Abstract
Molecularly targeted therapy frequently induces remissions in cancer, but rarely achieves complete disease eradication, with resulting risk of disease relapse/progression. Chronic myeloid leukemia (CML) is a good example, with rare, propagating stem cells (SCs) that are incompletely eradicated by BCR-ABL directed tyrosine kinase inhibitor (TKI) therapy in most cases. Consequently, CML relapse usually occurs following treatment discontinuation. Further, some patients fail to achieve a satisfactory response to TKI therapy and are at risk of disease progression. Ultimately, the depth of response to TKIs is dictated by CML-SCs, however, it has proven challenging to characterise this crucial population of cells as they reside in the same compartment as their normal hematopoietic stem cell (HSC) counterparts from which they cannot be reliably distinguished throughout the disease course.
Advances in single cell transcriptomics are opening up unprecedented opportunities to unravel heterogeneity in cell populations. However, to date, this technology has primarily been used to analyse normal tissues, partly reflecting the lack of sensitivity for detection of somatic mutations using current techniques. Herein, we developed a novel protocol allowing targeted amplification of BCR-ABL during single cell cDNA library generation using a modification of a published protocol (PMID:24385147). We then applied this method to analyse 1082 bone marrow SCs (defined as Lin-CD34+CD38-) from a cohort of 10 patients with CML and 3 normal controls.
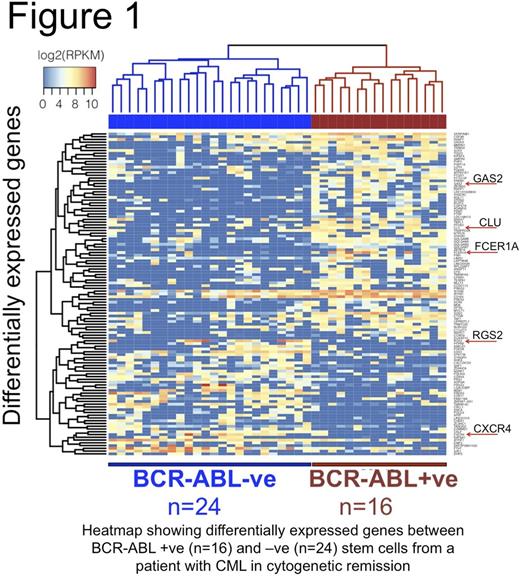
We first validated our method using the BCR-ABL +ve K562 cell line. Surprisingly, using standard methods, BCR-ABL was only detected in a small minority of cells. With our modified technique, we demonstrate robust detection of BCR-ABL in 100% of single K562 cells with parallel whole transcriptome amplification from the same cell. Further, using a plasmid Òspike-inÓ, we demonstrate sensitivity to detect a single molecule of BCR-ABL per cell, without false +ve BCR-ABL amplification in 142 cells tested. We then analysed 40 SCs from a patient with CML who was in a cytogenetic remission (CyR) following 3 months of TKI therapy. BCR-ABL was detected in 16 cells and was absent in 24 cells. With an average of 3.3 million mapped reads per single cell, we detected over 4600 genes per cell with good correlation of single cell ensemble data with bulk analysis. Using this approach we were uniquely able to compare BCR-ABL +ve and -ve cells and identify a large number of differentially expressed genes, which would not have been detected through bulk analysis. We selected a number of genes of biologic interest for validation by Q-PCR and 80% of them were confirmed as differentially expressed, a number of these are highlighted in the heatmap (see Figure).
We then analysed 717 single SCs from 8 CML diagnostic samples. This analysis identified marked heterogeneity of BCR-ABL expression with frequencies comparable to the published literature (9% to 92%). Unsupervised clustering analysis revealed many genes that were differentially expressed between BCR-ABL +ve and -ve SCs e.g. CML-SCs showed upregulation of cell cycle signatures and decreased expression of HSC-affiliated genes. We next analysed 223 single SCs from a series of 5 patients in CyR following TKI initiation. BCR-ABL positive SCs could still be detected, albeit at a reduced frequency (0.5% to 45%). In contrast to the diagnostic samples, CML-SCs showed quiescence and HSC signatures that were comparable to normal HSCs. Furthermore, through analysis of serial samples from diagnosis to remission from the same patient, this single-cell approach uniquely allowed analysis of gene expression specifically of the CML-SCs which evade TKI therapy, facilitating the identification of a number of candidate gene-sets including cell surface markers and potentially ÒdruggableÓ targets. Finally, through analysis of a patient with early blast crisis transformation of CML during TKI therapy, we illustrate how single cell RNA sequencing might be applied to predict such early disease progression events.
We herein describe a new approach for single cell RNA sequencing of CML-SCs that might be applied to fate-map persistent CML-SCs during and following treatment for discovery research and also to refine precision medicine in CML. This approach could be applied across a range of clonal disorders with potential broader relevance for cancer research.
No relevant conflicts of interest to declare.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal