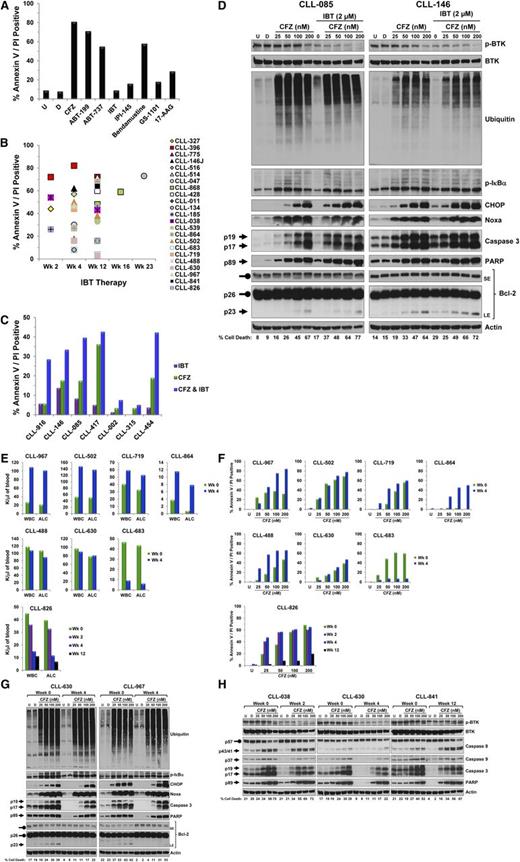
To the editor:
Ibrutinib, an orally bioavailable Bruton tyrosine kinase (BTK) inhibitor,1 has shown significant clinical benefit for chronic lymphocytic leukemia (CLL) patients.2,3 However, ibrutinib monotherapy often leads to partial responses and extended lymphocytosis,4 which led us to question whether combination therapy could provide complete clinical responses. To identify pharmacologic agents that complement ibrutinib therapy, we performed an ex vivo drug screen using targeted agents on CLL cells isolated from ibrutinib-treated patients (Table 1). We identified carfilzomib (PR-171), a second-generation proteasome inhibitor5 and ABT-1996 (a Bcl-2 antagonist) as the most cytotoxic agents, as indicated by increased annexin V/propidium iodide (PI) double positivity after 24-hour incubation (Figure 1A7 ). Results were consistent in samples from 7 additional patients (data not shown). Carfilzomib cytotoxic effect after 24-hour treatment was confirmed in samples from 23 patients treated with ibrutinib for 2 to 23 weeks (Figure 1B).
Patient characteristics
| Pt . | Sex . | Age, y . | RAI stage . | No. of prior therapy . | B2M . | ATM* . | p53* . | ZAP70 IHC . | IgVH gene . | Ibrutinib treatment . | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| WBC . | ALC . | ||||||||||||||||||
| W0 . | W4 . | W12 . | W16 . | W20 . | W0 . | W4 . | W12 . | W16 . | W20 . | ||||||||||
| 327 | F | 72 | 1 | 0 | 3.3 | 0 | 0 | NEG | MUT | 129 | 136 | 81 | 71 | 36 | 114 | 128 | 69 | 63 | 33 |
| 396 | M | 71 | 4 | 0 | 3.7 | 1 | 0 | POS | UNMUT | 12 | 97 | 83 | 39 | 40 | 5 | 81 | 78 | 34 | 32 |
| 775 | F | 74 | 1 | 2 | 2.3 | 0 | 88 | POS | ND | 167 | 30 | 15 | 10 | 12 | 158 | 24 | 13 | 8 | 9 |
| 146J | F | 55 | 4 | 1 | 2.5 | 0 | 38 | NEG | UNMUT | 97 | 117 | 53 | 56 | 28 | 88 | 107 | 49 | 53 | 24 |
| 516 | F | 72 | 3 | 3 | 8.2 | 0 | 0 | NEG | MUT | 7 | 18 | 13 | 14 | 14 | 2 | 14 | 11 | 12 | 11 |
| 514 | F | 68 | 1 | 0 | ND | 0 | 0 | NEG | MUT | 123 | ND | 145 | 120 | 99 | 115 | ND | 138 | 118 | 85 |
| 047 | M | 72 | 1 | 1 | 3.8 | 0 | 13 | POS | UNMUT | 4 | 14 | 5 | 7 | 6 | 1 | 13 | 3 | 4 | 3 |
| 868 | M | 61 | 4 | 1 | 3.3 | 0 | 96 | POS | UNMUT | 365 | 265 | 55 | 25 | 16 | 339 | 233 | 44 | 20 | 9 |
| 428 | F | 55 | 0 | 1 | ND | 0 | 68 | POS | UNMUT | 6 | 16 | 13 | 10 | 8 | 2 | 12 | 13 | 4 | 6 |
| 011 | F | 59 | 1 | 3 | 3.0 | 0 | 54 | ND | UNMUT | 43 | 39 | 18 | 13 | 10 | 38 | 35 | 17 | 11 | 6 |
| 134 | M | 50 | 1 | 1 | ND | 0 | 0 | ND | UNMUT | 49 | 203 | 117 | 70 | 55 | 41 | 183 | 109 | 67 | 49 |
| 841 | F | 61 | 3 | 2 | 7.9 | 0 | 25 | POS | UNMUT | 187 | 256 | 259 | 240 | 212 | 178 | 217 | 259 | 235 | 208 |
| 185 | M | 72 | 4 | 1 | 9.8 | 93 | 0 | NEG | UNMUT | 71 | 51 | 23 | 16 | 14 | 65 | 48 | 21 | 14 | 9 |
| 038 | M | 51 | 0 | 1 | 2.2 | 37 | 0 | POS | UNMUT | 117 | 60 | 28 | 14 | 13 | 107 | 55 | 26 | 12 | 9 |
| 539 | M | 68 | 2 | 4 | 6.4 | 61 | 0 | POS | UNMUT | 7 | 183 | 128 | 130 | 120 | 6 | 171 | 118 | 117 | 111 |
| 502 | M | 79 | 4 | 1 | 6.6 | 93 | 0 | POS | UNMUT | 53 | 130 | 71 | 55 | 38 | 51 | 126 | 66 | 50 | 32 |
| 826 | M | 68 | 2 | 0 | 2.0 | 0 | 0 | NEG | UNMUT | 45 | 15 | 11 | 10 | 8 | 40 | 12 | 7 | 5 | 4 |
| 967 | M | 53 | 1 | 0 | 1.9 | 0 | 58 | POS | UNMUT | 27 | 106 | 95 | 74 | 84 | 21 | 98 | 89 | 59 | 77 |
| 630 | F | 72 | 3 | 2 | 3.3 | 0 | 92 | NEG | UNMUT | 97 | 90 | 84 | 71 | 56 | 79 | 81 | 79 | 65 | 54 |
| 488 | M | 55 | 0 | 6 | 4.2 | 22 | 24 | ND | UNMUT | 118 | 108 | 67 | 47 | 25 | 108 | 90 | 50 | 43 | 21 |
| 719 | M | 65 | 4 | 1 | 2.5 | 65 | 0 | NEG | UNMUT | 41 | 54 | 43 | 30 | 27 | 33 | 50 | 37 | 24 | 22 |
| 864 | M | 65 | 1 | 1 | 1.3 | 7 | 0 | POS | MUT | 4 | 12 | 23 | 12 | 9 | 1 | 8 | 18 | 9 | 4 |
| 683 | M | 68 | 2 | 1 | 3.2 | 0 | 0 | POS | UNMUT | 47 | 9 | 4 | 5 | 3 | 44 | 6 | 2 | 3 | 2 |
| 916 | F | 60 | 1 | 0 | 2.4 | 0 | 0 | NEG | MUT | ||||||||||
| 146 | M | 72 | 1 | 0 | 4.9 | 34 | 0 | NEG | ND | ||||||||||
| 085 | F | 64 | 4 | 6 | 9.3 | 0 | 0 | NEG | MUT | ||||||||||
