Abstract
To assess the therapeutic value of sequential high-dose therapy (SHDT) including autologous stem cell transplantation in chronic lymphocytic leukemia (CLL) we performed a risk-matched comparison between 66 patients who had undergone a uniform SHDT regimen and a database of 291 patients treated conventionally. Matching variables were age, Binet stage, IgVH (variable region of the immunoglobulin heavy chain) gene mutational status, and lymphocyte count. Forty-four pairs fully matched for all 4 variables were identified. Patient groups were well balanced for additional risk factors including adverse genomic abnormalities and CD38 expression. With an overall median follow-up time of 70 and 86 months, respectively, survival was significantly longer for the SHDT patients than for the conventionally treated patients when calculated from diagnosis (hazard ratio [HR] 0.39; P = .03 [log rank]) or from study entry (HR 0.32; P = .006). The benefit for the SHDT group remained significant when the analyses were restricted to those 58 patients who had an unmutated VH status. Cox regression analysis confirmed SHDT as independent favorable prognostic factor for survival from diagnosis (HR 0.38, P = .04) as well as from study entry (HR 0.38, P = .03). These data suggest a survival benefit for patients with poor-risk CLL receiving SHDT during the course of their disease. (Blood. 2004;103:2850-2858)
Introduction
Autologous stem cell transplantation (SCT) has been increasingly used in younger patients with chronic lymphocytic leukemia (CLL) during the past decade. Although early studies indicated that this approach might be a potentially curative treatment in this otherwise incurable disease,1-4 more mature data do not support that SCT can result in eradication of the leukemic clone in a relevant proportion of patients.5-8 Relapse appears to be inevitable, especially in those individuals with high-risk CLL as defined by an unmutated VH (variable region of the heavy chain) gene status.9 Not being curative, however, does not necessarily mean not being beneficial, but pursuing a toxic and expensive procedure like SCT requires evidence of its clinical value in terms of improved remission duration or survival. Comparative studies between SCT and standard palliative treatment are lacking to date. Since the results of prospective randomized studies will not be available in the near future, we sought to retrospectively compare the outcome of patients who had undergone SCT with conventionally treated controls by a risk-matched analysis.
The assignment of proper controls to SCT recipients has been difficult in CLL because the natural course of the disease is highly variable. To this end, recent developments in genetic risk assessment have greatly facilitated the prediction of the patients' individual prognosis. Relevant prognostic parameters are genomic abnormalities as determined by fluorescence in situ hybridization (FISH), CD38 expression, and, in particular, the mutational status of the VH gene,10-13 which allow segregation of patients with indolent and aggressive course, with a high degree of accuracy. Taking advantage of these novel tools, we performed a risk-matched comparison between SCT and standard treatment using 2 large data sets for which comprehensive information on genetic, biologic, and clinical characteristics was available.
Patients and methods
Patients
Two databases containing data on consecutive patients were screened for cases having complete information available on the following variables: morphologic and immunologic diagnosis of B-cell chronic lymphocytic leukemia according to the REAL/WHO (Revised European-American Classification of Lymphoid Neoplasms/World Health Organization) classification; VH mutational status; age; Binet stage; lymphocyte count; time of diagnosis; time, type, and duration of individual CLL-specific treatment; follow-up for survival; cause of death. The SCT database consisted of 73 consecutive patients who had undergone sequential high-dose therapy (SHDT) including SCT between January 1993 and January 2001 according to a uniform protocol at the Universities of Kiel (n = 67) and Heidelberg (n = 6).3 The transplantation strategy used was characterized by a straightforward dose-escalating design (ie, the cytoreductive treatment prior to transplantation was designed to allow for successful and effective SCT). Briefly, patients were eligible for SHDT if they had a history of either symptomatic disease according to the revised National Cancer Institute (NCI) guidelines (Binet stage B with B symptoms and/or massive or progressive lymphadenopathy or splenomegaly, Binet stage C)14 or by asymptomatic disease with adverse prognostic factors, namely lymphocyte count higher than 50 × 109/L, lymphocyte doubling time (LDT) shorter than 12 months, diffuse bone marrow (BM) infiltration, and/or presence of a 11q22.3-q23.1 deletion. After initial cytoreduction with 1 to 6 cycles of conventional chemotherapy (CHOP[cyclophosphamide/doxorubicin/vincristine/prednisolone], fludarabine, or fludarabine/cyclophosphamide), autologous hematopoietic stem cells where mobilized with the Dexa-BEAM regimen (dexamethasone, BCNU [1,3-bis(2-chloroethyl)-1-nitrosourea], etoposide, cytarabine, melphalan), as previously described.3 The collection products underwent positive, negative, or positive/negative immunomagnetic purging with the MaxSep, Isolex, or Clinimacs systems. Subsequent myeloablative therapy consisted of fractionated total body irradiation (TBI; 12-14.4Gy) and cyclophosphamide (total dose 2 × 60 mg/kg) followed by reinfusion of at least 2 × 106 immunoselected CD34+ cells/kg. The protocol was approved by the responsible institutional review boards.
The conventional treatment (CT) database comprised 325 consecutive patients enrolled at the University of Heidelberg between October 1990 and August 1998 in a study of the impact of genomic aberrations on survival. Detailed clinical characteristics together with cytogenetic and molecular data were described previously.10,13 Two hundred forty-eight patients were untreated, 39 patients had received 1, and 38 patients had received 2 or more chemotherapeutic regimens prior to study entry. Standard clinical criteria14,15 were used for defining the indication for initiation of therapy, which was usually alkylator based at first line and mostly fludarabine at second line.
Altogether, 66 SHDT cases and 291 CT cases fulfilled the criteria mentioned at the top of this section for subsequent identification of matches. Essential characteristics of these patients are summarized in Table 1.
Patient characteristics of the data sets prior to matching
. | SHDT . | CT . |
|---|---|---|
| n | 66 | 291 |
| Female sex (%) | 16 (24) | 116 (40) |
| Age at study entry, y (range) | 51 (29-64) | 63 (30-87) |
| Binet stage at study entry (%) | ||
| A | 19 (29) | 155 (53) |
| B | 30 (45) | 89 (31) |
| C | 17 (26) | 46 (16) |
| Not available | 0 | 1 (0) |
| Lymphocyte count higher than 50 × 109/L (%) | 36 (55) | 87 (30) |
| VH homology less than 98% (%) | 22 (33) | 133 (46) |
| Deaths (%) | 8 (12) | 104 (36) |
| Median survival from diagnosis, mos | —* | 105 |
| Median follow-up, mos | 65 (11-185) | 68 (1-186) |
. | SHDT . | CT . |
|---|---|---|
| n | 66 | 291 |
| Female sex (%) | 16 (24) | 116 (40) |
| Age at study entry, y (range) | 51 (29-64) | 63 (30-87) |
| Binet stage at study entry (%) | ||
| A | 19 (29) | 155 (53) |
| B | 30 (45) | 89 (31) |
| C | 17 (26) | 46 (16) |
| Not available | 0 | 1 (0) |
| Lymphocyte count higher than 50 × 109/L (%) | 36 (55) | 87 (30) |
| VH homology less than 98% (%) | 22 (33) | 133 (46) |
| Deaths (%) | 8 (12) | 104 (36) |
| Median survival from diagnosis, mos | —* | 105 |
| Median follow-up, mos | 65 (11-185) | 68 (1-186) |
SHDT indicates sequential high-dose therapy including autologous stem cell transplantation; CT, conventional chemotherapy; —, not reached.
Median values and ranges are given for age at study entry.
Last observed death at 139 months (estimated survival probability: 0.61).
Interphase cytogenetic analysis
Interphase cytogenetic analysis was performed centrally for both data sets with a probe set allowing to screen for the following deletions, partial trisomies, and translocations: +(3q26), del(6q21), +(8q24), del(11q22-q23), t(11;14)(q13;q32), +(12q13), del(13q14), t(14;18)(q32;q21), t(14q32), and del(17p13) as previously described.10 Samples for interphase cytogenetics were available only for a part of the SHDT cases.
Immunoglobulin VH (IgVH) identification, sequencing, and mutational analysis
Details of the VH amplification and sequencing methodologies were published elsewhere.9,13 Basically, genomic DNA was isolated from blood or bone marrow samples and subjected to amplification of the VDJ (variable/diversity/joining) gene rearrangements by polymerase chain reaction (PCR) using appropriate (5′-VH) framework region (FR) I and (3′-JH) FRIV primers. The PCR products were spin column-purified (Qiagen, Hilden, Germany) and directly sequenced using dye terminator chemistry (Big Dye Kit; Applied Biosystems, Weiterstadt, Germany) with a genetic analyzer (models ABI 310 or ABI 377; Applied Biosystems). All analyses were performed in 2 to 4 replicates using independent PCR products.
The VDJ nucleotide sequences where aligned to published germ line VH, D, and JH genes (EMBL/GenBank, V-base directory; IMGT database, http://imgt.cines.fr/cgi-bin/IMGTdnap.jv?livret = 0&Option = humanIg). Mutational status was calculated as percent deviation from the closest matching germ line VH segment. Sequences showing 98% or more homology to the nearest germ line gene were assigned as unmutated. To ensure data quality and consistency a proportion of samples was analyzed twice in 2 independent institutions (Kiel and Ulm) and revealed identical results.
CD38 expression
CD38 expression was analyzed independently in both institutions in a proportion of cases from fresh or cryopreserved cells by triple-color flow cytometry analysis as described previously.13 In brief, cells were stained with anti-CD5, anti-CD19, and anti-CD38 using appropriate antibody conjugates. Isotype-matched negative controls were used in all assays to determine positive from negative cells. The percentage of CD38+ cells was measured in the CD19/CD5 positive fraction. Flow cytometric analyses were performed on FACScan flow cytometers (Becton Dickinson, Heidelberg, Germany).
Risk set matching
For risk set matching we used the “optimal” method of Rosenbaum16 as supplied by Bergstralh and Kosanke.17 The variables chosen for matching are listed in Table 2. These factors had been identified as being of independent prognostic significance for survival in a preceding analysis of the CT data set used here. In that analysis, VH homology had the strongest prognostic impact apart from distinct cytogenetic abnormalities (17p deletion and 11q deletion).13 Cytogenetic parameters were not considered as matching variables in the present study due to missing data in the SHDT cohort. For age, a maximum difference of 5 years was tolerated; for the other variables only exact matches were accepted. For definition of study entry see “Statistical analysis.”
Matching variables
Variable . | Categories . |
|---|---|
| Age at study entry | Years* |
| Binet stage at study entry | A |
| B | |
| C | |
| VH homology | ≥ 98% |
| < 98% | |
| Lymphocyte count | < 50 × 109/L |
| ≥ 50 × 109/L |
Variable . | Categories . |
|---|---|
| Age at study entry | Years* |
| Binet stage at study entry | A |
| B | |
| C | |
| VH homology | ≥ 98% |
| < 98% | |
| Lymphocyte count | < 50 × 109/L |
| ≥ 50 × 109/L |
A maximum difference of 5 years was tolerated.
Statistical analysis
The primary end point for comparison of the 2 risk-matched treatment groups identified by the matching procedure was survival from the time of diagnosis. Additional end points were survival from time of study entry, survival from time of symptomatic disease, and time from symptomatic disease to re-treatment. Study entry was defined as time of referral for high-dose therapy for the SHDT group and as time of referral for genetic study for the CT group. Symptomatic disease was defined as time of commencement of first-line therapy according to the criteria mentioned in “Patients.” Time to re-treatment was defined as time from first-line conventional treatment for symptomatic disease (CT group) or start of SHDT (symptomatic SHDT patients) to re-treatment or CLL-related death. CT patients without any treatment as well as SHDT patients who underwent treatment prior to development of symptoms were not considered for analyses related to time of symptomatic disease. Patients undergoing allogeneic SHDT for salvage treatment were censored as alive at the time of transplantation. The survival time distributions were plotted using Kaplan-Meier estimates. The median duration of follow-up was calculated according to the method of Korn.18 Treatment-related (SHDT vs CT) survival time distributions were compared by log-rank tests. Proportional hazards models (Cox regression) were fitted to investigate the confounding effects of prognostic factors. As explanatory variables for survival from diagnosis, treatment (SHDT vs CT), VH mutational status, presence of 11q deletion, age at diagnosis, and stage at diagnosis were included in the regression model. For survival from study entry, treatment (SHDT vs CT), VH mutational status, presence of 11q deletion, age at study entry, stage at study entry, lines of pretreatment before study entry, and lymphocyte count at study entry were considered. Missing data were estimated using a multiple-imputation technique with 10 random draws.
Groupwise comparisons of distributions of clinical, laboratory, and genetic data were performed with the Kruskal-Wallis or Mann-Whitney U test (quantitative variables) and Fisher exact test (categoric variables). All tests were 2 sided. An effect was considered statistically significant at a P value less than .05. To provide quantitative information on the relevance of statistically significant results, 95% confidence intervals for hazard ratios were computed. Pairwise correlations were estimated by the Pearson product moment correlation coefficient. The statistical analyses were performed with the following software packages: StatXact, version 5 (Cytel Software Corporation, Cambridge, MA); SAS, release 8.1 (SAS Institute Inc, Cary, NC); S-Plus (Insightful Corp, Seattle, WA), together with the Design software library19 and GraphPad Prism versions 3.00 and 3.02 (GraphPad Software, San Diego, CA).
Results
Matching
A total of 44 pairs of patients fully matched for all 4 variables were identified. In both cohorts, 70% of the patients were in advanced stage, 66% showed an unmutated VH status, and 57% had a high lymphocyte count, indicating predominance of poor-risk cases. Patient groups were also well balanced in terms of genomic abnormalities, CD38 expression, age at diagnosis, stage at diagnosis, prevalence of symptoms at study entry, time from diagnosis to symptomatic disease, time from diagnosis to study entry, sex, pretreatment before study entry, and fludarabine exposure prior to study entry, but SHDT patients had a later time of study entry (Table 3).
Patient and disease characteristics of matches
. | SHDT . | CT . | P . |
|---|---|---|---|
| n | 44 | 44 | |
| Age at study entry,* y (range) | 52 (40-64) | 54 (44-63) | |
| Age at diagnosis, y (range) | 49 (38-62) | 52 (42-60) | .20 |
| Binet stage at study entry*(%) | |||
| A | 13 (30) | 13 (30) | |
| B | 21 (48) | 21 (48) | |
| C | 10 (23) | 10 (23) | |
| Binet stage at diagnosis (%) | .63 | ||
| A | 22 (50) | 22 (50) | |
| B | 15 (34) | 14 (32) | |
| C | 5 (11) | 2 (5) | |
| Not available | 2 (5) | 6 (14) | |
| Lymphocyte count higher than 50 × 109/L* (%) | 25 (57) | 25 (57) | |
| VH homology less than 98%* (%) | 15 (34) | 15 (34) | |
| 11q deletion (%) | 7/34 (21) | 12 (27) | .60 |
| 17p deletion | 0 | 0 | |
| CD38+ more than 30% of CLL cells (%) | 16/31 (52) | 12/25 (48) | .99 |
| Female sex (%) | 12 (27) | 15 (34) | .64 |
| Symptomatic disease at study entry (%) | 32 (73) | 34 (77) | .81 |
| Months from diagnosis to symptoms | |||
| All patients, log rank | 18 | 30 | .12 |
| Symptomatic patients, Mann-Whitney U (range) | 7 (1-89) | 12 (1-136) | .65 |
| Months from diagnosis to study entry (range) | 16 (0-113) | 22 (0-106) | .52 |
| Chemotherapy naive at study entry (%) | 32 (73) | 28 (63) | .49 |
| Previous exposure to fludarabine (%) | 2 (5) | 2 (5) | .99 |
| Year of study entry (range) | 1998 (1994-2001) | 1994 (1991-1998) | <.001 |
. | SHDT . | CT . | P . |
|---|---|---|---|
| n | 44 | 44 | |
| Age at study entry,* y (range) | 52 (40-64) | 54 (44-63) | |
| Age at diagnosis, y (range) | 49 (38-62) | 52 (42-60) | .20 |
| Binet stage at study entry*(%) | |||
| A | 13 (30) | 13 (30) | |
| B | 21 (48) | 21 (48) | |
| C | 10 (23) | 10 (23) | |
| Binet stage at diagnosis (%) | .63 | ||
| A | 22 (50) | 22 (50) | |
| B | 15 (34) | 14 (32) | |
| C | 5 (11) | 2 (5) | |
| Not available | 2 (5) | 6 (14) | |
| Lymphocyte count higher than 50 × 109/L* (%) | 25 (57) | 25 (57) | |
| VH homology less than 98%* (%) | 15 (34) | 15 (34) | |
| 11q deletion (%) | 7/34 (21) | 12 (27) | .60 |
| 17p deletion | 0 | 0 | |
| CD38+ more than 30% of CLL cells (%) | 16/31 (52) | 12/25 (48) | .99 |
| Female sex (%) | 12 (27) | 15 (34) | .64 |
| Symptomatic disease at study entry (%) | 32 (73) | 34 (77) | .81 |
| Months from diagnosis to symptoms | |||
| All patients, log rank | 18 | 30 | .12 |
| Symptomatic patients, Mann-Whitney U (range) | 7 (1-89) | 12 (1-136) | .65 |
| Months from diagnosis to study entry (range) | 16 (0-113) | 22 (0-106) | .52 |
| Chemotherapy naive at study entry (%) | 32 (73) | 28 (63) | .49 |
| Previous exposure to fludarabine (%) | 2 (5) | 2 (5) | .99 |
| Year of study entry (range) | 1998 (1994-2001) | 1994 (1991-1998) | <.001 |
Matching variables.
Treatment
Treatment details are described in Tables 4 and 5. In the CT group, 10 patients (23%) remained untreated throughout the duration of follow-up. In the 34 patients (77%) developing symptoms, first-line treatment essentially consisted of alkylating agents (chlorambucil- or cyclophosphamide-based regimens; 65%) or of fludarabine-containing regimens (14%). Second-line treatment was necessary in 27 CT patients (63%) and again comprised alkylating agents (35%) and purine analoges (28%). Altogether, CT patients received a median number of 3 (range, 0-9) treatment lines, which was significantly more than in the SHDT group (1; range, 1-5 treatment lines, including salvage therapy after SHDT; P = .02, Mann-Whitney U test). Fludarabine-based regimens were given in 74% of treated patients in the CT group but only in 33% of the SHDT group at any time during the course of their disease (P = .03).
Clinical course and treatment details of matches for SHDT patients
UPN . | Sex/age, y . | % VH homology . | Binet stage . | Lymphocyte count higher than 50 × 109/L . | FISH karyotype . | Time to symptoms, mos . | Time to study entry, mos . | First-line therapy . | Second-line therapy . | Additional salvage therapy . | Outcome (follow-up from diagnosis) . | Cause of death . |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 97021 | M/56 | 100 | C | Yes | + 12 | 85 | 114 | Clb × 22 | SHDT (Flud × 3) | No | Alive (185+) | ND |
| 98024 | F/54 | 100 | C | Yes | NA | 36 | 92 | Flud × 3 | SHDT (CHOP × 6) | No | Dead (109) | TRM |
| 98006 | M/54 | 100 | C | Yes | +12 | 73 | 78 | Clb × 2 | SHDT (Flud × 2) | No | Alive (130+) | ND |
| 2000098 | F/51 | 100 | C | Yes | NA | 18 | 18 | SHDT (CHOP × 8) | Clb × 3 | No | Dead (48) | CLL |
| 99122 | F/51 | 98.3 | C | Yes | 13q14 | 7 | 7 | SHDT (CHOP × 6) | No | No | Alive (47+) | ND |
| 98078 | F/49 | 100 | C | Yes | 11q23; t(2;13) | 1 | 3 | Clb × 4 | SHDT (CHOP × 2) | No | Alive (45+) | ND |
| 2000136 | F/48 | 100 | C | Yes | Normal | 0 | 0 | SHDT (CHOP × 3) | No | No | Alive (27+) | ND |
| 2000038 | M/57 | 100 | C | No | NA | 26 | 26 | SHDT (Flud × 3) | No | No | Alive (46+) | ND |
| 97076 | M/60 | 100 | B | Yes | 6q21; 13q14 | 24 | 83 | Clb × 31 | SHDT (CHOP × 3) | Flud × 1; CD20 × 2; prednisolone | Dead (139) | CLL |
| 99137 | M/59 | 100 | B | Yes | NA | 1 | 89 | Clb × 21 | SHDT (CHOP × 3) | No | Alive (62+) | ND |
| 2000101 | M/57 | 100 | B | Yes | Normal | 48 | 48 | SHDT (FC × 2) | No | No | Alive (66+) | ND |
| 98075 | M/56 | 100 | B | Yes | 13q14 | 3 | 3 | SHDT (CHOP × 3) | No | No | Alive (49+) | ND |
| 97099 | F/53 | 100 | B | Yes | 13q14 | 51 | 51 | SHDT (CHOP × 4) | No | No | Alive (108+) | ND |
| 97096 | F/52 | 100 | B | Yes | Normal | 1 | 38 | COP × 6 | Clb × 2 | Flud × 5; SHDT (FC × 1); FC × 1 | Dead (90) | CLL |
| 98105 | M/51 | 100 | B | Yes | 11q23; 13q14 | 7 | 15 | SHDT (CHOP × 6) | No | No | Alive (64+) | ND |
| HD006 | M/50 | 100 | B | Yes | +8q24; 11q23 | 48 | 48 | SHDT (CHOP × 3) | No | No | Alive (88+) | ND |
| 99039 | M/49 | 100 | B | Yes | 13q14 | 1 | 1 | SHDT (CHOP × 3) | No | No | Alive (43+) | ND |
| 98099 | M/49 | 98.7 | B | Yes | NA | 2 | 97 | Clb × 6 | SHDT (CHOP × 6) | No | Alive (139+) | ND |
| 230 | M/48 | 100 | B | Yes | 13q14 | 8 | 35 | Clb × 19 | SHDT (CHOP × 2) | No | Alive (112+) | ND |
| 97031 | M/56 | 100 | B | No | +12 | 2 | 2 | SHDT (CHOP × 3) | No | No | Alive (70+) | ND |
| 2000050 | M/51 | 99.2 | B | No | NA | 1 | 9 | MCP × 6 | SHDT (no) | No | Alive (26+) | ND |
| 99076 | M/47 | 100 | B | No | 11q23; 13q14 | 1 | 1 | SHDT (CHOP × 3) | No | No | Dead (13) | TRM |
| 98049 | M/40 | 100 | B | No | Normal | 13 | 13 | SHDT (CHOP × 3) | No | No | Alive (34+) | ND |
| 2000092 | M/58 | 100 | A | Yes | +12 | NA | 12 | SHDT (FC × 2) | No | No | Alive (42+) | ND |
| 98002 | M/55 | 99.5 | A | Yes | 13q14 | NA | 15 | SHDT (CHOP × 3) | No | No | Alive (61+) | ND |
| 2000064 | M/53 | 100 | A | Yes | +12 | NA | 12 | SHDT (FC × 3) | No | No | Alive (33+) | ND |
| 98025 | M/58 | 100 | A | No | 11q23; 13q14 | NA | 69 | SHDT (Flud × 2) | No | No | Alive (112+) | ND |
| 2000061 | M/54 | 100 | A | No | Normal | NA | 11 | SHDT (FC × 1) | No | No | Alive (34+) | ND |
| 99102 | F/51 | 99.1 | A | No | 11q23 | NA | 11 | SHDT (CHOP × 2) | No | No | Alive (52+) | ND |
| 2000014 | M/64 | 97.4 | C | No | NA | 18 | 18 | SHDT (CHOP × 3) | No | No | Dead (38) | Mesothelioma |
| 256 | M/51 | 94.4 | C | No | Normal | 2 | 2 | SHDT (no) | No | No | Alive (79+) | ND |
| 2001020 | M/61 | 89 | B | Yes | NA | 16 | 47 | Clb × 15 | SHDT (Flud × 6) | No | Alive (64+) | ND |
| HD003 | F/46 | 87.2 | B | Yes | 13q14 | 62 | 76 | Clb × 22 | SHDT (no) | No | Alive (123+) | ND |
| 236 | M/54 | Low | B | No | NA | NA | 2 | SHDT (no) | No | No | Alive (79+) | ND |
| 99 | F/51 | 93.5 | B | No | Normal | 2 | 44 | Clb × 13 | SHDT (no) | Flud 5; CD20 × 4; FC × 2 | Alive (139+) | ND |
| 98073 | M/51 | 93.4 | B | No | +12 | 2 | 2 | SHDT (CHOP ×3) | No | No | Alive (49+) | ND |
| 206 | M/48 | 86.1 | B | No | Normal | 3 | 3 | SHDT (CHOP × 5) | No | No | Alive (88+) | ND |
| 99119 | M/57 | 93.6 | A | Yes | Normal | 89* | 89 | SHDT (CHOP × 4) | No | No | Alive (124+) | ND |
| 238 | F/49 | 91.7 | A | Yes | 11q23 | NA | 48 | SHDT (COP × 2) | No | No | Alive (134+) | ND |
| 176 | M/57 | 92 | A | No | Normal | NA | 16 | SHDT (no) | No | No | Alive (100+) | ND |
| 2000074 | M/57 | 93.7 | A | No | 13q14 | NA | 25 | SHDT (CHOP × 3) | No | No | Alive (58+) | ND |
| 97013 | M/54 | 89.5 | A | No | NA | 3† | 3 | SHDT (no) | No | No | Dead (11) | TRM |
