Key Points
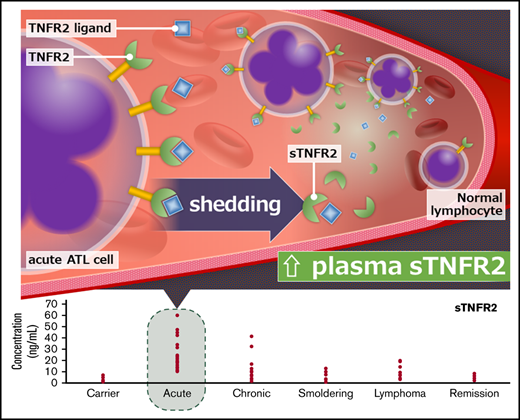
A distinct sTNFR2 range is observed in acute ATL patients (10-60 ng/mL) vs ACs (1-8 ng/mL) and those in remission (2-9 ng/mL).
High cell surface expression of TNFR2 on acute ATL cells suggests the clinical utility of sTNFR2 as a diagnostic biomarker for acute ATL.
Abstract
Adult T-cell leukemia/lymphoma (ATL) is a human T-cell leukemia virus type 1 (HTLV-1)–associated T-cell malignancy with generally poor prognosis. Although only ∼5% of HTLV-1 carriers progress to ATL, early diagnosis is challenging because of the lack of ATL biomarkers. In this study, we analyzed blood plasma profiles of asymptomatic HTLV-1 carriers (ACs); untreated ATL patients, including acute, lymphoma, smoldering, and chronic types; and ATL patients in remission. Through SOMAscan, expression levels of 1305 plasma proteins were analyzed in 85 samples (AC, n = 40; ATL, n = 40; remission, n = 5). Using gene set enrichment analysis and gene ontology, overrepresented pathways in ATL vs AC included angiogenesis, inflammation by cytokines and chemokines, interleukin-6 (IL-6)/JAK/STAT3, and notch signaling. In selecting candidate biomarkers, we focused on soluble tumor necrosis factor receptor 2 (sTNFR2) because of its active role in enriched pathways, extreme significance (Welch’s t test P < .00001), high discrimination capacity (area under the curve >0.90), and novelty in ATL research. Quantification of sTNFR2 in 102 plasma samples (AC, n = 30; ATL, n = 68; remission, n = 4) using enzyme-linked immunosorbent assay showed remarkable elevations in acute ATL, at least 10 times those of AC samples, and return of sTNFR2 to AC state levels after achieving remission. Flow cytometry and immunostaining validated the expression of TNFR2 in ATL cells. No correlation between sIL-2 and sTNFR2 levels in acute ATL was found, suggesting the possibility of sTNFR2 as an independent biomarker. Our findings represent the first extensive blood-based proteomic analysis of ATL, suggesting the potential clinical utility of sTNFR2 in diagnosing acute ATL.
Introduction
Adult T-cell leukemia/lymphoma (ATL) is a mature T-cell neoplasm associated with human T-cell leukemia virus type 1 (HTLV-1).1-4 The classification of ATL into acute, lymphoma, chronic, and smoldering clinical subtypes was proposed based on prognostic factors, clinical features, and natural history of the disease.5 The “Revised Adult T-Cell Leukemia-Lymphoma International Consensus Meeting Report” also includes a recently proposed variant of the lymphoma type ATL called the extranodal primary cutaneous variant,6 which has a fatal clinical course and is considered aggressive ATL.7 Patients with aggressive ATL (ie, acute, lymphoma, and unfavorable chronic types) are frequently treated with intensive multidrug chemotherapy with or without mogamulizumab, a defucosylated anti–CC chemokine receptor 4 monoclonal antibody. Aggressive ATL typically has a very poor prognosis, with a median survival time of 8 to 10 months. Median survival time with the VCAP-AMP-VECP regimen (ie, vincristine, cyclophosphamide, doxorubicin, and prednisone [VCAP]; doxorubicin, ranimustine, and prednisone [AMP]; and vindesine, etopside, carboplatin, and prednisone [VECP]), which showed the best results for chemotherapy in patients with untreated aggressive ATL in the phase 3 Japan Clinical Oncology Group (JCOG) 9801 trial (1998-2003), was only 13 months.8 Indolent ATL (ie, favorable chronic and smoldering types) generally progresses slowly, and it is therefore recommended that patients undergo monitoring through watchful waiting or treatment with interferon-α and zidovudine.9 However, most patients with indolent ATL will eventually die after progression to aggressive ATL during the chronic course of illness; the prognosis is not good, and there is no plateau phase in the survival curve.10
HTLV-1 is a retrovirus currently endemic in southwest Japan, sub-Saharan Africa, South America, the Caribbean, parts of the Middle East, and Australo-Melanesia; the estimated prevalence of infection is 10 to 20 million worldwide.11-15 HTLV-1 generally does not cause clinical features in a majority of infected individuals; in fact, ∼95% of HTLV-1 carriers remain asymptomatic throughout their lives.15 The lifetime risk of developing ATL in HTLV-1 carriers in Japan is ∼6% to 7% for men and ∼2% to 3% for women, with 1000 new ATL cases diagnosed each year.12
Because of the poor prognosis of ATL, it is vital to identify HTLV-1 carriers at high risk of developing ATL to establish early interventional treatment methods. A nationwide prospective study of 1218 asymptomatic HTLV-1 carriers (ACs) in Japan revealed that none developed ATL among those with a baseline proviral load lower than ∼4 copies per 100 peripheral blood mononuclear cells (PBMCs); higher proviral load, advanced age, family history of ATL, and first opportunity for HTLV-1 testing during treatment for another disease not related to HTLV-1 were independent risk factors for the progression of ATL in multivariate Cox analyses.16 In Japan, median age at diagnosis of ATL was reported as 68 years (range, 34-100 years),17 which is older than that in other ATL endemic areas such as the Caribbean (median, 54 years; range, 28-87 years).18 Geographical differences in the age at ATL onset may be explained not only by genetic factors, but also by several epigenetic and environmental factors that begin to have an effect during the 60-year latency period.12 Many of the epigenetic factors involved in the progression of ATL remain unknown, prompting us to explore the less researched field of ATL epigenetics: proteomics.
Plasma proteins play key roles in various biological processes, such as signaling, transport, growth, repair, and defense mechanisms, which are often dysregulated in disease states. Analyses of plasma proteins prove valuable for developing disease biomarkers with potential clinical utility, because plasma and other components of blood remain the predominant specimens for routine analysis. Previously, a high-throughput mass spectrometric plasma protein analysis of patients with HTLV-1–associated myelopathy was performed to describe the proteomic content of extracellular vesicles in comparison with the AC state.19 However, comparative proteomic profiling for ACs and ATL patients has not yet been performed. To further understand the biological processes in the progression of ATL, we explored the disease, remission, and carrier states with a proteomic approach. In this study, we aimed to identify potential protein biomarkers for: (1) the onset of ATL by comparing plasma profiles of ACs vs ATL patients; (2) the progression of ATL to its aggressive forms by comparing plasma profiles of acute, lymphoma, smoldering, and chronic types; and (3) the achievement of remission or response to chemotherapy in ATL patients by comparing plasma profiles of pre- and postremission states. Here, we show the discovery of soluble tumor necrosis factor receptor 2 (sTNFR2) as a novel and promising diagnostic biomarker for acute ATL.
Methods
Patients and specimens
Samples were obtained from the following: (1) ACs confirmed with anti-HTLV–1 antibodies through the particle-agglutination method; (2) ATL patients diagnosed based on the criteria proposed by the JCOG5 and confirmed with monoclonally integrated HTLV-1 proviral genome using the Southern blot hybridization method, as described previously20 ; and (3) previously diagnosed ATL patients achieving complete remission for >4 weeks, where remission was judged based on a modified version21 of the World Health Organization response criteria.22 Samples were procured from 7 institutions in Okinawa Prefecture, Japan (University of the Ryukyus Hospital, Heart Life Hospital, Nakagami Hospital, Naha City Hospital, Nanbu Medical Center, Chubu Tokushukai Hospital, and Kariyushi Hospital) from November 2016 to November 2019. Plasma and PBMCs were separated from blood samples and stored at −80°C until use. Lymph node and subcutaneous skin lesions from ATL patients were also harvested.
Proteomic profiling
Expression levels of 1305 proteins from cryopreserved plasma samples (1 mL each) were determined. The SOMAscan assay utilizes new-generation protein capture slow off-rate modified aptamer (SOMAmer) reagents, which are modified nucleotides with amino acid– or protein-like side chains that have affinities for proteins and, at the same time, are recognizable by DNA hybridization probes and measurable by fluorescence.23,24 The 1305 proteins analyzed were listed using their protein coding gene IDs (eg, TNFα, TNFR1, and TNFR2 are reflected as TNF, TNFRSF1A, and TNFRSF1B, respectively).
Statistical considerations
Significance levels of proteins in ACs vs ATLs and ATLs vs remissions were calculated using Welch’s t test, and discrimination capacities were determined by calculating the area under the receiver operating characteristic curve (AUC). We illustrated elevations and decreases in AC, ATL, and remission states using box-and-whisker and dot plots. All statistical analyses were performed using Python 3.6.2 (Python Software Foundation, Beaverton, OR) with the following packages: Statsmodels 0.8.0 for t tests and Matplotlib 2.0.2 for box-and-whisker plots.
Pathway enrichment
To generate heatmap visualizations and determine significantly enriched pathways among various parameters, such as ATL/AC/remission states, age, and ATL subtypes, we used gene set enrichment analysis (GSEA version 3.0; Broad Institute; Molecular Signatures Database: hallmarks and curated gene sets) with a false discovery rate cutoff of <25%, run at 1000 permutations. We also determined overrepresented pathways in ATL states using gene ontology (GO PANTHER Pathways) by inputting proteins categorized as extremely significant. Protein names in the heatmaps are also reflected as protein coding gene IDs.
Plasma protein concentrations
To determine plasma protein concentration levels of selected candidate biomarkers, the following enzyme-linked immunosorbent assay (ELISA) kits were sourced: sTNFα, sTNFR1, and sTNFR2 (R&D Systems, Minneapolis, MN) and sTNFRSF8 (RayBiotech, Peachtree Corners, GA).
Flow cytometric analysis
We proceeded with flow cytometry to determinne if elevations in plasma levels of sTNFR2 were correlated with ATL cell surface expression of TNFR2. We performed flow cytometry with the BD FACSCalibur cell analyzer, and data were analyzed with CellQuest Pro software (BD Biosciences, San Jose, CA). Briefly, cryopreserved PBMCs were retrieved by rapid thawing of cryotubes in a 37°C water bath, Fc-blocked with 2 ng/mL of human immunoglobulin G and resuspended in fluorescence-activated cell sorting buffer (phosphate-buffered saline containing 2% fetal calf serum and 0.1% sodium azide), and then washed. Cell surface staining was performed on ice for 30 minutes using the following antibody combinations: fluorescein isothiocyanate–conjugated rabbit anti-SynCAM TLSC1/CADM1 monoclonal antibody (MBL Co., Ltd, Nagoya, Japan), PE-cyanine7 mouse anti-human CD7 (BD Biosciences), Alexa Fluor 647 anti-human CD4 antibody (Biolegend, San Diego, CA), and phycoerythrin-conjugated anti-human TNFR2 (Miltenyi Biotec, Bergisch Gladbach, Germany). Cells were then washed and resuspended in 1% paraformaldehyde in phosphate-buffered saline (Wako Pure Chemical Industries, Ltd, Osaka, Japan). The gating strategy for selecting CD4+ T-cell lymphocyte subpopulations is shown in in Figure 3A. Because CADM1 expression is a marker for aggressive ATL and stepwise downregulation of CD7 is closely associated with clonal expansion of HTLV-1–infected cells in ATL,25 we also plotted CD4 against CD7 and TNFR2 against CADM1.
Immunostaining
Immunostaining with polyclonal antibodies against TNFR2 (Enzo Life Sciences, Farmingdale, NY) for tissue specimens including lymph nodes and subcutaneous skin lesions obtained from ATL patients was performed with heat-mediated antigen retrieval of formalin-fixed paraffin-embedded sections of the samples.
mRNA expression
Finally, to determine TNFR2 messenger RNA (mRNA) expression in ACs and ATL patients, semiquantitative reverse transcription polymerase chain reaction was performed. Total cellular RNA from PBMCs was extracted using RNeasy Mini Kit (Qiagen, Hilden, Germany) according to the manufacturer’s instructions. Reverse transcription of 500 ng of RNA to complementary DNA was carried out using SuperScript III First-Strand Synthesis System (Invitrogen, Carlsbad, CA), with oligo(dT)20 primer. Real-time polymerase chain reaction was completed on StepOnePlus (Applied Biosystems, Foster City, CA) using TaqMan Fast Advanced Master Mix (Thermo Fisher Scientific, Waltham, MA). mRNA expression of TNFR2 was calculated using the 2−ΔΔCT method. TaqMan primer-probe sets human TNFR2 (Hs00153550_m1) and housekeeping gene β-actin (Hs01060665_g1) were used in this assay (Thermo Fisher Scientific).
Ethical considerations
All samples and information of stored cryopreserved samples were obtained under prior comprehensive consent for storage and use in correlative studies after receiving approval from the institutional review board of each institution in accordance with the Declaration of Helsinki. The research plan was approved by the institutional review board of the University of the Ryukyus and NEC Corporation Japan.
Results
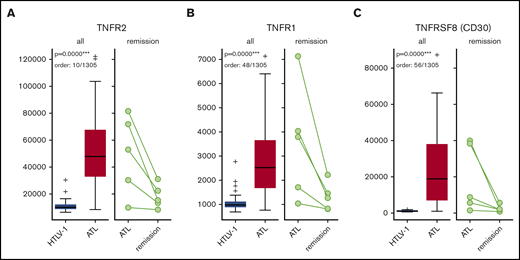
A total of 85 plasma samples (ACs, n = 40; ATLs, n = 40; remissions, n = 5) were selected for protein analysis using the SOMAscan assay. Demographic and clinical information of ATL patients is provided in supplemental Table 1. We categorized differences in protein levels in ACs vs ATLs as extremely significant, very significant, significant, and nonsignificant (Table 1) and listed the top 10 elevated or decreased proteins and their functions (Table 2). We also determined significance levels of 11 proteins overrepresented in ATLs vs remissions (Table 3) and listed these proteins with their functions (Table 4). The complete list of all 1305 plasma proteins measured is shown in supplemental Table 2, which includes P values and AUC values. We depicted the differences in plasma protein expression levels of some of the candidate biomarkers in AC vs ATL states, as well as in ATL vs remission states, in box-and-whisker plots and dot plots, as shown in Figure 1.
Number of plasma proteins with significantly different levels in HTLV-1 vs ATL
| P . | Interpretation . | No. of proteins . | Cumulative frequency . |
|---|---|---|---|
| <.00001 | Extremely significant | 176 | 176 |
| .00001 ≤ and < .0001 | 58 | 234 | |
| .0001 ≤ and < .001 | 99 | 333 | |
| .001 ≤ and < .01 | Very significant | 149 | 482 |
| .01 ≤ and < .05 | Significant | 149 | 631 |
| ≥.05 | Not significant | 674 | 1305 |
| P . | Interpretation . | No. of proteins . | Cumulative frequency . |
|---|---|---|---|
| <.00001 | Extremely significant | 176 | 176 |
| .00001 ≤ and < .0001 | 58 | 234 | |
| .0001 ≤ and < .001 | 99 | 333 | |
| .001 ≤ and < .01 | Very significant | 149 | 482 |
| .01 ≤ and < .05 | Significant | 149 | 631 |
| ≥.05 | Not significant | 674 | 1305 |
Top 10 proteins with significantly different levels in HTLV-1 vs ATL
| . | Protein name . | Elevation (up) or decrease (down) . | Protein function . | Protein coding gene ID . | UniProt . | P . | AUC . |
|---|---|---|---|---|---|---|---|
| 1 | Coagulation factor Xa | Up in ATL | Blood coagulation | F10 | P00742 | 1.82E-16 | 0.99 |
| 2 | Coagulation factor X | Up in ATL | Blood coagulation | F10 | P00742 | 4.13E-16 | 0.99 |
| 3 | Cadherin-6 | Down in ATL | Cell adhesion | CDH6 | P55285 | 9.32E-14 | 0.93 |
| 4 | Insulin-like growth factor binding protein 4 | Up in ATL | IGF regulation | IGFBP4 | P22692 | 2.90E-12 | 0.91 |
| 5 | Lysozyme C | Up in ATL | Hydrolysis | LYZ | P61626 | 5.65E-12 | 0.89 |
| 6 | BDNF/NT-3 growth factor receptor | Down in ATL | Neuronal development and regulation | NTRK2 | Q16620 | 1.70E-11 | 0.88 |
| 7 | Granulins | Up in ATL | Cell growth regulation | GRN | P28799 | 2.73E-11 | 0.91 |
| 8 | RAS GTPase-activating protein 1 | Down in ATL | Cell proliferation and differentiation | RASA1 | P20936 | 2.77E-11 | 0.95 |
| 9 | Interleukin-18-binding protein | Up in ATL | Cell immunity induction | IL18BP | O95998 | 5.03E-11 | 0.93 |
| 10 | TNFR2 | Up in ATL | Apoptosis modulation and signaling | TNFRSF1B | P20333 | 6.82E-11 | 0.92 |
| . | Protein name . | Elevation (up) or decrease (down) . | Protein function . | Protein coding gene ID . | UniProt . | P . | AUC . |
|---|---|---|---|---|---|---|---|
| 1 | Coagulation factor Xa | Up in ATL | Blood coagulation | F10 | P00742 | 1.82E-16 | 0.99 |
| 2 | Coagulation factor X | Up in ATL | Blood coagulation | F10 | P00742 | 4.13E-16 | 0.99 |
| 3 | Cadherin-6 | Down in ATL | Cell adhesion | CDH6 | P55285 | 9.32E-14 | 0.93 |
| 4 | Insulin-like growth factor binding protein 4 | Up in ATL | IGF regulation | IGFBP4 | P22692 | 2.90E-12 | 0.91 |
| 5 | Lysozyme C | Up in ATL | Hydrolysis | LYZ | P61626 | 5.65E-12 | 0.89 |
| 6 | BDNF/NT-3 growth factor receptor | Down in ATL | Neuronal development and regulation | NTRK2 | Q16620 | 1.70E-11 | 0.88 |
| 7 | Granulins | Up in ATL | Cell growth regulation | GRN | P28799 | 2.73E-11 | 0.91 |
| 8 | RAS GTPase-activating protein 1 | Down in ATL | Cell proliferation and differentiation | RASA1 | P20936 | 2.77E-11 | 0.95 |
| 9 | Interleukin-18-binding protein | Up in ATL | Cell immunity induction | IL18BP | O95998 | 5.03E-11 | 0.93 |
| 10 | TNFR2 | Up in ATL | Apoptosis modulation and signaling | TNFRSF1B | P20333 | 6.82E-11 | 0.92 |
Number of significantly different proteins in remission state
| HTLV-1 and ATL P < .00001 (176 proteins) . | Remission and ATL . | ||||||
|---|---|---|---|---|---|---|---|
| <.00001 . | <.0001 . | <.001 . | <.01 . | <.05 . | ≥.05 . | ||
| Remission and HTLV-1 | ≥.05 | 11 | 5 | 15 | 24 | 20 | 32 |
| HTLV-1 and ATL P < .00001 (176 proteins) . | Remission and ATL . | ||||||
|---|---|---|---|---|---|---|---|
| <.00001 . | <.0001 . | <.001 . | <.01 . | <.05 . | ≥.05 . | ||
| Remission and HTLV-1 | ≥.05 | 11 | 5 | 15 | 24 | 20 | 32 |
We identified a total of 75 proteins with significantly different expression levels in postremission vs ATL, which were also not significantly different in remission vs HTLV-1, to determine which proteins returned to the HTLV-1 carrier state after achieving remission.
Expression levels of 11 proteins that returned to HTLV-1 state after achieving remission in ATL patients with P > .00001
| . | Protein name . | Elevation (up) or decrease (down) . | Protein function . | Protein coding gene . | UniProt . | P . |
|---|---|---|---|---|---|---|
| 1 | Soluble L-selectin | Down in remission | Leukocyte-endothelial cell adhesion | SELL | P14151 | 9.98E-8 |
| 2 | Cell adhesion molecule 1 | Down in remission | Cell adhesion | CADM1 | Q9BY67 | 1.26E-7 |
| 3 | TNFRSF8 | Down in remission | Regulation of cell growth, activated lymphoblast transformation | TNFRSF8 | P28908 | 1.62E-7 |
| 4 | LDL receptor related protein 8 | Down in remission | Cholesterol transport receptor | LRP8 | Q14114 | 6.03E-7 |
| 5 | Lymphocyte activation gene | Down in remission | Lymphocyte activation | LAG3 | P18627 | 6.23E-7 |
| 6 | Tissue inhibitor of metallopeptidase-1 | Down in remission | Promotion of erythroid progenitor stem cell proliferation | TIMP1 | P01033 | 9.17E-7 |
| 7 | Angiopoietin-2 | Down in remission | Vascular remodeling, proliferation | ANGPT2 | O15123 | 8.05E-06 |
| 8 | T-lymphocyte surface antigen Ly-9/CD229 | Down in remission | Innate immune response | LY9 | Q9HBG7 | 1.07E-6 |
| 9 | Deoxycytidine-triphosphatase 1 | Down in remission | DNA replication | DCTPP1 | Q9H773 | 1.14E-6 |
| 10 | T-cell cytokine receptor (WSX-1) TCCR | Down in remission | Induction of Th-1 type immune response | IL27RA | Q6UWB1 | 2.99E-6 |
| 11 | Ephrin A4 | Down in remission | Metastasis | EFNA4 | P52798 | 4.51E-6 |
| . | Protein name . | Elevation (up) or decrease (down) . | Protein function . | Protein coding gene . | UniProt . | P . |
|---|---|---|---|---|---|---|
| 1 | Soluble L-selectin | Down in remission | Leukocyte-endothelial cell adhesion | SELL | P14151 | 9.98E-8 |
| 2 | Cell adhesion molecule 1 | Down in remission | Cell adhesion | CADM1 | Q9BY67 | 1.26E-7 |
| 3 | TNFRSF8 | Down in remission | Regulation of cell growth, activated lymphoblast transformation | TNFRSF8 | P28908 | 1.62E-7 |
| 4 | LDL receptor related protein 8 | Down in remission | Cholesterol transport receptor | LRP8 | Q14114 | 6.03E-7 |
| 5 | Lymphocyte activation gene | Down in remission | Lymphocyte activation | LAG3 | P18627 | 6.23E-7 |
| 6 | Tissue inhibitor of metallopeptidase-1 | Down in remission | Promotion of erythroid progenitor stem cell proliferation | TIMP1 | P01033 | 9.17E-7 |
| 7 | Angiopoietin-2 | Down in remission | Vascular remodeling, proliferation | ANGPT2 | O15123 | 8.05E-06 |
| 8 | T-lymphocyte surface antigen Ly-9/CD229 | Down in remission | Innate immune response | LY9 | Q9HBG7 | 1.07E-6 |
| 9 | Deoxycytidine-triphosphatase 1 | Down in remission | DNA replication | DCTPP1 | Q9H773 | 1.14E-6 |
| 10 | T-cell cytokine receptor (WSX-1) TCCR | Down in remission | Induction of Th-1 type immune response | IL27RA | Q6UWB1 | 2.99E-6 |
| 11 | Ephrin A4 | Down in remission | Metastasis | EFNA4 | P52798 | 4.51E-6 |
Box-and-whisker plots for sTNFR1, sTNFR2, and sTNFRSF8 in HTLV-1 ACs vs ATL patients and ATL patients vs those in remission. Numbers on the left side are in relative fluorescence units. sTNFR1 (A), sTNFR2 (B), and sTNFRSF8 (C).
Box-and-whisker plots for sTNFR1, sTNFR2, and sTNFRSF8 in HTLV-1 ACs vs ATL patients and ATL patients vs those in remission. Numbers on the left side are in relative fluorescence units. sTNFR1 (A), sTNFR2 (B), and sTNFRSF8 (C).
Enriched pathways and heatmap visualization
Heatmap visualization and pathway enrichment analysis for the following phenotype comparisons were generated via GSEA: (1) ATL vs AC, (2) ATL patients age <70 vs ≥70 years, (3) acute vs AC, (4) acute vs nonacute (including lymphoma, smoldering, and chronic), (5) acute vs chronic, (6) acute vs lymphoma, (7) acute vs smoldering, (8) chronic vs AC, (9) chronic vs lymphoma, (10) chronic vs smoldering, (11) lymphoma vs AC, (12) lymphoma vs smoldering, (13) smoldering vs AC, (14) preremission ATL vs remission (same patients followed up), (15) preremission ATL vs ATL (patients who did not achieve remission), (16) remission vs ATL (patients who did not achieve remission), and (17) AC vs remission (supplemental Figure 1A-Q for heatmap visualizations of each phenotype comparison; supplemental Tables 3-19 for enriched pathways). Overrepresented pathways in ATL vs AC, as generated by gene ontology, included inflammation by cytokine and chemokine signaling, IL signaling pathway, notch signaling, angiogenesis pathway, p53 pathway by glucose deprivation, blood coagulation, and ras pathway, among others (supplemental Figure 2).
Plasma protein levels of sTNFα and TNF superfamily receptors
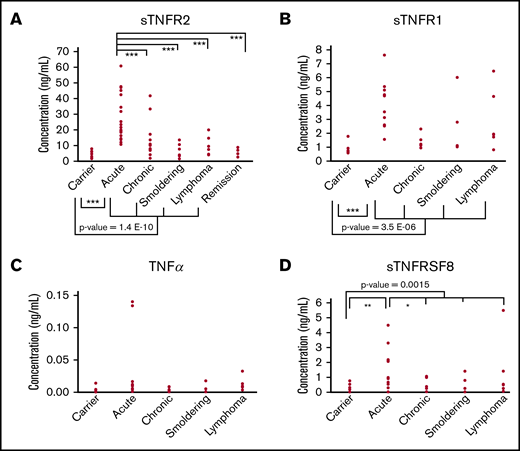
Proteins with active roles in the enriched pathways, extreme significance levels (P < .00001), and high discrimination capacities (AUC >0.90) were deemed candidate biomarkers in this study. Among the candidate biomarkers, we focused on sTNFR1, sTNFR2, and sTNFRSF8, which are receptors belonging to a superfamily of proteins: the TNF superfamily. We also included TNFα in our analysis, because it is the ligand of TNFR1 and TNFR2. For sTNFR2, a total of 102 plasma samples (AC, n = 30; ATL, n = 68 [acute, n = 33; lymphoma, n = 9; chronic, n = 12; smoldering, n = 14], and remission, n = 4) were confirmed using ELISA. A remarkable increase in sTNFR2 could be seen in ATL patients vs ACs (P < .001), especially in patients with acute ATL vs ACs (range, 10-60 ng/mL vs 1-8 ng/mL; P < .001; Figure 2A). A significant increase in sTNFR2 could also be seen in lymphoma ATL (P < .01); however, the values were quite varied, with some patients having low sTNFR2 levels (range, 1.5-32.7 ng/mL). Significantly higher sTNFR2 levels were found in acute ATL patients compared with those with other ATL subtypes. Interestingly, sTNFR2 levels of ACs and ATL patients who achieved remission were similar (range, 2-9 ng/mL in patients in remission). For sTNFR1, sTNFα, and sTNFRSF8, we tested 38 plasma samples (AC, n = 9; ATL, n = 29 [acute, n = 13; lymphoma, n = 6; chronic, n = 6; smoldering, n = 4]) using ELISA. Significant increases in sTNFR1 were apparent in ATL patients compared with ACs, especially in acute ATL patients (Figure 2B). Regarding sTNFα protein levels, no significant differences were found between ATL patients vs ACs (Figure 2C). For sTNFRSF8, significant increases in acute ATL patients vs ACs, as well as in acute vs chronic ATL patients, can be seen (Figure 2D). Although elevated levels of sTNFR1 and sTNFRSF8 in acute ATL patients were also apparent, the cutoff point is not as distinct as that of sTNFR2. sTNFR2 concentration values for all samples can be found in supplemental Table 20.
Plasma protein levels of TNF and TNF superfamily receptor proteins in HTLV-1 ACs, ATL patients, and patients in remission. sTNFR2 (n = 102) (A), sTNFR1 (n = 38) (B), TNFα (n = 38) (C), and sTNFRSF8 (n = 38) (D). Statistically significant P values in ACs vs ATL patients are shown. *P < .05, **P < .01, ***P < .001.
Plasma protein levels of TNF and TNF superfamily receptor proteins in HTLV-1 ACs, ATL patients, and patients in remission. sTNFR2 (n = 102) (A), sTNFR1 (n = 38) (B), TNFα (n = 38) (C), and sTNFRSF8 (n = 38) (D). Statistically significant P values in ACs vs ATL patients are shown. *P < .05, **P < .01, ***P < .001.
Cell surface expression of TNFR2 on PBMCs of acute ATL cells
Of the proteins tested using ELISA, we focused on TNFR2 because of its remarkable and consistent increase in acute ATL patients. Flow cytometric analysis of PBMCs from 12 samples (AC, n = 6; acute ATL, n = 6) showed significantly higher TNFR2+CADM1+ populations in acute ATL patients (mean, 54.9%; range, 33.3%-80.3%) compared with ACs (mean, 26.5%; range, 24.7%-28.9%; Figure 3B-D). Plasma sTNFR2 levels and TNFR2 cell surface expression levels were positively correlated, with a correlation coefficient of 0.63 (Figure 3E).
TNFR2 marker expression in CD4+ T cells in HTLV-1 ACs vs ATL patients. (A) CD4+ T-cell lymphocyte gating strategy. (B) TNFR2+CADM1+ asymptomatic HTLV-1 carriers. (C) TNFR2+CADM1+ T-cell expression among acute ATL patients. (D) Percentage of TNFR2 expression in CADM1+ (up) and CD4+ T cells (down). (E) Correlation of plasma sTNFR2 concentration and cell surface TNFR2+CADM1+ T-cell expression (n = 12 [AC, n = 6; acute ATL, n = 6]). **P < .01.
TNFR2 marker expression in CD4+ T cells in HTLV-1 ACs vs ATL patients. (A) CD4+ T-cell lymphocyte gating strategy. (B) TNFR2+CADM1+ asymptomatic HTLV-1 carriers. (C) TNFR2+CADM1+ T-cell expression among acute ATL patients. (D) Percentage of TNFR2 expression in CADM1+ (up) and CD4+ T cells (down). (E) Correlation of plasma sTNFR2 concentration and cell surface TNFR2+CADM1+ T-cell expression (n = 12 [AC, n = 6; acute ATL, n = 6]). **P < .01.
mRNA expression of TNFR2 on HTLV-1 carriers and ATL patients
TNFR2 mRNA expression analysis of RNA extracted from 12 PBMCs (AC, n = 3; acute ATL, n = 9) showed no significant increase in ACs vs ATL patients (supplemental Figure 3).
Discussion
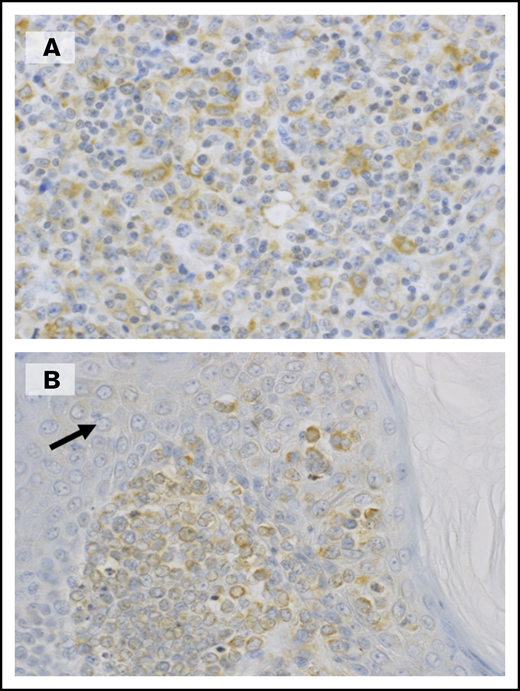
In this study, we have demonstrated a novel approach in searching for ATL biomarkers through the use of a SOMAmer-based high-throughput proteomic assay, followed by an extensive proteomic pathway analysis and confirmed using standard laboratory techniques such as ELISA, flow cytometry, and immunostaining. This approach led us to the discovery of candidate biomarkers belonging to the TNF receptor superfamily, namely sTNFR1, sTNFR2, and sTNFRSF8. Among them, sTNFR2 was deemed to be the most prominent in comparing AC vs ATL. Plasma protein analysis of sTNFR2 showed: (1) remarkable and distinct elevations in acute ATL (10-60 ng/mL), approximately at least 10 times higher than levels in ACs (1-9 ng/mL) and reference values of healthy controls26 (1-5 ng/mL), and (2) a return of sTNFR2 to levels similar to carrier state levels after achieving remission (2-8 ng/mL). Flow cytometric analysis showed higher cell surface expression of TNFR2 in ATL patients vs ACs (Figure 3B-D), and sTNFR2 concentration levels correlated with cell surface TNFR2 expression (Figure 3E). TNFR2 was immunohistochemically positive in atypical lymphoid cells (Figure 4A), as well as in skin infiltrates (Figure 4B), both from tumor cells of patients diagnosed with lymphoma ATL. However, the circulating levels of sTNFR2 among lymphoma ATL patients seemed to vary, and the range of concentration values was not as distinct compared with the range in acute ATL patients. These results indicate the potential of sTNFR2 as a clinically useful diagnostic biomarker for acute ATL.
Expression of TNFR2 in tumor cells from ATL patients. (A) A biopsied lymph node shows numerous atypical large lymphoid cells positive for TNFR2. (B) In this skin lesion, infiltrating tumor cells were positive for TNFR2, whereas epithelial cells (arrow) were negative. (A-B) Original magnification ×400, immunostaining with polyclonal antibodies against TNFR2.
Expression of TNFR2 in tumor cells from ATL patients. (A) A biopsied lymph node shows numerous atypical large lymphoid cells positive for TNFR2. (B) In this skin lesion, infiltrating tumor cells were positive for TNFR2, whereas epithelial cells (arrow) were negative. (A-B) Original magnification ×400, immunostaining with polyclonal antibodies against TNFR2.
TNFR1, TNFR2, and TNFRSF8, which are all receptors of the TNF superfamily, consist of proteins that are activated by various TNF-like cytokine ligands, including TNFα itself and lymphotoxin-α.27 In contrast to TNFR1, TNFR2 lacks the death domain and recruits TNFR-associated factor 2, which promotes cell activation, proliferation, and survival,28,29 likely via NF-κB activation. Previously, a comprehensive molecular analysis of ATL by Kataoka et al30,31 described NF-κB activation and multiple gene mutations in the ATL state to have a substantial impact on ATL progression. Of note, some of the overrepresented proteins found in the present study corresponded with some of the mutated protein coding genes in the aforementioned molecular analysis, including FYN and VAV1. In our analysis, FYN and VAV1 both have decreased expression levels in ATL patients vs ACs (supplemental Figure 1A). Another NF-κB–activating TNF superfamily receptor significantly overrepresented in this study was TNFRSF4 (OX40; supplemental Table 2). Interestingly, high levels of soluble OX40 has recently been associated with acute ATL patients,32 and shedding of OX40 is speculated to be involved in skin infiltration of leukemic ATL cells.33,34 Other overrepresented proteins in our analysis, such as CADM1, L-selectin, VCAM1, NOTCH1, β2M, and CD163 (elevated in ATL), as well as SPARC (decreased in ATL), have been previously implicated in ATL by virtue of gene mutation, protein deregulation, or gene network–based analyses,31,35-42 indicating the reproducibility of our proteomic profiling.
The TNFα-TNFR2 interaction has been associated with inflammatory conditions such as rheumatoid arthritis, allergies, and autoimmune diseases such as psoriasis.43 In recent years, TNFR2 has been considered a novel target for cancer immunotherapy because it can be found on the surface of potent regulatory T cells and is aberrantly expressed in various human tumor cells.44 TNFR2 expression has also been associated in patients with acute myeloid leukemia45 and Sèzary syndrome.46 A recent study revealed that peripheral T-cell non-Hodgkin lymphoma patients with high levels of circulating TNFR2 (≥2.16 ng/mL) had a twofold increased relative risk for shorter overall survival.47 Although sTNFR2 levels seem to be associated with other non-ATL peripheral T-cell lymphomas, sTNFR2 levels among acute ATL patients enrolled in our study seemed to be more prominently elevated, with concentration values ∼9 times higher than those of the patients in the aforementioned study (18.7 vs 2.16 ng/mL).
It is worth noting that TNFR2 is highly shed from the cell surface after binding with TNFα, and this TNFα-TNFR2 complex separates immediately in plasma. Because neither TNFα plasma levels nor TNFR2 mRNA expression levels in ACs vs ATL patients were significantly different, we are considering the possibility of TNFR2 binding to a ligand other than TNFα or TNF receptor shedding. To shed light on the mechanisms of TNFR2 expression, we have currently initiated an analysis of plasma lymphotoxin-α levels and ADAM17, a sheddase involved in the shedding of TNFR2.48,49 Interestingly, TNFR1, TNFR2, and TNFRSF8 are all substrates of ADAM17.50 TNFRSF8, sometimes referred to as CD30, has been previously associated with ATL, in both cell expression studies51 and plasma protein analyses52 ; however, in this study, some acute ATL patients had low levels of sTNFRSF8, similar to levels in ACs (Figure 2D), in contrast to sTNFR2, where a range of values for acute ATL could be established.
Our pathway analysis revealed enrichment of IL-2 STAT5 signaling, IL6/JAK/STAT3 signaling, epithelial mesenchymal transition, interferon-γ response, and angiogenesis pathways (supplemental Table 3). One of the roles of the HTLV-1 Tax oncoprotein is the induction of cytokines, including IL-2, which has long been known for its role in ATL leukemogenesis.53 We compared sTNFR2 levels among acute ATL patients with sIL-2R levels obtained from existing patient clinical data and found no correlation (Pearson’s coefficient r = 0.23), suggesting the possibility of sTNFR2 as an independent biomarker for acute ATL.
Among other cytokine receptors, IL-6 receptor subunit α was also an overrepresented protein in ATL, possibly having a role in activation of the JAK/STAT3 pathway, which is known to be aberrantly activated in hematopoietic malignancies and solid tumors.54-56 Older studies have shown that elevated levels of IL-6 are linked with inflammatory responses and poor general status among ATL patients.57 In HTLV-1–infected T cells, both IL-6 and IL-6 receptor have been reported to be elevated and associated with Tax activity.58
As for other candidate biomarkers, overrepresentation of various angiogenic factors (osteopontin, thrombospondin 2, TIMP1, angiopoietin 2, neuropilin 1, and follistatin-like 1) in ATL patients contributes to the enrichment of the angiogenesis pathway. Existing antiangiogenic treatments such as bevacizumab, a humanized monoclonal antibody against vascular endothelial growth factor, have been studied in ATL mice models, in which combination with CHOP (cyclophosphamide, doxorubicin, vincristine, and prednisone) therapy led to significantly prolonged overall survival.59 However, protein expression of vascular endothelial growth factor was not significantly different in ATL patients vs ACs in our study. This suggests that the angiogenic process in tumor cell promotion and metastasis for ATL cells may be a contribution of not only a single factor but rather many angiogenic factors that may replace the function of others in times of aberration. The roles of the abovementioned angiogenic factors in ATL progression should be further investigated.
Because acute ATL variants generally have a more rapid progression than other subtypes, we compared acute ATL patients with nonacute ATL patients (supplemental Figure 1D; supplemental Table 6). In comparing each subtype with AC (supplemental Tables 5, 10, 13, and 15), we showed elevations of different sets of cell adhesion molecules: CADM1, L-selectin, VCAM-1, and E-selectin for acute types; ICAM-1 for lymphoma types; L-selectin and CADM1 for chronic types; and CADM1 and ALCAM for smoldering types. Additional analyses of the proteomic profiles of all ATL subtypes are needed to provide more accurate subclassifications and elucidate the nature of ATL progression specific to each ATL subtype.
As for remission states, the decrease in sTNFR2 levels suggests that sTNFR2 may prove useful in monitoring chemotherapy sensitivity or progress in achieving remission. However, analyzing sTNFR2 levels in various treatment strategies using a larger sample is necessary to confirm whether sTNFR2 can be used as a chemotherapy-sensitivity marker.
The role of TNFR2 in ATL pathogenesis is yet to be explored and may prove to be pivotal in ATL research. With the developments in proteomic research and technology, it will be vital to analyze proteins for the improvement of diagnosis and treatment of disease, in the hope of improving patient survival outcomes.
Presented in part as an oral presentation at the 61st annual meeting of the American Society of Hematology, Orlando, FL, 9 December 2019 (oral abstract 0660).
For all original data, please contact the corresponding author, Takuya Fukushima (e-mail: fukutaku@med.u-ryukyu.ac.jp).
Acknowledgments
The authors thank all the patients who participated in this study, as well as the staff of Kariyushi Hospital, Ishigaki, Okinawa, for their contributions.
This work is partially supported by funds from the Advanced Medical Research Center, Faculty of Medicine, University of the Ryukyus.
Authorship
Contribution: T.F. designed the study; C.L.H.G. and T.F. wrote the manuscript; C.L.H.G. and Y.Y. had access to the raw data and analyzed the data; C.L.H.G., Y.Y., M.K., N.I., and T.F. conceptualized the visual abstract; C.L.H.G., M.M., N.I., and S.S. prepared samples and managed the data; C.L.H.G., M.K., and N.I. performed ELISA; M.K. and Y.T. performed flow cytometry; K. Karube performed immunostaining; K. Karube and Y.T. contributed reagents, materials, and analysis tools; N.I. performed mRNA analysis; M.H., T.M., K. Karimata, J.U., K.O., J.T., S.N., S.M., H.M., and T.F. managed the patients and collected samples; T.F. had full access to all the data in this study and had final responsibility for the decision to submit for publication; and all authors reviewed the manuscript.
Conflict-of-interest disclosure: T.F. and Y.Y. have patent rights related to findings from this study. NEC Corporation has a patent for the proteomic data (patent 2019-021539), and the University of the Ryukyus has a patent for data regarding sTNFR2 as an ATL biomarker (patent 2019-200986). The remaining authors declare no competing financial interests.
Correspondence: Takuya Fukushima, Laboratory of Hematoimmunology, Graduate School of Health Sciences, University of the Ryukyus, 207 Uehara, Nishihara, Okinawa, 903-0215 Japan; e-mail: fukutaku@med.u-ryukyu.ac.jp.
References
Author notes
The full-text version of this article contains a data supplement.

![TNFR2 marker expression in CD4+ T cells in HTLV-1 ACs vs ATL patients. (A) CD4+ T-cell lymphocyte gating strategy. (B) TNFR2+CADM1+ asymptomatic HTLV-1 carriers. (C) TNFR2+CADM1+ T-cell expression among acute ATL patients. (D) Percentage of TNFR2 expression in CADM1+ (up) and CD4+ T cells (down). (E) Correlation of plasma sTNFR2 concentration and cell surface TNFR2+CADM1+ T-cell expression (n = 12 [AC, n = 6; acute ATL, n = 6]). **P < .01.](https://ash.silverchair-cdn.com/ash/content_public/journal/bloodadvances/4/6/10.1182_bloodadvances.2019001429/3/m_advancesadv2019001429f3.png?Expires=1768240132&Signature=pwMavfom1MS-PXbfvAk9bBF7amLkBytZiZy~3hvBF1mAIh6uayqty~PrVV7RZ~gg2lO8RTca1AwLAB90WELKMz8VXDAbD4B2m35eocr6ZIczgoc-vv4dFVVXEk5sZuet7vu2yMkJ93h6Y7ws6DMTekpTjWu9thIZ54S6~jHlhZxOX7an68FoiqnsK1ZknFYKlXnJcKBhH7mLmMvNWcMPySArE~H8zqJqR0p57X76LKOG2wW0xV~-DgXaiCWuWN7StH7Ev0HQJo~kwpuIbQNAePgEv7EgxkfyYliantraLN0Y-ygmgWBHqwkooYjx8yavL~ydETlFh0i4NCmq6fKoKg__&Key-Pair-Id=APKAIE5G5CRDK6RD3PGA)