In this issue of Blood, Desmond et al present an extension of their earlier phase 2 study of the thrombopoietin receptor (TPO-R) agonist eltrombopag to treat 43 patients with refractory severe aplastic anemia (SAA). Hematologic responses, including trilineage response, were maintained despite later discontinuation of the drug. They propose that eltrombopag directly stimulates residual hematopoietic stem cells (HSCs) in SAA. This represents a novel approach to the treatment of SAA.1,2
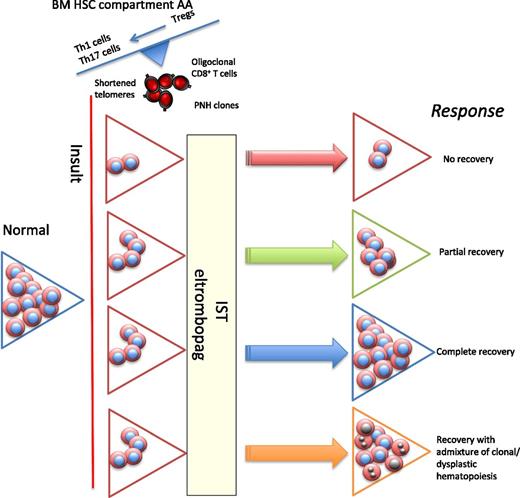
In acquired AA, following an insult (likely viral), there is immune-mediated damage to stem cells, resulting in an inflammatory immune response. There is oligoclonal expansion of cytotoxic CD8 T cells, but the more important component comprises the CD4 T cells, with expansion of Th1 (clonal) and Th17 cells and reduced and dysfunctional Tregs. Two approaches to drug treatment are (1) IST with ATG and ciclosporin and (2) eltrombopag, resulting in direct stem cell stimulation. The different possible responses to treatment are scenario 1, where there are too few HSCs to respond to either IST or eltrombopag (the role of combination therapies is being explored). In scenario 2, there is almost a complete peripheral blood recovery, but biopsy usually shows a hypocellular BM, and often patients have macrocytosis. Scenario 3 shows complete recovery of blood and BM. In scenario 4, there is recovery with emergence of clones (such as −7 or other myeloid-specific mutations), which have a growth advantage, eventually resulting in features of hypocellular MDS. PNH, paroxysmal nocturnal hemoglobinuria.
In acquired AA, following an insult (likely viral), there is immune-mediated damage to stem cells, resulting in an inflammatory immune response. There is oligoclonal expansion of cytotoxic CD8 T cells, but the more important component comprises the CD4 T cells, with expansion of Th1 (clonal) and Th17 cells and reduced and dysfunctional Tregs. Two approaches to drug treatment are (1) IST with ATG and ciclosporin and (2) eltrombopag, resulting in direct stem cell stimulation. The different possible responses to treatment are scenario 1, where there are too few HSCs to respond to either IST or eltrombopag (the role of combination therapies is being explored). In scenario 2, there is almost a complete peripheral blood recovery, but biopsy usually shows a hypocellular BM, and often patients have macrocytosis. Scenario 3 shows complete recovery of blood and BM. In scenario 4, there is recovery with emergence of clones (such as −7 or other myeloid-specific mutations), which have a growth advantage, eventually resulting in features of hypocellular MDS. PNH, paroxysmal nocturnal hemoglobinuria.
The immune basis of acquired SAA for most patients is now well established in vitro, characterized by clonal expansion of CD8+ T cells, Th1 cells, reduced regulatory T cells (Tregs) that are also dysfunctional in their ability to suppress T effectors, and increased Th2 and Th17 cells.3,4 From clinical observations, response to immunosuppressive therapy (IST) with antithymocyte globulin and ciclosporin occurs in approximately two-thirds of patients. The reasons for nonresponse to IST in the remainder have remained an enigma. An alternative diagnosis of constitutional aplastic anemia accounts for only 5% to 10% of patients.3 Lessons learned from using hematopoietic growth factors (HGFs),5 such as granulocyte colony-stimulating factor (G-CSF), granulocyte-macrophage colony-stimulating factor, stem cell factor, and thrombopoietin, in an (erroneous) attempt to treat the underlying disorder, showed that they were ineffective, but it was also assumed that there were too few HSC remaining in SAA following the initial insult to the bone marrow (BM), despite supraphysiological doses of circulating HGF. However, the results of this study appear to negate this latter assumption.
This study first confirms the earlier observations from the phase 2 study that eltrombopag can not only stimulate the platelet count in SAA but also induce bi- or trilineage hematologic responses and that overall responses occur in 40% of patients. Second—and this is an even more exciting observation—out of 14 responders who continued the drug, 5 patients later discontinued eltrombopag and maintained sustained hematologic response. What is the possible explanation for these observations? Direct stimulation of HSC is likely because the TPO-R, c-mpl, is present not only on megakaryocytes but also on HSC and progenitor cells and HSCs are deficient in c-mpl knockout mice.6,7 This study suggests that the remaining few HSCs in SAA can be stimulated by high eltrombopag levels and/or by moving them from a quiescent state. However, future studies using long-term culture initiating cells before and after eltrombopag may provide more evidence for this. The authors propose that a critical number of HSCs is required for IST response as a possible explanation for lack of response to IST. But could there be other explanations, such as an off-target effect of eltrombopag? Could eltrombopag have an immune-modulatory role, analogous to improvement in Treg function, demonstrated by the suppression of autologous T effectors seen in chronic immune thrombocytopenic purpura patients responding to eltrombopag?8
Alongside these important observations, however, is the concern regarding clonal evolution to myelodysplasia. Out of 43 patients, 8 developed clonal cytogenetic abnormalities, most frequently monosomy 7, known to be associated with a high risk of transformation to myelodysplastic syndrome (MDS)/acute myeloid leukemia (AML) in SAA. Furthermore, abnormal clones were detected very early after starting eltrombopag, most frequently by 3 months. This is much earlier than the onset of clonal evolution following antithymocyte globulin (ATG) treatment. The normocellularity of BM trephines seen in responding patients is also different from typical marrow appearances after ATG, where some degree of residual hypocellularity is more frequent. The changes observed with eltrombopag may reflect a greater recovery of stem cells, but careful morphologic and molecular characterization is required to exclude changes due to MDS. Although only 2 patients showed dyserythropoiesis, follow up of patients is too short, and it is too small a series because 5 of the 8 patients were subsequently transplanted. In SAA, the remaining HSCs are under constant pressure to support adequate hematopoiesis and peripheral blood counts. Combined with shortened telomeres, this predisposes to genomic instability, with acquired somatic mutations and risk of transformation to MDS/AML (see figure). Recent observations from our group using massively parallel targeted gene sequencing have identified acquired somatic mutations typical of myeloid disorders, most frequently ASXL1 and DNMT3A, in 20% of patients with aplastic anemia (AA) without BM morphologic changes of MDS.9 It is possible that low-level clones, below the sensitivity of metaphase cytogenetics and SNP-A karyotyping, are present at diagnosis, and eltrombopag may stimulate their expansion. This has been shown from eloquent in vitro studies with G-CSF, demonstrating expansion of small monosomy 7 clones in AA and MDS.10 The use of targeted high-throughput DNA sequencing to detect even smaller clones may help to identify those SAA patients with small clones at diagnosis that might be excluded from treatment with eltrombopag.
Future potential uses of this exciting agent include treatment of constitutional BM failure and graft failure after allogeneic hematopoietic stem cell transplantation (HSCT). Two prospective randomized studies using eltrombopag with upfront ATG and ciclosporin are in progress or about to commence, respectively. The “proof of the pudding” here will be whether addition of eltrombopag to IST will improve response in the one-third of patients who are expected to fail to respond to IST. For SAA patients who are ineligible for HSCT, new treatments are urgently needed. The unexpected observation of trilineage hematologic response in some patients represents a novel approach to therapy. However, eltrombopag should only be used in the context of clinical trials with rigorous and long-term monitoring for clonal evolution.
Conflict-of-interest disclosure: J.C.W.M. and G.J. have applied for research funding from GSK. G.J.M. was an independent advisor for GSK.