| 271 | M/47 | 92.9 | A | No | Normal | NA | 8 | SHDT (no) | No | No | Alive (86+) | ND |
| 98069 | M/47 | 95.2 | A | No | +12 | NA | 7 | SHDT (CHOP × 3) | No | No | Alive (62+) | ND |
UPN . | Sex/age, y . | % VH homology . | Binet stage . | Lymphocyte count higher than 50 × 109/L . | FISH karyotype . | Time to symptoms, mos . | Time to study entry, mos . | First-line therapy . | Second-line therapy . | Additional salvage therapy . | Outcome (follow-up from diagnosis) . | Cause of death . |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 97021 | M/56 | 100 | C | Yes | + 12 | 85 | 114 | Clb × 22 | SHDT (Flud × 3) | No | Alive (185+) | ND |
| 98024 | F/54 | 100 | C | Yes | NA | 36 | 92 | Flud × 3 | SHDT (CHOP × 6) | No | Dead (109) | TRM |
| 98006 | M/54 | 100 | C | Yes | +12 | 73 | 78 | Clb × 2 | SHDT (Flud × 2) | No | Alive (130+) | ND |
| 2000098 | F/51 | 100 | C | Yes | NA | 18 | 18 | SHDT (CHOP × 8) | Clb × 3 | No | Dead (48) | CLL |
| 99122 | F/51 | 98.3 | C | Yes | 13q14 | 7 | 7 | SHDT (CHOP × 6) | No | No | Alive (47+) | ND |
| 98078 | F/49 | 100 | C | Yes | 11q23; t(2;13) | 1 | 3 | Clb × 4 | SHDT (CHOP × 2) | No | Alive (45+) | ND |
| 2000136 | F/48 | 100 | C | Yes | Normal | 0 | 0 | SHDT (CHOP × 3) | No | No | Alive (27+) | ND |
| 2000038 | M/57 | 100 | C | No | NA | 26 | 26 | SHDT (Flud × 3) | No | No | Alive (46+) | ND |
| 97076 | M/60 | 100 | B | Yes | 6q21; 13q14 | 24 | 83 | Clb × 31 | SHDT (CHOP × 3) | Flud × 1; CD20 × 2; prednisolone | Dead (139) | CLL |
| 99137 | M/59 | 100 | B | Yes | NA | 1 | 89 | Clb × 21 | SHDT (CHOP × 3) | No | Alive (62+) | ND |
| 2000101 | M/57 | 100 | B | Yes | Normal | 48 | 48 | SHDT (FC × 2) | No | No | Alive (66+) | ND |
| 98075 | M/56 | 100 | B | Yes | 13q14 | 3 | 3 | SHDT (CHOP × 3) | No | No | Alive (49+) | ND |
| 97099 | F/53 | 100 | B | Yes | 13q14 | 51 | 51 | SHDT (CHOP × 4) | No | No | Alive (108+) | ND |
| 97096 | F/52 | 100 | B | Yes | Normal | 1 | 38 | COP × 6 | Clb × 2 | Flud × 5; SHDT (FC × 1); FC × 1 | Dead (90) | CLL |
| 98105 | M/51 | 100 | B | Yes | 11q23; 13q14 | 7 | 15 | SHDT (CHOP × 6) | No | No | Alive (64+) | ND |
| HD006 | M/50 | 100 | B | Yes | +8q24; 11q23 | 48 | 48 | SHDT (CHOP × 3) | No | No | Alive (88+) | ND |
| 99039 | M/49 | 100 | B | Yes | 13q14 | 1 | 1 | SHDT (CHOP × 3) | No | No | Alive (43+) | ND |
| 98099 | M/49 | 98.7 | B | Yes | NA | 2 | 97 | Clb × 6 | SHDT (CHOP × 6) | No | Alive (139+) | ND |
| 230 | M/48 | 100 | B | Yes | 13q14 | 8 | 35 | Clb × 19 | SHDT (CHOP × 2) | No | Alive (112+) | ND |
| 97031 | M/56 | 100 | B | No | +12 | 2 | 2 | SHDT (CHOP × 3) | No | No | Alive (70+) | ND |
| 2000050 | M/51 | 99.2 | B | No | NA | 1 | 9 | MCP × 6 | SHDT (no) | No | Alive (26+) | ND |
| 99076 | M/47 | 100 | B | No | 11q23; 13q14 | 1 | 1 | SHDT (CHOP × 3) | No | No | Dead (13) | TRM |
| 98049 | M/40 | 100 | B | No | Normal | 13 | 13 | SHDT (CHOP × 3) | No | No | Alive (34+) | ND |
| 2000092 | M/58 | 100 | A | Yes | +12 | NA | 12 | SHDT (FC × 2) | No | No | Alive (42+) | ND |
| 98002 | M/55 | 99.5 | A | Yes | 13q14 | NA | 15 | SHDT (CHOP × 3) | No | No | Alive (61+) | ND |
| 2000064 | M/53 | 100 | A | Yes | +12 | NA | 12 | SHDT (FC × 3) | No | No | Alive (33+) | ND |
| 98025 | M/58 | 100 | A | No | 11q23; 13q14 | NA | 69 | SHDT (Flud × 2) | No | No | Alive (112+) | ND |
| 2000061 | M/54 | 100 | A | No | Normal | NA | 11 | SHDT (FC × 1) | No | No | Alive (34+) | ND |
| 99102 | F/51 | 99.1 | A | No | 11q23 | NA | 11 | SHDT (CHOP × 2) | No | No | Alive (52+) | ND |
| 2000014 | M/64 | 97.4 | C | No | NA | 18 | 18 | SHDT (CHOP × 3) | No | No | Dead (38) | Mesothelioma |
| 256 | M/51 | 94.4 | C | No | Normal | 2 | 2 | SHDT (no) | No | No | Alive (79+) | ND |
| 2001020 | M/61 | 89 | B | Yes | NA | 16 | 47 | Clb × 15 | SHDT (Flud × 6) | No | Alive (64+) | ND |
| HD003 | F/46 | 87.2 | B | Yes | 13q14 | 62 | 76 | Clb × 22 | SHDT (no) | No | Alive (123+) | ND |
| 236 | M/54 | Low | B | No | NA | NA | 2 | SHDT (no) | No | No | Alive (79+) | ND |
| 99 | F/51 | 93.5 | B | No | Normal | 2 | 44 | Clb × 13 | SHDT (no) | Flud 5; CD20 × 4; FC × 2 | Alive (139+) | ND |
| 98073 | M/51 | 93.4 | B | No | +12 | 2 | 2 | SHDT (CHOP ×3) | No | No | Alive (49+) | ND |
| 206 | M/48 | 86.1 | B | No | Normal | 3 | 3 | SHDT (CHOP × 5) | No | No | Alive (88+) | ND |
| 99119 | M/57 | 93.6 | A | Yes | Normal | 89* | 89 | SHDT (CHOP × 4) | No | No | Alive (124+) | ND |
| 238 | F/49 | 91.7 | A | Yes | 11q23 | NA | 48 | SHDT (COP × 2) | No | No | Alive (134+) | ND |
| 176 | M/57 | 92 | A | No | Normal | NA | 16 | SHDT (no) | No | No | Alive (100+) | ND |
| 2000074 | M/57 | 93.7 | A | No | 13q14 | NA | 25 | SHDT (CHOP × 3) | No | No | Alive (58+) | ND |
| 97013 | M/54 | 89.5 | A | No | NA | 3† | 3 | SHDT (no) | No | No | Dead (11) | TRM |
| 271 | M/47 | 92.9 | A | No | Normal | NA | 8 | SHDT (no) | No | No | Alive (86+) | ND |
| 98069 | M/47 | 95.2 | A | No | +12 | NA | 7 | SHDT (CHOP × 3) | No | No | Alive (62+) | ND |
UPN indicates unique patient number; M, male; SHDT, sequential high-dose therapy: cytoreduction, DexaBEAM, total body irradiation/CY + autotransplant; the cytoreductive regimen prior to DexaBEAM is given in parentheses; Clb, chlorambucil with or without prednisolone; Flud, fludarabine; ND, not dead; F, female; NA, not applicable; CHOP, cyclophosphamide, doxorubicin, vincristine, prednisolone; TRM, treatment-related mortality; CD20, rituximab; FC, fludarabine and cyclophosphamide; COP, cyclophosphamide, vincristine, prednisolone; MCP, mitoxantron.
B symptoms.
Leukemic pericardial involvement.
Clinical course and treatment details of matches for CT patients
UPN . | Sex/age, y . | % VH homology . | Binet stage at study . | Lymphocyte count higher than 50 × 109/L . | FISH karyotype . | Time to symptoms, mos . | Time to study entry, mos . | First-line therapy . | Second-line therapy . | Additional salvage therapy . | Outcome (follow-up from diagnosis) . | Cause of death . |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 91PB024 | M/57 | 100 | C | Yes | 13q14 | 48 | 93 | Clb × 24 | COP × 2 | Flud × 12; COP × 1 | Dead (134) | CLL |
| 95PB021 | F/55 | 100 | C | Yes | 11q23 | 1 | 24 | COP × 7 | Flud × 7 | Idarubicine × 3; 2CDA × 2; FE × 1; Paclitaxel × 1 | Dead (37) | CLL |
| 95PB324 | M/54 | 100 | C | Yes | 6q21; 11q23; 13q14 | 5 | 17 | Clb × 5 | Flud × 1 | No | Dead (45) | CLL |
| 96PB959 | M/52 | 100 | C | Yes | 13q14 | 46 | 69 | Clb × 5 | Clb × 3 | COP × 1; CHOP × 4; DexaBEAM × 1; CY × 1 | Alive (100+) | ND |
| 98PB547 | M/59 | 100 | C | Yes | Normal | 1 | 0 | Flud × 6 | No | No | Alive (10+) | ND |
| 91PB084 | F/47 | 100 | C | Yes | Normal | 2 | 9 | Clb × 2 | Flud × 12 | Flud × 3; PmM × 5; Idarubicin × 2; COP × 2; 2CDA × 3; Paclitaxel × 1 | Dead (78) | CLL |
| 91LK1 | M/45 | 100 | C | Yes | 11q23 | 0 | 16 | Clb × 10 | COP × 7 | CY × 4; Flud × 1; IMVP16 × 2 | Dead (21) | CLL |
| 91PB079 | M/58 | 99.3 | C | No | Normal | 5 | 23 | Clb × 4 | COP × 4 | PmM × 4; Flud × 9; VP16 × 2; CHOP × 1 | Dead (66) | Pulmonary embolism |
| 93PB212 | F/61 | 98.6 | B | Yes | 11q23; 13q14 | 13 | 14 | COP × 3 | Idarubicine × 3 | Flud × 4; Clb × 4 | Alive (78+) | ND |
| 92PB198 | F/59 | 99.6 | B | Yes | Normal | 85 | 90 | Clb × 2 | Flud × 8 | Flud × 2; CD20 × 1; 2CDA × 1; pentostatin × 1 | Alive (166+) | ND |
| 96PB816 | M/58 | 100 | B | Yes | 13q14 | 51 | 53 | Flud × 2 | COP × 6 | Clb × 2; COP × 3; DexaBEAM × 1; Clb × 1; CY × 1 | Alive (85+) | ND |
| 91PB132 | M/56 | 99.5 | B | Yes | Normal | 1 | 12 | Clb × 3 | COP × 2 | Flud × 4 | Dead (34) | Infection |
| 93PB194 | M/55 | 100 | B | Yes | 11q23 | 1 | 2 | COP × 4 | Flud × 6 | Flud × 3; Clb × 4; FC × 2; CD20 × 4; Clb × 2; CHOP × 1; Bendamustin × 1 | Dead (70) | CLL |
| 93PB170 | F/58 | 100 | B | Yes | 11q23; + 12 | 1 | 3 | Clb × 3 | Flud × 6 | Flud × 5; FC × 4; Clb × 1; CD20 × 4; CHOP × 1; Vindesine × 1 | Dead (56) | CLL |
| 98PB909 | F/56 | 100 | B | Yes | 13q14 | 62 | 76 | Clb × 22 | No | No | Alive (123+) | ND |
| 95PB433 | M/56 | 100 | B | Yes | 6q21; 11q23; 13q14 | 1 | 44 | COP × 6 | Flud × 5 | Clb × 2; COP × 2; 2CDA × 2; COP × 2; CD20 × 4; CHOP × 2 | Dead (83) | CLL |
| 94PB125 | M/53 | 99.6 | B | Yes | 13q14 | 88 | 73 | Flud × 7 | CHOP × 6 | DexaBEAM × 2 | Alive (134+) | ND |
| 91PB140 | F/53 | 99.6 | B | Yes | 11q23; 13q14 | 1 | 20 | Clb × 3 | CHOP × 3 | COP × 3; PmM × 1; Flud × 4 | Dead (34) | CLL |
| 93PB091 | M/50 | 100 | B | Yes | 11q23; + 12 | 31 | 17 | Flud × 6 | Idarubicine × 2 | COP × 2; 2CDA × 3; Clb × 1 | Dead (64) | CLL |
| 95PB540 | M/60 | 100 | B | No | + 12 | 113 | 79 | Clb × 1 | No | No | Dead (119) | CLL |
| 91PB138 | M/51 | 100 | B | No | 11q23; 13q14 | 11 | 24 | COP × 3 | Flud × 6 | Flud × 6 | Dead (63) | CLL |
| 92PB195 | M/51 | 100 | B | No | + 12 | 1 | 1 | COP × 3 | 2CDA × 1 | Clb × 6; Flud × 2; 2CDA × 1; DexaBEAM × 1 | Alive (70+) | ND |
| 94PB156 | F/44 | 100 | B | No | + 12 | 2 | 1 | COP × 6 | Flud × 3 | DexaBEAM × 1; Flud × 3; FC × 2; MCP × 2; IMVP16 × 1 | Dead (49) | CLL |
| 91PB124 | F/49 | 99.5 | A | Yes | 11q23; 13q14 | 1 | 19 | COP × 3 | CHOP × 3 | Clb × 3; Flud × 3 | Dead (53) | CLL |
| 92PB073 | F/57 | 100 | A | Yes | Normal | 27 | 21 | Prednimustin × 3 | Flud × 6 | Flud × 1 | Dead (73) | CLL |
| 94PB068 | F/52 | 100 | A | Yes | 6q21; + 12 | 79 | 79 | Flud × 6 | Clb × 6 | Flud × 2 | Alive (143+) | ND |
| 98PB277 | M/58 | 100 | A | No | 11q23; 13q14 | NA | 0 | No | No | No | Alive (14+) | ND |
| 96PB515 | M/54 | 100 | A | No | 13q14 | NA | 15 | No | No | No | Alive (51+) | ND |
| 93PB142 | M/53 | 100 | A | No | 13q14 | 66 | 17 | Clb × 4 | Clb × 1 | No | Alive (86+) | ND |
| 94PB141 | M/59 | 90.8 | C | No | 13q14 | 107 | 107 | COP × 1 | Clb × 5 | No | Dead (152) | Meningitis |
| 95PB384 | M/52 | 97.1 | C | No | Normal | 1 | 41 | Clb × 4 | Flud × 6 | CHOP × 6; HAM × 2 | Dead (67) | CLL |
| 93PB103 | M/63 | 90.9 | B | Yes | 13q14 | NA | 103 | No | No | No | Alive (176+) | ND |
| 94PB169 | M/45 | 92.6 | B | Yes | 13q14 | 62 | 28 | Clb × 3 | No | No | Alive (86+) | ND |
| Prostate | ||||||||||||
| 93PB155 | M/54 | 87.5 | B | No | + 12 | 30 | 42 | COP × 1 | No | No | Dead (84) | cancer |
| 93PB165 | M/53 | 90.8 | B | No | + 12; 13q14 | 106 | 78 | Flud × 2 | No | No | Alive (145+) | ND |
| 96PB908 | M/52 | 91.4 | B | No | + 12 | 27 | 4 | Clb × 2 | No | No | Alive (35+) | ND |
| 98PB235 | Low | B | No | NA | 1 | No | No | No | Alive (18+) | ND | ||
| 96PB453 | F/61 | 94.3 | A | Yes | 13q14 | NA | 105 | No | No | No | Alive (143+) | ND |
| 91PB070 | F/49 | 94.2 | A | Yes | + 12 | 43 | 79 | Prednimustin × 3 | COP × 1 | Clb × 17 | Alive (175+) | ND |
| 98PB136 | F/57 | 90.1 | A | No | 13q14 | NA | 3 | No | No | No | Alive (19+) | ND |
| 95PB432 | F/57 | 92.3 | A | No | 13q14 | NA | 28 | No | No | No | Alive (75+) | ND |
| 94PB168 | F/54 | 95.5 | A | No | 13q14 | NA | 24 | No | No | No | Alive (85+) | ND |
| 96PB905 | M/48 | 96.6 | A | No | 13q14 | NA | 33 | No | No | No | Alive (65+) | ND |
| 97PB260 | F/47 | 94.5 | A | No | + 12 | NA | 0 | No | No | No | Alive (24+) | ND |
UPN . | Sex/age, y . | % VH homology . | Binet stage at study . | Lymphocyte count higher than 50 × 109/L . | FISH karyotype . | Time to symptoms, mos . | Time to study entry, mos . | First-line therapy . | Second-line therapy . | Additional salvage therapy . | Outcome (follow-up from diagnosis) . | Cause of death . |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 91PB024 | M/57 | 100 | C | Yes | 13q14 | 48 | 93 | Clb × 24 | COP × 2 | Flud × 12; COP × 1 | Dead (134) | CLL |
| 95PB021 | F/55 | 100 | C | Yes | 11q23 | 1 | 24 | COP × 7 | Flud × 7 | Idarubicine × 3; 2CDA × 2; FE × 1; Paclitaxel × 1 | Dead (37) | CLL |
| 95PB324 | M/54 | 100 | C | Yes | 6q21; 11q23; 13q14 | 5 | 17 | Clb × 5 | Flud × 1 | No | Dead (45) | CLL |
| 96PB959 | M/52 | 100 | C | Yes | 13q14 | 46 | 69 | Clb × 5 | Clb × 3 | COP × 1; CHOP × 4; DexaBEAM × 1; CY × 1 | Alive (100+) | ND |
| 98PB547 | M/59 | 100 | C | Yes | Normal | 1 | 0 | Flud × 6 | No | No | Alive (10+) | ND |
| 91PB084 | F/47 | 100 | C | Yes | Normal | 2 | 9 | Clb × 2 | Flud × 12 | Flud × 3; PmM × 5; Idarubicin × 2; COP × 2; 2CDA × 3; Paclitaxel × 1 | Dead (78) | CLL |
| 91LK1 | M/45 | 100 | C | Yes | 11q23 | 0 | 16 | Clb × 10 | COP × 7 | CY × 4; Flud × 1; IMVP16 × 2 | Dead (21) | CLL |
| 91PB079 | M/58 | 99.3 | C | No | Normal | 5 | 23 | Clb × 4 | COP × 4 | PmM × 4; Flud × 9; VP16 × 2; CHOP × 1 | Dead (66) | Pulmonary embolism |
| 93PB212 | F/61 | 98.6 | B | Yes | 11q23; 13q14 | 13 | 14 | COP × 3 | Idarubicine × 3 | Flud × 4; Clb × 4 | Alive (78+) | ND |
| 92PB198 | F/59 | 99.6 | B | Yes | Normal | 85 | 90 | Clb × 2 | Flud × 8 | Flud × 2; CD20 × 1; 2CDA × 1; pentostatin × 1 | Alive (166+) | ND |
| 96PB816 | M/58 | 100 | B | Yes | 13q14 | 51 | 53 | Flud × 2 | COP × 6 | Clb × 2; COP × 3; DexaBEAM × 1; Clb × 1; CY × 1 | Alive (85+) | ND |
| 91PB132 | M/56 | 99.5 | B | Yes | Normal | 1 | 12 | Clb × 3 | COP × 2 | Flud × 4 | Dead (34) | Infection |
| 93PB194 | M/55 | 100 | B | Yes | 11q23 | 1 | 2 | COP × 4 | Flud × 6 | Flud × 3; Clb × 4; FC × 2; CD20 × 4; Clb × 2; CHOP × 1; Bendamustin × 1 | Dead (70) | CLL |
| 93PB170 | F/58 | 100 | B | Yes | 11q23; + 12 | 1 | 3 | Clb × 3 | Flud × 6 | Flud × 5; FC × 4; Clb × 1; CD20 × 4; CHOP × 1; Vindesine × 1 | Dead (56) | CLL |
| 98PB909 | F/56 | 100 | B | Yes | 13q14 | 62 | 76 | Clb × 22 | No | No | Alive (123+) | ND |
| 95PB433 | M/56 | 100 | B | Yes | 6q21; 11q23; 13q14 | 1 | 44 | COP × 6 | Flud × 5 | Clb × 2; COP × 2; 2CDA × 2; COP × 2; CD20 × 4; CHOP × 2 | Dead (83) | CLL |
| 94PB125 | M/53 | 99.6 | B | Yes | 13q14 | 88 | 73 | Flud × 7 | CHOP × 6 | DexaBEAM × 2 | Alive (134+) | ND |
| 91PB140 | F/53 | 99.6 | B | Yes | 11q23; 13q14 | 1 | 20 | Clb × 3 | CHOP × 3 | COP × 3; PmM × 1; Flud × 4 | Dead (34) | CLL |
| 93PB091 | M/50 | 100 | B | Yes | 11q23; + 12 | 31 | 17 | Flud × 6 | Idarubicine × 2 | COP × 2; 2CDA × 3; Clb × 1 | Dead (64) | CLL |
| 95PB540 | M/60 | 100 | B | No | + 12 | 113 | 79 | Clb × 1 | No | No | Dead (119) | CLL |
| 91PB138 | M/51 | 100 | B | No | 11q23; 13q14 | 11 | 24 | COP × 3 | Flud × 6 | Flud × 6 | Dead (63) | CLL |
| 92PB195 | M/51 | 100 | B | No | + 12 | 1 | 1 | COP × 3 | 2CDA × 1 | Clb × 6; Flud × 2; 2CDA × 1; DexaBEAM × 1 | Alive (70+) | ND |
| 94PB156 | F/44 | 100 | B | No | + 12 | 2 | 1 | COP × 6 | Flud × 3 | DexaBEAM × 1; Flud × 3; FC × 2; MCP × 2; IMVP16 × 1 | Dead (49) | CLL |
| 91PB124 | F/49 | 99.5 | A | Yes | 11q23; 13q14 | 1 | 19 | COP × 3 | CHOP × 3 | Clb × 3; Flud × 3 | Dead (53) | CLL |
| 92PB073 | F/57 | 100 | A | Yes | Normal | 27 | 21 | Prednimustin × 3 | Flud × 6 | Flud × 1 | Dead (73) | CLL |
| 94PB068 | F/52 | 100 | A | Yes | 6q21; + 12 | 79 | 79 | Flud × 6 | Clb × 6 | Flud × 2 | Alive (143+) | ND |
| 98PB277 | M/58 | 100 | A | No | 11q23; 13q14 | NA | 0 | No | No | No | Alive (14+) | ND |
| 96PB515 | M/54 | 100 | A | No | 13q14 | NA | 15 | No | No | No | Alive (51+) | ND |
| 93PB142 | M/53 | 100 | A | No | 13q14 | 66 | 17 | Clb × 4 | Clb × 1 | No | Alive (86+) | ND |
| 94PB141 | M/59 | 90.8 | C | No | 13q14 | 107 | 107 | COP × 1 | Clb × 5 | No | Dead (152) | Meningitis |
| 95PB384 | M/52 | 97.1 | C | No | Normal | 1 | 41 | Clb × 4 | Flud × 6 | CHOP × 6; HAM × 2 | Dead (67) | CLL |
| 93PB103 | M/63 | 90.9 | B | Yes | 13q14 | NA | 103 | No | No | No | Alive (176+) | ND |
| 94PB169 | M/45 | 92.6 | B | Yes | 13q14 | 62 | 28 | Clb × 3 | No | No | Alive (86+) | ND |
| Prostate | ||||||||||||
| 93PB155 | M/54 | 87.5 | B | No | + 12 | 30 | 42 | COP × 1 | No | No | Dead (84) | cancer |
| 93PB165 | M/53 | 90.8 | B | No | + 12; 13q14 | 106 | 78 | Flud × 2 | No | No | Alive (145+) | ND |
| 96PB908 | M/52 | 91.4 | B | No | + 12 | 27 | 4 | Clb × 2 | No | No | Alive (35+) | ND |
| 98PB235 | Low | B | No | NA | 1 | No | No | No | Alive (18+) | ND | ||
| 96PB453 | F/61 | 94.3 | A | Yes | 13q14 | NA | 105 | No | No | No | Alive (143+) | ND |
| 91PB070 | F/49 | 94.2 | A | Yes | + 12 | 43 | 79 | Prednimustin × 3 | COP × 1 | Clb × 17 | Alive (175+) | ND |
| 98PB136 | F/57 | 90.1 | A | No | 13q14 | NA | 3 | No | No | No | Alive (19+) | ND |
| 95PB432 | F/57 | 92.3 | A | No | 13q14 | NA | 28 | No | No | No | Alive (75+) | ND |
| 94PB168 | F/54 | 95.5 | A | No | 13q14 | NA | 24 | No | No | No | Alive (85+) | ND |
| 96PB905 | M/48 | 96.6 | A | No | 13q14 | NA | 33 | No | No | No | Alive (65+) | ND |
| 97PB260 | F/47 | 94.5 | A | No | + 12 | NA | 0 | No | No | No | Alive (24+) | ND |
UPN indicates unique patient number; M, male; Clb, chlorambucil with or without prednisolone; COP, cyclophosphamide, vincristine, prednisolone; Flud, fludarabine; F, female; 2CDA, 2-chlor-desoxyadenosine; FE, fludarabine, epirubicine; CHOP, cyclophosphamide, doxorubicin, vincristine, prednisolone; ND, not dead; DexaBEAM, dexamethason (Dexa), carmustin, etoposide (VP16), Ara-C, melphalan; CY, cyclophosphamide; PmM, prednimustin, mitoxantron; IMVP16, ifosfamide, methotrexate, VP16; VP16, etoposide; CD20, rituximab; FC, fludarabine, CY; MCP, mitoxantron, Clb, prednisolone; NA, not applicable; and HAM, high-dose ara-C, mitoxantron.
In the SDHT cohort, high-dose therapy was administered as first-line (70%) or second-line treatment (28%) in almost all cases. Salvage therapy due to symptomatic relapse after SHDT was necessary in only 4 patients. One of these is alive after consecutive fludarabine, rituximab, and fludarabine/cyclophosphamide regimens; 3 patients died due to progressive disease after salvage therapy (chlorambucil; fludarabine/cyclophosphamide; and fludarabine, rituximab, prednisolone, respectively).
Survival
The median observation time from diagnosis was shorter for the 44 SHDT than for the 44 CT patients (70 months; range, 11-185 months vs 86 months; range, 10-176 months; P = .03). The median follow-up time from study entry was not significantly different between the 2 groups (46 months; range, 8-97 months vs 62 months; range, 5-96 months; P = .58). Seven patients in the SHDT cohort and 20 patients in the CT cohort died. Causes of death are described in Table 6, showing that the excess mortality in the CT group was entirely due to CLL-related causes.
Causes of death
. | SHDT . | CT . | P . |
|---|---|---|---|
| n | 44 | 44 | |
| CLL (%) | 3 (7) | 16 (36) | .001 |
| Non-CLL (%) | 4 (9) | 4 (9) | NS |
| Infection | 0 | 2 (5) | |
| Second malignancy | 1 (2) | 1 (2) | |
| Treatment-related | 3 (7) | 0 | |
| Other | 0 | 1 (2) | |
| Total (%) | 7 (16) | 20 (45) | .005 |
. | SHDT . | CT . | P . |
|---|---|---|---|
| n | 44 | 44 | |
| CLL (%) | 3 (7) | 16 (36) | .001 |
| Non-CLL (%) | 4 (9) | 4 (9) | NS |
| Infection | 0 | 2 (5) | |
| Second malignancy | 1 (2) | 1 (2) | |
| Treatment-related | 3 (7) | 0 | |
| Other | 0 | 1 (2) | |
| Total (%) | 7 (16) | 20 (45) | .005 |
NS indicates not significant.
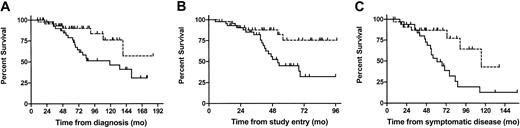
Accordingly, survival was significantly longer for the SHDT than for the CT patients when calculated from diagnosis: with last death occurring at 139 months and a corresponding survival rate of 0.57, the median survival was not reached in the SHDT cohort, whereas median survival was 119 months in the CT group. This translates into a hazard ratio (HR) of 0.394 (95% confidence interval [95%CI] 0.16-0.92) in favor of the SHDT group (P = .03). Similarly, SHDT patients had a survival advantage over CT patients when calculated from study entry (median survival not reached with last death at 56 months with a corresponding survival rate of 0.75 vs 52 months, HR 0.32 [95%CI 0.14-0.76], P = .006) and from time of symptomatic disease (median survival time 115 vs 61 months, HR 0.34 [95%CI 0.14-0.80], P = .01; for 32 SHDT and 34 CT patients; Figure 1).
The survival advantage of SHDT patients over CT patients. Survival from diagnosis (A; n = 88), from time of study entry (B; n = 88), and from time of symptomatic disease (C; n = 66) of all patients treated with SHDT (- - -) and CT (—).
The survival advantage of SHDT patients over CT patients. Survival from diagnosis (A; n = 88), from time of study entry (B; n = 88), and from time of symptomatic disease (C; n = 66) of all patients treated with SHDT (- - -) and CT (—).
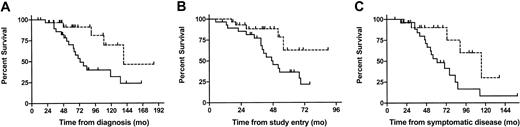
The benefit for the SHDT group remained significant when the analyses were restricted to those 58 patients who had an unmutated VH status (median survival time from diagnosis 139 vs 73 months, HR 0.31 [95%CI 0.11-0.84], P = .02; median survival time from study entry not reached with last death at 56 months with a corresponding survival rate of 0.63 vs 47 months, HR 0.32 [95%CI 0.12-0.86], P = .02; median survival from time of symptomatic disease 115 vs 61 months, HR 0.31 [95%CI 0.11-0.85], P = .02; for 23 SHDT and 27 CT patients; Figure 2). Since only 5 events occurred in the 30 patients with mutated VH (SHDT 2, CT 3), in this subset a significant survival difference between SCT and CT patients could not be shown.
Survival advantage of patients with SHDT over patients with CT (unmutated patients only). Survival from diagnosis (A; n = 58), from time of study entry (B; n = 58), and from time of symptomatic disease (C; n = 50) of patients with unmutated VH treated with SHDT (- - -) and CT (—).
Survival advantage of patients with SHDT over patients with CT (unmutated patients only). Survival from diagnosis (A; n = 58), from time of study entry (B; n = 58), and from time of symptomatic disease (C; n = 50) of patients with unmutated VH treated with SHDT (- - -) and CT (—).
Time to re-treatment
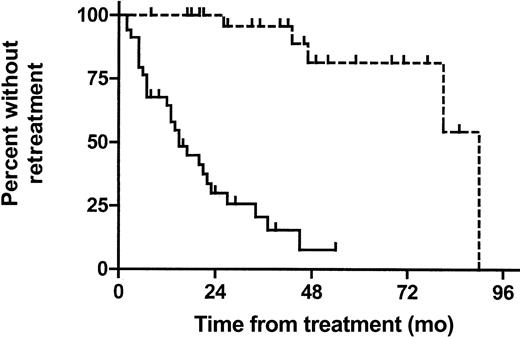
In the 32 symptomatic patients who underwent autografting we calculated the time from SHDT to re-treatment and compared it with the time from first-line therapy to salvage treatment or CLL-related death in the 34 symptomatic CT patients by log-rank testing. Time to re-treatment was considerably longer in the SHDT group than in the CT group (90 vs 15 months, HR 0.10 [95%CI 0.03-0.17], P < .001; Figure 3).
Time to re-treatment in symptomatic patients. Time from start of SHDT to first salvage therapy after transplantation (n = 32; - - -). Time from first-line treatment to first salvage therapy in CT patients (n = 34; —). Patients dying due to unrelated causes are censored at the time of death.
Time to re-treatment in symptomatic patients. Time from start of SHDT to first salvage therapy after transplantation (n = 32; - - -). Time from first-line treatment to first salvage therapy in CT patients (n = 34; —). Patients dying due to unrelated causes are censored at the time of death.
Multivariate analysis
Cox proportional hazards regression confirmed SHDT as an independent favorable predictor for overall survival from diagnosis and study entry (P = .04 and P = .03, respectively). In addition, the proportional hazards regression models identified 11q deletion (P = .005) as significant prognostic factor for survival from diagnosis and 11q deletion (P = .001) and Binet stage (P = .01) as prognostic factors for survival from study entry. The hazard ratios together with their 95% confidence limits are shown in Table 7.
Cox regression analysis of survival time from diagnosis and study entry in 88 risk-matched patients
. | Hazard ratio for death (95% CI) . |
|---|---|
| Overall survival from diagnosis | |
| Treatment SHDT | 0.38 (0.15-0.97)* |
| 11q deletion | 3.82 (1.51-9.67)* |
| Stage at diagnosis B | 0.88 (0.36-2.11) |
| Stage at diagnosis C | 1.69 (0.35-8.13) |
| VH mutated | 0.44 (0.15-1.28) |
| Age at diagnosis | 1.50 (0.54-4.18) |
| Overall survival from study entry | |
| Treatment SHDT | 0.38 (0.15-0.93)* |
| 11q deletion | 4.91 (1.84-13.08)* |
| Lymphocyte count at study entry higher than 50 × 109/L | 0.43 (0.17-1.10) |
| Stage at study entry B | 1.65 (0.44-6.10) |
| Stage at study entry C | 5.90 (1.42-24.44)* |
| VH mutated | 0.74 (0.23-2.41) |
| Age at study entry | 1.19 (0.48-2.93) |
| Chemotherapeutic pretreatment at study entry | 2.25 (0.91-5.52) |
. | Hazard ratio for death (95% CI) . |
|---|---|
| Overall survival from diagnosis | |
| Treatment SHDT | 0.38 (0.15-0.97)* |
| 11q deletion | 3.82 (1.51-9.67)* |
| Stage at diagnosis B | 0.88 (0.36-2.11) |
| Stage at diagnosis C | 1.69 (0.35-8.13) |
| VH mutated | 0.44 (0.15-1.28) |
| Age at diagnosis | 1.50 (0.54-4.18) |
| Overall survival from study entry | |
| Treatment SHDT | 0.38 (0.15-0.93)* |
| 11q deletion | 4.91 (1.84-13.08)* |
| Lymphocyte count at study entry higher than 50 × 109/L | 0.43 (0.17-1.10) |
| Stage at study entry B | 1.65 (0.44-6.10) |
| Stage at study entry C | 5.90 (1.42-24.44)* |
| VH mutated | 0.74 (0.23-2.41) |
| Age at study entry | 1.19 (0.48-2.93) |
| Chemotherapeutic pretreatment at study entry | 2.25 (0.91-5.52) |
For age, hazard ratios and confidence intervals (CIs) are computed for 10-year increments.
P < .05.
Discussion
With a median survival time of about 10 years, the natural course of CLL is often indolent. In a proportion of cases, however, prognosis is considerably worse under standard palliative care,20,21 justifying novel treatment strategies in these patients. To this end, myeloblative therapy with autologous SCT has been explored during recent years in younger patients with poor-risk CLL. Initial hopes that this approach might have curative potential have remained largely unfulfilled as clinical and molecular data provide evidence of disease persistence in the vast majority of autografted patients.5-9,22 Nevertheless, similar to the situation in multiple myeloma, SCT could have a major beneficial impact on the prognosis even if it does not result in cure.23 Thus, proper assessment of the possible clinical benefit of SCT is urgently needed. In the absence of controlled prospective comparative studies the best approach to this appears to be to perform risk-matched analyses of patient populations who have undergone SCT and standard treatment.
Risk-matched comparisons for CLL transplantations have been hampered by several serious limitations including the following: small patient numbers with sufficient follow-up after SCT; lack of standardized inclusion criteria for transplantation; high age of conventionally treated populations; and, most importantly, the great variability in prognosis. Taking advantage of recent major achievements in genetic risk assessment, in particular the determination of the mutational status of the VH gene, we were able to largely circumvent these obstacles by having access to a reasonable number of patients who had been uniformly treated with SCT according to defined inclusion criteria as well as to a large cohort of patients who had been managed by standard palliative care in a single center. For both data sets comprehensive information on genetic and biologic risk factors, clinical course, treatment, and follow-up was available. In addition, the number of events was high enough to allow the choice of overall survival from diagnosis as primary end point.
The matching of the patients was extensive, taking into account 4 variables that covered all essential prognostic factors for CLL, as previously validated on these and independent data sets,9,11-13 except for cytogenetic abnormalities. The latter could not be considered for the matching procedure due to missing data in the SDHT cohort. Another important prognostic factor that may be useful to evaluate the comparability of individual cohorts of patients with CLL is response. The use of response as a discriminating factor for the prognosis of certain CLL patient populations, however, requires that these populations must have received the same treatment at a defined time point during the course of the disease. This was clearly not the case in the 2 cohorts studied here since the SHDT patients were generally treated more aggressively than the CT patients from the very beginning of the treatment line for which transplantation was considered (Tables 4-5). Thus, response could not be considered in this comparison.
A genuine problem of matched comparisons in CLL transplantations is defining the time of matching (ie, finding a time point during the course of the disease in the CT patients that corresponds to the time of transplantation in the SCT recipients). Alternatively, one could use the time of diagnosis or the time of first treatment; however, at these time points genetic and clinical information was not complete for the 2 data sets analyzed here. Thus, we decided to define the time of matching by the event of first genetic study. This was identical with the time of preparation for SCT in the transplantation patients and corresponded to the time of referral for evaluation of first- or second-line treatment in both cohorts. The fact that after matching both groups turned out to be highly similar with regard to all relevant risk factors as well as to the clinical course until time of study entry confirms that the matching accuracy obtained by this approach was high.
With survival and disease control in the CT group in the range of what could be expected from previous studies for this high-risk population,12,13,24-27 the outcome in the SHDT cohort was significantly better for all end points considered. As suggested by a longer time to re-treatment, by much less need for salvage therapy, and by the fact that the excess mortality in the CT group was entirely due to CLL, the outcome benefits of SHDT appear to be essentially caused by better disease control. Interestingly, the survival advantage became evident no earlier than after 3 years of follow-up. This observation resembles findings after autografting for myeloma23,28 and might reflect the impact of early deaths due to treatment-related mortality in the SHDT group. It could also be an explanation for the fact that a survival benefit for SHDT was not seen in the preliminary analysis of another comparative study on CLL transplantations that had a much shorter follow-up.29
The favorable effect of SHDT remained when the analyses were restricted to the 58 patients who had an unmutated VH status and translated into a survival benefit of more than 5 years from diagnosis. Although we have previously shown that an unmutated VH status has an adverse effect on posttransplantation outcome,9 this observation implies that SHDT still may add substantial benefit in comparison to merely conventional treatment in this high-risk subset of patients. On the other hand, the fact that 10 CT matches did not require any treatment during follow-up indicates that the eligibility criteria for SHDT used here were not stringent enough to select high-risk patients only. Eight of these 10 patients had less than 98% VH homology. Despite that the present study was underpowered to evaluate the role of SHDT in mutated CLL, these observations strongly suggest that SCT should currently not be recommended to patients with mutated VH status prior to the development of symptoms.
Although this analysis for the first time shows a clinical benefit for SCT in CLL in a comparative setting, it has several limitations that have to be considered in the interpretation of our findings. The first is the selection bias inherent to transplantation studies (ie, only patients who meet the performance and remission status requirements for transplantation are included, implying that favorable cases are selected). In the SHDT database used here about 20% of the individuals originally scheduled for transplantation did not proceed to SCT due to various reasons.30 These patients could not be considered for matching on an intent-to-treat basis because their VH status was not available. Inclusion of these cases might have thinned the SHDT effect in the present study. However, the thorough matching strategy employed including biologic risk factors should have prevented critical bias caused by this effect. This assumption is supported by the fact that cases with 17p deletion, who are at the highest risk for pretransplantation failure (as well as for failure at all), have not been selected by the matching process. Furthermore, patients with pretransplantation failure had a significantly longer time from diagnosis, had received more lines of pretreatment, had a higher lymphocyte count, and had a more advanced stage in comparison to the cases selected for matching, confirming that patients with pretransplantation failure might represent a risk group different from that investigated here.30
Another limitation is that our findings are restricted to the SHDT strategy used here (early transplantation, extensive ex vivo purging, use of TBI). It is unknown how variations of these variables might influence the outcome. It has to be stressed that what we investigated here was not the effectiveness of autografting as a consolidating treatment in a defined response situation. Instead, the aim of this study was to compare the principal therapeutic approach of early sequential high-dose therapy including autotransplantation with the classical strategy of standard palliative care.
Finally, the results of our study might have been biased by the imbalances in terms of study entry, which may translate into differences in favor of the SHDT group of supportive care and, particularly, salvage therapy. However, the vast majority of CT patients requiring salvage treatment received fludarabine during the course of their disease, which still can be considered as the most active single agent for the treatment of CLL.26,27 Moreover, only very few SHDT SCT patients actually needed salvage treatment until the end of follow-up, making a relevant effect of this issue unlikely. Nevertheless, novel therapeutic modalities, such as fludarabine combinations and monoclonal antibodies, may improve the results of conservative treatment.31-34 As their potential impact on the natural course of CLL is still unclear, the present study suggests that SHDT might be the appropriate control in future prospective comparative trials aiming at evaluation of these agents.
In conclusion, this study provides evidence that SHDT might prolong survival of younger patients with high-risk CLL. Although patients with unmutated VH status have a significantly poorer outcome after SHDT than those with mutated VH, the present analysis indicates that patients with unmutated VH will, nevertheless, particularly benefit from high-dose therapy due to the extremely poor results of conventional treatment in this subset. Because of the limitations inherent to this kind of study, our data do not definitely prove a survival advantage but provide for the first time a solid rationale for prospective randomized trials on this issue.
Prepublished online as Blood First Edition Paper, December 11, 2003; DOI 10.1182/blood-2003-05-1549.
Supported by the Deutsche José Carreras Leukämie-Stiftung (Grant DJCLS-R16); the European Union (EU) (QLG2-CT-1999-00786), and Bundesmini sterium für Bildung und Forschung (BMBF) (01KW9938).
The publication costs of this article were defrayed in part by page charge payment. Therefore, and solely to indicate this fact, this article is hereby marked “advertisement” in accordance with 18 U.S.C. section 1734.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal