Key Points
Patients with AML often undergo code status transitions near the end of life; median time from last code status change to death was 2 days.
Patients participated in only 60.5% of final code status transitions, highlighting a need for earlier conversations to improve involvement.
Abstract
Patients with high-risk acute myeloid leukemia (AML) often experience intensive medical care at the end of life (EOL), including high rates of hospitalizations and intensive care unit (ICU) admissions. Despite this, studies examining code status transitions are lacking. We conducted a mixed-methods study of 200 patients with high-risk AML enrolled in supportive care studies at Massachusetts General Hospital between 2014 and 2021. We defined high-risk AML as relapsed/refractory or diagnosis at age ≥60. We used a consensus-driven medical record review to characterize code status transitions. At diagnosis, 86.0% (172/200) of patients were “full code” (38.5% presumed, 47.5% confirmed) and 8.5% had restrictions on life-sustaining therapies. Overall, 57.0% of patients experienced a transition during the study period. The median time from the last transition to death was 2 days (range, 0-350). Most final transitions (71.1%) were to comfort measures near EOL; only 60.5% of patients participated in these last transitions. We identified 3 conversation types leading to transitions: informative conversations focusing on futility after clinical deterioration (51.0%), anticipatory conversations at the time of acute deterioration (32.2%), and preemptive conversations (15.6%) before deterioration. Younger age (B = 0.04; P = .002) and informative conversations (B = −2.79; P < .001) were associated with shorter time from last transition to death. Over two-thirds of patients were “presumed full code” at diagnosis of high-risk AML, and most experienced code status transitions focused on the futility of continuing life-sustaining therapies near EOL. These results suggest that goals-of-care discussions occur late in the illness course for patients with AML and warrant interventions to increase earlier discussions regarding EOL preferences.
Background
Patients with acute myeloid leukemia (AML) have a poor prognosis and intense health care utilization, especially at the end of life (EOL).1 Care needs are especially intense for patients with high-risk disease, particularly older individuals, those with poor cytogenetics and molecular risk profile, and patients with relapsed or refractory disease.1,2 In fact, up to one-third of patients with high-risk AML will require intensive care unit (ICU) admission, and over 50% will die in the hospital.3-6 Furthermore, patients with leukemia who experience a cardiac arrest and receive CPR have an extremely limited prognosis.7 Despite their poor prognosis, patients with AML often have substantial misperceptions of their treatment goals and prognosis.8 This may lead to difficult EOL discussions, particularly regarding the potential role of intensive medical procedures such as cardiopulmonary resuscitation or intubation (ie, code status discussions). Prior studies have shown that patients often feel that they have a very short timeframe between hearing key prognostic information and being asked to make major treatment and EOL decisions, which impacts their ability to process and make informed decisions about their EOL care.9
Patients with advanced solid tumors who actively engage in early discussions regarding their preferences for care, including code status discussions, are more likely to report better quality of life, lower caregiver distress, and are more likely to receive goal-concordant care at the EOL.10-12 However, studies examining code status discussions in patients with high-risk AML are lacking.13,14 Prior studies have underscored the need to further understand the nature of communication during difficult EOL and code status transitions for patients with AML, and these findings in solid tumor patients suggest that mechanisms of increasing patient participation in EOL discussions may yield similar benefits in the AML population.13
In this study, we used mixed qualitative and quantitative methodology to characterize code status transitions for patients with high-risk AML and identify factors associated with time from code status transition to death in this population.
Methods
Study procedures
We conducted a secondary analysis of 200 patients with high-risk AML enrolled in 3 supportive care studies at Massachusetts General Hospital between 1 July 2014 and 1 June 2021. All 3 studies enrolled patients with high-risk AML with similar eligibility criteria and data collection methods, allowing the study team to combine the data for this secondary analysis. One study was a prospective longitudinal study of older patients (≥60 years) describing the quality of life and mood in this population.15 The other 2 studies were randomized trials assessing the efficacy of a palliative care intervention for patients with high-risk AML (clinical trials.gov: NCT02975869 and NCT03310918).16,17 We did not see an effect of the palliative care intervention on code-status transitions in these studies, but nonetheless, we controlled for random assignment to the palliative care intervention in all analyses.
Study participants
The eligibility criteria for all 3 studies were identical, allowing us to combine the data for this secondary data analysis. Patient eligibility criteria included age ≥18 years old, ability to read and complete questionnaires in English with minimal assistance, and diagnosis with high-risk AML. High-risk AML was defined as a new diagnosis at age ≥60 years, AML with an antecedent hematologic disorder, therapy-related AML, or relapsed/refractory AML. All 3 studies excluded patients with active psychiatric conditions or comorbid conditions that the oncology clinician believed prohibited the ability of patients to provide informed consent. This study was approved by the Dana-Farber Harvard Cancer Center’s Institutional Review Board, and all participants provided written informed consent.
Code status transitions
We conducted a review of the Electronic Health Record (EHR) to identify code status transitions between the time of diagnosis and death. We identified code statuses as “presumed full” (not discussed), “confirmed full” (discussed with the patient and/or family), “restricted” (ie, do not resuscitate/do not intubate [DNR/DNI] or do not resuscitate/OK to intubate [DNR/OK to I]), or “comfort measures only” (CMO). Notably, at our institution, all patients admitted to the hospital must have a code status entered into the system, and the admitting clinician cannot bypass code status entry. We obtained dates and times of code status entry from the EHR. Code status at the time of diagnosis was identified as the code status up to 14 days before diagnosis of AML or the first subsequent code status documented; if both were available, the code status after diagnosis was used. Code statuses before this time point were not considered in the analysis. We also used the EHR to collect information about the clinician signing the code status order as well as the context and location of the code status entry. We used our EHR’s standardized “Advance Care Planning” tab, which aggregates all code status orders, healthcare proxy documentation, and entries into a “Serious Illness Conversation” module to collect all code status transitions as well as detailed information regarding any conversations that led to code status transitions, including the clinicians involved, clinical setting (ie, ICU, oncology floor, emergency department), and whether patients, families, or palliative care clinicians participated in these conversations.
Code status transition characterization
Two physicians (A.E.-J. and H.A.) independently reviewed the EHR for 10 patients to develop a coding schema for characterizing the type of conversation leading to code status transitions. A.E.-J. is a board-certified oncologist who cares for patients with hematologic malignancies; H.A. is an internal medicine resident physician. The 2 physicians then refined their coding schema and reviewed 20 additional cases independently to ensure consensus regarding the characterization of the conversations resulting in code status transition, achieving high concordance (κ = 0.97). The resulting coding system, classifying conversations as predominantly “preemptive,” “anticipatory,” or “informative,” was then applied to characterize the conversations leading up to code status transition across the entire study cohort. For conversations that included elements of both anticipatory and informative conversations, the resulting outcome of the conversation dictated the final classification of the conversation as “anticipatory” or “informative” (eg, a time-limited trial of life-sustaining therapy or transition to CMO). Clinical deterioration and timing of conversations relative to clinical deterioration were abstracted from the medical record, defined by the clinicians reviewing the record, and annotated for each transition. Examples of clinical events that frequently were associated with code status transitions were infection, progression of leukemia, symptom progression such as worsening altered mental status or dyspnea, and development of hemodynamic or respiratory instability. A similar research methodology was used in prior studies examining code status transitions in patients with advanced solid tumors.18
Demographic and clinical data
At the time of study enrollment, participants self-reported their gender, race, ethnicity, income, marital status, and education level. We used the EHR to gather information on treatment regimen, age, and date of death.
Statistical analysis
We performed all analyses using STATA 9.4 (StataCorp, College Station, TX). We described patient characteristics, code status at the time of diagnosis, and code status transitions using frequencies/percentages for categorical variables and medians/ranges for continuous variables. We also used descriptive statistics to describe the type of conversations leading to code status transitions. We used a chi-square test of independence to assess the relationship between initial code status and treatment type. We grouped these conversations into 3 types: (1) preemptive conversations before any clinical change; (2) anticipatory conversations at the time of an acute clinical deterioration; and (3) informative conversations after acute clinical deterioration, focused on withdrawing life-sustaining therapies. Binomial logistic regression was used to assess the relationship between the length of time from diagnosis to death and whether a preemptive code status conversation occurred. We then used univariate Poisson regression analyses to explore factors that were associated with time from the last code status transition to death, given the distribution of the time from the last code status transition to death. Factors of interest were identified a priori based on prior literature and included age (continuous variable), sex, race, income, marital status, education level, the intensity of AML therapy (intensive 7 + 3 or similar regimen vs nonintensive therapy), and type of conversations involved in code status transitions (preemptive, anticipatory, or informative, as identified by characterization process described above).18 Factors identified in the univariate analysis with P < .25 were then included in multivariable Poisson regression analyses that adjusted for random assignment to the palliative care intervention in the supportive care studies.
Results
Baseline characteristics
Table 1 details the demographic and baseline clinical data of our study cohort. The median age of the cohort was 70 years (range, 20-100), 57.0% (114/200) identified as men, and 61.5% (123/200) underwent treatment with intensive chemotherapy. Overall, 57.0% (114/200) patients experienced a code status transition during the study period, and 43.0% (86/200) did not have a code status transition. The median time from diagnosis to death was 262 days for those patients (n = 138) who died during the study period and had information on the date of death available. There were no significant demographic differences between patients who experienced a code status change during their illness course compared with those who did not. Patients who had a code status change were more likely to receive nonintensive therapy for their AML.
Baseline characteristics of the study cohort
| . | (n = 200) . | (n = 114) . | (n = 86) . |
|---|---|---|---|
| Age (yr), median (range) | 69.7 (19.7-100.3) | 70.6 (22.8-100.3) | 68.7 (19.7-89.2) |
| Gender, n (%) | |||
| Male | 114 (57.0) | 67 (58.8) | 47 (41.2) |
| Female | 86 (43.0) | 47 (41.2) | 39 (34.2) |
| Relationship status, n (%) | |||
| Single | 16 (8.0) | 11 (9.6) | 5 (4.4) |
| Married | 144 (72.0) | 83 (72.8) | 61 (53.5) |
| Divorced | 17 (8.5) | 9 (7.9) | 8 (7.0) |
| Widowed | 17 (8.5) | 8 (7.0) | 9 (7.9) |
| Unknown | 6 (3.0) | 3 (2.6) | 3 (2.6) |
| Education, n (%) | |||
| High school or less | 55 (27.5) | 35 (30.7) | 20 (17.5) |
| College | 85 (42.5) | 44 (38.6) | 41 (36.0) |
| Postgraduate | 54 (27.0) | 32 (28.1) | 22 (19.3) |
| Unknown | 6 (3.0) | 3 (2.6) | 3 (2.6) |
| Race, n (%) | |||
| American Indian or Alaskan Native | 1 (0.5) | 0 (0.0) | 1 (1.2) |
| Asian | 4 (2.0) | 2 (1.8) | 2 (2.3) |
| African American or Black | 3 (1.5) | 2 (1.8) | 1 (1.2) |
| Native Hawaiian or other Pacific Islander | 1 (0.5) | 0 (0.0) | 1 (1.2) |
| White | 186 (93.0) | 108 (94.7) | 78 (90.7) |
| Other | 5 (2.5) | 2 (1.8) | 3 (3.5) |
| Hispanic or Latino ethnicity, n (%) | |||
| Hispanic or Latino | 18 (9.0) | 9 (7.9) | 9 (10.5) |
| Not Hispanic or Latino | 181 (90.5) | 104 (91.2) | 77 (89.5) |
| Unknown | 1 (0.5) | 1 (0.9) | 0 (0.0) |
| Initial treatment, n (%) | |||
| Intensive (7 + 3 or similar) | 123 (61.5) | 64 (56.1) | 57 (66.3) |
| Nonintensive therapy | 66 (33) | 45 (39.5) | 23 (26.7) |
| Unknown | 11 (5.5) | 5 (4.4) | 6 (7.0) |
| . | (n = 200) . | (n = 114) . | (n = 86) . |
|---|---|---|---|
| Age (yr), median (range) | 69.7 (19.7-100.3) | 70.6 (22.8-100.3) | 68.7 (19.7-89.2) |
| Gender, n (%) | |||
| Male | 114 (57.0) | 67 (58.8) | 47 (41.2) |
| Female | 86 (43.0) | 47 (41.2) | 39 (34.2) |
| Relationship status, n (%) | |||
| Single | 16 (8.0) | 11 (9.6) | 5 (4.4) |
| Married | 144 (72.0) | 83 (72.8) | 61 (53.5) |
| Divorced | 17 (8.5) | 9 (7.9) | 8 (7.0) |
| Widowed | 17 (8.5) | 8 (7.0) | 9 (7.9) |
| Unknown | 6 (3.0) | 3 (2.6) | 3 (2.6) |
| Education, n (%) | |||
| High school or less | 55 (27.5) | 35 (30.7) | 20 (17.5) |
| College | 85 (42.5) | 44 (38.6) | 41 (36.0) |
| Postgraduate | 54 (27.0) | 32 (28.1) | 22 (19.3) |
| Unknown | 6 (3.0) | 3 (2.6) | 3 (2.6) |
| Race, n (%) | |||
| American Indian or Alaskan Native | 1 (0.5) | 0 (0.0) | 1 (1.2) |
| Asian | 4 (2.0) | 2 (1.8) | 2 (2.3) |
| African American or Black | 3 (1.5) | 2 (1.8) | 1 (1.2) |
| Native Hawaiian or other Pacific Islander | 1 (0.5) | 0 (0.0) | 1 (1.2) |
| White | 186 (93.0) | 108 (94.7) | 78 (90.7) |
| Other | 5 (2.5) | 2 (1.8) | 3 (3.5) |
| Hispanic or Latino ethnicity, n (%) | |||
| Hispanic or Latino | 18 (9.0) | 9 (7.9) | 9 (10.5) |
| Not Hispanic or Latino | 181 (90.5) | 104 (91.2) | 77 (89.5) |
| Unknown | 1 (0.5) | 1 (0.9) | 0 (0.0) |
| Initial treatment, n (%) | |||
| Intensive (7 + 3 or similar) | 123 (61.5) | 64 (56.1) | 57 (66.3) |
| Nonintensive therapy | 66 (33) | 45 (39.5) | 23 (26.7) |
| Unknown | 11 (5.5) | 5 (4.4) | 6 (7.0) |
Code status at diagnosis
At diagnosis, 38.5% (77/200) of patients had a “presumed full” code status, 47.5% (95/200) had a “confirmed full” status, 8.5% (17/200) had a “restricted” status (ie, DNR/DNI or DNR/OK to I), and 5.5% (11/200) had statuses that were unknown. Overall, 40.0% (80/200) of initial code statuses were documented during a hospital admission where the AML diagnosis was established, whereas 42.5% (85/200) were documented on subsequent hospitalizations, usually after an outpatient diagnosis of AML. Notably, 9.0% (18/200) of initial code statuses were documented during a clinical decompensation, and 3.0% (6/200) occurred in other or unrelated contexts. Overall, 68.5% (122/178) of patients were treated with intensive chemotherapy (such as “7 + 3” or a similar intensive chemotherapy regimen). Patients receiving intensive chemotherapy were more likely to have an initial full code status (P < .001).
Transitions through illness course
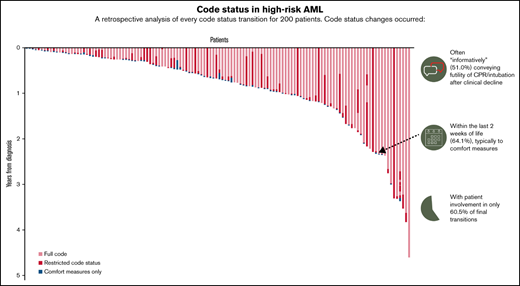
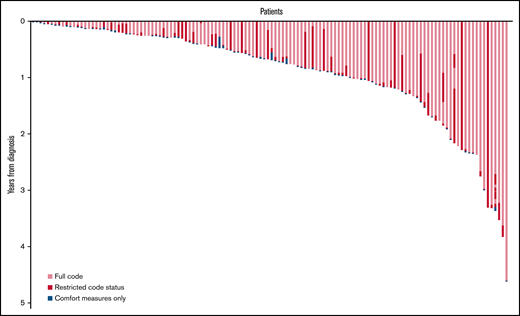
The median number of code status transitions per patient was 2 (range, 1-8). Of 206 code status transitions, 98 (47.6%) were changes from a full code status to a restricted code status such as DNR/DNI, 65 (31.6%) were from restricted code status to CMO, 16 (7.8%) were changes from a full code to CMO, and 11 (5.3%) were from a restricted code status to another more restricted code status (eg, transitioning from DNR/OK to I to DNR/DNI). Of note, only 7.8% (16/200) of transitions were reversals from restricted code status to a less-restricted status, such as a transition from DNR/DNI to full code. Code status orders were predominantly documented and signed by a member of the oncology team (153/206 [74.3%]), while a minority were ordered by nononcology clinicians (52/206 [25.2%]) such as emergency department and ICU clinicians. Palliative care clinicians were involved in 31.1% (64/206) of all code status transitions, including 50% (37/74) of transitions for patients who received the palliative care intervention in the supportive care randomized clinical trials. Data on the number of hospitalizations were available for 162 patients; for these patients, the median number of hospitalizations during the study period was 3 (range, 0-14). The majority of patients (134/206 [65.0%]) and families (162/206 [78.6%]) were involved in code status transitions. However, patients and families were simultaneously involved in only 44.2% (91/206) of code status transitions, and families participated in 34.4% (71/206) transitions alone. Sixteen percent (33/206) of all transitions occurred on the final day of life, 42.7% of all transitions occurred in the final 3 days of life, and 64.1% (132/206) of all transitions occurred within the final 2 weeks of life.
Type of conversations leading to code status transitions
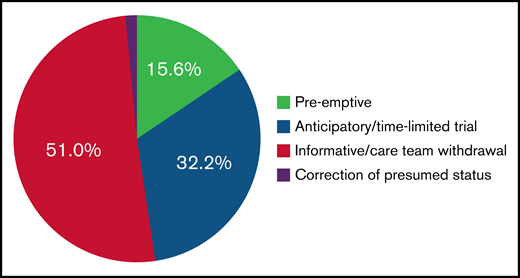
We developed 3 categories to describe the processes underlying code status transitions for patients with AML, as noted in Figure 1. Table 2 provides examples of processes leading to code status transitions in this population. Code status transitions occurred as a result of (1) preemptive conversations before any clinical change (32/206 [15.6%]); (2) anticipatory conversations at the time of acute clinical deterioration (66/206 [32.2%]); and (3) informative conversations after acute clinical deterioration, focused on withdrawing life-sustaining therapies (105/206 [51.0%]). Patients who had a longer length of time between diagnosis and death were more likely to have a preemptive conversation during their illness course (P = .006).
Distribution of types of conversations resulting in code status transitions.
Examples depicting code status conversation categories
| Code Status Transition Process . | Example #1 . | Example #2 . | Example #3 . |
|---|---|---|---|
| Preemptive conversations before clinical change | “[When asked about] code status, patient states he has form in his bag. Is DNR/DNI” (On routine admission). | (During routine palliative care visit during reinduction admission) “[Patient] raised topic of changing code status to DNR/DNI. He has been thinking about it for some time […] [Patient] doesn’t believe that resuscitation would provide any improvement in his quality of life, and given his poor prognosis, he would prefer to change code status to DNR/DNI.” | “Discussed, informally, code status. Can readdress with [primary oncologist], but she clearly stated her wish to be DNR/DNI.” |
| Anticipatory conversations at the time of acute clinical deterioration | “We reviewed her course thus far and acknowledged a very low chance of long-term success given her current situation. I outlined all options for care. We agreed to continue with current measures [but that] at this point that heroic measures such as CPR and intubation would not be in her best interests.” | “Held family meeting in light of [patient’s] acute change in clinical status. He has developed bacteremia with GPC [and] increased GI bleeding overnight.[…] Minimally arousable this AM. Family in agreement that if his condition worsens, that [patient] would not want to be transferred to the MICU for pressors or intubation. They also do not want him to undergo CPR or shock in the event of a cardiac arrest. They agree that we should continue all current measures, including antibiotics, blood products, and supportive care, in hopes that he will rebound from current infection.” | “We reviewed the treatment for his leukemia […] Unfortunately, he has developed progressive pulmonary infection [and] he is also developing renal dysfunction. It is possible that his clinical status may deteriorate. We reviewed options, including intubation vs if [he] opts for noninvasive means of therapy […] We discussed we support whatever decision he would like to proceed with. [Patient] opted for DNR/DNI status and would like to continue all noninvasive medical measures at this time.” |
| Informative conversations after acute clinical deterioration | “Pt is critically ill and may be dying. Team met with family yesterday, who understand this and have changed his code status to DNR” | “Prognosis is markedly poor now that WBC is rising […] I brought up that CPR, shocks, intubation would not meaningfully change prognosis if she were to have an arrest and would potentially cause suffering. She asked us not to pursue those measures.” | “I met with [patient] and his family today to review his hospital course to date [and] the results of the bone marrow biopsy, which are highly suggestive of disease recurrence. I explained that he is too debilitated to receive additional chemotherapy. We discussed that [he] wishes to return home for Christmas. We agreed that at this point, we will work on getting [him] home as soon as possible […] He wishes to be DNR.” |
| Code Status Transition Process . | Example #1 . | Example #2 . | Example #3 . |
|---|---|---|---|
| Preemptive conversations before clinical change | “[When asked about] code status, patient states he has form in his bag. Is DNR/DNI” (On routine admission). | (During routine palliative care visit during reinduction admission) “[Patient] raised topic of changing code status to DNR/DNI. He has been thinking about it for some time […] [Patient] doesn’t believe that resuscitation would provide any improvement in his quality of life, and given his poor prognosis, he would prefer to change code status to DNR/DNI.” | “Discussed, informally, code status. Can readdress with [primary oncologist], but she clearly stated her wish to be DNR/DNI.” |
| Anticipatory conversations at the time of acute clinical deterioration | “We reviewed her course thus far and acknowledged a very low chance of long-term success given her current situation. I outlined all options for care. We agreed to continue with current measures [but that] at this point that heroic measures such as CPR and intubation would not be in her best interests.” | “Held family meeting in light of [patient’s] acute change in clinical status. He has developed bacteremia with GPC [and] increased GI bleeding overnight.[…] Minimally arousable this AM. Family in agreement that if his condition worsens, that [patient] would not want to be transferred to the MICU for pressors or intubation. They also do not want him to undergo CPR or shock in the event of a cardiac arrest. They agree that we should continue all current measures, including antibiotics, blood products, and supportive care, in hopes that he will rebound from current infection.” | “We reviewed the treatment for his leukemia […] Unfortunately, he has developed progressive pulmonary infection [and] he is also developing renal dysfunction. It is possible that his clinical status may deteriorate. We reviewed options, including intubation vs if [he] opts for noninvasive means of therapy […] We discussed we support whatever decision he would like to proceed with. [Patient] opted for DNR/DNI status and would like to continue all noninvasive medical measures at this time.” |
| Informative conversations after acute clinical deterioration | “Pt is critically ill and may be dying. Team met with family yesterday, who understand this and have changed his code status to DNR” | “Prognosis is markedly poor now that WBC is rising […] I brought up that CPR, shocks, intubation would not meaningfully change prognosis if she were to have an arrest and would potentially cause suffering. She asked us not to pursue those measures.” | “I met with [patient] and his family today to review his hospital course to date [and] the results of the bone marrow biopsy, which are highly suggestive of disease recurrence. I explained that he is too debilitated to receive additional chemotherapy. We discussed that [he] wishes to return home for Christmas. We agreed that at this point, we will work on getting [him] home as soon as possible […] He wishes to be DNR.” |
Final code status transition before death
One hundred thirty-eight (69.0%) patients died during the study period, and 114 (57.0%) patients transitioned code status. The median number of days between diagnosis and death was 262 (range, 18-4524). Among 114 final code status transitions occurring before death, families were involved in 87.7% (100/114) of these transitions, while patients were involved in only 60.5% (69/114). Patients and families were simultaneously involved in 48.2% (55/114) of these final conversations. Palliative care clinicians were also involved in a substantial minority of these last code status transitions (48/114 [42.1%]). Most of these final code status transitions (81/114 [71.1%]) were transitions from either full or restricted statuses to CMO. The final code status transitions occurred mostly as informative conversations after an acute clinical deterioration (89/114 [78.1%]), with only 13.2% (15/114) occurring as anticipatory conversations at the time of an acute decline and 7.9% (9/114) occurring preemptively before any clinical change. One (0.9%) was a correction of presumed status. The median time from diagnosis to first code status transition was 212 days (range, 7-4507). The date of death was available for 108 patients, and the median time from the last code status transition to death for patients who died during the study period was 2 days (range, 0-350). As depicted in Figure 2, most code status transitions near the time of death were transitions to CMO.
Factors associated with time from final code status transition to death
In univariate analyses, patients who experienced a longer time interval between their final code status transition to death were older (B = 0.08; P < .001) and more likely to receive nonintensive therapy (B = 1.42; P = .003). Patients whose code status transition occurred as a result of an informative conversation after acute clinical deterioration were more likely to have a shorter time from their final code status transition to death relative to other conversation types (B = −2.84; P < .001). We found no association between the time of final code status transition to death with sex, race, ethnicity, marital status, or education.
In multivariable analyses, older patients were more likely to have a longer time from the last code status transition to death (B = 0.04; P = .002), whereas patients whose code status transition occurred as a result of an informative conversation after acute clinical deterioration were more likely to have a shorter time from their final code status transition to death (B = −2.79; P < .001) (Table 3).
Multivariate analysis of factors associated with time between last code status and death
| . | Coefficient . | 95% CI . | P . |
|---|---|---|---|
| Age | 0.04 | (0.01 to 0.06) | .002 |
| Sex | 0.24 | (−0.42 to 0.89) | .482 |
| Nonintensive therapy | −0.16 | (−0.93 to 0.62) | .692 |
| “Informative”-type last conversation | −2.79 | (−3.46 to −2.12) | <.001 |
| . | Coefficient . | 95% CI . | P . |
|---|---|---|---|
| Age | 0.04 | (0.01 to 0.06) | .002 |
| Sex | 0.24 | (−0.42 to 0.89) | .482 |
| Nonintensive therapy | −0.16 | (−0.93 to 0.62) | .692 |
| “Informative”-type last conversation | −2.79 | (−3.46 to −2.12) | <.001 |
CI, confidence interval.
Model adjusted for random assignment to palliative care intervention on supportive care trials.
Discussion
We found that patients with AML experienced high rates of code status transitions throughout their illness course, with most transitions clustering close to the EOL. Code status transitions near the EOL were commonly changes from full code to DNR/DNI or CMO after an acute clinical decompensation. The majority of patients had a code status of presumed full or confirmed full at the time of diagnosis, and 1 in 4 transitions occurred in nononcology settings such as the emergency department or ICU. Although older patients were less likely to experience rapid code status transition at the EOL, the median time from the last code status transition to death was only 2 days across the entire cohort, and over half of all transitions occurred in the final 2 weeks of life, underscoring the high rate of these transitions in the final few days of life for this population.
In this study, we also characterized the type of conversations leading to code status transition in patients with AML. Unfortunately, we noted that code status transitions often occurred very late in the illness course, despite a population that experiences intense healthcare utilization, multiple hospital admissions, and potential opportunities to address their code status earlier in the illness course, given their limited prognosis. In fact, most code status transitions occurred in an “informative” or “futility” dynamic between patients or families and providers, wherein care team members provided prognostic information after an acute clinical deterioration. Only a minority of code status transition conversations occurred preemptively before a clinical change. These findings corroborate what patients have described previously, that many people experience a short time frame between hearing key prognostic information and being asked to make decisions.9 The finding that patients with a longer time from diagnosis to death were more likely to have a preemptive conversation about code status should be interpreted in the context of immortal time bias but may reflect that these patients have more opportunities to discuss code status outside of the pressured environment of clinical decompensation. Further, code status transitions occurred very late within the overall illness course, giving patients and families little time to prepare before a clinical decompensation. These late changes point to a critical need for more preemptive conversations about EOL wishes in patients with high-risk AML.19,20 We also found an overall low rate of palliative care involvement in code status transitions, which may reflect less common use of palliative care services in the care of patients with hematologic malignancies and may suggest a need for earlier referrals.21,22 More research is necessary to assess how these short time frames affect the grieving and quality of life of patients and families; however, previous studies in patients with advanced solid tumors have shown that even small increases in the time that patients and families spend together preparing for the future can improve the sense of comfort and preparedness for death.23
One consequence of abrupt deterioration is that patients and family members often do not have time to talk with each other or their clinicians about their EOL wishes. We noted in our study that patients were only involved in 65% of all code status transitions, which declined further in the final code status transition before death. Moreover, fewer than half of all final code status transitions included the input of both patients and families. Since these conversations predominantly occurred after an acute clinical deterioration, patients were likely too ill to participate in the decision-making process. This places a significant burden on caregivers, and prior studies have clearly shown that many patients with advanced cancer prefer to be involved in their own EOL decision-making.24-26 Furthermore, when conversations occur too late in the illness course, family members are often faced with the immense challenge of making difficult decisions on behalf of their loved ones.27 Earlier preemptive conversations about EOL care preferences could enable patients to have a voice in decision-making processes even when they are too ill to participate and may mitigate the uncertainty of caregivers about what patients would want.28 Future research should aim to increase patient participation in EOL decision-making by increasing preemptive and anticipatory conversations for patients with high-risk AML.
