Abstract
IL-33 is a recently discovered cytokine involved in induction of Th2 responses and functions as an alarmin. Despite numerous recent studies targeting IL-33, its role in vivo is incompletely understood. Here we investigated inflammatory responses to intraperitoneal IL-33 injections in wild-type and mast cell–deficient mice. We found that wild-type mice, but not mast cell–deficient Wsh/Wsh mice, respond to IL-33 treatment with neutrophil infiltration to the peritoneum, whereas other investigated cell types remained unchanged. In Wsh/Wsh mice, the IL-33–induced innate neutrophil response could be rescued by local reconstitution with wild-type but not with T1/ST2−/− mast cells, demonstrating a mast cell–dependent mechanism. Furthermore, we found this mechanism to be partially dependent on mast cell–derived TNF, as we observed reduced neutrophil infiltration in Wsh/Wsh mice reconstituted with TNF−/− bone marrow–derived mast cells compared with those reconstituted with wild-type bone marrow–derived mast cells. In agreement with our in vivo findings, we demonstrate that humanneutrophils migrate toward the supernatant of IL-33–treated human mast cells. Taken together, our findings reveal that IL-33 activates mast cells in vivo to recruit neutrophils, a mechanism dependent on IL-33R expression on peritoneal mast cells. Mast cells activated in vivo by IL-33 probably play an important role in inflammatory reactions.
Key Points
IL-33–induced neutrophil recruitment in vivo is mast cell– dependent. This is partly mediated through the mast cell release of TNF-α.
IL-33–treated human mast cells induce neutrophil migration in vitro.
Introduction
IL-33 is a recently described cytokine of the IL-1 family that is expressed by a variety of cell types, most notably by epithelial and endothelial cells.1 IL-33 promotes Th2 responses2,3 and has been suggested to function as an alarmin when released on necrosis.4-7 IL-33 is inactivated during apoptosis,8 and unlike IL-1β and IL-18, it does not require proteolytical processing for activation.4,9 Recently, we demonstrated that of all endogenous compounds released on necrosis, IL-33 alone is a key alarmin responsible for mast-cell activation.5 Furthermore, IL-33 has emerged as a potent regulator of mast cell activity; inducing cytokine release, adhesion, maturation, and IgE-mediated degranulation.10-17 In addition to mast cells, IL-33 activates several other cell types by signaling through the IL-33 receptor (IL-33R), including eosinophils,18-20 basophils,21-24 and dendritic cells.25-27 Furthermore, a new family of IL-33R–positive innate lymphoid cells, including nuocytes, has been described during the last years.28
Moreover, IL-33 has been implicated in the pathogenesis of several diseases (reviewed by Liew et al1 ), including asthma29,30 and arthritis.31,32 Importantly, mast cells might have a central role in IL-33–associated diseases, as IL-33 for instance has been shown to exacerbate collagen-induced arthritis through a mast cell–dependent mechanism.32 On the other hand, IL-33 has been ascribed several beneficial functions, including a cardioprotective function,33 a protective role in atherosclerosis,34 as well as an important role in helminth infections.3 Thus, IL-33, similar to mast cells, exerts beneficial or detrimental effects depending on the local environment, which makes IL-33 an interesting cytokine with therapeutic potential.
Despite this accumulated knowledge, much remains to be investigated regarding IL-33 functions in vivo. It was previously reported that IL-33 participates in the recruitment of mononuclear cells,35 and a recent study reported that mice injected intravenously with IL-33 before cecal ligation and puncture recruited more neutrophils to the peritoneum than did mice treated with PBS,36 thus displaying a reduced sepsis mortality rate.
In this study we investigated the cellular responses to intraperitoneal IL-33 injections in mice and further addressed the responsiveness of human mast cells to IL-33. Our data demonstrate that a large proportion of the IL-33R+ cells in the peritoneal cavity are c-Kit+FcϵRI+ mast cells, and that IL-33 induces neutrophil influx in the peritoneum of mice through a mast cell–dependent mechanism partly dependent on mast cell–derived TNF.
Methods
Immunohistochemistry
Mouse mesenterium was prepared as previously described.37 Mesentery samples were first preincubated with blocking solution (1% BSA, 0.3% Triton X-100 in PBS) for 1 hour at RT, then treated overnight at 4°C with a combination of primary antibodies diluted in blocking solution. Samples were double-stained with antibodies against IL-33 (1:200, R&D Systems) and 1 of 2 markers for endothelial cells; either CD31 (1:100, Serotec) or Griffonia simplicifolia (Bandeiraea) isolectin B4 (IB4) directly conjugated with FITC (1:50, Jackson ImmunoResearch Laboratories). After washing in 0.25% Triton X-100 in PBS, the secondary antibody combination was applied and incubated for 4 hours at RT. These were donkey anti–mouse-FITC and/or donkey anti–goat-Cy3 (both 1:200, Jackson ImmunoResearch Laboratories). Slides were washed twice and mounted with DTG mounting media (2.5% DABCO [Sigma-Aldrich], 50mM Tris-HCl pH 8.0, 90% glycerol) with 0.375 ng/mL DAPI (Sigma-Aldrich). A Carl Zeiss AxioImager M2 fluorescent microscope (Carl Zeiss) with a 20× objective with a 0.8 numeric aperture was used for the images. The images were processed and arranged in Adobe Photoshop.
Cell cultures and experimental animals
Bone marrow cells were isolated from C57BL/6 wild-type, T1/ST2−/−38 and TNF−/− mice and bone marrow–derived mast cells (BMMCs) were generated as previously described.39 Human cord blood–derived human mast cells were generated as previously described.40 Bone marrow from TNF−/− mice was a kind gift from Kerstin Steinbrink (Johannes Gutenberg-Universität, Mainz, Germany). Peritoneal cavity mast cells (PCMCs) were generated by obtaining peritoneal cells from C57BL/6 mice by PBS-flushing and subsequent culturing in OptiMEM supplemented with 10% FCS, 1% PeSt, 1% l-glutamine, and 4% SCF conditioned medium for 30 days. Mast cell purity was ensured by toluidine blue staining and FACS analysis. For in vivo experiments, wild-type C57BL/6 and Wsh/Wsh mast cell–deficient mice41 on C57BL/6 background were used. All animal experiments were approved by the Swedish local ethics committee for animal welfare.
In vitro stimulations and monitoring of mast cell responses
For in vitro stimulations of PCMCs, 1 × 106 cells were seeded in 96-well plates and incubated with 10 or 100 ng/mL recombinant IL-33 (rIL-33, Alexis or Biolegend; reported endotoxin level: < 0.1EU/μg purified protein as determined by LAL test, Bio Whittaker) for 24 hours, after which supernatants were collected and stored at −20°C until analyzed. IL-6 and TNF release was monitored using ELISA (Biolegend and BioSource, respectively), and release of IL-13, GM-CSF, MIP-2, KC, MIP-1α, and MCP-1 was monitored using Luminex (BioRad).
In vivo administration of IL-33 and flow cytometry
Wild-type C57BL/6 mice and mast cell–deficient Wsh/Wsh mice were injected intraperitoneally with PBS or 0.1 to 1000 ng rIL-33. After 0.5 to 6 hours, the mice were killed and peritoneal lavage was collected. Peritoneal cells were analyzed by flow cytometry using a FACSCalibur (Becton Dickinson). The following antibodies were used: anti–mouse CD16/CD32 (2.4G2; BioLegend), APC-conjugated anti–mouse FcϵRIα (MAR-1; Biolegend), PerCP-Cy5.5–conjugated anti–mouse CD117 (2B8; BioLegend), FITC-conjugated anti–mouse Ly-6G (1A8; BD Pharmingen), APC-conjugated anti–mouse CD19 (1D3; BD Pharmingen), PE-conjugated anti–mouse CD11b (M1/70; BD Pharmingen), FITC-conjugated anti–mouse T1/ST2 (DJ8; MDBiosciences), and FITC-conjugated rat IgG1 isotype control (R&D Systems). For reconstitution experiments, Wsh/Wsh mice were injected intraperitoneally with wild-type, T1/ST2−/− or TNF−/− BMMCs (2.5 × 106 per mouse). Four weeks later, the mice were injected with rIL-33 as previously described. For blocking experiments, 10 μg rat-anti–mouse KC/CXCL1 neutralizing antibody (clone 124014) or isotype control (Rat IgG2A, clone 54447) was used (both from R&D Systems). In experiments where zileuton was used, a concentration of 35 mg/kg was used. The experiments were approved by the Swedish local ethics committee for animal welfare.
Human neutrophil migration assay
Migration studies were performed using the disposable 96-well chemotaxis chamber (ChemoTx; Neuroprobe) with a polycarbonate filter with a pore size of 5 μm and a filter width/well of 3.2 mm. Twenty-nine μL of the attractants and controls were added to the wells and 25 μL cell suspension (250 000 cells) were placed on top of the filter. After 1 hour in 37°C, 5% CO2, the cells were wiped off the filter with a cell harvester and the plate was centrifugated at 200g for 5 minutes before the filter was removed and the supernatants aspirated. Migrated cells were quantified by measurement of the neutrophil marker enzyme myeloperoxidase (MPO). Freshly prepared substrate buffer containing 0.4 mg/mL
o-phenylenediamine dihydrochloride (Sigma-Aldrich), 0.05M phosphate-citrate buffer with 0.1% TritonX-100 was added to each well (25 μL). After 10 minutes, the absorbance was measured at 450 nm (Perkin Elmer EnSpire 2300 Multilabel Reader).
Statistical analysis
The Mann-Whitney test, 1-way ANOVA (Kruskal-Wallis test) or Student t test were used for statistical analyses, with a P value of < .05 being considered statistically significant.
Results
The majority of the peritoneal IL-33R+ population is c-Kit+FcϵRI+ mast cells; which are dose dependently activated by IL-33 in vitro to secrete cytokines and chemokines
We studied the presence of IL-33–expressing cells in the mesentery by analyzing the expression pattern of IL-33 in relation to classic markers for endothelial cells. We found nuclear expression of IL-33 in the majority of mesentery double-layer cells (Figure 1A). Using CD31 (Figure 1A) and Griffonia lectin (Figure 1B-C), we were able to visualize small capillaries as well as medium-sized vessels of the mesentery. We could not observe any coexpression of IL-33 with either of the tested endothelial markers. However, numerous IL-33–positive cells with elongated nuclei were found attached to medium-sized vessels (Figure 1C).
IL-33 is expressed in the mouse mesentery. (A) Representative immunohistochemistry image depicting double-layer mesentery exhibiting strong IL-33 expression (red) in a majority of the cells, (green CD31, blue DAPI nuclear stain). (B) No endothelial cells (green, IB4) expressing IL-33 (red) were found in small or (C) medium-size blood vessels, (blue DAPI nuclear stain). (C) Numerous IL-33–positive cells (red) with elongated nuclei were found attached to medium-size vessels (green, IB4). Scale bars 50 μm.
IL-33 is expressed in the mouse mesentery. (A) Representative immunohistochemistry image depicting double-layer mesentery exhibiting strong IL-33 expression (red) in a majority of the cells, (green CD31, blue DAPI nuclear stain). (B) No endothelial cells (green, IB4) expressing IL-33 (red) were found in small or (C) medium-size blood vessels, (blue DAPI nuclear stain). (C) Numerous IL-33–positive cells (red) with elongated nuclei were found attached to medium-size vessels (green, IB4). Scale bars 50 μm.
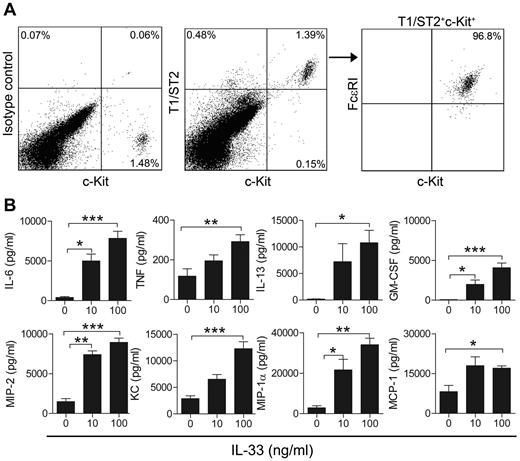
We next investigated the proportion of mast cells in peritoneal lavage of wild-type C57BL/6 mice and observed that approximately 1% of the peritoneal cells were c-Kit+ mast cells (1.15% ± 0.13% c-Kit+, n = 10, data not shown), and that the mast cells express the T1/ST2 subunit of the IL-33R (Figure 2A). Because several innate immune cell populations, such as nuocytes and natural helper cells, have been recently shown to be c-Kit+ (reviewed in Saenz et al42 ), we stained peritoneal cells for IL-33R and c-Kit together with an antibody recognizing FcϵRI to rule out the possibility of mistaking nuocytes/natural helper cells for mast cells. Using this approach, we found that all T1/ST2 c-Kit+ cells are FcϵRI+ (Figure 2A), thus proving the cells in this population to be mast cells. We next cultured peritoneal cells in vitro until obtaining a pure population of PCMCs. When treating PCMCs with 10 or 100 ng/mL rIL-33, we observed a dose-dependent secretion of cytokines (IL-6, TNF, IL-13, and GM-CSF; Figure 2B top panel), and chemokines (MIP-2, KC, MIP-1α, and MCP-1; Figure 2B bottom panel). In contrast, we could not detect any secretion of IL-1β, IL-10, IL-17, or IFN-γ after 24 hours (data not shown). Taken together, these results demonstrate that mouse mesothelial cells of the abdominal cavity express IL-33, and that mast cells constitute the majority of the IL-33R+ cells in the peritoneum, and that these cells respond to IL-33 in vitro by secreting proinflammatory mediators.
A large percentage of peritoneal IL-33R+ cells are mast cells that are activated in vitro to secrete cytokines and chemokines. (A) Expression of T1/ST2, c-Kit, and FcϵRI on peritoneal cells. (B) Secretion of cytokines (top panel) and chemokines (bottom panel) in PCMCs treated with 10 or 100 ng/mL rIL-33 in vitro. In panel A, 1 representative of 3 experiments is shown. In panel B, values are presented as mean ± SEM (n = 4-8) *P < .05, **P < .01, ***P < .001.
A large percentage of peritoneal IL-33R+ cells are mast cells that are activated in vitro to secrete cytokines and chemokines. (A) Expression of T1/ST2, c-Kit, and FcϵRI on peritoneal cells. (B) Secretion of cytokines (top panel) and chemokines (bottom panel) in PCMCs treated with 10 or 100 ng/mL rIL-33 in vitro. In panel A, 1 representative of 3 experiments is shown. In panel B, values are presented as mean ± SEM (n = 4-8) *P < .05, **P < .01, ***P < .001.
Intraperitoneal IL-33 administration induces rapid neutrophil recruitment
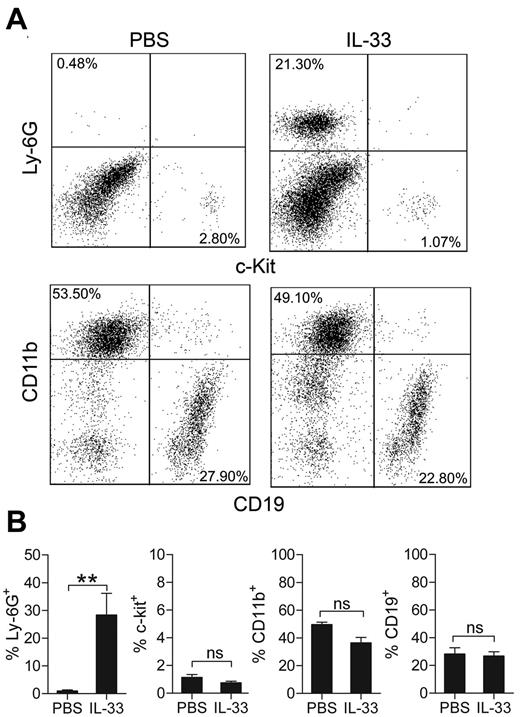
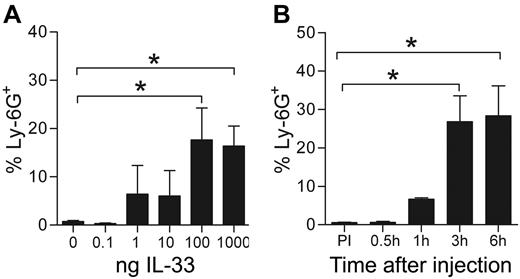
Although many recent studies have investigated various aspects of IL-33 biology, the initial mechanisms for IL-33–induced cell recruitment in vivo are still not fully explored or described. To address this, we first investigated acute responses to intraperitoneally IL-33 injections by monitoring peritoneal cellular composition 1 to 6 hours after injection. Using a neutrophil-specific antibody targeting Ly-6G (clone 1A8), we observed strong neutrophil infiltration in the peritoneum compared with PBS-injected mice (28.5% ± 7.8% Ly-6G+ neutrophils versus 0.98% ± 0.4%, P = .0079, n = 5) 6 hours after injection with 0.1 μg IL-33, whereas the levels of other investigated cell types (mast cells, macrophages, and B lymphocytes) remained unchanged (Figure 3A-B). These results suggest an innate function for IL-33 in early neutrophil recruitment in vivo, and we therefore further investigated dose and time dependency of the observed response. We found that administration of small amounts of IL-33 (1-10 ng) generated a small but insignificant recruitment of neutrophils, whereas administration of 0.1 to 1 μg generated a strong neutrophilic influx (Figure 4A). Furthermore, we found a significant neutrophil infiltration into the peritoneum already 1 hour after IL-33 injection and maximal peritoneal neutrophil infiltration was observed 3 to 6 hours after injection (Figure 4B).
Intraperitoneal administration of IL-33 causes neutrophil influx in C57BL/6 mice. (A) Wild-type C57BL/6 mice were injected intraperitoneally with PBS or 0.1 μg IL-33 and peritoneal cells were analyzed 6 hours later by flow cytometry. Neutrophils are represented by a Ly-6G+ population (a neutrophil specific clone of the Ly-6G antibody was used: 1A8), mast cells by a c-Kit+ population, macrophages by a CD11bhigh population and B cells by a CD19+ population. (B) Quantification of panel A; results are presented as percentage of total peritoneal cells in peritoneal lavage obtained from injected mice. In panel A, representative plots are shown. Values are presented as mean ± SEM (n = 4-5) **P < .01. ns = not significant.
Intraperitoneal administration of IL-33 causes neutrophil influx in C57BL/6 mice. (A) Wild-type C57BL/6 mice were injected intraperitoneally with PBS or 0.1 μg IL-33 and peritoneal cells were analyzed 6 hours later by flow cytometry. Neutrophils are represented by a Ly-6G+ population (a neutrophil specific clone of the Ly-6G antibody was used: 1A8), mast cells by a c-Kit+ population, macrophages by a CD11bhigh population and B cells by a CD19+ population. (B) Quantification of panel A; results are presented as percentage of total peritoneal cells in peritoneal lavage obtained from injected mice. In panel A, representative plots are shown. Values are presented as mean ± SEM (n = 4-5) **P < .01. ns = not significant.
Neutrophils are rapidly recruited to the peritoneum following IL-33 administration. (A) Percent neutrophils in peritoneal lavage of mice treated for 6 hours with 0, 0.1, 1, 10, 100, or 1000 ng IL-33. (B) Neutrophil infiltration over time in mice injected with 100 ng IL-33. Values are presented as mean ± SEM (n = 4-5) *P < .05. ns = not significant.
Neutrophils are rapidly recruited to the peritoneum following IL-33 administration. (A) Percent neutrophils in peritoneal lavage of mice treated for 6 hours with 0, 0.1, 1, 10, 100, or 1000 ng IL-33. (B) Neutrophil infiltration over time in mice injected with 100 ng IL-33. Values are presented as mean ± SEM (n = 4-5) *P < .05. ns = not significant.
IL-33–induced neutrophil influx is mast cell–dependent
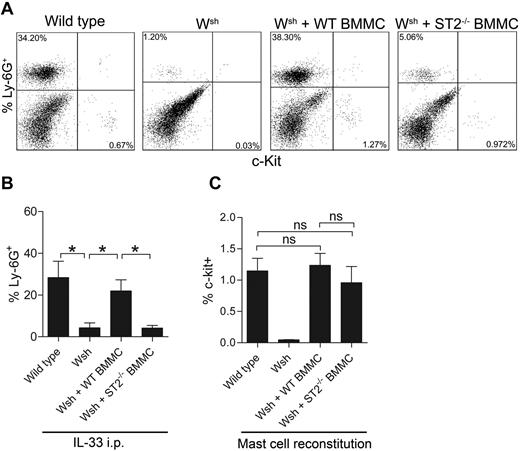
Because IL-33 is a potent activator of mast cells and that we showed mast cells to constitute a major part of the IL-33R+ cells in the peritoneum, we speculated that mast cells might have a significant role in the neutrophil recruitment observed in response to IL-33. Therefore, we injected wild-type C57BL/6 and mast cell–deficient Wsh/Wsh-mice intraperitoneally with rIL-33 and monitored cells in the peritoneum 6 hours after injection. rIL-33 caused a potent influx of neutrophils in the peritoneal cavity in wild-type but not in mast cell–deficient mice (28.35% ± 7.82% Ly-6G+ versus 4.32% ± 2.31%, P = .031, n = 5; Figure 5A-B). The percentage of Ly-6G+ cells in PBS control Wsh/Wsh-mice was 0.6 ± 0.37%. The defective neutrophil response in Wsh/Wsh-mice was rescued by reconstitution with wild-type but not with T1/ST2−/− BMMCs (21.94% ± 5.39% Ly-6G+ vs 4.15% ± 1.28%, P = .016, n = 5), thus clearly indicating that neutrophil influx into the peritoneal cavity in response to rIL-33 is induced through a mast cell–dependent mechanism. There were no significant differences in mast cell numbers between wild-type mice and Wsh/Wsh-mice reconstituted with wild-type or T1/ST2−/− BMMCs (Figure 5C). Our data generated in vivo thus show that innate responses to rIL-33 are mast cell–dependent, as mast cell–deficient mice fail to initiate acute inflammatory responses, manifested by neutrophil infiltration.
IL-33–induced peritoneal neutrophil influx is mast cell–dependent. (A) Flow cytometry plots of c-Kit+ mast cells and Ly-6G+ neutrophils in peritoneal fluid from wild-type mice, Wsh/Wsh-mice, Wsh/Wsh mice reconstituted with wild-type BMMCs and Wsh/Wsh mice reconstituted with T1/ST2−/− BMMCs and injected intraperitoneally with 0.1 μg IL-33. Representative plots are shown. (B) Quantification of panel A (n = 4-5). (C) Percentage of mast cells in the peritoneum of wild-type, Wsh/Wsh-mice, and Wsh/Wsh mice reconstituted with wild-type or T1/ST2−/− BMMCs (n = 5-11). Values are presented as mean ± SEM, *P < .05, ns = not significant.
IL-33–induced peritoneal neutrophil influx is mast cell–dependent. (A) Flow cytometry plots of c-Kit+ mast cells and Ly-6G+ neutrophils in peritoneal fluid from wild-type mice, Wsh/Wsh-mice, Wsh/Wsh mice reconstituted with wild-type BMMCs and Wsh/Wsh mice reconstituted with T1/ST2−/− BMMCs and injected intraperitoneally with 0.1 μg IL-33. Representative plots are shown. (B) Quantification of panel A (n = 4-5). (C) Percentage of mast cells in the peritoneum of wild-type, Wsh/Wsh-mice, and Wsh/Wsh mice reconstituted with wild-type or T1/ST2−/− BMMCs (n = 5-11). Values are presented as mean ± SEM, *P < .05, ns = not significant.
IL-33–induced neutrophil influx is partially dependent on mast cell–derived TNF
We next attempted to elucidate the mechanism behind the observed mast cell–dependent neutrophil recruitment in response to IL-33. To do this, we first repeated an experiment performed by Hueber et al in a recently published article.43 Here, we treated wild-type mice with rIL-33 in the presence of a neutralizing antibody targeting KC/CXCL1. In contrast to results presented by Hueber et al, we could not obtain a significant decrease in neutrophil infiltration in mice treated with anti-KC antibody compared with mice treated with corresponding isotype (Rat IgG2A) control (10.4% ± 0.8602% Ly-6G+ vs 13.86% ± 3.01%, P = .46, n = 5-6; Figure 6A). Next, we subjected wild-type mice to zileuton, a leukotriene inhibitor, to investigate the possibility that LTB4 might be involved in the neutrophil recruitment. However, despite treatment with 35 mg/kg zileuton, IL-33 induced similar recruitment of neutrophils compared with vehicle-treated animal (data not shown), thus suggesting other mediators to be responsible for the neutrophil recruitment. To explore a possible role for TNF, we reconstituted Wsh/Wsh-mice with wild-type or TNF−/− BMMCs and treated these mice with IL-33. Here, we observed a significant decrease in neutrophil recruitment in mice reconstituted with TNF−/− BMMCs, compared with mice reconstituted with wild-type BMMCs (2.361% ± 0.786% Ly-6G+ versus 4.87% ± 1.2%, P = .046, n = 6-12; Figure 6B). No significant differences in mast cell numbers between wild-type mice and Wsh/Wsh-mice reconstituted with wild-type or TNF−/− BMMCs were observed (Figure 6C). Taken together, our data imply that neutrophil influx in response to IL-33 is partially dependent on TNF secreted by mast cells.
Neutrophil infiltration in response to IL-33 is partially dependent on mast cell–derived TNF. (A) Percentage of Ly-6G+ neutrophils in wild-type C57BL/6 mice injected with IL-33 intraperitoneally in the presence of 10 μg anti-KC antibody or rat IgG2A isotype control (n = 5-6). (B) Percentage of Ly-6G+ neutrophils in Wsh/Wsh mice reconstituted with wild-type BMMCs and Wsh/Wsh mice reconstituted with TNF−/− BMMCs and injected intraperitoneally with IL-33 (n = 7-12). (C) Percentage of mast cells in the peritoneum of Wsh/Wsh mice reconstituted with wild-type or TNF−/− BMMCs (n = 7-12). Values are presented as mean ± SEM, *P < .05, ns = not significant.
Neutrophil infiltration in response to IL-33 is partially dependent on mast cell–derived TNF. (A) Percentage of Ly-6G+ neutrophils in wild-type C57BL/6 mice injected with IL-33 intraperitoneally in the presence of 10 μg anti-KC antibody or rat IgG2A isotype control (n = 5-6). (B) Percentage of Ly-6G+ neutrophils in Wsh/Wsh mice reconstituted with wild-type BMMCs and Wsh/Wsh mice reconstituted with TNF−/− BMMCs and injected intraperitoneally with IL-33 (n = 7-12). (C) Percentage of mast cells in the peritoneum of Wsh/Wsh mice reconstituted with wild-type or TNF−/− BMMCs (n = 7-12). Values are presented as mean ± SEM, *P < .05, ns = not significant.
Human neutrophils migrate toward supernatant of human cord blood–derived mast cells treated with IL-33
To test the importance of the IL-33 mast-cell axis on neutrophil migration in a human system, we generated human cord blood–derived mast cells (CBMCs) and treated these in vitro with 10 ng/mL IL-33. The supernatant of IL-33–treated CBMCs was then used in a migration assay with peripheral human blood neutrophils from healthy blood donors. Neutrophil migration was assayed as a measurement of MPO activity. In this experiment, we found that migration was increased in neutrophils migrating toward the supernatant of IL-33–treated CBMCs compared with those migrating toward supernatant from untreated CBMCs (Figure 7). Although CBMCs spontaneously released a chemotactic factor which induced neutrophil migration, the migration was significantly increased if the CBMCs had been treated with IL-33. Neutrophils did not migrate toward the negative control (medium alone) or toward IL-33 alone (10 ng/mL), although migration toward 2 different positive controls (1 ng/mL IL-8 and 10% zymosan activated serum) was observed. This taken together with our earlier results thus demonstrates that both murine and human mast cells treated with IL-33 induce neutrophil migration.
The supernatant of IL-33–treated human mast cells induces migration of human neutrophils. Human blood neutrophils were obtained from 2 different donors and assayed in triplicates in a chemotaxis assays. Neutrophil migration toward untreated or IL-33–treated CBMCs or IL-33 alone was then quantified by measurement of MPO, and shown as O.D 450-620 nm. Unconditioned medium alone or was used as negative controls, and 1 ng/mL IL-8 and 10% ZAS were used as positive controls. Values are presented as mean ± SEM, **P < .01. CBMCs = cord blood–derived mast cells, ZAS = zymosan activated serum.
The supernatant of IL-33–treated human mast cells induces migration of human neutrophils. Human blood neutrophils were obtained from 2 different donors and assayed in triplicates in a chemotaxis assays. Neutrophil migration toward untreated or IL-33–treated CBMCs or IL-33 alone was then quantified by measurement of MPO, and shown as O.D 450-620 nm. Unconditioned medium alone or was used as negative controls, and 1 ng/mL IL-8 and 10% ZAS were used as positive controls. Values are presented as mean ± SEM, **P < .01. CBMCs = cord blood–derived mast cells, ZAS = zymosan activated serum.
Discussion
We report here that IL-33 activates PCMCs in vitro to secrete proinflammatory cytokines and chemokines, and that intraperitoneal IL-33 administration in vivo induces rapid neutrophil recruitment to the peritoneal cavity through a mast cell–dependent mechanism. In addition, we found an abundant number of IL-33–positive cells in the mesothelium. A few of these had elongated nuclei and were seen in close contact with vessels, but they were stained for neither CD31 nor lectin. This is in accordance with previous report that also found mouse endothelial to be IL-33–negative.44 Thus, the peritoneal cavity represents a sound choice of anatomical location for studies of acute responses to elevated IL-33 levels. The finding that mast cells are required for the early recruitment of neutrophils in response to IL-33 helps delineating the mechanism behind IL-33–induced immune responses, and also highlights the role of mast cells in innate immune responses. Although many studies have shown that mouse mast cells are activated by IL-33 to release cytokines and chemokines,10,12,13,16,32 many of these studies used mast cell lines or bone marrow–derived mast cells, which do not represent fully mature mast cells. Therefore, we here investigated whether mast cells obtained from the peritoneal cavity (PCMCs) display similar responses to IL-33. Our results show that IL-33 induces release of several proinflammatory cytokines and chemokines, including TNF, GM-CSF, MIP-2, and KC dose dependently.
It was recently described that mice injected with IL-33 before CLP display improved bacterial clearance, increased neutrophil influx, and reduced mortality compared with PBS-treated mice.36 Given our observation that c-Kit+FcϵRI+ mast cells constitute a major part of the IL-33R+ population in the peritoneal cavity, we investigated whether mast cells play a role in neutrophil infiltration into the peritoneum. Interestingly, our results demonstrate that this process is entirely mast cell–dependent, as mast cell–deficient mice lack the neutrophil response to IL-33 administration seen in wild-type mice. This response could be rescued in mast cell–deficient mice by reconstituting the peritoneal cavity with wild-type mast cells, but not with mast cells lacking the IL-33R, T1/ST2. Mast cells thus appear to be important for neutrophil influx in the peritoneum. However, our results are in contrast to a recent study by Verri et al, where it was demonstrated that IL-33 participates in the orchestration of neutrophil migration in arthritis after intra-articular injections.45 Opposite to our data, the authors show that the neutrophil infiltration in this setting was induced through a mast cell-independent mechanism, and could instead attribute this effect to synoviocytes and macrophages. In addition, it was recently demonstrated that IL-33 injections into the ears of mice induces inflammatory skin lesions, partly through a mast cell–dependent mechanism and that IL-33 recruited neutrophils into the ear.43 Consequently, our and others' data thus suggest that neutrophil recruitment in response to IL-33 treatment is not only dose and time-dependent, but also highly dependent on the tissue in which IL-33 is administered. This together with our data showing that IL-33–mediated neutrophil influx is mast cell–dependent might have important implications for future studies seeking to investigate various potentially therapeutic aspects of IL-33 biology. Given the fact that IL-33 has the potential to exert harmful as well as protective functions in vivo,1 administration route/location, dosage, and timing will be of vital importance in such studies.
