adaptive immunity, we designed an ongoing phase I/II trial aimed to test the safety and efficacy of post-transplant infusion of donor T-cells transduced with the iC9 suicide gene (BPX-501 cells) in children with either malignant or non-malignant disorders (ClinicalTrials.gov identifier: NCT02065869). As the transduced gene contains sequences for the CD19 marker, BPX-501 cells (CD3+/CD19+) can be easily tracked in peripheral blood. We report on 10 children with hemoglobinopathies and ED who were enrolled in the phase II portion of the study.
Patients and methods: Five patients were males and 5 were females, and median age at diagnosis and at HSCT was 5.34 and 9.52 years (range 2.33-11.71), respectively. Seven patients had TM (all bo/bo), 2 DBA and 1 SCD. All 10 patients were transfusion-dependent and receiving iron-chelation therapy before haplo-HSCT. Among the thalassemia patients, 4 patients belonged to class I and 3 to class II of the Pesaro classification. All patients were transplanted from a parent. Median number of CD34+ and αβ+ T-cells infused was 22.5 x 106/kg and 0.3 x 105/kg, respectively. In all patients, conditioning regimen included busulfan (16 mg/Kg), thiotepa (10 mg/Kg) and fludarabine (160 mg/m2). Rabbit ATG (12 mg/Kg over 3 days, from day -4 to day -2) was administered to prevent graft-versus-host disease (GvHD) and graft failure and Rituximab (200 mg/ m2 on day -1) was administered to prevent EBV-related lymphoproliferative disorders. No post-transplantation GvHD prophylaxis was given. Median follow-up is 301 days (range 41-420 days). Basic phenotyping of circulating lymphocytes was assessed by flow cytometry on fresh heparinized peripheral blood samples at 10, 20, 30, 60, 90, 120 and 150 days post haplo-HSCT.
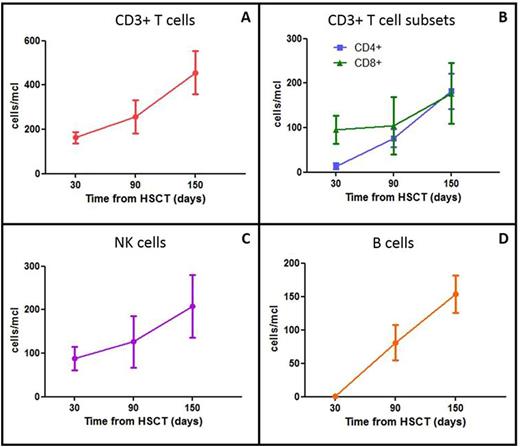
Results: After haplo-HSCT, the median time to reach neutrophil and platelet recovery was 14 days (range 11-28) and 10 days (range 8-12), respectively. After engraftment of the allograft, BPX-501 cells were infused (dose: 1x106 cells/kg) at a median time of 13.5 days after HSCT (range 10-26). Nine of the 10 patients maintained sustained donor engraftment, reaching full chimerism. The patient who experienced secondary graft failure was successfully re-transplanted from the same parent and he is full donor chimeric with transfusion-independence. Grade I/II skin acute GvHD occurred in 2 patients (at 31 and 59 days after HSCT, respectively). There was no occurrence of chronic GVHD. Remarkably, no patient has died and none of the patients have been re-hospitalized after initial discharge. The last erythrocyte transfusion was administered on day +7 post-transplant (range 4-33 days). At last follow-up, the median hemoglobin value of these patients was 11.35 gr/dL (range 10.2-13.4). BPX-501 cells expanded after infusion and still persist in all patients at last follow-up. All children are alive and transfusion-independent. Details on T cell, NK cell and B cell recovery are shown in Figure 1 (Panel A-D).
Conclusions: Children with hemoglobinopathies and DBA can benefit from curative haplo-HSCT after depletion of α/β T-cells followed by infusion of BPX-501 cells, which, expanding and persisting over time, contribute to speed immune recovery of adaptive T-cell immunity, thus rendering the procedure safer.
French:Bellicum Pharmaceuticals: Employment, Membership on an entity's Board of Directors or advisory committees. Moseley:Bellicum Pharmaceuticals: Employment, Membership on an entity's Board of Directors or advisory committees.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal