Key Points
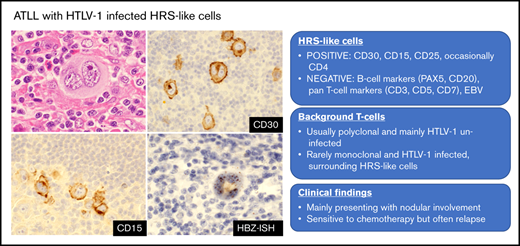
ATLL with HTLV-1–infected HRS-like cells is a new pathological variant of ATLL, distinct from conventional Hodgkin-like variant of ATLL.
Diffuse HBZ-ISH positivity and negativity for PAX5 and EBV are key features distinguishing this variant from other morphological mimics.
Abstract
Hodgkin and Reed-Sternberg (HRS) cells, a hallmark of classic Hodgkin lymphoma (CHL), are occasionally detected in non-Hodgkin lymphomas, including adult T-cell leukemia/lymphoma (ATLL), a lymphoid neoplasm caused by human T-cell leukemia virus type 1 (HTLV-1). HRS-like cells associated with ATLL have been described to be of B-cell lineage and infected with Epstein-Barr virus (EBV), not HTLV-1. We herein describe clinicopathological findings in 8 cases (4 males and 4 females; median age, 73 years [range, 55-81 years]) of ATLL with HTLV-1–infected HRS-like cells identified by ultrasensitive RNA in situ hybridization for HTLV-1 basic leucine zipper factor (HBZ-ISH), a specific viral transcript of HTLV-1. All patients showed nodal or mediastinal lesions, and 5 of the 8 patients were at an advanced disease stage. HRS-like cells were positive for CD30, CD15, MUM1, CD25, and HBZ-ISH and negative for B-cell markers, including PAX5, pan-T-cell antigens, and EBV in all cases. Five cases were positive for CD4, and 6 cases were positive for fascin. HBZ was identified in both HRS-like cells and surrounding lymphoid cells in 1 case with an aggressive clinical course and only HRS-like cells in 7 cases, most of whom showed a clinical response regardless of the chemotherapeutic regimen. Even though the definitive lineage typing of the HTLV-1–infected HRS cells is one of the limitations of this study in the absence of single-cell microdissection for polymerase chain reaction analysis, the combination of diffuse HBZ-ISH positivity and negativity for PAX5 and EBV deemed these cases distinct from CHL arising in HTLV-1 carriers.
Introduction
Hodgkin and Reed-Sternberg (HRS) cells, a hallmark of classic Hodgkin lymphoma (CHL), are characterized by a large nucleus and are often multinucleated, with prominent nucleoli. They are accompanied by various inflammatory infiltrates such as lymphocytes, histiocytes, and eosinophils. The “HRS phenotype” includes CD15 and CD30 positivity and occasional association with Epstein-Barr virus (EBV), as well as features of the B-cell lineage, such as weak positivity for PAX5 and sporadic CD20 expression.1 HRS-like cells are occasionally detected in non-Hodgkin lymphomas and lymphoproliferative disorders such as angioimmunoblastic T-cell lymphoma (AITL)2 ; chronic active EBV infection of the T-cell/natural killer (NK) cell type, systemic form (CAEBV-T/NK-S)3 ; and peripheral T-cell lymphoma, not otherwise specified (PTCL-NOS).4 However, the characteristics of HRS-like cells differ according to the background disease. HRS-like cells in AITL usually exhibit a B-cell phenotype and positivity for EBV-encoded small RNA by in situ hybridization (EBER-ISH),2,5 whereas those in CAEBV-T/NK-S and PTCL-NOS show a T- or NK-cell phenotype.3,4
Adult T-cell leukemia/lymphoma (ATLL) is a T-cell neoplasm caused by human T-cell leukemia virus type 1 (HTLV-1).1 ATLL is subdivided into 4 clinical subtypes: smoldering, chronic, lymphoma, and acute.6,7 Smoldering ATLL is indolent, whereas the lymphoma and acute subtypes are aggressive8 and often harbor lymph node lesions infiltrated by neoplastic ATLL cells. Affected lymph nodes in ATLL usually show diffuse effacement of nodal architecture with proliferation of medium-sized or large pleomorphic cells.1 However, various morphological variants have been identified that mimic anaplastic large-cell lymphoma, AITL, and dermatopathic lymphadenopathy.9-11 The Hodgkin-like variant is accompanied by HRS-like cells.12 The initial case report described 3 out of 4 cases that transformed to aggressive ATLL within 2 years, suggesting that this variant is an incipient phase of ATLL.13 The pathogenesis of Hodgkin-like ATLL is thought to involve a combination of EBV-infected polyclonal B cells mimicking HRS-like cells and clonal HTLV-1–infected CD4+ T lymphocytes.12,14-17 However, to date, only 5 cases have been analyzed in detail by single-cell sorting or microdissection methods.16,17 Therefore, it remains unclear whether the same mechanism applies to all lesions with CHL-like morphology in HTLV-1 carrier patients.
The recent development of ultrasensitive RNA in situ hybridization (RNAscope) has facilitated the identification and localization of viral transcripts in tissue specimens.18 Using this technique, we reported the first case of HRS-like cells infected with HTLV-1 surrounded by HTLV-1–uninfected lymphocytes.19 In this study, we added 7 similar cases to the first case and analyzed their detailed clinicopathological findings, focusing on the difference from CHL and conventional ATLL.
