Key Points
Patients with iTTP in remission had pathologically increased blood–brain barrier permeability, which improved but did not resolve 6 months later.
Abstract
Immune-mediated thrombotic thrombocytopenic purpura (iTTP) is a rare, life-threatening disorder of systemic microthrombosis and organ ischemia. The etiology of chronic cerebrovascular outcomes in iTTP survivors is largely unknown. In this pilot study, we measured blood–brain barrier (BBB) permeability in patients with iTTP at the start of remission and 6 months later. This prospective pilot study included 7 adult patients with incident iTTP. Eligibility criteria included ADAMTS13 activity < 10% and detectable inhibitor at diagnosis. Patients were recruited from London Health Sciences Centre in Canada (2017-2019) within 3 days of hospital admission and followed for 6 months after remission (defined as normalization of platelet count and lactate dehydrogenase with no clinical signs or symptoms of microvascular injury for more than 30 days after the last plasma exchange). All patients had cerebral computed tomography perfusion scans with BBB permeability surface product measurements. Patients (5 women, 2 men) had a mean age of 48 years (range, 21-77 years). At diagnosis, patients had a mean platelet count of 22 (standard deviation [SD], 25) × 109/L. At the start of remission, mean BBB permeability surface product was 0.91 (0.30) mL/min/100 g. Six months later, the mean permeability surface product was 0.56 (0.22) mL/min/100 g, with a mean difference of −0.312 mL/min/100 g (95% confidence interval: −0.4729 to −0.1510; P = .0032). In this pilot study of patients with iTTP, pathologically increased BBB permeability was evident, and although there was some improvement, this persisted 6 months after remission. Future work will explore the chronicity of these findings and their clinical implications.
Introduction
Immune-mediated thrombotic thrombocytopenic purpura (iTTP) is a rare and life-threatening blood disorder caused by antibody-mediated inhibition of the proteolytic enzyme ADAMTS13 (A Disintegrin And Metalloproteinase with a Thrombospondin type 1 motif, member 13). Severe deficiency of ADAMTS13 activity leads to accumulated ultra-large von Willebrand factor multimers, increased platelet aggregation, and subsequent microvascular thrombosis within many organs.1-5 Therapeutic plasma exchange (TPE), with or without corticosteroids and/or other immunosuppressive therapies, has been shown to significantly reduce the 30-day mortality rate from 90% to 20%.6-13
The current standard of care for patients with iTTP focuses primarily on the normalization of hematologic parameters (ie, platelets, lactate dehydrogenase [LDH]); however, patients who survive the initial iTTP episode may be at increased risk for long-term cerebrovascular dysfunction, cognitive impairment, and depression.14,15 The 8-year mortality rate is 33%, and a significant proportion of deaths is attributed to cerebrovascular disease.14 The pathologic mechanism of these complications in iTTP is unclear. However, blood–brain barrier (BBB) disruption and damage have been linked to the development of depression and early dementia,16,17 and its relationship to ADAMTS13 deficiency in the context of stroke models has also recently been published. Therefore, we elected to assess this specifically in our patients with iTTP.
Although some studies have also used magnetic resonance imaging (MRI) technology to assess BBB integrity, it cannot quantify the degree of permeability. The computed tomography (CT) perfusion technique for the brain used in our pilot study was developed (by coauthor T.-Y.L.) to quantitatively assess brain perfusion and BBB integrity, where the mean permeability surface (PS) product of the brain (measured in mL/min/100 g) was determined.18-20 Higher PS product scores indicate greater permeability (ie, worse BBB leakage). Using this CT perfusion technique to assess 12 sequential stroke patients, it was found that their mean BBB PS products were 2.24 ± 1.61 mL/min/100 g on the ischemic side of the brain and 1.59 ± 0.67 mL/min/100 g on the contralateral region at the time of stroke (unpublished data). We used these novel 256-slice CT perfusion scans to assess the gross structural pathology and cerebral vascular system, including measuring the permeability of the BBB. Cerebral blood flow, cerebral blood volume, and mean transit time were also measured, which could be potentially affected by microvascular thrombosis.
In this prospective observational pilot study, we sought to assess for structural and ischemic injuries in iTTP who had achieved remission and characterize their BBB integrity using novel imaging techniques. We hypothesize that patients with iTTP have long-term risk of depression and neurocognitive changes. We hypothesize that patients with iTTP may have persistent increased BBB permeability after their acute episode despite hematologic remission. Understanding and monitoring neurocognitive function is particularly relevant in this young patient population with a median age of 40 years at the time of their first iTTP episode,21 although effective strategies for this remain unclear. Characterizing the pathophysiology and progression of cerebrovascular injury in iTTP may help to improve the long-term care and prognosis of our patients with iTTP and guide future targeted therapies to halt or even reverse the damage.
Materials and methods
Study design and setting
We conducted a prospective observational pilot study of patients with iTTP. Patients were enrolled between 1 January 2018 and 1 March 2019 at the London Health Sciences Centre (London, ON, Canada). Patients were approached by a research coordinator at the time of hospital admission and diagnosis. All patients in this study provided written informed consent (all consents were obtained before hospital discharge). The study received ethics approval from Western University’s Health Sciences Research Ethics Board (HSREB# 108273).
Eligibility criteria
Patients were eligible if they were age ≥ 18 years and had a new diagnosis of iTTP, defined as (1) platelet count < 150 × 109/L; (2) microangiopathic hemolytic anemia (presence of schistocytes on peripheral blood film); (3) LDH > 1.25 × the upper limit of normal; (4) ADAMTS13 activity < 10%; and (5) positive anti-ADAMTS13 inhibitor. Diagnosis required all 5 criteria be met. Patients were excluded if they had a prior diagnosis of iTTP, if they had an alternative diagnosis of thrombotic microangiopathy (eg, hemolytic uremic syndrome, disseminated intravascular coagulopathy, or malignant hypertension), or if they had iTTP secondary to drugs or connective tissue disease. Patients with a history of any of the following conditions were also excluded: hypertension, diabetes mellitus, cardiovascular disease, cerebrovascular disease, vasculitis of central nervous system, or a history of contrast allergies, severe asthma, and/or advanced kidney dysfunction (glomerular filtration rate < 30 mL/min/1.75 m2).
Data collection
Clinical characteristics during the acute iTTP phase were obtained from medical charts as follows: ADAMTS13 activity (enzyme-linked immunosorbent assay; reference range, 41%-130% [Technozym, Vienna, Austria]), complete blood counts, creatinine, peripheral blood film (as reported by hematologist), LDH, and the TPE regimen. Information on other treatments received was also recorded (eg, corticosteroids, rituximab, cyclophosphamide, cyclosporine, vincristine, bortezomib, and splenectomy). Data were collected within 1 month of remission from iTTP and again 6 months later. Remission was defined as platelet count > 150 × 109/L, LDH < 2× the upper limit of normal (=214 U/L), and no clinical evidence of iTTP for at least 30 days since the last TPE.22
Imaging protocol
Cerebral CT perfusion scans were done within 1 month of remission from TTP and again 6 months later. The CT protocol for the brain was developed by a collaborator (T.-Y.L.) to quantitatively assess brain perfusion and BBB integrity, reported as the PS product [ml/min/100g].18-20 Images were acquired on the 256-slice GE Healthcare Revolution CT scanner (Waukesha, WI) using the following protocol: 5-second delay from the start of intravenous contrast injection (370 mg/mL iodine) at a dosage of 0.7 mL/kg body weight and at an injection rate of 3 mL/s; 19 scans at 2.5-second intervals, followed by 7 scans at 15-second intervals using 80 kV, 45 mAs, and 16 slices of 5-mm thickness for each scan. The total scan time was 150 seconds. We have shown previously that a shorter scan time would lead to inaccuracy (>10%) in the measurement of BBB PS product.23 Images were reconstructed with an iterative algorithm (ASIRv, GE Healthcare) to reduce CT noise from the low radiation dose used. The radiation dose was estimated to be 69.0 mGy for CTDIvol and 552.7 mGy⋅cm for dose length product. The estimated effective radiation dose was 1.2 mSv (the annual background radiation dose is approximately 3 mSV, which is lower than that of a conventional CT scan [4 mSV]).
Imaging analysis
From the cerebral CT perfusion images, localization images of the head were read by the study radiologist (M.T.J.) to assess for any gross pathology. One operator (F.S.) used prototype software developed in T.-Y.L.’s laboratory (CTP5; GE Healthcare) to calculate parametric maps of cerebral blood flow, cerebral blood volume, mean transit time, and PS product from the first- and second-phase CT perfusion data. Arterial input and venous time-density curves (TDCs) were derived from an internal carotid artery in the cavernous sinus and the posterior superior sagittal sinus, respectively. To avoid operator bias, 2 collaborators (M.T.J. and F.S.) were blinded from patients’ clinical data. We corrected partial volume averaging of the arterial input TDCs by scaling the curve with the ratio of the area under the venous TDCs to the area under the arterial input TDCs. Maps were calculated by deconvolving the arterial input TDCs from the brain tissue TDCs from 3 × 3 pixel blocks of CT source images with use of the Johnson-Wilson-Lee model to estimate cerebral blood flow, cerebral blood volume, mean transit time, and PS product.24 BBB permeability should be close to zero, and a recent study has shown that its standard deviation is 0.24 mL/min/100 g.25 For quantitative assessment, the parametric maps were analyzed by the same operator (F.S.) using custom software (Interactive Display Language v6.1; Research Systems, Boulder, CO).
Statistical analysis
Means and standard deviations (SD) are reported for normally distributed data and medians and interquartile ranges (25th, 75th percentiles) for nonnormally distributed data. Patient and clinical characteristics are summarized at the time of diagnosis and at the start of remission. The mean PS products over the left and right cerebral hemispheres are reported at the start of remission and 6 months later. Mean differences between measurements taken at these 2 time points (ie, time 1 minus time 2) are reported with 95% confidence intervals (CIs). We also report mean cerebral blood flow, cerebral blood volume, and mean transit time, stratified by age and ADAMTS13 activity. For normally distributed data, a 1-sample t test was used to test whether the mean change in outcomes was significantly different from zero. All analyses were conducted using SAS software version (9.4).
Results
Of 12 patients approached to participate in the study, 1 patient did not consent, and a second was diagnosed with lupus-related secondary iTTP and therefore excluded from the pilot. Of the 10 patients who agreed to participate (7 women and 3 men), 7 had a cerebral CT perfusion scan done within 30 days of remission and again at 6 months. Three of the 10 patients had the first CT scan but not the second scan, which was cancelled because of the COVID-19 pandemic–related institutional restrictions. The final analyzable sample therefore included 7 patients with complete data.
Patient characteristics at the time of hospital admission are shown in Table 1. At the time of diagnosis, patients were between 21 and 77 years of age (mean, 48 years). Mean laboratory values included a platelet count of 22 (SD, 25) × 109/L, hemoglobin of 84 (SD, 18) g/L, and LDH of 1465 (SD, 813) U/L. All patients had ADAMTS13 activity < 1% with detectable anti-ADAMTS13 antibodies. TPE was started on the first day of diagnosis, and all patients received prednisone; 6 of the 7 also received rituximab for refractory disease. Two patients received vincristine, again for refractory disease, and 1 patient received a placebo drug as part of a separate clinical trial. At the time of the first scan (Table 1), patients had a mean platelet count of 251 (SD, 41) × 109/L, hemoglobin of 126 (SD, 13) g/L, and LDH of 254 (SD, 114) U/L. The median ADAMTS13 activity was 81% (19%, 91%), although it ranged from <1% to 96%. Of note, all patients maintained their remission status throughout the study period.
Clinical characteristics of iTTP patients at baseline, within 30 days of remission (month 0) and 6 months after remission (month 6)
| . | Baseline . | Month 0 . | Month 6 . |
|---|---|---|---|
| Demographic characteristics (N = 7)* | |||
| Women, n (%) | 5 (71%) | ||
| Age at symptom onset, y | |||
| Mean (SD) | 48 (19) | ||
| Median (25th, 75th percentiles) | 50 (30, 59) | ||
| Range | 21-77 | ||
| Body mass index, kg/m2 | |||
| Mean (SD) | 36 (8) | ||
| Median (25th, 75th percentiles) | 33 (33, 41) | ||
| Range | 25-48 | ||
| Clinical characteristics | |||
| Platelet count*, × 109/L† | |||
| Mean (SD) | 22 (25) | 251 (41) | 239 (44) |
| Median (25th, 75th percentiles) | 11 (10, 26) | 248 (229, 290) | 239 (223, 261) |
| Range | <10-76 | 183-299 | 161-306 |
| Hemoglobin, g/L | |||
| Mean (SD) | 84 (18) | 126 (13) | 128 (17) |
| Median (25th, 75th percentiles) | 79 (78, 102) | 126 (122, 141) | 121 (116, 149) |
| Range | 53-106 | 102, 142 | 109 – 153 |
| Lactate dehydrogenase, U/L (upper limit of normal, 214 U/L) | |||
| Mean (SD) | 1465 (813) | 254 (114) | 217 (83) |
| Median (25th, 75th percentiles) | 1420 (671, 2256) | 249 (161, 339) | 197 (143, 263) |
| Range | 417-2500 | 79, 413 | 134-371 |
| ADAMTS13,† % | |||
| Mean (SD) | <1‡ | 61 (41)§ | 75 (40)§ |
| Median (25th, 75th percentiles) | <1 | 81 (19, 91) | 89 (52, 114) |
| Range | <1 | <1, 96 | <1->114 |
| Serum creatinine, µmol/L | |||
| Mean (SD) | 123 (40) | 100 (56) | 75 (18) |
| Median (25th, 75th percentiles) | 122 (95, 157) | 73 (66, 107) | 71 (64, 76) |
| Range | 65-186 | 58, 220 | 62-115 |
| Missing, n (%) | 0 (0%) | 0 (0%) | |
| Immunosuppressive therapy | |||
| Prednisone, n (%) | 7 (100%) | ||
| Rituximab, n (%) | 6 (86%) | ||
| Vincristineǁ | 2 (29%) |
| . | Baseline . | Month 0 . | Month 6 . |
|---|---|---|---|
| Demographic characteristics (N = 7)* | |||
| Women, n (%) | 5 (71%) | ||
| Age at symptom onset, y | |||
| Mean (SD) | 48 (19) | ||
| Median (25th, 75th percentiles) | 50 (30, 59) | ||
| Range | 21-77 | ||
| Body mass index, kg/m2 | |||
| Mean (SD) | 36 (8) | ||
| Median (25th, 75th percentiles) | 33 (33, 41) | ||
| Range | 25-48 | ||
| Clinical characteristics | |||
| Platelet count*, × 109/L† | |||
| Mean (SD) | 22 (25) | 251 (41) | 239 (44) |
| Median (25th, 75th percentiles) | 11 (10, 26) | 248 (229, 290) | 239 (223, 261) |
| Range | <10-76 | 183-299 | 161-306 |
| Hemoglobin, g/L | |||
| Mean (SD) | 84 (18) | 126 (13) | 128 (17) |
| Median (25th, 75th percentiles) | 79 (78, 102) | 126 (122, 141) | 121 (116, 149) |
| Range | 53-106 | 102, 142 | 109 – 153 |
| Lactate dehydrogenase, U/L (upper limit of normal, 214 U/L) | |||
| Mean (SD) | 1465 (813) | 254 (114) | 217 (83) |
| Median (25th, 75th percentiles) | 1420 (671, 2256) | 249 (161, 339) | 197 (143, 263) |
| Range | 417-2500 | 79, 413 | 134-371 |
| ADAMTS13,† % | |||
| Mean (SD) | <1‡ | 61 (41)§ | 75 (40)§ |
| Median (25th, 75th percentiles) | <1 | 81 (19, 91) | 89 (52, 114) |
| Range | <1 | <1, 96 | <1->114 |
| Serum creatinine, µmol/L | |||
| Mean (SD) | 123 (40) | 100 (56) | 75 (18) |
| Median (25th, 75th percentiles) | 122 (95, 157) | 73 (66, 107) | 71 (64, 76) |
| Range | 65-186 | 58, 220 | 62-115 |
| Missing, n (%) | 0 (0%) | 0 (0%) | |
| Immunosuppressive therapy | |||
| Prednisone, n (%) | 7 (100%) | ||
| Rituximab, n (%) | 6 (86%) | ||
| Vincristineǁ | 2 (29%) |
There are no missing values for these demographic and clinical characteristics.
Platelet count < 10 × 109/L (our laboratory does not quantify < 10 × 109/L); a value of 5 × 109/L was given for this summary.
All patients had ADAMTS13 levels < 10% and inhibitor levels > 14 U/mL. All patients had detectable inhibitor level.
A value of <1% was replaced with a value of 1 for this summary; a value of >114 was replaced with 114 for this summary. Inhibitors were not run because most patients had normalized their ADAMTS13 activity.
Used in cases of refractory iTTP not responsive to TPE and corticosteroids alone.
Average map
The average map is the average of all source images of a CT perfusion study. It provides a high signal-to-noise, contrast-enhanced image of the head that accentuates the contrast between gray and white matter. All patients had normal average map images except for a 77-year-old man who had chronic ischemic changes with some volume loss reported.
Cerebral CT perfusion studies of the head
On the initial scans (ie, at remission), the mean whole brain mean PS product was 0.91 ± 0.30 mL/min/100 g, with 0.92 ± 0.32 mL/min/100 g on the right side and 0.89 ± 0.29 mL/min/100 g on the left (Table 2). The whole brain mean PS product at 6 months was 0.59 ± 0.22 mL/min/100 g, and the mean change from the first study to the second was −0.311 mL/min/100 g (95% CI: −0.4729 to −0.1510 mL/min/100 g; P = .0032). Cerebral blood flow, cerebral blood volume, and mean transit time did not show significant changes between initial and 6-month scans. Figure 1 shows the average cerebral blood flow, cerebral blood volume, mean transit time, and PS product map (5.0-mm slice) of 1 patient at the start of remission and 6 months later. Figure 2 shows the PS product map of all 7 patients at both time points. The PS product in patients who normalized their ADAMTS13 activity in remission had significant improvement at 6 months. Those with ADAMTS13 activity < 40% at the time of remission did not demonstrate a significant difference (supplemental Table 1). However, there were only 2 patients in this subgroup.
BBB permeability surface product, cerebral blood flow, cerebral volume, and mean transit time measurements at the time of remission (month 0) and 6 months later (month 6)
| . | Month 0 (N = 7) . | Month 6 (N = 7) . | Mean change(95% CI) . | P* . |
|---|---|---|---|---|
| PS (ml/min/100 g), weighted average (mean [SD]) | ||||
| Right side | 0.92 (0.32) | 0.61 (0.22) | −0.3075 (−0.4428, −0.1722) | .0014 |
| Left side | 0.89 (0.29) | 0.58 (0.23) | −0.3138 (−0.5122, −0.1155) | .0083 |
| Overall† | 0.91 (0.30) | 0.59 (0.22) | −0.3119 (−0.4729, −0.1510) | .0032 |
| CBF (ml/100 g), weighted average (mean [SD]) | ||||
| Right side | 46.8 (4.7) | 43.2 (2.8) | −3.5709 (−8.9584, 1.8165) | .1560 |
| Left side | 47.8 (5.8) | 43.3 (4.9) | −4.5457 (−9.5221, 0.4307) | .0668 |
| Overall† | 48.2 (3.9) | 44.1 (4.0) | −4.0461 (−9.0938, 1.0015) | .0975 |
| CBV (ml/100 g), weighted average (mean [SD]) | ||||
| Right side | 2.84 (0.30) | 2.61 (0.24) | −0.2313 (−0.5939, 0.1314) | .1696 |
| Left side | 2.91 (0.32) | 2.60 (0.38) | −0.3041 (−0.6141, 0.0060) | .0533 |
| Overall† | 2.93 (0.25) | 2.66 (0.32) | −0.2699 (−0.5957, 0.0559) | .0890 |
| MTT (s), weighted average (mean [SD]) | ||||
| Right side | 3.65 (0.12) | 3.62 (0.13) | −0.0275 (0.1801, 0.1250) | .6743 |
| Left side | 3.66 (0.11) | 3.60 (0.14) | −0.0610 (−0.2210, 0.0989) | .3865 |
| Overall† | 3.73 (0.25) | 3.68 (0.19) | −0.0488 (−0.2086, 0.1110) | .4833 |
| . | Month 0 (N = 7) . | Month 6 (N = 7) . | Mean change(95% CI) . | P* . |
|---|---|---|---|---|
| PS (ml/min/100 g), weighted average (mean [SD]) | ||||
| Right side | 0.92 (0.32) | 0.61 (0.22) | −0.3075 (−0.4428, −0.1722) | .0014 |
| Left side | 0.89 (0.29) | 0.58 (0.23) | −0.3138 (−0.5122, −0.1155) | .0083 |