Previous studies of code status transitions in patients with advanced cancer have categorized transitions based on mechanisms of change and conversation content,18 or the degree of documentation and involvement.29 In this study, we described the dynamic between clinicians and patients in each transition. We identified categories of preemptive conversations, anticipatory conversations at the time of clinical deterioration, and informative conversations focused on withdrawing life-sustaining therapies. These categories correspond to existing models for communication for seriously ill patients. Early preemptive conversations are known to be associated with improved quality-of-life outcomes,11,30,31 and anticipatory conversations, sometimes conceptualized as time-limited trials, are a key intermediary structure for improving communication and reducing invasive interventions in patients who do not survive ICU care.32 A large subset of patients progressed from full code to restricted status and then CMO. This may reflect the desire of the clinician to implement time-limited trials of life-sustaining interventions at the time of clinical deterioration in the care of patients with advanced hematologic malignancies, and final transitions to CMO likely account for the very short time interval between final code status change and death in our population. Importantly, we achieved high interrater reliability in characterizing these types of transitions, suggesting that our categorization schema is reproducible and may be used to characterize code status transitions across other contexts and populations.
We also identified that older patients were more likely to experience a longer time from code status transition to death. This may reflect the capacity of the clinician to prognosticate more accurately in this population, as these patients are less likely to benefit from intensive medical care at EOL.33,34 Conversely, we found that patients who had “informative”-type final code status conversations had shorter time frames between final code status transition to death. This is an anticipated finding and likely reflects that these conversations often occur after clinical deterioration as patients are imminently dying. These findings underscore the need to engage patients with discussions regarding their EOL care preferences earlier in their clinical course in a preemptive or anticipatory fashion to avoid informative conversations occurring late after acute clinical deterioration.
This study has several limitations. We studied code status transitions at a tertiary care center caring for a predominantly White and highly-educated population in Boston, which may not be generalizable across other populations or care settings. Furthermore, the limited racial, ethnic, and religious diversity in our population may also affect EOL communication among patients, families, and healthcare providers. We also relied on the EHR to describe code status transitions, and this may not capture all conversations occurring between patients and clinicians throughout the illness course that are not documented in the EHR, particularly conversations occurring in the outpatient setting; however, because patients with AML spend nearly one-third of their life after diagnosis in the hospital, outpatient changes in status are likely detected before they become clinically relevant.1 EHR documentation may also have a limited capacity to capture nuances of these code status conversations.
Conclusions
Our study reveals several opportunities for improving EOL communication in patients with AML. We found that patient code statuses often changed very late in their illness course, with a median of 2 days from the last code status change to death. Most final code status conversations were informative discussions in which clinicians conveyed the futility of further medical care near EOL. Patients frequently could not participate in these late conversations about code status, and fewer than half of all final code status transitions included both patients and families. Further studies should assess whether these short time frames between code status transitions and death correlate with patient- and caregiver-reported measures of coping and bereavement. Overall, these findings highlight a pressing need to address code status and EOL preferences earlier in patients with high-risk AML.
Acknowledgment
The authors wish to thank the Leukemia and Lymphoma Society, for which A.E.-J. is a Scholar in Clinical Research.
Authorship
Contribution: All authors made substantial contributions to the conception and design, acquisition of data, or analysis and interpretation of data; M.W.L., M.J.R., A.C.W., K.B., T.W.L., and A.E.-J. contributed significantly to data collection and studies, of which this study is a secondary analysis; H.R.A. and A.E.-J. conducted the chart review and analysis described and drafted the article; and all authors were involved in revising the article critically for important intellectual content, provided final approval of the manuscript, and agreed to be accountable for all aspects of the work.
Conflict-of-interest disclosure: The authors declare no competing financial interests.
Correspondence: Hannah Abrams, 55 Fruit St, Boston, MA 02114; e-mail: hrabrams@partners.org.
References
Author notes
Contact the corresponding author for data sharing: hrabrams@partners.org.
Presented in abstract form at the 63rd annual meeting of the American Society of Hematology, Atlanta, GA, 11 December 2021.