In this issue of Blood, Kim et al demonstrate, in a mouse model of in utero hematopoietic cell transplantation (HCT), a novel approach to enhancing donor hematopoietic stem cell (HSC) engraftment by mobilizing host HSCs from their endogenous niche and then infusing the donor HSCs during the period of maximal mobilization.1 Much like a game of “musical chairs”—where everyone tries to sit down when the music stops—when the agents causing mobilization wear off, the donor HSCs have many more empty niches to potentially occupy, thereby resulting in enhanced multilineage engraftment.
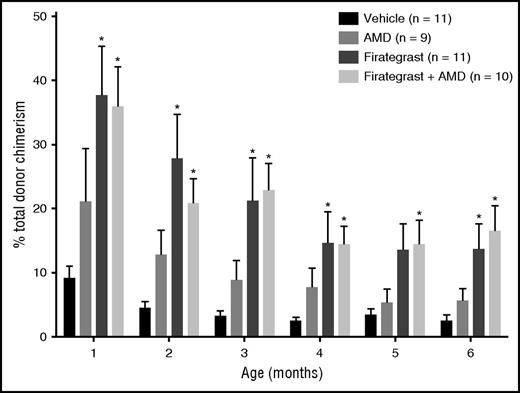
Whole blood donor chimerism over time in mice following in utero HCT with various pre-HCT “conditioning” strategies. Six-month-old mice are roughly equivalent to 20- to 30-year-old humans. See Figure 2B in the article by Kim et al that begins on page 2457.
Whole blood donor chimerism over time in mice following in utero HCT with various pre-HCT “conditioning” strategies. Six-month-old mice are roughly equivalent to 20- to 30-year-old humans. See Figure 2B in the article by Kim et al that begins on page 2457.
Some infants with genetic diseases of HSCs begin to demonstrate clinical sequelae shortly after birth, a classic example being severe combined immunodeficiency (SCID), in which patients are at immediate risk for the development of serious infections.2 Other infants, such as those with α-thalassemia, begin to manifest symptoms of their disease even before birth.3 Hence, when there is prenatal knowledge that such a disease exists, in utero HCT holds great promise for very early correction of the defect.4 Unfortunately, clinical application of in utero HCT has been significantly limited by the inability to administer the traditional bone marrow “space-making” chemotherapeutics used in most postnatal HCT settings. Therefore, the only successful human reports have been in fetuses with SCID, where there is a profound defect in the patient’s ability to reject donor cells. In these infants, only donor T-cell chimerism has been produced,5 analogous to the standard outcome seen following postnatal nonconditioned HCT for SCID. However, to expand the practical application of this technique to other diseases, donor engraftment resulting in multilineage cell production needs to be achieved.
To accomplish this, Kim et al hypothesized that blocking the pathways that maintain fetal HSCs in their normal niche (the fetal liver) would open sufficient space for donor HSCs to engraft. To accomplish this, they studied both the CXCR4 inhibitor AMD3100 (plerixafor), as well as firategrast, a small molecule that antagonizes α4β1-integrin. When administered to the pregnant mothers, both agents rapidly crossed the placenta into the fetal circulation, with firategrast crossing more rapidly and achieving higher levels. Either because of this, or perhaps because of a more important role for α4β1-integrin in HSC retention within the fetal liver, firategrast demonstrated superior mobilization of HSCs compared with plerixafor alone. The administration of plerixafor, firategrast, or both, to the mothers was shown to be globally safe, with no increase in postnatal deaths compared with vehicle control.
Subsequently, the fetuses were injected with allogeneic donor HSCs, and the amount of donor cells in the fetal liver was evaluated 24 hours later. This demonstrated a low, but significantly superior, percentage of cells of donor origin in the firategrast-treated group, compared with either the plerixafor- or vehicle-treated groups. The in utero transplanted mice were subsequently born and followed with monthly chimerism analysis for 6 months. In the animals treated with firategrast, whole blood chimerism of 30% to 40% was seen at 1 month, followed by a drop to 10% to 20% donor by 4 months, and then apparent stabilization over the following 2 months (see figure). At 6 to 9 months of age, donor chimerism was demonstrated in peripheral blood B cells, monocytes, and granulocytes; furthermore, there were no signs of graft-versus-host disease.
The experiments were fairly conclusive that plerixafor on its own was insufficient to open sufficient niches to improve donor cell engraftment, a finding that mirrors that of a clinical trial in infants with SCID.6 However, the experiments were not completely definitive regarding whether the addition of plerixafor augmented the effect of firategrast, and more work will need to be done to fully optimize this approach. Plerixafor has been widely used by the HCT community to mobilize HSCs since its approval in 2008, and appears to have minimal transient side effects in adults and older children,7 although its published use in infants is relatively limited.6 Firategrast has been shown to have minimal side effects in a trial of adults with multiple sclerosis,8 but there is no published pediatric experience. Although expected to be safe for the mothers, as the authors rightly point out, both agents require further safety testing in larger animal models before their use in trials of human in utero HCT can commence.
Presuming that the 10% to 20% donor chimerism that Kim et al produced with this approach in their 6-month-old mice ultimately translates to a similar percentage in a 20- or 30-year-old human, this may be a sufficient level to fully correct the manifestations of many genetic hematologic diseases. However, other diseases will likely benefit from or require higher levels of donor chimerism for complete cure. As such, additional work will be essential to determine whether the approach of blocking of α4β1-integrin (+/−CXCR4) can be further enhanced by combining it with other agents. One such agent might be a monoclonal antibody against the c-kit receptor (ACK2) for stem cell factor. ACK2 transiently depletes HSCs and has been demonstrated to produce similar degrees of donor chimerism when administered as the sole conditioning agent before congenic HCT in immunodeficient mice.9 It has also been shown that ACK2 can be safely injected directly into fetal mice; when followed by a transplant of congenic HSCs, it results in approximately 15% multilineage donor engraftment.10
Such a combined approach would consist of the music starting (mobilization of host HSCs from the endogenous niche via blockade of α4β1-integrin), followed by removal of some of the participants from the game (partial depletion of host HSCs via anti-c-kit), and then followed by addition of the new contestants (infusion of the donor HSCs). Once the music stops (fading of the α4β1-integrin blockade), the overabundance of donor HSCs would hopefully have a competitive advantage for successfully sitting down into the chairs (the HSC niche). “Augmented musical chairs” might sound like a mild diversion, but for the fetus treated in this manner, the eventual ability to correct a genetic hematologic disease before birth (and therefore before the onset of organ damage or infections) without using chemotherapeutic agents (with their known long-term risks) would not be a game; it would be a triumph.
Conflict-of-interest disclosure: The author reports consulting for Jazz Pharmaceuticals.