| 417 | M | 41 | 2 | 0 | 2.5 | 0 | 0 | NEG | MUT | ||||||||||
| 002 | F | 67 | 1 | 0 | 1.6 | 19 | 0 | POS | ND | ||||||||||
| 315 | M | 59 | 3 | 0 | 6.5 | 0 | 91 | NEG | UNMUT | ||||||||||
| 454 | M | 57 | 1 | 0 | 6.1 | 38 | 0 | POS | UNMUT | ||||||||||
| Pt . | Sex . | Age, y . | RAI stage . | No. of prior therapy . | B2M . | ATM* . | p53* . | ZAP70 IHC . | IgVH gene . | Ibrutinib treatment . | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| WBC . | ALC . | ||||||||||||||||||
| W0 . | W4 . | W12 . | W16 . | W20 . | W0 . | W4 . | W12 . | W16 . | W20 . | ||||||||||
| 327 | F | 72 | 1 | 0 | 3.3 | 0 | 0 | NEG | MUT | 129 | 136 | 81 | 71 | 36 | 114 | 128 | 69 | 63 | 33 |
| 396 | M | 71 | 4 | 0 | 3.7 | 1 | 0 | POS | UNMUT | 12 | 97 | 83 | 39 | 40 | 5 | 81 | 78 | 34 | 32 |
| 775 | F | 74 | 1 | 2 | 2.3 | 0 | 88 | POS | ND | 167 | 30 | 15 | 10 | 12 | 158 | 24 | 13 | 8 | 9 |
| 146J | F | 55 | 4 | 1 | 2.5 | 0 | 38 | NEG | UNMUT | 97 | 117 | 53 | 56 | 28 | 88 | 107 | 49 | 53 | 24 |
| 516 | F | 72 | 3 | 3 | 8.2 | 0 | 0 | NEG | MUT | 7 | 18 | 13 | 14 | 14 | 2 | 14 | 11 | 12 | 11 |
| 514 | F | 68 | 1 | 0 | ND | 0 | 0 | NEG | MUT | 123 | ND | 145 | 120 | 99 | 115 | ND | 138 | 118 | 85 |
| 047 | M | 72 | 1 | 1 | 3.8 | 0 | 13 | POS | UNMUT | 4 | 14 | 5 | 7 | 6 | 1 | 13 | 3 | 4 | 3 |
| 868 | M | 61 | 4 | 1 | 3.3 | 0 | 96 | POS | UNMUT | 365 | 265 | 55 | 25 | 16 | 339 | 233 | 44 | 20 | 9 |
| 428 | F | 55 | 0 | 1 | ND | 0 | 68 | POS | UNMUT | 6 | 16 | 13 | 10 | 8 | 2 | 12 | 13 | 4 | 6 |
| 011 | F | 59 | 1 | 3 | 3.0 | 0 | 54 | ND | UNMUT | 43 | 39 | 18 | 13 | 10 | 38 | 35 | 17 | 11 | 6 |
| 134 | M | 50 | 1 | 1 | ND | 0 | 0 | ND | UNMUT | 49 | 203 | 117 | 70 | 55 | 41 | 183 | 109 | 67 | 49 |
| 841 | F | 61 | 3 | 2 | 7.9 | 0 | 25 | POS | UNMUT | 187 | 256 | 259 | 240 | 212 | 178 | 217 | 259 | 235 | 208 |
| 185 | M | 72 | 4 | 1 | 9.8 | 93 | 0 | NEG | UNMUT | 71 | 51 | 23 | 16 | 14 | 65 | 48 | 21 | 14 | 9 |
| 038 | M | 51 | 0 | 1 | 2.2 | 37 | 0 | POS | UNMUT | 117 | 60 | 28 | 14 | 13 | 107 | 55 | 26 | 12 | 9 |
| 539 | M | 68 | 2 | 4 | 6.4 | 61 | 0 | POS | UNMUT | 7 | 183 | 128 | 130 | 120 | 6 | 171 | 118 | 117 | 111 |
| 502 | M | 79 | 4 | 1 | 6.6 | 93 | 0 | POS | UNMUT | 53 | 130 | 71 | 55 | 38 | 51 | 126 | 66 | 50 | 32 |
| 826 | M | 68 | 2 | 0 | 2.0 | 0 | 0 | NEG | UNMUT | 45 | 15 | 11 | 10 | 8 | 40 | 12 | 7 | 5 | 4 |
| 967 | M | 53 | 1 | 0 | 1.9 | 0 | 58 | POS | UNMUT | 27 | 106 | 95 | 74 | 84 | 21 | 98 | 89 | 59 | 77 |
| 630 | F | 72 | 3 | 2 | 3.3 | 0 | 92 | NEG | UNMUT | 97 | 90 | 84 | 71 | 56 | 79 | 81 | 79 | 65 | 54 |
| 488 | M | 55 | 0 | 6 | 4.2 | 22 | 24 | ND | UNMUT | 118 | 108 | 67 | 47 | 25 | 108 | 90 | 50 | 43 | 21 |
| 719 | M | 65 | 4 | 1 | 2.5 | 65 | 0 | NEG | UNMUT | 41 | 54 | 43 | 30 | 27 | 33 | 50 | 37 | 24 | 22 |
| 864 | M | 65 | 1 | 1 | 1.3 | 7 | 0 | POS | MUT | 4 | 12 | 23 | 12 | 9 | 1 | 8 | 18 | 9 | 4 |
| 683 | M | 68 | 2 | 1 | 3.2 | 0 | 0 | POS | UNMUT | 47 | 9 | 4 | 5 | 3 | 44 | 6 | 2 | 3 | 2 |
| 916 | F | 60 | 1 | 0 | 2.4 | 0 | 0 | NEG | MUT | ||||||||||
| 146 | M | 72 | 1 | 0 | 4.9 | 34 | 0 | NEG | ND | ||||||||||
| 085 | F | 64 | 4 | 6 | 9.3 | 0 | 0 | NEG | MUT | ||||||||||
| 417 | M | 41 | 2 | 0 | 2.5 | 0 | 0 | NEG | MUT | ||||||||||
| 002 | F | 67 | 1 | 0 | 1.6 | 19 | 0 | POS | ND | ||||||||||
| 315 | M | 59 | 3 | 0 | 6.5 | 0 | 91 | NEG | UNMUT | ||||||||||
| 454 | M | 57 | 1 | 0 | 6.1 | 38 | 0 | POS | UNMUT | ||||||||||
ALC, absolute lymphocyte count (K/mL of blood); ATM, Ataxia telangiectasia mutated; B2M, b-2-microglobulin level (mg/L); F, female; IgVH, immunoglobulin variable region heavy chain; IHC, immunohistochemistry; M, male; MUT, mutated; ND, not determined; NEG, negative; POS, positive; Pt, patient; UNMUT, unmutated; W0, week 0; W4, week 4; W12, week 12; W16, week 16; W20, week 20; WBC, white blood cell count (K/mL of blood); ZAP70, Zeta-chain-associated protein kinase 70.
Percentage of positive cell with cytogenetic abnormality for the corresponding locus.
Biological and molecular consequences of adding CFZ to IBT in primary CLL lymphocytes. Peripheral blood was obtained from CLL patients who were either IBT-naive or undergoing IBT therapy and who had given written informed consent in accordance with the Declaration of Helsinki and under a protocol approved by the Institutional Review Board of The University of Texas MD Anderson Cancer Center. PBMCs were separated by Ficoll-Hypaque density centrifugation (Atlanta Biologicals). Cells were cultured at 1 × 107/mL in complete RPMI medium containing 10% human serum and were either untreated or treated with the indicated dose of drugs. (A) Pharmacological screening of IBT-treated CLL cells with 8 therapeutic agents. CLL cells isolated from a patient treated with IBT for 12 weeks were left untreated or were incubated for 24 hours with vehicle (DMSO) or the indicated agents at which point cell death was assessed by annexin V/PI double positivity. For apoptosis assay, 1 × 106 cells were stained with annexin V (BD Biosciences) and PI (Sigma) and analyzed by flow cytometry as previously described.7 CFZ (50 nM, proteasome inhibitor; Selleck Chemicals); ABT-199 (5 nM, Bcl-2 antagonist; Xcess Biosciences); ABT-737 (5 nM, Bcl-2 and Bcl-xL antagonist); IBT (5 μM, BTK inhibitor; Selleck Chemicals); IPI-145 (2.5 μM, PI3Kδ/γ inhibitor); bendamustine (30 μM, alkylating agent); GS-1001 (5 μM, PI3Kδ inhibitor); 17-AAG (5 μM, HPS90 inhibitor; Sigma). (B) Ex vivo cytotoxicity of CFZ in CLL cells post-IBT therapy. PMBCs were isolated from 23 CLL patients who had received IBT for the indicated weeks and were then incubated with CFZ (50 nM) for 24 hours; cell death was assessed by annexin V/PI double positivity (percentage of cell death from DMSO treatment was subtracted from all samples). (C) In vitro cytotoxic effect of IBT, CFZ, or their combination. PBMCs from 7 patients diagnosed with CLL were incubated for 16 hours with CFZ (50 nM) or IBT (2 μM) alone or in combination and cell death was then evaluated by annexin V/PI staining (percentage of cell death from DMSO treatment was subtracted from all samples). (D) Impact of IBT, CFZ, or their combination on the expression level of different proteins. PBMCs from 2 patients with CLL were isolated and treated with the indicated concentrations of CFZ only or in combination with IBT for 16 hours and cell death was assessed by annexin V/PI double positivity (bottom numbers). Treated cells at 16 hours were collected, lysed, and the cell lysates were subjected to immunoblot analysis with the indicated antibodies. Immunoblot analysis was performed as previously described8 by using the following antibodies: phospho-BTK (Tyr223), BTK, phospho-IκBα (Ser32/36), C/EBP homologous protein (CHOP), cleaved poly(ADP-ribose) polymerase (PARP), and cleaved caspase 3 from Cell Signaling Technology; ubiquitin (Santa Cruz Biotechnology Inc); Noxa (EMD Millipore); β-actin (Cytoskeleton Inc); and Bcl-2 (DAKO). Arrows and rounded arrows indicate cleaved and full-length forms, respectively, of the indicated proteins. (E) Impact of IBT therapy on peripheral WBC count and lymphocyte count. WBC count (K/μL) and ALC (K/μL) were plotted for 8 CLL patients prior to (week 0) and 4 weeks (week 4) or 2 (week 2), 4, and 12 (week 12) weeks (CLL-826) after the initiation of IBT treatment. (F) Comparison of CFZ-induced apoptosis in CLL cells pre- and post-IBT therapy. PBMCs from 8 patients were isolated at baseline (week 0) and after 4 weeks (week 4) or after 2, 4, and 12 weeks (CLL-826) of IBT therapy and treated with the indicated concentrations of CFZ (nM). Cell death was measured by annexin V/PI staining at 24 hours or 16 hours (CLL-826) (percentage of cell death from DMSO treatment was subtracted from all samples). No correlation could be made between prognostic markers (Table 1) and CFZ cytotoxic response. (G) Comparison of CFZ-induced changes on the expression level of proteins in CLL cells pre- and post-IBT therapy. The cells from 2 patients obtained at baseline and 4 weeks after IBT therapy were treated as above for 16 hours and then collected for cell death assessment by annexin V/PI double positivity (bottom numbers). Treated cells were lysed and cell lysates were then subjected to immunoblot analysis with the indicated antibodies as described for Figure 1D. Arrows and rounded arrows indicate cleaved and full-length forms, respectively, of the indicated proteins. (H) Evaluation of the cytotoxic effect of CFZ on CLL cells isolated from patients prior to and following the initiation of IBT therapy. For each patient, PBMCs were isolated before IBT therapy (week 0) and at the treatment weeks indicated. The cells were treated with the indicated concentration of CFZ (nM) for 16 hours, and cell death was assessed by annexin V/PI double positivity (bottom numbers). Treated cells at 16 hours were collected, lysed, and the cell lysates were subjected to immunoblot analysis with the indicated antibodies as described in Figure 1D (caspase 8 and cleaved caspase 9; Cell Signaling). Arrows and rounded arrows indicate cleaved and full-length forms, respectively, of the indicated proteins. CFZ, carfilzomib; D, DMSO, dimethyl sulfoxide; IBT, ibrutinib; LE, long exposure; PBMC, peripheral blood mononuclear cell; SE, short exposure; U, untreated; WBC, white blood cell.
Biological and molecular consequences of adding CFZ to IBT in primary CLL lymphocytes. Peripheral blood was obtained from CLL patients who were either IBT-naive or undergoing IBT therapy and who had given written informed consent in accordance with the Declaration of Helsinki and under a protocol approved by the Institutional Review Board of The University of Texas MD Anderson Cancer Center. PBMCs were separated by Ficoll-Hypaque density centrifugation (Atlanta Biologicals). Cells were cultured at 1 × 107/mL in complete RPMI medium containing 10% human serum and were either untreated or treated with the indicated dose of drugs. (A) Pharmacological screening of IBT-treated CLL cells with 8 therapeutic agents. CLL cells isolated from a patient treated with IBT for 12 weeks were left untreated or were incubated for 24 hours with vehicle (DMSO) or the indicated agents at which point cell death was assessed by annexin V/PI double positivity. For apoptosis assay, 1 × 106 cells were stained with annexin V (BD Biosciences) and PI (Sigma) and analyzed by flow cytometry as previously described.7 CFZ (50 nM, proteasome inhibitor; Selleck Chemicals); ABT-199 (5 nM, Bcl-2 antagonist; Xcess Biosciences); ABT-737 (5 nM, Bcl-2 and Bcl-xL antagonist); IBT (5 μM, BTK inhibitor; Selleck Chemicals); IPI-145 (2.5 μM, PI3Kδ/γ inhibitor); bendamustine (30 μM, alkylating agent); GS-1001 (5 μM, PI3Kδ inhibitor); 17-AAG (5 μM, HPS90 inhibitor; Sigma). (B) Ex vivo cytotoxicity of CFZ in CLL cells post-IBT therapy. PMBCs were isolated from 23 CLL patients who had received IBT for the indicated weeks and were then incubated with CFZ (50 nM) for 24 hours; cell death was assessed by annexin V/PI double positivity (percentage of cell death from DMSO treatment was subtracted from all samples). (C) In vitro cytotoxic effect of IBT, CFZ, or their combination. PBMCs from 7 patients diagnosed with CLL were incubated for 16 hours with CFZ (50 nM) or IBT (2 μM) alone or in combination and cell death was then evaluated by annexin V/PI staining (percentage of cell death from DMSO treatment was subtracted from all samples). (D) Impact of IBT, CFZ, or their combination on the expression level of different proteins. PBMCs from 2 patients with CLL were isolated and treated with the indicated concentrations of CFZ only or in combination with IBT for 16 hours and cell death was assessed by annexin V/PI double positivity (bottom numbers). Treated cells at 16 hours were collected, lysed, and the cell lysates were subjected to immunoblot analysis with the indicated antibodies. Immunoblot analysis was performed as previously described8 by using the following antibodies: phospho-BTK (Tyr223), BTK, phospho-IκBα (Ser32/36), C/EBP homologous protein (CHOP), cleaved poly(ADP-ribose) polymerase (PARP), and cleaved caspase 3 from Cell Signaling Technology; ubiquitin (Santa Cruz Biotechnology Inc); Noxa (EMD Millipore); β-actin (Cytoskeleton Inc); and Bcl-2 (DAKO). Arrows and rounded arrows indicate cleaved and full-length forms, respectively, of the indicated proteins. (E) Impact of IBT therapy on peripheral WBC count and lymphocyte count. WBC count (K/μL) and ALC (K/μL) were plotted for 8 CLL patients prior to (week 0) and 4 weeks (week 4) or 2 (week 2), 4, and 12 (week 12) weeks (CLL-826) after the initiation of IBT treatment. (F) Comparison of CFZ-induced apoptosis in CLL cells pre- and post-IBT therapy. PBMCs from 8 patients were isolated at baseline (week 0) and after 4 weeks (week 4) or after 2, 4, and 12 weeks (CLL-826) of IBT therapy and treated with the indicated concentrations of CFZ (nM). Cell death was measured by annexin V/PI staining at 24 hours or 16 hours (CLL-826) (percentage of cell death from DMSO treatment was subtracted from all samples). No correlation could be made between prognostic markers (Table 1) and CFZ cytotoxic response. (G) Comparison of CFZ-induced changes on the expression level of proteins in CLL cells pre- and post-IBT therapy. The cells from 2 patients obtained at baseline and 4 weeks after IBT therapy were treated as above for 16 hours and then collected for cell death assessment by annexin V/PI double positivity (bottom numbers). Treated cells were lysed and cell lysates were then subjected to immunoblot analysis with the indicated antibodies as described for Figure 1D. Arrows and rounded arrows indicate cleaved and full-length forms, respectively, of the indicated proteins. (H) Evaluation of the cytotoxic effect of CFZ on CLL cells isolated from patients prior to and following the initiation of IBT therapy. For each patient, PBMCs were isolated before IBT therapy (week 0) and at the treatment weeks indicated. The cells were treated with the indicated concentration of CFZ (nM) for 16 hours, and cell death was assessed by annexin V/PI double positivity (bottom numbers). Treated cells at 16 hours were collected, lysed, and the cell lysates were subjected to immunoblot analysis with the indicated antibodies as described in Figure 1D (caspase 8 and cleaved caspase 9; Cell Signaling). Arrows and rounded arrows indicate cleaved and full-length forms, respectively, of the indicated proteins. CFZ, carfilzomib; D, DMSO, dimethyl sulfoxide; IBT, ibrutinib; LE, long exposure; PBMC, peripheral blood mononuclear cell; SE, short exposure; U, untreated; WBC, white blood cell.
Next, we evaluated carfilzomib and ibrutinib synergetic cytotoxic effect on ibrutinib-naive CLL cells during in vitro incubation. Cells isolated from 7 patients (Table 1) were treated for 16 hours with concentrations of ibrutinib and carfilzomib that resulted in modest toxicity individually but, when combined, showed at least an additive cytotoxic effect (Figure 1C). The response to single agents and the combination varied between patients. Western blot analysis of CLL cells isolated from 2 patients confirmed the inhibitory effect of carfilzomib on the proteasome machinery, as illustrated by the accumulation of polyubiquitinated proteins and the stabilization of a short-lived protein (ie, p-IκBα) in a dose-dependent manner (Figure 1D8 ). Furthermore, carfilzomib induced a dose-dependent activation of caspase 3 and accumulation of cleaved PARP and Bcl-2 (Figure 1D), both of which were consistent with a previous study where CLL samples with 17p del also responded well to the cytotoxic effect of carfilzomib.9 Ibrutinib treatment resulted in decreased p-BTK at tyr-223, with a minor effect on caspase activation; however, when combined with carfilzomib, an enhancement of cleaved caspase 3 was notably observed with low doses of carfilzomib (Figure 1D). Notably, increasing concentrations of carfilzomib caused accumulation of the transcription factor CHOP and the proapoptotic BH3-only protein Noxa (Figure 1D), which is consistent with evidence that proteasome inhibition leads to activation of the endoplasmic reticulum stress response and accumulation of apoptotic regulators.10,11 Interestingly, Noxa was previously shown to play a critical role in bortezomib-induced apoptosis in CLL; however, treatment with this reversible inhibitor did not result in increased of CHOP protein,12 suggesting different modes of action between bortezomib and carfilzomib. Here, ibrutinib cotreatment did not influence carfilzomib’s ability to induce accumulation of polyubiquitinated proteins, p-IκBα, Noxa, and CHOP (Figure 1D).
To compare carfilzomib-induced cytotoxic responses between untreated and ibrutinib-treated samples, we isolated CLL cells from 8 patients prior to and 4 weeks after the initiation of ibrutinib therapy, which was previously reported as the time line for maximum transient lymphocytosis.2 With ibrutinib therapy, 4 patients exhibited signs of lymphocytosis as previously reported2 : 2 remained unchanged and 2 patients showed a >80% decrease in absolute lymphocyte count (ALC) (Figure 1E). No notable differences were observed in carfilzomib responses between CLL cells isolated before and after ibrutinib treatment, except 3 patients (CLL-967, CLL-864, and CLL-488) whose cells showed increased sensitivity to carfilzomib in ibrutinib-treated samples (Figure 1F). In contrast, the ibrutinib-treated sample from patient CLL-683 had a marked decrease in apoptosis (Figure 1F), possibly due to a >80% reduction in his ALC (Figure 1E), and similar results were observed in patient CLL-826, 12 weeks following ibrutinib treatment (Figure 1E). These observations are consistent with the report that carfilzomib is less cytotoxic to normal lymphocytes than CLL cells.9 We next used western blot analysis to assess the effects of carfilzomib treatment on CLL cells isolated before and after ibrutinib treatment from 2 patients, 1 with therapy-related lymphocytosis (CLL-967) and 1 without (CLL-630). Cells from both patients responded in a similar manner to carfilzomib treatment, irrespective of ibrutinib therapy, as indicated by the accumulation of polyubiquitinated proteins, p-IκBα, CHOP, and Noxa (Figure 1G). Additionally, carfilzomib treatment in both untreated and ibrutinib-treated cells showed caspase 3 activation and accumulation of cleaved PARP and Bcl-2 (Figure 1G). We extended these findings with cells isolated from 3 patients before and after 2, 4, or 12 weeks of ibrutinib treatment, respectively; in all cases, the cells exhibited decreased phosphorylation of BTK (Figure 1H). Furthermore, both the extrinsic and the intrinsic apoptotic pathways were activated by carfilzomib treatment, as indicated by increases in cleaved caspase 8 and caspase 9, respectively (Figure 1H), and to some extent the cells appeared more resistant to spontaneous endogenous apoptosis ex vivo following ibrutinib therapy.
In summary, this pilot study provides some foundation to further investigate carfilzomib-ibrutinib combination therapy for CLL.
Authorship
Acknowledgments: The authors gratefully acknowledge Dr Bryant G. Darnay for critical comments and for reading the manuscript, and Kathryn B. Carnes for editing the manuscript. The authors thank Benjamin Hayes for collection, Yuling Chen, Min Fu, and Vrushali Datar for transportation of samples, and Susan Smith for providing patient characteristics. V.G. and W.G.W. are members of the CLL Research Consortium.
This work was supported in part by grant P01 CA81534 from the National Cancer Institute at the National Institutes of Health, a CLL Global Research Foundation Alliance grant award, a sponsored research agreement from Pharmacyclics, and generous philanthropic contributions to The University of Texas MD Anderson Cancer Center Moon Shot Program.
Contribution: B.L. designed and performed most of the experiments, analyzed the data, and wrote the manuscript; F.C.-G. performed part of the experiments corresponding to Figure 1A-B; M.S., M.J.K., and W.G.W. provided clinical and patient-related input; V.G. conceptualized and coordinated the project, supervised F.C.-G., and obtained funding; and all authors reviewed and approved the final version of the manuscript.
Conflict-of-interest disclosure: V.G. received a sponsored research agreement from Pharmacyclics. The remaining authors declare no competing financial interests.
Correspondence: Varsha Gandhi, Department of Experimental Therapeutics, The University of Texas MD Anderson Cancer Center, Unit 1950, 1901 East Rd, Houston, TX 77054; e-mail: vgandhi@mdanderson.org.