Methods
Patients and samples
We searched the archives from the pathology departments of 4 hospitals in Okinawa prefecture (Ryukyu University Hospital, Heartlife Hospital, Okinawa Prefectural Nanbu Medical Center and Children’s Medical Center, and Nakagami Hospital), an HTLV-1–endemic area of Japan, for HTLV-1–seropositive cases in which the term “Hodgkin lymphoma” or “Hodgkin and Reed-Sternberg cells” was used in the diagnostic report. The anaplastic variant of ATLL, which accounts for 6% to 13% of lymphomatous ATLL cases and is characterized by sheets of large anaplastic lymphoid cells often accompanied by HRS-like cells, was excluded from our case series.20,21 We initially selected 21 HTLV-1–seropositive cases with CHL-like morphology (supplemental Table 1). This study was approved by the institutional ethics committees of the University of the Ryukyus and other participating institutions. The study protocol adhered to the principles of the Declaration of Helsinki.
Histological and immunohistochemical evaluation
Tissue histology was examined by hematoxylin and eosin staining. Immunohistochemistry was performed with the Benchmark ULTRA system (Roche, Tokyo, Japan) according to the manufacturer’s protocol. The antibodies used along with staining conditions are summarized in supplemental Table 2. EBER-ISH was performed using a commercial kit (Agilent Technologies, Tokyo, Japan). Thresholds were defined as 30% for positivity and 10% to 30% for focal positivity. The Ki-67 (MIB-1) labeling index of the background cells was calculated as the percentage of expression by counting positive cells among the nucleated cells, excluding obvious HRS/HRS-like cells, using a high-power objective (×40). In situ hybridization for HTLV-1 basic leucine zipper factor (HBZ-ISH), a specific viral transcript of HTLV-1, along with peptidylprolyl isomerase (PPIB) as a positive control, was performed using RNAscope (ACDBio, Newark, CA), according to a recently published protocol.22 Samples without the positive control signal were defined as nonevaluable. Cutoff criteria for positivity in HBZ-ISH was determined according to a previous report.22 Clear positive signals (single dots or clusters) that were identified at least in a subset of tumor cells were evaluated as positive. In some cases, a few positive signals were observed in high-magnification view, but when they were difficult to distinguish as tumor cells, they were regarded as negative. These cutoff criteria provided a sensitivity of 76% and a false-positive rate of 0%, although the sensitivity was dependent on the age of the tissue samples.22 Immunohistochemistry data provided by cooperating institutions were also included in the analysis.
Flow cytometry and molecular analyses
Flow cytometry, detection of clonal rearrangement of the T-cell receptor γ (TCR-γ) chain gene and immunoglobulin heavy chain (IGH) gene, and Southern blot analysis of HTLV-1 provirus integration were performed as previously described.9,23,24
Statistical analysis
We used the Student t test for comparative analysis of the Ki-67 index.
Results
Clinical characteristics of patients
HBZ-ISH was performed to detect HTLV-1 in tissue specimens from 21 HTLV-1–seropositive cases with CHL-like morphology (supplemental Table 1). Three cases were nonevaluable due to negativity for the positive control (PPIB). HRS-like cells were negative for HBZ-ISH in 10 cases. Among them, 3 cases showed HRS-like cells positive for EBER-ISH and/or PAX5 as well as monoclonal TCR-γ chain gene rearrangement or loss of pan-T-cell antigens in the background cells, corresponding to the currently recognized pathological features of Hodgkin-like ATLL.16,17 Thus, 8 cases, including a recently described case (case 1)19 with a positive HBZ-ISH signal in HRS-like cells, were considered to be ATLL with HTLV-1–infected HRS-like cells. The clinical characteristics of these cases are shown in Table 1. The median age of the patients (4 males and 4 females) was 73 years (range, 55-81 years). All patients showed nodal or mediastinal lesions. Five patients (63%) were in an advanced stage of disease (Ann Arbor stage III or IV) at presentation. Bone marrow and hepatosplenic involvement was observed in 3 and 2 cases, respectively, but no other extranodal lesions were present in any of the cases. All patients except for case 6, who was treated with palliative care because of the co-occurrence of severe liver cirrhosis, received systemic combination chemotherapy. Two patients (cases 1 and 3) were mainly treated with ABVD, a standard therapy for CHL, and 5 patients were treated with a combination chemotherapy regimen such as CHOP. All cases, with enough follow-up period, treated with chemotherapy achieved complete remission, except for case 4, but a longer remission (>2 years) was achieved in 2 patients treated with CHOP. One patient (case 4) had been followed up for chronic-type ATLL before lymph node biopsy. This patient had very high serum levels of soluble interleukin-2 receptor, a prognostic biomarker for poor outcome,8 was refractory to systemic chemotherapy, and died 3 months after diagnosis (Table 1).
Clinical characteristics of ATLL patients with HTLV-1–infected HRS-like cells
| Case . | Age, y/sex . | Affected organs . | Ann Arbor stage . | Background status . | sIL2R. U/mL . | LDH, IU/L . | Clinical course . | Status at the last follow-up . | ||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 1* | 69/M | Generalized lymphadenopathy, splenomegaly, bone marrow | IV | Smoldering type ATLL (10 y)† | 6502 | 275 | Treated with ABVD chemotherapy; in partial remission for 6 months and relapsed in bone marrow; treated with brentuximab vedotin (anti-CD30 antibody) | Alive with disease (18 mo) | ||||
| 2 | 55/M | Axillary lymph node‡ | I‡ | HTLV-1 carrier | 6024 | 296 | Treated with VCAP-AMP-VECP | Alive with disease (4 mo) | ||||
| 3 | 79/F | Mediastinal mass, bone marrow | IV | HTLV-1 carrier | 3205 | 322 | Treated with 1 cycle of CHOP followed by ABVD chemotherapy | Alive in CR (7 mo) | ||||
| 4 | 55/F | Generalized lymphadenopathy, hepatosplenomegaly, bone marrow | IV | Chronic-type ATLL (7 mo)† | 65713 | 320 | Treated with VCAP-AMP-VECP and mogamulizumab (anti-CCR4 antibody), but disease status was progressive | Died of disease (3 mo) | ||||
| 5 | 80/F | Cervical and supraclavicular lymphadenopathy | II | HTLV-1 carrier | 635 | 307 | Treated with CHOP chemotherapy followed by oral etoposide therapy; stable disease for 16 mo followed by recurrence | Died of disease (18 mo) | ||||
| 6 | 74/M | Generalized lymphadenopathy§ | IIIB§ | HTLV-1 carrier | 5660 | 188 | Treated with palliative care; no chemotherapy because of accompanying liver cirrhosis | Died of disease (2 mo) | ||||
| 7 | 72/F | Inguinal lymphadenopathy | I | HTLV-1 carrier | 2493 | 212 | Treated with CHOP | Alive in CR (28 mo) | ||||
| 8 | 81/M | Generalized lymphadenopathy§ | IIIA§ | HTLV-1 carrier | 5732 | 486 | Treated with CHOP | Alive in CR (28 mo) | ||||
| Case . | Age, y/sex . | Affected organs . | Ann Arbor stage . | Background status . | sIL2R. U/mL . | LDH, IU/L . | Clinical course . | Status at the last follow-up . | ||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 1* | 69/M | Generalized lymphadenopathy, splenomegaly, bone marrow | IV | Smoldering type ATLL (10 y)† | 6502 | 275 | Treated with ABVD chemotherapy; in partial remission for 6 months and relapsed in bone marrow; treated with brentuximab vedotin (anti-CD30 antibody) | Alive with disease (18 mo) | ||||
| 2 | 55/M | Axillary lymph node‡ | I‡ | HTLV-1 carrier | 6024 | 296 | Treated with VCAP-AMP-VECP | Alive with disease (4 mo) | ||||
| 3 | 79/F | Mediastinal mass, bone marrow | IV | HTLV-1 carrier | 3205 | 322 | Treated with 1 cycle of CHOP followed by ABVD chemotherapy | Alive in CR (7 mo) | ||||
| 4 | 55/F | Generalized lymphadenopathy, hepatosplenomegaly, bone marrow | IV | Chronic-type ATLL (7 mo)† | 65713 | 320 | Treated with VCAP-AMP-VECP and mogamulizumab (anti-CCR4 antibody), but disease status was progressive | Died of disease (3 mo) | ||||
| 5 | 80/F | Cervical and supraclavicular lymphadenopathy | II | HTLV-1 carrier | 635 | 307 | Treated with CHOP chemotherapy followed by oral etoposide therapy; stable disease for 16 mo followed by recurrence | Died of disease (18 mo) | ||||
| 6 | 74/M | Generalized lymphadenopathy§ | IIIB§ | HTLV-1 carrier | 5660 | 188 | Treated with palliative care; no chemotherapy because of accompanying liver cirrhosis | Died of disease (2 mo) | ||||
| 7 | 72/F | Inguinal lymphadenopathy | I | HTLV-1 carrier | 2493 | 212 | Treated with CHOP | Alive in CR (28 mo) | ||||
| 8 | 81/M | Generalized lymphadenopathy§ | IIIA§ | HTLV-1 carrier | 5732 | 486 | Treated with CHOP | Alive in CR (28 mo) | ||||
ABVD, Adriamycin, bleomycin, vinblastine, and dacarbazine; AMP, doxorubicin, ranimustine, and prednisone; CHOP, cyclophosphamide, doxorubicin, vincristine, and prednisone; F, female; LDH, lactate dehydrogenase; M, male; sIL2R, soluble interleukin 2 receptor; VCAP, vindesine, cyclophosphamide, doxorubicin, and prednisone; VECP, vindesine, etoposide, carboplatin, and prednisone.
Pathological features of this case have been reported previously.19
Duration from the diagnosis of indolent type ATLL to the development of ATLL with HTLV-1–infected HRS-like cells.
This patient showed increased lymphocytes in the cerebrospinal fluid. However, no HRS-like large cells were recognized.
Bone marrow involvement was not assessed in these patients.
Pathological characteristics of patients
Detailed pathological findings are described in Tables 2 and 3 and Figures 1 and 2. All cases showed scattered HRS-like cells surrounded by numerous inflammatory infiltrates (Figure 1A). Diffuse and nodular architecture was observed in 5 and 2 cases, respectively; 4 cases were accompanied by dense collagenous or hyalinized bands, and 1 patient (case 2) showed preserved normal architecture with an expanded paracortex harboring HRS-like cells. In all cases, inflammatory cells surrounding the HRS-like cells mainly consisted of lymphocytes and histiocytes. Eosinophil infiltration was evident in cases 2, 6, 7, and 8, whereas other cases showed no, or very few, eosinophils. No necrotic foci were observed in any of the cases.
Pathological findings of ATLL with HTLV-1–infected HRS-like cells: morphology and immunophenotype
| . | Morphology . | Immunophenotype of HRS-like cells . | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Case . | Tissue . | Architecture . | Sclerosis . | CD30 . | CD15 . | CD3 . | CD4 . | CD5 . | CD7 . | CD8 . | CD25 . | PAX5 . | CD20 . | PD-L1 . |
| 1 | Cervical LN | Diffuse | + | + | + | − | +, f | − | − | − | + | − | − | +, f |
| 2 | Axillary LN | Partial infiltration | − | + | + | − | + | − | − | − | + | − | − | + |
| 3 | Mediastinal mass | Nodular | + | + | +, f | − | − | − | − | − | + | − | − | +, f |
| 4 | Cervical LN | Diffuse | − | + | + | − | + | − | − | − | + | − | − | − |
| 5 | Cervical LN | Diffuse | − | + | +, f | − | − | − | − | − | + | − | − | − |
| 6 | Cervical LN | Nodular | + | + | + | − | − | − | − | − | + | − | − | − |
| 7 | Inguinal LN | Diffuse | − | + | +, f | − | + | − | − | − | + | − | − | + |
| 8 | Supraclavicular LN | Diffuse | + | + | +, f | − | +, f | − | − | − | + | − | − | +, f |
| . | Morphology . | Immunophenotype of HRS-like cells . | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Case . | Tissue . | Architecture . | Sclerosis . | CD30 . | CD15 . | CD3 . | CD4 . | CD5 . | CD7 . | CD8 . | CD25 . | PAX5 . | CD20 . | PD-L1 . |
| 1 | Cervical LN | Diffuse | + | + | + | − | +, f | − | − | − | + | − | − | +, f |
| 2 | Axillary LN | Partial infiltration | − | + | + | − | + | − | − | − | + | − | − | + |
| 3 | Mediastinal mass | Nodular | + | + | +, f | − | − | − | − | − | + | − | − | +, f |
| 4 | Cervical LN | Diffuse | − | + | + | − | + | − | − | − | + | − | − | − |
| 5 | Cervical LN | Diffuse | − | + | +, f | − | − | − | − | − | + | − | − | − |
| 6 | Cervical LN | Nodular | + | + | + | − | − | − | − | − | + | − | − | − |
| 7 | Inguinal LN | Diffuse | − | + | +, f | − | + | − | − | − | + | − | − | + |
| 8 | Supraclavicular LN | Diffuse | + | + | +, f | − | +, f | − | − | − | + | − | − | +, f |
f, focal (10%-30% of cells positive); LN, lymph node.
Pathological findings of ATLL with HTLV-1–infected HRS-like cells: immunophenotype, background cell characteristics, and molecular findings
| . | Immunophenotype of HRS-like cells . | Characteristics of background cells . | Molecular findings . | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Case . | TIA-1 . | Fascin . | CCR4 . | MUM1 . | HBZ-ISH . | EBER . | T-cell component . | Eosinophils . | Abnormal T-phenotype* . | Ki-67 index, % . | TCRG . | IGH . | HTLV-1 SBH . |
| 1 | − | + | − | + | + | −† | CD4 > CD8 | − | − | 32 | Polyclonal | Polyclonal | − |
| 2 | − | − | − | + | + | −† | CD4 = CD8 | + | − | 38 | Polyclonal | Polyclonal | ND |
| 3 | − | − | − | + | + | −† | CD4 > CD8 | − | − | 24 | Polyclonal | Polyclonal | − |
| 4 | − | + | − | + | + | −† | CD4 (monotonous) | − | + | 26 | Clonal | Polyclonal | + |
| 5 | − | + | − | + | + | −† | CD4 > CD8 | − | − | 30 | NE | Polyclonal | ND |
| 6 | − | + | − | + | + | −† | CD4 > CD8 | + | − | 22 | NE | NE | +/−‡ |
| 7 | − | + | − | + | + | −† | CD4 = CD8 | + | − | 43 | Clonal§ | Polyclonal | +/−‡ |
| 8 | − | + | + | + | + | −† | CD4 = CD8 | + | − | 35 | Clonal§ | Polyclonal | ND |
| . | Immunophenotype of HRS-like cells . | Characteristics of background cells . | Molecular findings . | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Case . | TIA-1 . | Fascin . | CCR4 . | MUM1 . | HBZ-ISH . | EBER . | T-cell component . | Eosinophils . | Abnormal T-phenotype* . | Ki-67 index, % . | TCRG . | IGH . | HTLV-1 SBH . |
| 1 | − | + | − | + | + | −† | CD4 > CD8 | − | − | 32 | Polyclonal | Polyclonal | − |
| 2 | − | − | − | + | + | −† | CD4 = CD8 | + | − | 38 | Polyclonal | Polyclonal | ND |
| 3 | − | − | − | + | + | −† | CD4 > CD8 | − | − | 24 | Polyclonal | Polyclonal | − |
| 4 | − | + | − | + | + | −† | CD4 (monotonous) | − | + | 26 | Clonal | Polyclonal | + |
| 5 | − | + | − | + | + | −† | CD4 > CD8 | − | − | 30 | NE | Polyclonal | ND |
| 6 | − | + | − | + | + | −† | CD4 > CD8 | + | − | 22 | NE | NE | +/−‡ |
| 7 | − | + | − | + | + | −† | CD4 = CD8 | + | − | 43 | Clonal§ | Polyclonal | +/−‡ |
| 8 | − | + | + | + | + | −† | CD4 = CD8 | + | − | 35 | Clonal§ | Polyclonal | ND |
HBZ-ISH, RNAscope for HTLV-1 bZIP factor; MIB-1 LI, MIB-1 labeling index; NE, not evaluable; ND, not done; SBH, Southern blot hybridization; TCRG, TCR-γ chain gene.
At least one of the pan-T-cell markers deletions, including CD2, CD3, CD5, and CD7, recognized by immunohistochemistry and/or flow cytometry.
Few positive cells were observed in all cases, but positive cells were small to medium-sized, and HRS-like cells were negative.
Very faint bands were identified.
Monoclonal peak was identified with polyclonal backgrounds.
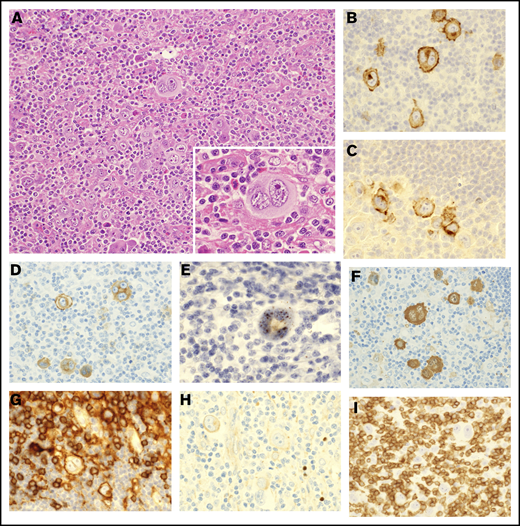
Pathological findings of lymph nodes in ATLL with HTLV-1–infected HRS-like cells. (A) Lymph nodes of case 7 were effaced by infiltration of HRS-like cells (inset) in a background of lymphocytes, histiocytes, and eosinophils (hematoxylin and eosin staining). (B-H) The HRS-like cells were positive for CD30 (B), CD15 (C), CD25 (D), HBZ-ISH (E), fascin (F), and CD4 (G) and negative for PAX5 (H). Most of the infiltrating small lymphocytes were T cells positive for CD3 (I) and negative for HBZ-ISH (E). Original magnification ×200 (A) and ×400 (A, inset, and B-I).
Pathological findings of lymph nodes in ATLL with HTLV-1–infected HRS-like cells. (A) Lymph nodes of case 7 were effaced by infiltration of HRS-like cells (inset) in a background of lymphocytes, histiocytes, and eosinophils (hematoxylin and eosin staining). (B-H) The HRS-like cells were positive for CD30 (B), CD15 (C), CD25 (D), HBZ-ISH (E), fascin (F), and CD4 (G) and negative for PAX5 (H). Most of the infiltrating small lymphocytes were T cells positive for CD3 (I) and negative for HBZ-ISH (E). Original magnification ×200 (A) and ×400 (A, inset, and B-I).
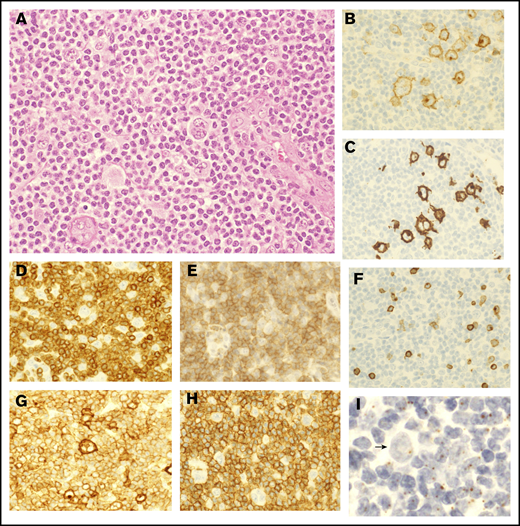
Pathological findings of lymph nodes in ATLL with HTLV-1–infected HRS-like cells and abnormal medium-sized lymphoid cells. (A) In the lymph nodes of case 4, HRS-like cells were surrounded by relatively monotonous medium-sized lymphoid cells with mild nuclear atypia (hematoxylin and eosin staining). (B-C) The HRS-like cells were positive for CD30 (B) and CD15 (C). (D-F) The medium-sized lymphoid cells were positive for CD3 (D) and CD4 (E) but lacked CD7 expression (F), indicating an abnormal T-cell phenotype. (G-H) Both the HRS-like cells and the medium-sized lymphoid cells were positive for CD25 (G), whereas only the medium-sized lymphoid cells were diffusely positive for CCR4 (H). (I) HBZ-ISH revealed that both HRS-like cells and medium-sized lymphoid cells were infected with HTLV-1. Arrow indicates an HRS-like cell. Original magnification ×400 (A-I).
Pathological findings of lymph nodes in ATLL with HTLV-1–infected HRS-like cells and abnormal medium-sized lymphoid cells. (A) In the lymph nodes of case 4, HRS-like cells were surrounded by relatively monotonous medium-sized lymphoid cells with mild nuclear atypia (hematoxylin and eosin staining). (B-C) The HRS-like cells were positive for CD30 (B) and CD15 (C). (D-F) The medium-sized lymphoid cells were positive for CD3 (D) and CD4 (E) but lacked CD7 expression (F), indicating an abnormal T-cell phenotype. (G-H) Both the HRS-like cells and the medium-sized lymphoid cells were positive for CD25 (G), whereas only the medium-sized lymphoid cells were diffusely positive for CCR4 (H). (I) HBZ-ISH revealed that both HRS-like cells and medium-sized lymphoid cells were infected with HTLV-1. Arrow indicates an HRS-like cell. Original magnification ×400 (A-I).
The immunophenotype of HRS-like cells was similar across cases (Tables 2 and 3). HRS-like cells were positive for CD30 (Figure 1B), CD15 (Figure 1C), MUM1, CD25 (Figure 1D), and HBZ-ISH (Figure 1E) in all cases. HBZ-ISH, CD30, MUM1, and CD25 were homogeneously positive in HRS-like cells, whereas the CD15+ rate varied between 10% and 90%. A total of 6 and 5 cases showed positivity (at least focally) for fascin (Figure 1F), and CD4 (Figure 1G), respectively. HRS-like cells were negative for PAX5 (Figure 1H) and CD20 as well as Oct-2 and Bob.1, TIA1 and granzyme B, and ALK (data not shown). CCR4, a target protein of mogamulizumab that is expressed in the majority of conventional ATLL cases,21 was positive in HRS-like cells in only 1 case. Background cells were mainly CD3+ T cells (Figure 1I), with variable proportions of CD4+ or CD8+ cells, and did not show any abnormal loss of pan-T-cell antigens by immunohistochemistry or flow cytometry, except for case 4. In the molecular analysis, no cases showed clonal IGH gene rearrangement, whereas clonal TCR-γ chain gene rearrangement was observed in 3 of the 6 cases in which informative results were obtained (supplemental Figure 1).
Only case 4 showed diffuse monotonous infiltration of medium-sized lymphoid cells that outnumbered HRS-like cells (Figure 2A). The immunophenotype of the latter was similar to that in the other cases, including positivity for CD30 (Figure 2B) and CD15 (Figure 2C). On the other hand, the medium-sized lymphoid cells surrounding the HRS cells were positive for CD3 (Figure 2D) and CD4 (Figure 2E) but lacked CD7 expression (Figure 2F), indicating an abnormal T-cell phenotype. Both cell populations were homogeneously positive for CD25 (Figure 2G), whereas only medium-sized lymphoid cells were positive for CCR4 (Figure 2H). HBZ-ISH revealed that both populations were infected with HTLV-1 (Figure 2I). This was different from findings in the other 7 cases, which had only a few CCR4+ background cells and showed no loss of pan-T-cell antigens.
Comparison with cases with CHL-like morphology in HTLV-1 carriers and noncarriers
In the initial case series, 3 cases were determined to be conventional Hodgkin-like ATLL, with HBZ-ISH-negative HRS-like cells accompanied by neoplastic background T cells (supplemental Table 1). In these cases, HRS-like cells showed a B-cell phenotype and/or EBER-ISH positivity, and in 1 case, the background T cells showed loss of CD7. In the other 7 cases (cases 13-15 and 17-20), HRS-like cells were negative for HBZ-ISH, and no definitive T-cell malignant potential was observed. Although they were designated as “not confirmative,” as the sensitivity of HBZ-ISH depends on the age of samples and pseudo-negativity was not completely denied, these cases seemed to include bone fide CHL developed in HTLV-1 carriers. In 6 out of these 7 cases, HRS-like cells were positive for EBV or PAX5, an ordinary phenotype of HRS cells (supplemental Table 1). These findings were in stark contrast to ATLL with HTLV-1–infected HRS-like cells.
We further compared the pathological findings of ATLL with HTLV-1–infected HRS-like cells and CHL developed in HTLV-1–seronegative patients. The latter comprised 5 HTLV-1–uninfected cases with EBV(+) CHL, and 5 with EBV(−) CHL that were archived at the University of the Ryukyus Hospital (supplemental Table 3). All CHL cases were evaluated as nodular sclerosis or mixed cellularity as a morphological subtype. While HRS cells in CHL showed positivity for some B-cell markers, including PAX5, in all cases, no HTLV-1–infected HRS-like cells expressed these markers. Additionally, no HTLV-1–infected HRS-like cells showed EBV positivity. In 5 cases (63%), HTLV-1–infected HRS-like cells showed CD4 positivity, at least focally, whereas no HRS cells were positive for CD4 in CHL. CD25 was positive in HTLV-1–infected HRS-like cells in all cases, whereas it was positive in 6 out of 10 CHL cases. Although CCR4 is considered an important marker and therapeutic target in ATLL, positive rates were comparable between HRS cells in CHL and HTLV-1-infected HRS-like cells. Regarding surrounding lymphocytes, CD4+ T cells were predominant in the majority of CHL cases. No significant difference in the Ki-67 (MIB-1) labeling index of background cells was observed between cases with ATLL with HTLV-1–infected HRS-like cells and CHL (Tables 2 and 3; supplemental Figure 2 and supplemental Table 3).
Discussion
This study described 8 cases of ATLL with HTLV-1–infected HRS-like cells. HRS-like cells were negative for B-cell markers and EBER-ISH and occasionally positive for CD4 and CD25, and they were mostly surrounded by various inflammatory infiltrates accompanied by HTLV-1–negative T cells with a normal phenotype. This lesion appears to be a pathological variant distinct from CHL, “conventional” Hodgkin-like ATLL with HRS-like cells exhibiting a B-cell phenotype, and PTCL-NOS or CAEBV-T/NK-S with HRS-like cells exhibiting a T-cell phenotype.3,4,13,16,17
Although the diffuse pleomorphic type, in which medium-sized to large pleomorphic lymphoid cells obliterate tissue structures, is the representative histological variant of ATLL, the extreme morphological heterogeneity makes it difficult to distinguish ATLL from other T-cell lymphomas.10 Conventional Hodgkin-like ATLL, recognized as a rare morphological variant of ATLL, is characterized by HRS-like cells (reported to be EBV-associated B cells) accompanied by neoplastic CD4+ T lymphocytes infected with HTLV-1.13,16,17 Three cases (10, 16, and 21) in the initial case collection were concordant with this variant (supplemental Table 1). Among cases of ATLL with HTLV-1–infected HRS-like cells, case 4 showed monotonous infiltration of atypical medium-sized lymphoid cells that surrounded HRS-like cells, and exhibited an abnormal T-cell phenotype (CD3+CD5+CD7−), as well as strong positivity for CCR4 and HBZ-ISH, unlike the other 7 cases. These findings indicate that the medium-sized lymphoid cells were neoplastic HTLV-1–infected T cells, similar to those described in known cases of Hodgkin-like ATLL. However, in contrast to these previous cases, HRS-like cells in case 4 were positive for CD4, CD25, and HBZ-ISH and negative for B-cell markers and EBER-ISH.17 Although it was not possible to analyze the clonal association between surrounding medium-sized T cells and HRS-like cells, the phenotypic similarity suggests that the HRS-like cells in case 4 were transformed neoplastic T cells.
One of the differential diagnostic considerations of ATLL with HTLV-1–infected HRS cells is CHL developed in HTLV-1 carriers. Among the 21 HTLV-1–seropositive cases with CHL-like morphology, 13 cases were negative or not evaluable for HBZ (supplemental Table 1). Some of these results, especially 4 of the cases in which biopsies were performed >10 years ago (2009 or before), may be false negative due to the sensitivity of HBZ-ISH being dependent on the age of tissue samples.22 In case 18, where the sample was obtained 12 years ago, the HRS-like cells were negative for PAX5 and EBER, compatible with the phenotype of ATLL with HRS-like cells (supplemental Table 1). When the analyses were limited to the recent samples (2010 or after), HRS-like cells in all 6 “not confirmed” cases showed a typical HRS phenotype (positive for PAX5 or EBV), suggesting that these cases were bona fide CHL developed in HTLV-1 carriers.
It is also important to determine whether HTLV-1 infection in HRS-like cells is a simple secondary infection resulting from HRS cells of CHL developed in HTLV-1 carriers or contributes to the distinct pathogenesis of the neoplastic transformation. If HBZ-ISH was positive in a subset of HRS-like cells, it would seem to indicate a secondary infection of HTLV-1. For example, diffuse large B-cell lymphoma cases often show EBV positivity in <20% of the tumor cells, considered to be a secondary EBV infection in the established B-cell clone.25 Another case report of a secondary EBV infection of an established malignant T-cell clone has also been described.26 However, a secondary infection is unlikely in this case series, because HBZ-ISH was homogenously positive in HRS-like cells in all cases, indicating the pathological significance of HTLV-1 in the initial neoplastic transformation. Furthermore, the atypical phenotype of HTLV-1–infected HRS-like cells, namely occasional CD4 positivity and PAX5 and EBER-ISH negativity, also indicated that HTLV-1–infected HRS-like cells are biologically distinct from HRS cells in CHL. These atypical phenotypes may be useful for a differential diagnosis. Immunohistochemical analyses will reduce the risk of overlooking ATLL cases with HTLV-1–infected HRS-like cells in HTLV-1–nonendemic areas where HTLV-1 serological tests are not usually performed in cases with CHL-like morphology.
Accurate definition of the lineage of HRS-like cells in our cases was difficult because they were negative for most T- and B-cell markers. HTLV-1–infected HRS-like cells may be derived from CD4+ T cells, as they were often positive for CD4 and CD25 (the typical immunophenotype of ATLL) and CD4+ T cells are the normal counterpart in virtually all ATLL cases.1 On the other hand, HRS cells in CHL rarely showed loss of PAX5 and aberrant T-cell antigen expression.27 A single-cell polymerase chain reaction (PCR) analysis in combination with microdissection was attempted, but the results were not interpretable (data not shown). Single-cell sorting/microdissection in combination with PCR analysis will more clearly characterize HTLV-1–infected HRS-like cells; however, distinguishing them from HRS cells of CHL is still equivocal, even if monoclonal TCR rearrangement is observed. While the World Health Organization classification1 described that the postulated normal counterpart of HRS cells in CHL is usually germinal center B cells, the classification also states that “the HRS cells of rare CHL cases have been reported to harbor clonally rearranged TCR genes, indicating that exceptional cases with morphological features of CHL may be derived from T-cells.”28,29 Therefore, even the most accurate method for the distinction of cell lineage cannot totally exclude the possibility of CHL. We would thus prefer to emphasize the phenotypic characteristics and the proportion of HTLV-1–positive cells among HRS-like cells to explain the distinction between HRS-like cells and HRS cells in CHL as described above.
The neoplastic nature of background lymphoid cells was confirmed in case 4. In the other 7 cases, background HTLV-1–uninfected lymphoid cells seemed reactive rather than neoplastic. Nuclear atypia was not evident morphologically. No aberrant loss of T-cell markers was observed by immunohistochemistry or flow cytometry in any of the cases. The Ki-67 index of background cells was similar between ATLL with HTLV-1–infected HRS-like cells and CHL of mixed cellularity or nodular sclerosis variants (Tables 2 and 3; supplemental Figure 2 and supplemental Table 3). CD4+ cells were more abundant than CD8+ cells in 4 of 8 cases, but this is common in CHL and is not indicative of a neoplastic nature.30 Monoclonal rearrangement of the TCR-γ chain gene was observed in 3 cases, including case 4 with obviously neoplastic background cells. However, in the other 2 cases, it was not clear whether the clonal peaks were derived from neoplastic HRS-like cells, comprising a minor population of the whole tissue, or background cells (supplemental Figure 1). Even in CHL, a clonal peak was observed in ∼25% of cases as a result of improved sensitivity of the PCR protocol.31 These findings contrast with PTCL-NOS with HRS-like cells in which the background T cells are mostly neoplastic.4
Most of these patients showed a clinical response regardless of the chemotherapeutic regimen; however, recurrence occurred in 2 patients. In the present study, 3 of 8 patients died and another 3 survived for >18 months after diagnosis, suggesting a clinical course intermediate between CHL and aggressive ATLL, although the small number of cases in this series does not support a confident conclusion on the prognosis.20 It is unclear whether the pathogenesis of ATLL with HTLV-1–infected HRS cells is closer to that of CHL or ATLL, although case 4 was clinicopathologically similar to aggressive ATLL. This case did not respond to the combination of systemic chemotherapy and mogamulizumab (a CCR4 antibody) and succumbed to progressive disease 3 months after diagnosis, indicating an aggressive clinical course. Distinguishing between ATLL with HTLV-1–infected HRS-like cells, CHL, and other CHL-like lesions is important from a clinicopathological standpoint. Further clinical characterization with additional cases and detailed biological characterization including comprehensive genomic analyses of HRS-like cells is warranted in the future.
In conclusion, ATLL with HTLV-1–positive HRS-like cells is an important differential diagnosis for lymph node lesions with CHL-like morphology in HTLV-1 carriers. CD4 positivity and PAX5 and EBER-ISH negativity are key features for suspecting ATLL with HTLV-1–positive HRS-like cells, and diffuse HBZ-ISH positivity is critical for confirming the diagnosis. These findings expand the pathological spectrum of ATLL and provide guidance for appropriate case selection in future clinical trials.
A part of clinicopathological findings have been presented in Tsuruta et al.19
Acknowledgments
The authors thank Chikako Nagamine and the Pathology Unit staff of the University of the Ryukyus Hospital, Okinawa Prefectural Nanbu Medical Center and Children’s Medical Center, Heartlife Hospital, and Nakagami Hospital for providing samples and clinical data.
This study was supported by Grants-in-Aid for Scientific Research (KAKENHI) from the Ministry of Education, Culture, Sports, Science, and Technology of Japan (19K07438 [K.K.] and 19K17835 [S.S.]) and grants from Okinawa Prefecture (K.K.), Mochida Memorial Foundation for Medical and Pharmaceutical Research (K.K.), Japan Leukemia Research Fund (K.K.), Okinawa Internal Medicine Research Promotion Society (K.K.), Japanese Society of Hematology (K.K.), Takeda Science Foundation (K.K.), Life Medicine Research Promotion Foundation (K.K.), and The Shinnihon Foundation of Advanced Medical Treatment Research (K.K.).
Authorship
Contribution: K.K. contributed to study conception and design; M.T., K.K., S.S., and T.F. contributed to development of methodology; K.K., S.T., Y.T., T.M., N. Nakada, I.N., K.M., S.K., S.M., M.H., T.F., and H.M. contributed to data acquisition; K.K., M.T., Y.Y.K., and N. Nakamura contributed to data analysis and interpretation; K.K. contributed to manuscript writing and revision; K.K., M.T., S.S., Y.T., T.M., K.M., S.K., N. Nakada, M.H., S.T., I.N., K.O., N.I., Y.Y.K., N. Nakamura, S.M., H.M., and T.F. contributed to manuscript review; and S.T., T.M., N. Nakada, I.N., S.M., M.H., T.F., and H.M. provided administrative, technical, or material support.
Conflict-of-interest disclosure: The authors declare no competing financial interests.
Correspondence: Kennosuke Karube, Department of Pathology and Cell Biology, Graduate School of Medicine, University of the Ryukyus, 207 Uehara, Nishihara-cho, Okinawa 903-0215, Japan; e-mail: karube@med.u-ryukyu.ac.jp.
References
Author notes
The full-text version of this article contains a data supplement.