Background
TP53 mutations are associated with unfavorable outcomes in various cancer types and present an obstacle to achieving sustained remission in acute myeloid leukemia (AML). Even with allogeneic hematopoietic stem cell transplantation (HSCT), the risk of relapse approximates 80%, with dismal long-term survival. This could imply methods of immune escape in TP53 mutated leukemias that allow them to evade the graft-versus-leukemia (GVL) effect, which is a critical mechanism of disease control with HSCT. This study investigates the dynamics of immune reconstitution at single-cell resolution in TP53-mutated AML at pre- and multiple post-HSCT timepoints in patients who either remain in long-term remission or relapse following HSCT.
Methods
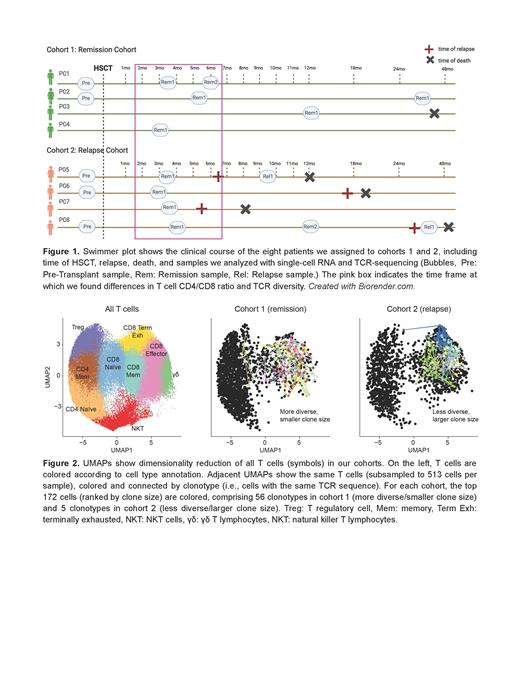
We collected 26 longitudinal bone marrow aspirates and 1 peripheral blood sample from 12 patients with TP53-mutated AML who underwent HSCT at the Dana-Farber Cancer Institute between 2015-21. All patients received HLA-matched T-cell replete HSCT in morphologic remission, with standard tacrolimus/methotrexate-based prophylaxis and peripheral blood stem cells. We categorized the patients into two cohorts based on their treatment response: long-term remission (>3.5 years, cohort 1, n=4) or relapse (cohort 2, n=8). We performed paired 5' single-cell RNA and T cell receptor (TCR) sequencing on sorted viable mononuclear from each sample and CD3+ T cells from selected samples using 10x Genomics procedures. In total, we analyzed 44 samples (27 mononuclear, 17 CD3+), resulting in 250,640 high-quality single-cell transcriptomes, including 137,649 T cells and NK cells. Data analysis was performed using Cell Ranger Multi (10x Genomics) and R 4.3 with the tidyverse 2.0, Seurat 5.1, and scRepertoire 1.1 packages.
Results
We focused our initial analysis on the 2-6-month post-HSCT period that is critical for immune reconstitution. This included eight samples from seven patients in the two cohorts (1: remission, 2: relapse, pink box in Figure 1). Of these 7 patients, 4 were in the relapse cohort, with these relapses occurring >4 months post-HSCT. No significant difference in T-cell chimerism was seen between these 7 patients in this 2-6 month period. Based on canonical gene expression signatures, we annotated cell types and selected T cells for further evaluation (Figure 2). First, we found that the CD4/CD8 ratio differed between the cohorts, indicating a relative CD8+ T cell expansion in the relapse cohort (cohort 1: 1.69, cohort 2: 0.55, P<0.0001). Second, we found lower TCR diversity in the relapse cohort (inverse Simpson index, cohort 1: 234.6, cohort 2: 83.0, P<0.0001). This difference remained significant when subsampling the same number of T cells per patient and when restricting all samples to the 3 to 4-month timepoint. Considering the disparity in CD8 T cell numbers between the cohorts, we also analyzed repertoire diversity, specifically in CD8+ T cells, and found that the observed significance persisted. To further investigate TCR diversity, we overlaid T cell clonotypes (cells that share the same TCR sequence) in a UMAP visualization, which highlighted increased hyperexpanded CD8+ T cells in the relapse cohort (Figure 2). Finally, we found that greater clonotype expansion was correlated with the expression of key T cell genes, including CD8A, CD8B, PRF1, NKG7, and GZMA. These results suggest that cytotoxic CD8+ T cells link lower TCR diversity to the likelihood of disease relapse.
Conclusions
By performing deep analysis of single-cell gene expression linked to TCR clonotypes, we discover new insights into the biology of hematopoietic reconstitution after HSCT of patients with TP53-mutated AML. Despite having analyzed a limited number of samples, we find that patients who remain in long-term remission have significantly higher CD8+ TCR diversity between 2-6 months post-HSCT, while those who relapsed between 5-18 months post-HSCT showed lower TCR diversity and hyperexpanded CD8+ clones at the same timepoints. This work suggests that early post-HSCT TCR diversity could be a biomarker associated with GVL and warrants further study. In ongoing and future analyses, we will mine the current dataset for further insights into the factors influencing survival in TP53-mutated AML patients. We also plan to extend the measurement of TCR diversity as a predictor of survival for validation in a larger cohort.
Disclosures
Cutler:Ruth L. Kirschstein Postdoctoral Individual National Research Service Award: Research Funding; Astellas: Consultancy; Rigel: Consultancy; Oxford Immune Algorithmics: Membership on an entity's Board of Directors or advisory committees; InhibRx: Consultancy; Pluristem Therapeutics: Other: DSMB; Allovir: Other: Data Safety Monitoring Board (DSMB); Sanofi: Consultancy; Cimeio: Membership on an entity's Board of Directors or advisory committees. Ho:Omeros: Consultancy; Jazz: Consultancy, Research Funding; Allovir: Consultancy; CareDx: Research Funding; Alexion: Consultancy. Koreth:BMS: Research Funding; Clinigen Labs: Consultancy, Research Funding; Amgen: Research Funding; Tr1x: Consultancy; Biolojic Design: Consultancy; Cue Biopharma: Consultancy; Gentibio: Consultancy; Equillium: Consultancy; Mallinckrodt: Membership on an entity's Board of Directors or advisory committees; Cugene: Membership on an entity's Board of Directors or advisory committees; Miltenyi Biotec: Research Funding; Regeneron: Research Funding; Equillium: Research Funding. Lindsley:Qiagen: Consultancy; Bluebird bio: Consultancy, Membership on an entity's Board of Directors or advisory committees; Takeda Pharmaceuticals: Consultancy; Sarepta Therapuetics: Consultancy; Verve Therapuetics: Consultancy; Jazz Pharmaceuticals: Consultancy; Vertex Pharmaceuticals: Consultancy. Luskin:Pfizer: Honoraria; Jazz: Honoraria; AbbVie: Research Funding; Novartis: Honoraria; Novartis: Research Funding. Garcia:AstraZeneca: Research Funding; AbbVie: Consultancy, Research Funding; Pfizer: Research Funding; New Wave: Research Funding; Astellas: Consultancy; Genentech: Consultancy, Research Funding; Bristol Myers Squibb: Consultancy; Gilead: Consultancy; Servier: Consultancy; Prelude: Research Funding. Livak:MBQ Pharma Inc: Consultancy; Standard BioTools Inc: Current equity holder in publicly-traded company. Soiffer:Juno Therapeutics/ BMS/Celgene USA: Other: Data Safety Monitoring Board; Smart Immune: Consultancy; Astellas: Consultancy; Jasper: Consultancy; Neovii: Consultancy; Bluesphere Bio: Consultancy; Vor Bipharma: Consultancy; NMPD - Be the Match, USA: Membership on an entity's Board of Directors or advisory committees. Ritz:Garuda Therapeutics: Consultancy, Current holder of stock options in a privately-held company, Membership on an entity's Board of Directors or advisory committees; Equillium: Research Funding; Kite/Gilead: Research Funding; Oncternal: Research Funding; LifeVault Bio: Consultancy, Current holder of stock options in a privately-held company, Membership on an entity's Board of Directors or advisory committees; Avrobio: Consultancy, Membership on an entity's Board of Directors or advisory committees; Novartis: Consultancy, Membership on an entity's Board of Directors or advisory committees, Research Funding; TScan Therapeutics: Consultancy, Current holder of stock options in a privately-held company, Membership on an entity's Board of Directors or advisory committees; Smart Immune: Consultancy, Membership on an entity's Board of Directors or advisory committees; Clade Therapeutics: Consultancy, Current holder of stock options in a privately-held company, Membership on an entity's Board of Directors or advisory committees; Akron Biotech: Consultancy, Membership on an entity's Board of Directors or advisory committees. Wu:BioNTech Inc: Current equity holder in publicly-traded company; Pharmacyclics: Research Funding. Lane:AbbVie: Research Funding; Stemline Therapeutics: Research Funding; Cimeio Therapeutics: Consultancy; IDRx: Consultancy; Jnana Therapeutics: Consultancy; ProeinQure: Consultancy; Qiagen: Consultancy; Medzown: Current equity holder in private company. Van Galen:ManaT Bio: Consultancy; Immunitas: Consultancy.