Abstract
Targeted therapy with imatinib and other selective tyrosine kinase inhibitors has transformed the treatment of chronic myeloid leukemia. Unlike chronic myeloid leukemia, chronic lymphocytic leukemia (CLL) lacks a common genetic aberration amenable to therapeutic targeting. However, our understanding of normal B-cell versus CLL biology points to differences in properties of B-cell receptor (BCR) signaling that may be amenable to selective therapeutic targeting. The applica-tion of mouse models has further expanded this understanding and provides information about targets in the BCR signaling pathway that may have other important functions in cell development or long-term health. In addition, overexpression or knockout of selected targets offers the potential to validate targets genetically using new mouse models of CLL. The initial success of BCR-targeted therapies has promoted much excitement in the field of CLL. At the present time, GS-1101, which reversibly inhibits PI3Kδ, and ibrutinib (PCI-32765), an irreversible inhibitor of Bruton tyrosine kinase, have generated the most promising early results in clinical trials including predominately refractory CLL where durable disease control has been observed. This review provides a summary of BCR signaling, tools for studying this pathway relevant to drug development in CLL, and early progress made with therapeutics targeting BCR-related kinases.
Introduction
The complex signaling responsible for the activation of B cells has been studied extensively. With the identification of numerous proteins responsible for signal transduction came the observation that many of these processes are tightly regulated in normal B cells and are aberrantly activated in select B-cell malignancies, including CLL and non-Hodgkin lymphoma (NHL). This knowledge has allowed for the development of inhibitors of the B-cell receptor (BCR) pathway, which have demonstrated promising early clinical activity in refractory chronic lymphocytic leukemia (CLL) and NHL. Although BCR signaling antagonists are exciting, the complexity of this pathway suggests that there may be many options for targeted inhibition, and the ideal target or specific kinase inhibitor may have yet to be identified. In addition, it is possible that CLL and different types of NHL will have divergent ideal targets based on their specific disease biology. The dramatic change in treatment landscape driven by the introduction of targeted therapy with imatinib in chronic myeloid leukemia (CML) followed by second-generation molecules with further improved properties provides great excitement of the potential of similar approaches in CLL, although the considerable genetic heterogeneity in CLL will probably mean that multiple drugs or multitargeted agents are necessary.
This review provides background on BCR signaling in normal mature B cells, with subsequent focus on differences observed in CLL cells that provide a therapeutic window for drug targeting in this disease. We then review preclinical and clinical data associated with the kinase inhibitors currently in clinical trials that are of great interest to the CLL community.
Normal mature B-cell signaling
Early activation: formation of the signalosome and BCR aggregation
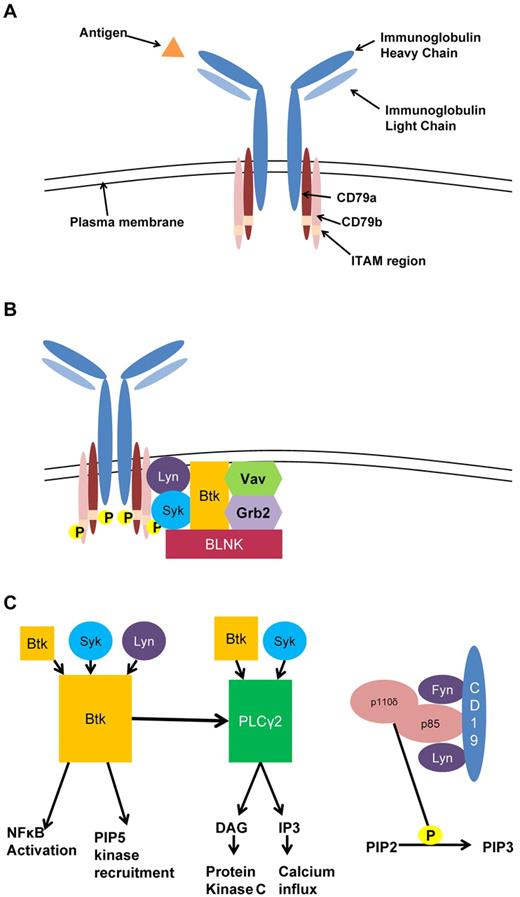
The BCR complex (Figure 1A) is composed of a membrane immunoglobulin (IgM) noncovalently bonded to a heterodimer composed of CD79a(Igα)/CD79b(Igβ). When IgM is ligated by a soluble or membrane-bound antigen, tyrosine residues in the cytoplasmic ITAM portion of CD79a and CD79b are phosphorylated by the Src family kinases Lyn1 and spleen tyrosine kinase (Syk).2 This phosphorylation results in recruitment of the signalosome, which includes many kinases and adaptor proteins, including the kinases Syk, Bruton tyrosine kinase (Btk), and Lyn, the guanine exchange factor Vav proteins, and the adaptor proteins Grb2 and B-cell linker (BLNK; Figure 1B). The protein tyrosine kinases are primarily responsible for amplification of this activation. Syk is responsible for the dual phosphorylation of CD79a/CD79b,2 and Lyn provides continued amplification of the BCR through recruitment of protein tyrosine kinases as well as formation of a complex with CD19 and other costimulatory molecules that reduce the threshold of B-cell activation.3 As these signals begin to propagate, antigen ligation results in BCR aggregation and microcluster formation in regions of the plasma membrane termed lipid rafts.4 Aggregation is promoted by the costimulatory receptor CD195 and activation of Btk.6
B-cell receptor signaling in CLL. (A) The BCR is composed of membrane immunoglobulin bound to CD79a/CD79b. Antigen binding induces CD79a/CD79b ITAM recruitment of Syk and Lyn initiating the signaling cascade. (B) Signalosome complex. Phosphorylation of CD79a/CD79b recruits a number of kinases and adaptor proteins, which form the initial signaling complex of the BCR. (C) Intermediate activation. Signalosome activation recruits a number of additional kinases leading to activation down 3 main pathways: Btk, PLC-γ2, and PI3K. Btk is phosphorylated by itself; Syk, and Lyn, which lead to phosphorylation of PLC-γ2, activation of NFκB; and recruitment of PIP5K. PLC-γ2 is phosphorylated by Btk and Syk and leads to production of DAG and IP3. PI3K activation leads to phosphorylation of PIP2 to PIP3.
B-cell receptor signaling in CLL. (A) The BCR is composed of membrane immunoglobulin bound to CD79a/CD79b. Antigen binding induces CD79a/CD79b ITAM recruitment of Syk and Lyn initiating the signaling cascade. (B) Signalosome complex. Phosphorylation of CD79a/CD79b recruits a number of kinases and adaptor proteins, which form the initial signaling complex of the BCR. (C) Intermediate activation. Signalosome activation recruits a number of additional kinases leading to activation down 3 main pathways: Btk, PLC-γ2, and PI3K. Btk is phosphorylated by itself; Syk, and Lyn, which lead to phosphorylation of PLC-γ2, activation of NFκB; and recruitment of PIP5K. PLC-γ2 is phosphorylated by Btk and Syk and leads to production of DAG and IP3. PI3K activation leads to phosphorylation of PIP2 to PIP3.
Activation propagation
Signal propagation from the BCR occurs via multiple pathways, notably through phospholipase C-γ2 (PLC-γ2), phosphatidalyinositol-3-kinase (PI3K), and Btk (Figure 1C). After Syk and Lyn are recruited to the phosphorylated ITAM of the BCR, BLNK is recruited to the non-ITAM portion of CD79a, where it binds via its Src homology 2 (SH2) domain7,8 and is rapidly phosphorylated by Syk. BLNK then serves as a scaffold protein to bind via the SH2 domain proteins, including PLC-γ2 and Btk.9-11 Once Syk, Btk, BLNK, and PLC-γ2 are in contact, PLC-γ2 is dually phosphorylated by Btk and Syk to produce the second messengers diacylglycerol (DAG) and inositol-1,4,5-triphosphate (IP3) from the plasma membrane lipid phosphatidylinositol 4,5-bisphophate (PIP2).12 DAG is a classic activator of protein kinase C, which is responsible for many downstream effects of BCR signaling. The generation of IP3 leads to enzyme activation and calcium influx from the endoplasmic reticulum as well as the extracellular compartment.13
Initial phosphorylation events after BCR ligation also activate the PI3K pathway. At rest, the regulatory PI3K subunit p85 complexes with the catalytic p110 subunit14 ; however, on BCR activation, p85 is recruited to the plasma membrane15 where it associates with the Src kinases Lyn and Fyn.16 In addition, Lyn phosphorylation of CD19 allows p85 to bind to CD19, and this coassociation activates the p110 subunit, which in B cells is p110 delta.17 PI3K then phosphorylates PIP2 to create phosphatidylinositol 3,4,5-triphosphate (PIP3). PIP3 at the plasma membrane results in recruitment of Btk and other kinases with a pleckstrin homology domain, resulting in continued BCR activation.
Btk is a member of the tyrosine protein kinase (Tec) family of kinases and has a critical role in the amplification of the BCR signal, as demonstrated by profound BCR signaling defects associated with its loss of function in X-linked agammaglobulinemia (XLA) and its mouse counterpart X-linked immunodeficiency (XID). In these conditions, mutation in the pleckstrin homology domain of Btk prevents effective membrane recruitment by PIP3 and abrogates the calcium flux associated with BCR activation. XLA is caused by point mutations in Btk and is characterized by a block at the B1 stage of development, lack of circulating immunoglobulins, and defective B cell signaling, all leading to profound humoral immune deficiency.18,19 The XID phenotype, while showing a similar block in B-cell development, produces a milder phenotype.20
BTK deficiency produces initial BCR phosphorylation event defects, whereas downstream effects of BCR signaling can be restored by increasing intracellular calcium, suggesting the role of Btk in the intermediate stages of signal transduction.21 After phosphorylation by Syk and Lyn at the Y551 site of the kinase domain, Btk activation is amplified through autophosphorylation of the Y223 site in the SH3 domain.22 In addition, Btk amplifies its own activation through a scaffolding role by recruiting the phosphatidylinositol-4-phosphate 5-kinases (PIP5Ks),23 which are responsible for synthesis of PIP2, which after phosphorylation by PI3K results in continued recruitment of Btk. In addition to its role in the PI3K/Akt pathway, Btk has been shown to activate IκB kinase, which phosphorylates the NF-κB inhibitor I-κBα, allowing NF-κB to translocate to the nucleus.24
Downstream activation pathways influenced by BCR
Activation of the mature BCR leads to multiple downstream effectors, which are differentially activated depending on the state of cell maturation and antigen ligated. Calcium influx directly activates a number of transcription factors, including NF-κB, Jun, and nuclear factor of activated T cells (NFAT), which vary with the duration and amplitude of calcium influx.25 NFAT proteins are activated through calcium-dependent activation of calcineurin, which dephosphorylates the NFAT proteins to allow nuclear translocation and subsequent regulation of cytokine production and other effectors of the immune response.26 NF-κB genes, which have a broad role in B lymphocyte proliferation and class switching27 as well as mature B-cell survival,28 are dependent on calcium influx, Vav proteins,29 and Btk for activation.
The mitogen-activated protein kinase (MAPK) pathway contributes to effector functions of BCR activation, including proliferation and cell survival. The MAPK pathway is activated after BCR ligation through a number of mechanisms, including DAG production leading to protein kinase C activation,30 and direct activation by Vav and Grb2. The MAPK pathway regulates a number of transcription factors, including Elk1 and c-Myc through Erk, c-Jun and ATF2 through JNK, and ATF2 and Max through p38 MAPK.31 Based on the maturational state of the B cell and its activation status, these transcription factors can lead to proliferation and survival or apoptosis.
The Akt pathway as well contributes to BCR-induced survival. Akt is activated when PIP3 is formed by PI3K, thereby phosphorylating and recruiting Akt to the plasma membrane.32 Akt then promotes survival by phosphorylating the proapoptotic proteins Bad and FOX03a and by enhancing nuclear accumulation of NFAT through phosphorylation/inhibition of glycogen synthase kinase 3.33 The canonical NF-κB pathway is also an important survival effector in BCR signaling. NF-κB exists inactive in the cytoplasm as a dimer (consisting of p50, p52, p65/RelA, RelB, or c-Rel with the most usual dimers being the p50/p65 heterodimer and the p50/p50 homodimer)34 bound to I-κB. On stimulation via Btk, PI3K, or Akt, the IκB kinase complex causes phosphorylation and subsequent proteasomal degradation of I-κB, facilitating nuclear translocation of NF-κB and gene transcription. NF-κB activates a wide variety of genes responsible for inflammation, proliferation, and B-cell survival. Importantly, these and other pathways have multiple crosstalk mechanisms between them, underscoring the complexity of BCR signaling.
Activation regulation
Regulation of the BCR is necessary to prevent the unrestrained activation that can result in development of autoimmune conditions and malignancies. Activation is tightly controlled via specific inhibitory regulators, such as FcγRIIb SH2 domain-containing inositol 5′-phosphatase (SHIP), SH2 domain-containing phosphatase (SHP-1), and kinases with differential activation and inhibitory properties, including Lyn and others.
FcγRIIb is a transmembrane receptor and potent negative regulator of BCR activation, transmitting signals through an ITIM in its cytoplasmic tail. FcγRIIb can be coligated with the BCR, especially in the case of self-reactive IgG-containing antigens, and induces a negative signal by recruiting SHIP to the plasma membrane,35 which eliminates the membrane binding of PLC-γ2, Btk, and Akt by hydrolyzing PIP3.36-38 In addition, inhibitory signals by SHIP and activating signals by Btk have been shown to be critical to the actin remodeling required for the initial formation of BCR microclusters on lipid rafts.6 SHIP thus both regulates the initial activation of the BCR and the calcium flux required for downstream protein activation. Similarly, SHP-1 can associate with a number of ITIM-containing molecules, and coligation of the BCR and FcγRIIb results in the reversal of SHP-1 autoinhibition, allowing activated SHP-1 to dephosphorylate various substrates. SHP-1 is also associated with the BCR at rest, and this association is disrupted by BCR stimulation, suggesting that SHP-1 is involved in preventing signal transduction in resting B cells.39
An important modulator of the BCR activation response is the Src family kinase Lyn, which has a positive and negative regulatory role in B-cell activation. Although the activation effects of Lyn are well described, it has been shown that Lyn knockout mice surprisingly demonstrate BCR hyper-responsiveness and develop lethal autoimmune glomerulonephritis.40,41 Subsequently, it was shown that Lyn is required for phosphorylation of both SHIP42 and FcγRIIb,43 making it a crucial kinase in the regulation of the BCR. This complex function of Lyn emphasizes also the challenges of therapeutically targeting BCR signaling.
BCR signaling and interface with integrins and adhesion
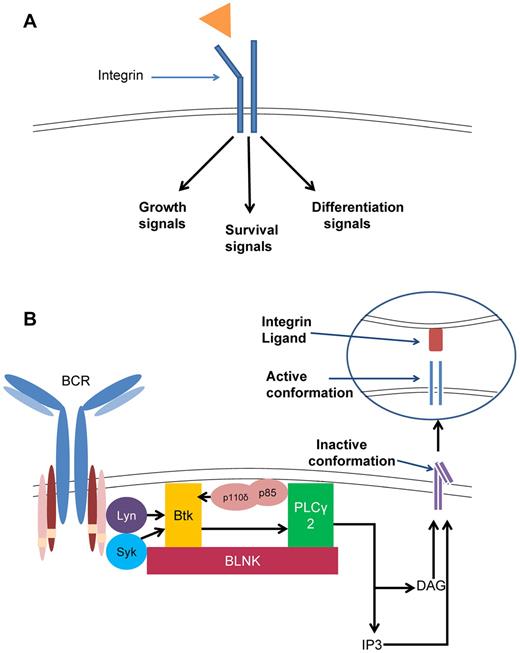
Integrins are transmembrane receptors that induce bidirectional signaling across the plasma membrane. During “inside-out” signaling (Figure 2A), signals generated by the BCR activate an integrin to induce a conformational change in the extracellular domain, resulting in adhesion or cellular migration. In contrast, during “outside-in” signaling (Figure 2B), ligands can bind to the extracellular domain, generating an intracellular signal, which can affect cellular growth, differentiation, and survival.44 In B cells, the BCR has been shown to control integrin-mediated adhesion through “inside-out” signaling, and requires the activation of Lyn, PI3K, Btk, and PLC-γ.45 In addition, the chemokine SDF-1 induces “outside-in” activation of Btk and PLC-γ, which results in integrin-mediated migration toward SDF-1. This migration in mature B cells is important for the migration of B cells into follicles and subsequent germinal center organization.46 These data underscore the importance of BCR signaling in the interaction between lymphocytes and the microenvironment.
Integrin signaling in CLL. (A) Inside-out integrin signaling. Signaling through the BCR, dependent on Btk and PLC-γ2, activates the cytoplasmic domain of the integrin, causing a conformational change to the open, active form. This form has high affinity for ligand binding and induces cell migration and adhesion. (B) Outside-in integrin signaling. Ligand binding to an integrin induces signals that can lead to growth, differentiation, survival, or apoptosis.
Integrin signaling in CLL. (A) Inside-out integrin signaling. Signaling through the BCR, dependent on Btk and PLC-γ2, activates the cytoplasmic domain of the integrin, causing a conformational change to the open, active form. This form has high affinity for ligand binding and induces cell migration and adhesion. (B) Outside-in integrin signaling. Ligand binding to an integrin induces signals that can lead to growth, differentiation, survival, or apoptosis.
Use of mouse models to evaluate BCR pathway proteins acceptable for therapeutic targeting
Mouse models engineered with specific aberrations can assist in determining whether a target is appropriate for therapeutic intervention. An example is the PI3K pathway, where isoforms have been extensively studied using mouse models. The α- and β-isoforms of p110, which are ubiquitously expressed, have been shown to be necessary for development, as knockout of either is embryonic lethal.47,48 Mice with homozygous deletion of the δ- and γ-isoforms, which are expressed primarily in hematopoietic cells, are viable but show defects in immune cell development. p110γ knockout mice have normal B-cell development but impaired T-cell activation and neutrophil function.49 Knockout of p110δ is associated with normal hematopoiesis, except for B cells. These mice show normal development through the pro-B stage but significantly reduced mature lymphocytes and peritoneal B1 lymphocytes, as well as low levels of immunoglobulins. In addition, these mice have abrogated BCR signaling.50 Notable long-term effects in this mouse model is a low frequency of inflammatory bowel disease,51 possibly resulting from the influence of this isoform of PI3K on T-regulatory cells.52 The B-cell phenotype of the PI3K-δ mouse is similar to what is seen with mice with impaired Btk because of mutation or deletion. XID mice with a mutation in the pleckstrin homology domain of BTK20 and BTK null mice53 both show disruption of B-cell development at the pro-B level, impaired proliferation in response to antigen stimulation, and disruption of B-cell signaling.54 The BTK knockout mouse has a more profound B-cell deficiency than XID, which underscores the adaptor role of this protein. In addition, XID mice have intrinsic defects in monocyte and neutrophil function that probably relate to Btk expression in these cells. Thus, the phenotype in knockout models suggests that both PI3K-δ and Btk may serve as good drug targets for lymphoid malignancies. In contrast, mice deficient in Lyn can respond to antigen as long as Blk and Fyn are present,55 but develop autoimmune disease40,41,56 because of dysregulated BCR signaling, and constitutive deficiency of Syk is embryonic lethal probably because of the role in vascular development,57,58 suggesting that the sole inhibition of these kinases may not be ideal.
BCR signaling in CLL
CLL has distinct BCR signaling compared with normal B cells that is characterized by low-level IgM expression, variable response to antigen stimulation, and tonic activation of antiapoptotic signaling pathways (further summarized in Table 1). CLL cells by gene expression profiling share many features with antigen-activated mature B cells, suggesting a role for activation of BCR signaling in the disease pathogenesis. In addition, CLL disease courses can be dichotomized depending on the presence or absence of somatic hypermutation in the immunoglobulin variable heavy chain region genes in the sIg which is strongly associated with overexpression of ZAP-70. ZAP-70 protein enhances BCR signaling of blood-derived CLL cells59 by an adaptor role independent of its kinase activity60 and as well enhances migration toward chemokines and response to survival stimuli from the microenvironment.61 A recent tissue-based comparison demonstrated enhanced up-regulation of the BCR pathway related genes by microarray in bone marrow and lymph nodes of all CLL patients compared with blood irrespective of ZAP-70 expression or immunoglobulin variable heavy chain region mutational status.62 Thus, enhanced BCR gene expression is present in all CLL patients in nodal and lymph node sites. In addition, patients with ZAP-70–positive disease have higher BCR responsiveness to IgM stimulation in vitro compared with ZAP70-negative disease. We discuss pathways and genes differentially regulated in CLL versus normal B cells and how these might be effectively targeted pharmacologically.
B-cell receptor signaling in CLL cells compared with normal B cells
| Characteristic . | CLL cells . |
|---|---|
| sIg expression | Low |
| CD79b expression | Low |
| Response to antigen stimulation | Variable |
| Syk/Lyn/Btk expression | Elevated |
| PI3K p110δ expression | Normal |
| PI3K kinase activity | Elevated |
| Calcium flux | Variable/generally low |
| Characteristic . | CLL cells . |
|---|---|
| sIg expression | Low |
| CD79b expression | Low |
| Response to antigen stimulation | Variable |
| Syk/Lyn/Btk expression | Elevated |
| PI3K p110δ expression | Normal |
| PI3K kinase activity | Elevated |
| Calcium flux | Variable/generally low |
Amplification of signal initiation
Dysregulation of the BCR signaling pathway in CLL is characterized by constitutively active phosphorylation of certain kinases and variable response to IgM stimulation. For instance, the tyrosine kinases Lyn and Syk have both been shown to be up-regulated in CLL. Lyn has been shown to be overexpressed at the protein level but not at the transcriptional level,63 whereas Syk has been shown to be overexpressed at both the mRNA and protein levels.64 Activity of these kinases has also been shown to be amplified in CLL. In vitro kinase assays indicate that Lyn is constitutively active in CLL.63 Baseline phosphorylation of Syk as well is higher in CLL cells than in normal B cells,64 and response to antigen stimulation via the BCR is variable.65 Inhibition of Syk in vitro by BAY61-3606 induces apoptosis, which is dependent on PKC-δ.66 Similarly, the Syk inhibitor R406 abrogates survival after IgM stimulation and reduces downstream targets of BCR signaling, including calcium mobilization and phosphorylation of Erk and Akt.67 The Lyn inhibitors PP2 and SU6656 promote CLL cell apoptosis in vitro with inhibition of Lyn kinase activity.63 These data suggest that clinical targeting of BCR proteins may be effective, even in patients with decreased BCR response to external stimulation.
Amplification of effector pathways
Aberrant activation of the PI3K/Akt pathway has been shown in CLL and is implicated in cell survival. Although protein expression of the catalytic 110δ-subunit, the predominant PI3K subunit in hematopoietic cells,14 is comparable between normal B cells and CLL cells, PI3K has been shown to be constitutively active in CLL by in vitro kinase assay.68,69 In addition, inhibition of PI3K by the pan-PI3K inhibitor LY29400268 and the PI3Kδ inhibitor CAL-10169 both promote CLL cell apoptosis in a caspase-dependent manner. PI3K-α isoform specific inhibitors were shown to be important to stromal cell-mediated protection of CLL cells and also were directly proapoptotic.70 Via upstream PI3K activity, the Akt pathway is also constitutively active in CLL. Inhibition of PI3K by CAL-101 or LY294002 inhibits Akt activation, decreases Mcl-1 expression,68,69 and inhibits protein expression of the bcl-2 family member bax and the antiapoptotic protein XIAP.68 Specific inhibition of Akt by A-443654 decreases phosphorylation of GSK3-β and expression of Mcl-1, thus promoting apoptosis.71 The Btk pathway has also been shown to be amplified in CLL and leads to prosurvival signals through its effects on PI3K, PLC-γ2, and NF-κB. Btk expression at the gene level is elevated in CLL compared with normal B cells.72 Inhibition of Btk by the kinase inhibitor PCI-32765 induces apoptosis in a caspase-dependent manner and inhibits both phosphorylation of Btk after IgM ligation as well as downstream targets of Btk activation, including Erk, NF-κB, and Akt.72 In addition, PCI32765 inhibits proliferation by CpG or microenvironment stimulation.72,73
NF-κB as well is dysregulated in CLL and leads to overexpression of antiapoptotic genes. CLL cells have high levels of nuclear NF-κB and exaggerated responses to stimulation with CD40L, including prolonged cell survival.74 In vitro stimulation through BCR ligation is variable but does induce increased NF-κB nuclear activity that is inversely correlated with spontaneous apoptosis.75 The constitutive activity of PI3K and AKT is important to this NF-κB up-regulation.
Finally, the MAPK pathway, specifically the antiapoptotic proteins Erk1/Erk2, is dysregulated in CLL. A subset of CLL patients demonstrates constitutive phosphorylation of Erk, which is associated with decreased responsiveness to BCR stimulation similar to anergic B cells.76 CLL cells, which lack constitutive phosphorylation, have shown inducible phosphorylation and cell survival in the presence of phorbol ester.77 Expression of the transcription factor Myc has been found to be dependent on Erk1/2 activation after BCR stimulation, suggesting that this pathway is important to CLL survival and proliferation.78
BCR signaling and interface with integrins, adhesion molecules, and microenvironment in CLL
Besides evidence of tonic BCR signaling activation, the tumor microenvironment that exists in the lymph nodes and bone marrow has been shown to promote CLL survival and proliferation, and these effects are partially the result of amplification of the BCR signaling pathway. The proliferating compartment of CLL exists in the bone marrow and lymph nodes, where these cells have close contact to a host of accessory cells, including nurse-like cells, T lymphocytes, and bone marrow stromal cells.79 Cross-talk between accessory cells and CLL cells enhances survival via cytokines, chemokines, and other factors that promote homing, adhesion, and proliferation. In addition, the tumor microenvironment has been shown to promote chemoresistance in CLL.80 Gene expression profiles from CLL cells obtained from blood versus lymph nodes shows that, although many genes are differentially expressed between these 2 populations, the gene set associated with the BCR pathway showed the most striking differential expression, with significant up-regulation in the lymph nodes.62 In addition, the canonical NF-κB pathway was significantly up-regulated in lymph node samples, and the expression of the inhibitory I-κBs was significantly lower.62 Coculture experiments with stromal cells have also shown activation of Syk81 and Akt.82 These data suggest that effectors in the BCR signaling pathway may be important to survival and proliferation signals in the bone marrow and lymph node niches.
Further evidence that the BCR signaling pathway members are important in microenvironment-induced survival signals comes from the in vitro effects of BCR kinase inhibitors. The Syk inhibitor R406 blocks Akt activation seen in stromal coculture, reduces the homing signals and Akt activation induced by CXCL12, and decreases stromal-induced Mcl-1 expression.81 Similarly, the PI3Kδ inhibitor CAL-101 inhibits CLL cell viability in coculture with nurse-like cells and down-regulates chemokines and cytokines amplified in this coculture.83 The Btk inhibitor PCI-37265 as well has been shown to antagonize microenvironment signals. Coculture with stromal cells results in cell survival that is blocked by PCI-32765, and treatment with PCI-32765 antagonizes the protective effect of the prosurvival microenvironment signals TNF-α and BAFF.72
Putting it all together: can mouse models help?
The TCL1 transgenic mouse model of CLL has been validated as a model similar to human CLL, providing an avenue to extend study of BCR signaling to the in vivo setting.84,85 These mice spontaneously develop a CD5+ B-cell leukemia with unmutated immunoglobulin variable heavy chain region resembling unmutated CLL.86 Testing therapeutics targeting BCR signaling for translation to CLL has been effectively performed with agents, such as fostamatinib87 and PCI-32765,73 providing insightful information relative to the mechanism of action and hints of which agents may be active in human disease. Other mouse models of CLL have not been explored in this context.
Clinical translation of BCR signaling inhibitors in CLL
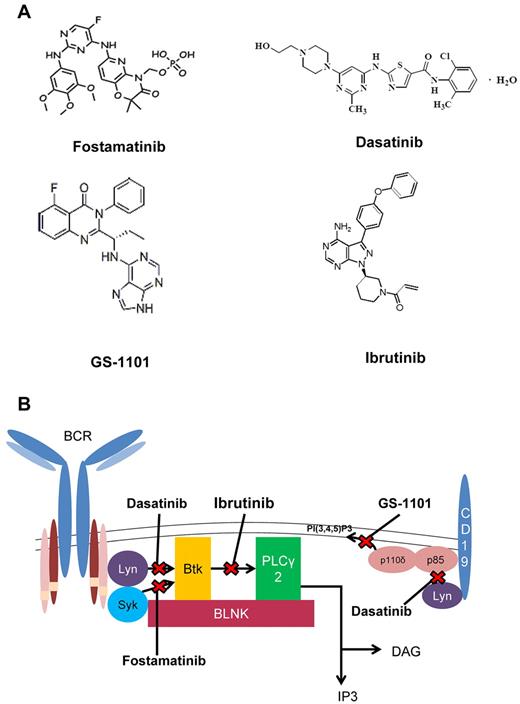
Although strong scientific rationale and compelling preclinical data can generate a flurry of excitement for the first trial targeting a specific pathway, a blizzard of activity can be expected to follow if early promising success is observed. This has clearly happened with early BCR signaling agents, and a plethora of successful trials have followed that, to date, have been presented only at scientific meetings. We focus on therapeutics with documented success targeting BCR with the caveat that, even the phase 1 data for the most exciting compounds, GS-1101 (formerly CAL-101) and ibrutinib (formerly PCI32765), have not been formally published. A comprehensive list of agents targeting BCR signaling is summarized in Table 2.
BCR signaling inhibitors currently in clinical trials in B-cell malignancies
| Drug . | Phase of study . |
|---|---|
| Ibrutinib | 3 |
| GS-1101 | 3 |
| Fostamatinib | 1/2 |
| Dasatinib | 2 |
| Sorafenib | 2 |
| AVL-292 | 1 |
| Enzastaurin | 1 |
| GDC-0941 | 1 |
| XL147 | 1 |
| Drug . | Phase of study . |
|---|---|
| Ibrutinib | 3 |
| GS-1101 | 3 |
| Fostamatinib | 1/2 |
| Dasatinib | 2 |
| Sorafenib | 2 |
| AVL-292 | 1 |
| Enzastaurin | 1 |
| GDC-0941 | 1 |
| XL147 | 1 |
Fostamatinib
Fostamatinib (Figure 3A-B), a prodrug of R406, is an orally bioavailable reversible inhibitor of Syk kinase and multiple alternative kinases. This agent has been studied in a phase 1/2 trial in CLL and NHL. The phase 1 portion established a dose of 200 mg twice daily at which 11 patients with CLL/small lymphocytic lymphoma were treated. Partial responses, based on lymphoma criteria that do not account for peripheral blood disease, were seen in 6 of these patients, with a progression-free survival of 6.4 months. All patients with CLL/small lymphocytic lymphoma exhibited an initial lymphocytosis that has since been recognized as a BCR antagonist class effect, probably because of disruption of CXCR4-SDF1 and other adhesion factors in the marrow and nodal sites leading to blood mobilization.81 This agent was well tolerated, with rare grade 3/4 toxicities. Hematologic toxicity was mild, and infections were uncommon.88
Kinase inhibitors in CLL. (A) Chemical structures of signal kinase inhibitors. (B) Kinase inhibitors and the BCR pathway.
Kinase inhibitors in CLL. (A) Chemical structures of signal kinase inhibitors. (B) Kinase inhibitors and the BCR pathway.
Dasatinib
Dasatinib (Figure 3A-B) is a reversible pan-Src kinase inhibitor currently approved for the treatment of CML, but which also inhibits Lyn, BTK, and a variety of other kinases.89 A phase 2 trial in patients with relapsed or refractory CLL treated with a daily dose of 140 mg for up to 24 months was performed. Of 15 patients enrolled, 9 experienced some reduction in lymph node size, with ≥ 50% decrease noted in 4 patients. Four patients experienced a response beyond 12 months. This agent was associated with grade 3/4 neutropenia in 67% of patients and grade 3/4 thrombocytopenia in 40%.90
GS-1101 (formerly CAL-101)
GS-1101 (Figure 3A-B) is a reversible inhibitor of the PI3K δ isoform of the p110 catalytic subunit with strong preclinical data to support early phase 1 exploration of this molecule in CLL.69,91 In a broad phase 1 trial in indolent B-cell malignancies, a dose of 150 mg twice daily was identified to be ideal for the phase 2 dose in CLL based on tolerability and minimal increase in plasma area under the curve at increasing doses. Of 54 CLL patients enrolled on this trial, 84% achieved a ≥ 50% decrease in lymph node and spleen size. A > 50% increase in peripheral lymphocyte count was seen in 58% of patients, which resolved over time in a subset of patients. As a consequence of the prolonged lymphocytosis, response across all patients enrolled was 24% by International Workshop on CLL response criteria.92 This response was independent of high-risk genetic features, bulky adenopathy, prior therapy, or presence of cytopenias. Median progression-free survival was 15 months, with 46% of patients continuing on therapy at the time of the presentation.93 Side effects with this agent have been mild and included rare cytopenias and pneumonia that was not clearly increased relative to a similarly refractory group of patients treated with traditional therapeutics. Approximately 5% of patients developed grade 3 or 4 transient liver function abnormalities during the early phase of treatment, which was reversible by holding therapy and generally did not recur with resumption at a lower dose level. Based on the lymphocytosis observed and potential to mobilize CLL cells from protected sites, such as the nodes and bone marrow, GS-110 has also been investigated in combination with either the anti-CD20 monoclonal antibodies rituximab (GR) or ofatumumab, or the chemotherapy agents bendamustine (GB), GB + rituximab (GBR), or fludarabine in the phase 1 setting. GS-1101 was dosed at 100 mg twice daily or 150 mg twice daily with other drugs administered as standard. Therapy was well tolerated, with no toxicities other than those expected with the single agents. Response data have been presented for the GR, GB, and GBR regimens. For GR and GB, 13 of 14 patients and 9 of 10 patients, respectively, achieved a ≥ 50% reduction in lymph node size. For 3 patients treated with GBR, all patients achieved a lymph node response.94 Using traditional CLL response criteria, > 80% of patients receiving each regimen met criteria for response. GS-1101 is currently being investigated in the phase 2 setting as a single agent and in the upfront setting for elderly patients in combination with rituximab. FDA registration studies with GS-1101 are also soon to begin in relapsed CLL.
Ibrutinib (formerly PCI-32765)
Ibrutinib (Figure 3A-B) is an irreversible inhibitor of Btk. Details of the compound, an elegant pharmacodynamic probe to examine drug occupancy of BTK enzyme in the cell, and preclinical efficacy in spontaneous dog lymphoma have been reported.95 These were followed by preclinical data supporting its exploration in CLL.72,73 The initial phase 1 study with this agent examined dose escalation in various B-cell malignancies. In this study, 47 patients were enrolled, including 15 patients with CLL. Objective responses were observed in 9 of these 15 patients. As seen with other BCR antagonists, lymphocytosis was observed that diminished either during time off drug (when an interrupted schedule was used) or with continued treatment. Ibrutinib was well tolerated at all dose levels examined, with only 5 of 47 patients discontinuing therapy for toxicity. A dose of 420 mg daily was established for further study. This dose was based on > 90% occupancy of Btk using a novel drug probe developed by Pharmacyclics.96 No dose-limiting toxicity was identified at higher doses explored. In a subsequent phase 1b/2 study that is ongoing for patients with relapsed or refractory CLL,97 61 patients were enrolled at 2 different doses (420 mg and 840 mg). Extended therapy with ibrutinib was well tolerated with common grade 1 and 2 adverse events, including diarrhea, cough, fatigue, upper respiratory infections, nausea, fever, peripheral edema, myalgias, and petechiae/ecchymosis. Serious grade 3 or 4 infections were noted in 25% of patients in the 420-mg cohort and 29% patients in the 840-mg cohort. Although human XLA and mouse XID are characterized by neutrophil development and functional defects98,99 in addition to B-cell dysfunction, neutropenia has not been seen as a common side effect of ibrutinib, and neutrophil function has yet to be fully examined. Similar to other BCR signaling inhibitors, treatment is associated with an early increase in lymphocytosis that begins at approximately 1 week and persists for less than 8 months in the majority of patients. The overall rate of response at 10 months of follow-up using the International Workshop on CLL 200892 in the 420-mg cohort was 67% (1 complete response, 17 partial response) and 68% (28 partial response) in the 840-mg cohort. In addition, 6 (22%) and 8 (24%) of patients in the 420-mg and 840-mg cohort had a nodal response with persistent lymphocytosis. In virtually all patients with persistent lymphocytosis at 12 months, only nodal response is noted, suggesting that this may be a surrogate endpoint to evaluate adequacy of response. The 1-year estimated progression-free survival for the patients enrolled in this study is 86%.97 Studies are currently ongoing with this agent in combination with ofatumumab, bendamustine, or rituximab in the relapsed setting. Based on the success of ibrutinib as a single agent, FDA registration studies with this agent in relapsed disease are being planned.
Conclusions and looking forward
The disrupted BCR signaling in CLL makes kinase inhibition a rational therapeutic approach, and the clinical activity of the kinase inhibitors to date represents an exciting proof of concept. Molecules, such as GS-1101 and ibrutinib, clearly demonstrate promising leads based on dramatic responses and durable remissions observed to date in refractory CLL patients. Moving forward, these agents have taught us that old response criteria defining progressive disease based on lymphocytosis will have to be altered as the lymphocytosis associated with these agents is clearly not a sign of disease progression. National Comprehensive Cancer Network guidelines have already eliminated progressive lymphocytosis as a sign of disease progression in the presence of reduction of spleen and lymph node size to address this, and consensus guidelines from other groups are likely to follow. These drugs appear to have the benefit of low toxicity and continued efficacy that allows (and perhaps requires) prolonged dosing, as is done with imatinib. As experience thus far has shown durability of remissions, which for the most part are partial remissions, the importance of attaining a complete response and minimal residual disease-negative disease status, as is the current goal with chemoimmunotherapy, will also need to be re-examined both in the context of single-agent or combination use. For this same reason, the long-term safety of these agents must be assessed. Finally, as we gain more experience with these agents, we will be better able to predict which patients will respond and which may need combination therapy or alternative strategies targeting other BCR target proteins. More accurate molecular classification of our patients may also assist in the choice of drug, leading to more personalized care.
Acknowledgments
The authors thank Drs David Lucas, Natarajan Muthusamy, and Michael Grever for review of the final manuscript. They acknowledge the many publications not cited in this paper because of the citation number allowed by the journal.
This work was supported by the Leukemia & Lymphoma Society and the National Cancer Institute (P50 CA140158, PO1 CA95426, PO1 CA81534, 1K12 CA133250), Mr and Mrs Michael Thomas, the Harry Mangurian Foundation, and the D. Warren Brown Foundation.
National Institutes of Health
Authorship
Contribution: J.A.W. wrote the first draft, revised subsequent versions, and approved the final paper; and A.J.J. and J.C.B. organized the structure of the manuscript, reviewed, edited, and revised all versions, and approved the final paper.
Conflict-of-interest disclosure: J.C.B. has served as an unpaid consultant for Genentech, Xencor, Merck, Pharmacyclics, Emergent Biosciences, and Gilead, has been a consultant for Calistoga, and has residual milestone payments for success of GS-1101; these have been committed to charity. The remaining authors declare no competing financial interests.
Correspondence: John C. Byrd, 455 Wiseman Hall, 410 West 12th Ave, Columbus, OH 43210; e-mail: john.byrd@osumc.edu.