| Overall† | 0.91 (0.30) | 0.59 (0.22) | −0.3119 (−0.4729, −0.1510) | .0032 |
| CBF (ml/100 g), weighted average (mean [SD]) | ||||
| Right side | 46.8 (4.7) | 43.2 (2.8) | −3.5709 (−8.9584, 1.8165) | .1560 |
| Left side | 47.8 (5.8) | 43.3 (4.9) | −4.5457 (−9.5221, 0.4307) | .0668 |
| Overall† | 48.2 (3.9) | 44.1 (4.0) | −4.0461 (−9.0938, 1.0015) | .0975 |
| CBV (ml/100 g), weighted average (mean [SD]) | ||||
| Right side | 2.84 (0.30) | 2.61 (0.24) | −0.2313 (−0.5939, 0.1314) | .1696 |
| Left side | 2.91 (0.32) | 2.60 (0.38) | −0.3041 (−0.6141, 0.0060) | .0533 |
| Overall† | 2.93 (0.25) | 2.66 (0.32) | −0.2699 (−0.5957, 0.0559) | .0890 |
| MTT (s), weighted average (mean [SD]) | ||||
| Right side | 3.65 (0.12) | 3.62 (0.13) | −0.0275 (0.1801, 0.1250) | .6743 |
| Left side | 3.66 (0.11) | 3.60 (0.14) | −0.0610 (−0.2210, 0.0989) | .3865 |
| Overall† | 3.73 (0.25) | 3.68 (0.19) | −0.0488 (−0.2086, 0.1110) | .4833 |
Results are presented as mean (standard deviation). There are no missing values. CBF, cerebral blood flow; CBV, cerebral blood volume; MTT, mean transit time.
A one-sample t test was used to test whether the mean change in each outcome was significantly different from zero.
The overall value is calculated as the weighted average of both the right and left sides.
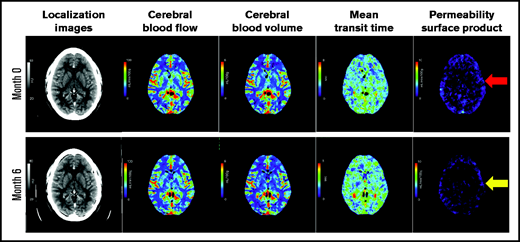
Representative cerebral CT perfusion imaging of a patient. Representative imaging slices of the cerebral CT perfusion scans of a patient. Images include the localization images, cerebral blood flow slices, cerebral blood volume slices, minute transit time images, and PS product images at the time of remission and 6 months later. Of note, images of a normal brain would have minimal contrast leakage through the BBB (ie, minimal blue color). The red arrow indicates diffuse BBB leakage at month 0 (1.26 ml/min/100 g) with modest improvement at month 6 (0.96 ml/min/100 g), shown with yellow arrow. Please see supplemental Figure 1 for comparison of PS product images of diffuse BBB leakage in a patient with TTP, localized BBB leakage in a stroke patient, and minimal BBB leakage in another stroke patient.
Representative cerebral CT perfusion imaging of a patient. Representative imaging slices of the cerebral CT perfusion scans of a patient. Images include the localization images, cerebral blood flow slices, cerebral blood volume slices, minute transit time images, and PS product images at the time of remission and 6 months later. Of note, images of a normal brain would have minimal contrast leakage through the BBB (ie, minimal blue color). The red arrow indicates diffuse BBB leakage at month 0 (1.26 ml/min/100 g) with modest improvement at month 6 (0.96 ml/min/100 g), shown with yellow arrow. Please see supplemental Figure 1 for comparison of PS product images of diffuse BBB leakage in a patient with TTP, localized BBB leakage in a stroke patient, and minimal BBB leakage in another stroke patient.
BBB PS map of all 7 patients at months 0 and 6. The yellow circle represents highest BBB PS product region in the 7 patients with iTTP (1.80 ml/min/100 g, at right parietal lobe). There is intense blue color in that region. For the same patient, the red and blue arrows represent a significant change between month 0 and 6 PS products (1.72-0.73 ml/min/100 g at the left frontal lobe). Please see supplemental Figure 1 for comparison of PS product images. Patient 4 had chronic ischemic changes on the CT images. Patient 5 had persistent low ADAMTS13 activities < 1% with inhibitor level > 94 U/mL, consistent with iTTP.
BBB PS map of all 7 patients at months 0 and 6. The yellow circle represents highest BBB PS product region in the 7 patients with iTTP (1.80 ml/min/100 g, at right parietal lobe). There is intense blue color in that region. For the same patient, the red and blue arrows represent a significant change between month 0 and 6 PS products (1.72-0.73 ml/min/100 g at the left frontal lobe). Please see supplemental Figure 1 for comparison of PS product images. Patient 4 had chronic ischemic changes on the CT images. Patient 5 had persistent low ADAMTS13 activities < 1% with inhibitor level > 94 U/mL, consistent with iTTP.
Discussion
Chronic neurocognitive changes in survivors of iTTP have been recognized for years, although the pathophysiology has remained unclear. In this pilot study of adult patients with iTTP in remission, we found that most do not have any gross pathologic changes. Previous work has shown that most patients will have normal conventional head CT imaging, even during acute episodes.26 Although CT studies can be normal in iTTP patients, Alwan et al27 demonstrated that hyperintense lesions can be identified on their MRI, which was associated with worse cognitive impairment. Hamilton et al28 also described similar hyperintense lesions in patients who have recovered from iTTP episodes. Further studies to assess the correlation between BBB permeability, MRI white matter changes, and neurocognitive impacts would be important.
Compared with conventional imaging, cerebral CT perfusion scans allow quantification of the PS product, an objective measurement for monitoring BBB disruption. In our study, the mean whole brain PS product was 0.91 ± 0.30 and 0.59 ± 0.22 mL/min/100 g at remission and 6 months later, respectively, with a mean change (ie, improvement) of −0.311 mL/min/100 g (95% CI: −0.4729 to −0.1510 mL/min/100 g; P = .0032). Although improvement was observed over this time, BBB permeability was still detected at 6 months. After acute stroke, BBB permeability typically peaks after 7 days and starts to improve afterward.29
Disruption of the BBB has been studied in other diseases. In multiple sclerosis, there are data suggesting dysregulation of the BBB and transendothelial migration of activated leukocytes are early signs of disease.30 The mechanism for BBB disruption in multiple sclerosis is not completely understood but may be driven by cytokine- and chemokine-mediated injury.31 Furthermore, a study of thrombocytopenia in patients with HIV suggested that platelets may have a “protagonist” role in BBB disruption.32 This may suggest additional mechanisms to BBB disruption in patients with iTTP. Although permeability measurement is not affected by renal dysfunction, the impact of therapeutic plasma exchange, corticosteroids, and other immunosuppressive treatments on BBB disruption/repair is not known. In a future larger study, treatment influence on BBB permeability will be evaluated, and long-term data will be collected to more fully assess BBB permeability chronically in this population.
Normal cerebral blood flow, cerebral blood volume, and mean transit time are 44.4 ± 6.5 mL/100 g, 3.8 ± 0.7 mL/100 g, and 3.2 to 4.4 seconds.33,34 Although we expected these parameters to be compromised in our patients, as seen in stroke or anemia, these values were still within the normal range.35 This may be because we assessed these parameters during their remission period. We will need to further study these factors along with along with BBB permeability during acute iTTP episodes for a complete comparison.
Reproducibility of BBB PS measurements, using the same theoretical basis in another disease area (liver tumor), has been reported to be 6.7% to 12.1% in both animal and human subjects.36,37 Contrast-enhanced MRIs of BBB PS product have relied mainly on subjective assessment of image intensity at the site of compromised BBB. Our CT perfusion technique, with objective measurement of BBB PS product in units of mL/min/100 g, has a physiologic basis, and the absolute PS values would facilitate an objective and more sensitive detection of changes in longitudinal studies.38 BBB PS as a marker of disruption has been implicated in the development of cognitive dysfunction in a recent study by Nation et al.17 Of note, the method of BBB PS measurement in this publication was the same CT-based method used in our pilot study and can serve both as clinical validation of our BBB PS measurement methodology and proving the association of abnormal PS product with development of insidious cognitive dysfunction outside of stroke.
The standard use of TPE and immunosuppression has led to significant improvements in survival for patients with iTTP; however, long-term complications persist even in those in remission.14 Most significantly, mood disorders, neurocognitive changes, and strokes are reported at much greater rates than in the general population, despite recovery of ADAMTS13 activity.39,40 The observation of persistent BBB leakage after the initial iTTP diagnosis contrasts with findings in ischemic stroke patients (supplemental Figure 1) and allows us to further explore the correlation between BBB leakage in iTTP and its long-term neuropsychiatric complications in a future study.41 It is unclear why patients with iTTP who are in hematologic remission still have elevated BBB PS products. Although we did not measure this in our study, Ferrari et al42 have shown that patients with iTTP may have persistent circulating ADAMST13 immune complexes. Also, serum S100β, primarily synthesized by astrocytes, is quickly released into the circulation when the BBB is disrupted.43 These and additional biomarkers to assess BBB permeability could be considered in future studies in iTTP.
Management of acute iTTP episodes includes TPE and immunosuppression with corticosteroids and other medications. Steroids may have some potential benefit in reducing inflammation and slowing down BBB leakage, although the extent of this effect is currently unknown. Other therapies such as caplacizumab and recombinant ADAMTS13 are potential new treatments for iTTP.44 Acutely, they may reduce the microthrombotic burden and thereby reduce BBB damage. Further assessment of the impact of these therapies on BBB leakage will be important for future studies.
This pilot study does have some limitations. First, it is possible that our findings could be explained by the coexistence of other conditions or comorbidities; however, to minimize such confounding, we excluded patients with secondary iTTP and other thrombotic microangiopathies. In addition, the radiologist who reported these images was blinded from the clinical data. None of the patients had known cerebrovascular conditions before their initial iTTP diagnosis. In addition, all patients were recruited at the time of their first presentation, thereby eliminating the potential of residual effects from prior episodes. Second, although our study did not include neurocognitive assessments, other studies have shown that many patients with iTTP demonstrate adverse neurocognitive effects for months and even years after their initial iTTP episode.14 Finally, we do not have healthy control imaging data because of the risk of radiation. However, as shown, the BBB PS values of 12 sequential stroke patients were higher on the ischemic side of the brain (2.24 ± 1.61 mL/min/100 g), which suggests that our patients may have had much higher BBB values during their acute episodes. We plan to conduct a larger study to further examine whether the degree and duration of BBB permeability and associated consequences (eg, white matter injury) correlates with depression and overall neurocognitive function. During this study, we will also capture the BBB permeability data during the acute iTTP episodes.
In conclusion, our pilot study provides preliminary evidence that pathologic BBB permeability is elevated in survivors of iTTP up to 6 months after hematologic remission. The relationship between neurocognitive outcomes, anatomical changes, and the degree and duration of increased BBB leakage requires ongoing evaluation, as do strategies to mitigate these consequences. We focused on neurologic outcomes, but because iTTP is a systemic disease, other areas of microvascular damage may also go unnoticed. New therapeutic options, including caplacizumab and recombinant ADAMTS13, may have such a role, but this is yet to be proven. Long-term implications of these findings will be examined in future research.
Acknowledgments
The authors thank the Bailey family for help in generation of quantitative analysis of the maps, Meaghan Knight and Jessica Sontrop for assistance in statistical analysis, and Kerri Gallo for help as a research coordinator.
This study received financial support from PSI and Answering TTP.
Authorship
Contribution: S.-H.S.H., K.P., and C.J.P. designed and conducted this pilot study; T.-Y.L., K.S.T., J.T., and J.D.T. designed the imaging protocols and obtained the images; T.-Y.L., M.T.J., and A.B. analyzed the imaging results; J.M., D.B., and C.L. assisted in designing the study protocol; and all authors contributed to the writing of the manuscript, provided intellectual content of critical importance to the work described, and approved the version of manuscript to be published.
Conflict-of-interest disclosure: T.-Y.L. licenses the CT Perfusion software to GE Healthcare and Neusoft. The remaining authors declare no competing financial interests.
Correspondence: Shih-Han Susan Huang, Department of Medicine, Division of Nephrology, Western University, 800 Commissioners Rd E, Rm A2-344, London, ON N6A 5W9, Canada; e-mail: shuang45@uwo.ca.
References
Author notes
For data sharing, contact the corresponding author at shuang45@uwo.ca.
The full-text version of this article contains a data supplement.