In our attempt to find the mechanism behind the mast cell–dependent neutrophil infiltration in response to IL-33, we first investigated a role for KC/CXCL1, because this chemokine recently was implicated in neutrophil recruitment after IL-33 administration.43 However, we could not find significant evidence for a role of KC/CXCL1 in this mechanism in contrast to results reported by Hueber et al, even though we used the same concentration of KC/CXCL1 neutralizing antibody.43 One possible explanation for this could be that we performed this experiment in C57BL/6 mice, whereas Hueber et al used Balb/c mice.43 Mast cell–derived leukotrienes have also been assigned an important role in neutrophil recruitment.46 Therefore, we pretreated mice with zileuton before IL-33 administration, to inhibit the formation of LTB4, a strong neutrophil chemoattractant,47 but we were unable to detect a significant decrease in neutrophil recruitment compared with vehicle-treated animals. Finally, we explored a possible role for mast cell–derived TNF, because this cytokine previously has been shown to be important in mast cell-mediated neutrophil recruitment.48-50 Here, we observed a significant reduction in neutrophil recruitment when TNF was absent, suggesting an important role for TNF in this mechanism (Figure 6C). Although the percentage of infiltrated Ly-6G+ cells in IL-33–treated WT mice differ in Figures 5 and 6, the absolute numbers of infiltrating Ly-6G+ cells did not differ significantly between these 2 experiments (supplemental Figure 1, available on the Blood Web site; see the Supplemental Materials link at the top of the online article).
Our results from in vivo and in vitro experiments using PCMCs, genetically modified mice, human mast cells and neutrophils, provide additional evidence for the notion that mast cells initiate inflammatory responses after IL-33 recognition, here manifested by neutrophil recruitment. Mast cell–mediated neutrophil recruitment in response to IL-33 might contribute to a beneficial outcome during for instance bacterial infection, but exaggerated neutrophil influx might on the other hand result in collateral damage to surrounding tissues.17
In summary, we show here for the first time, that when injected intraperitoneally, IL-33 induces neutrophil influx to the peritoneal cavity through a mast cell–dependent mechanism, as wild-type mice but not mast cell–deficient mice respond to IL-33 with neutrophil influx. In addition, we show that this mechanism is partially dependent on TNF. Further proof for this conclusion is that local reconstitution of mast cell–deficient mice with wild-type BMMCs, but not with T1/ST2−/− BMMCs, restored the neutrophil response to IL-33 injection in these mice. In addition, we show that mast cells constitute close to 75% of the IL-33R+ peritoneal cells. Our results thus demonstrate that mast cells are crucial for the important early recruitment of neutrophils during acute stages of inflammation induced by IL-33 in vivo. Mast cells activated in vivo by IL-33 probably play an important role in both beneficial but also detrimental actions of IL-33. Our work presented here provides novel insights regarding the biology of IL-33, and clearly highlights mast cells as important orchestrators of IL-33–induced innate immune responses.
The online version of this article contains a data supplement.
The publication costs of this article were defrayed in part by page charge payment. Therefore, and solely to indicate this fact, this article is hereby marked “advertisement” in accordance with 18 USC section 1734.
Acknowledgments
The authors thank Sara Lind Enoksson (Karolinska Institutet) for technical assistance, Kerstin Steinbrink and Talkea Schmidt (Johannes Gutenberg-Universität, Mainz, Germany) for kindly providing bone marrow from TNF−/− mice, and Mikael Adner (Karolinska Institutet) for kindly providing Zileuton.
This work was supported by the Swedish Research Council, the Swedish Cancer Foundation, the Ellen, Walter and Lennart Hesselman Foundation, the Ollie and Elof Ericsson Foundation, the King Gustaf V:s 80-years Foundation, the Åke Wiberg Foundation, and Karolinska Institutet.
Authorship
Contribution: M.E. planned and performed the experiments and wrote the paper; C.M.-W. performed experiments, G.W. and K.F.-N. performed experiments and wrote the paper; C.L.-A. planned and performed experiments and wrote the paper; P.G.F. wrote the paper and contributed important reagents; and G.N. conceived the study, planned experiments, and wrote the paper.
Conflict-of-interest disclosure: The authors declare no competing financial interests.
Correspondence: Gunnar Nilsson, Department of Medicine, Clinical Immunology and Allergy Unit, Karolinska Institutet KS L2:04 SE-171 76, Stockholm, Sweden; e-mail: gunnar.p.nilsson@ki.se.




This feature is available to Subscribers Only
Sign In or Create an Account Close Modal