Comment on Blumenthal et al, page 965
Wnt5a and its receptor Frizzled-5 are up-regulated in human macrophages upon Mycobacterium tuberculosis infection, and their expression promotes IL-12 production and T-cell activation, demonstrating a previously undescribed link between Wnt signaling and innate and acquired immune responses.
The Wnt family of ligands consists of evolutionarily highly conserved glycoproteins that function in embryonic induction, cell polarity, and cell differentiation. Moreover, disregulation of Wnt signaling has been implicated in various cancers.1,2 Despite a significant amount of data on the 19 different Wnt proteins and their receptors, very little is known on how these ligand-receptor complexes may regulate an immune response.3 In this issue of Blood, Blumenthal and colleagues have defined a novel role for Wnt5a and its receptor Frizzled-5 in promoting a macrophage immune response to various stimuli including LPS and Mycobacterium tuberculosis.FIG1
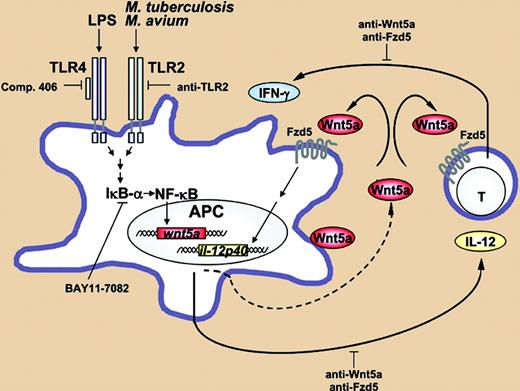
Model: Wnt5a and Fzd5 as modulators of the interplay between antigen-presenting cells and T lymphocytes. See the complete figure in the article beginning on page 965.
Model: Wnt5a and Fzd5 as modulators of the interplay between antigen-presenting cells and T lymphocytes. See the complete figure in the article beginning on page 965.
The studies stemmed from a microarray-based gene expression screen that revealed that Wnt5a mRNA expression was up-regulated 3- to 5-fold in human macrophages infected with M tuberculosis, or treated with LPS or Pam3CSK2. Of interest, the induction of Wnt5a by M tuberculosis and LPS was dependent on TLR2 and TLR4, respectively, and both stimuli required NF-κB activation. This is a previously undescribed pathway for inducing Wnt expression. The authors also established that the Wnt5a receptor Frizzled-5 was up-regulated in macrophages infected with M tuberculosis, although to a lesser extent relative to treatment with LPS. Moreover, unlike expression of Wnt5a, which was limited to macrophages and dendritic cells, stimulated lymphocytes also showed a 10-fold increase in expression of Frizzled-5 mRNA. In vivo studies showed the presence of Wnt5 mRNA and Frizzled-5 protein expression in granulomatous lesions isolated from pulmonary tuberculosis patients. This was an important finding since it demonstrates that Wnt5a and Frizzled-5 are expressed in the proper location (ie, the granuloma) to promote the host response to an M tuberculosis infection.
The crux of the study, however, is the functional link between Wnt5a/Frizzled-5 expression and macrophage and T-cell activation. The authors made the startling discovery that peripheral-blood mononuclear cells (PBMCs) isolated from healthy donors who were negative for “purified protein derivative of M tuberculosis” (PPD) were diminished in their production of PPD-induced IFN-γ in the presence of neutralizing anti-Wnt5a antibody. This suggests that the antigen-induced release of this essential cytokine from T cells was dependent on the production of Wnt5a by antigen-presenting cells (APCs). A similar decrease in IFN-γ production by PPD-treated PBMCs was observed following treatment with a neutralizing anti–Frizzled-5 antibody. The authors go on to show that IL-12 released by human macrophages upon M tuberculosis infection is blocked by anti-Wnt5a neutralizing antibody. The presence of IL-12 at the site of infection is essential for IFN-γ production by activated T cells (see figure).
These exciting studies suggest a new order of complexity in regulating the bridge between the innate and acquired immune responses. Since pathogenic organisms including M tuberculosis release various factors that can modulate an immune response, one might predict that interfering with the production of Wnt5a, its interaction with Frizzled-5, or the downstream signaling from the receptor might be important mechanisms by which pathogens limit the acquired immune response. ▪

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal