Key Points
ESAs can treat anemia of unknown etiology, and responses may be similar to those in CKD.
No statistically significant increase in cardiovascular events in those receiving ESAs was detected, but a type II error cannot be excluded.
Abstract
A specific cause of anemia cannot be identified in many elderly patients. Erythropoiesis-stimulating agents (ESAs) may play a role in treating these patients with anemia of unknown etiology (AUE). This study examines hemoglobin and cardiovascular outcomes among elderly anemic patients treated with ESAs. We conducted a retrospective cohort study that included all anemic patients older than age 60 years who had erythropoietin (EPO) measured between 2005 and 2013 at a single center. Three independent reviewers used defined criteria to assign each patient’s anemia to 1 of 4 groups: chronic kidney disease (CKD), myelodysplastic syndrome, AUE, or other etiology. Logistic regression was used to compare treatment response (defined per the International Working Group response criteria in myelodysplasia). Adjusted Cox regression analysis was used to calculate the cardiovascular event hazard ratios associated with ESA treatment. A total of 570 patients met the inclusion criteria, of whom 101 received ESAs. There was a nonstatistically significant but quantitatively better response in AUE (47%) and CKD (54%) compared with other etiologies (22%). The adjusted odds ratio for response in AUE compared with other etiologies was 3.3 (95% confidence interval, 0.838-13.0). A baseline EPO level <200 IU/L independently predicted treatment response. There was no statistically significant difference in cardiovascular events or cardiovascular event-free survival between the treated and untreated groups after adjusting for confounders. Our results suggest that ESAs may effectively treat AUE, and responses may be similar to those in CKD. We could not detect a statistically significant increase in cardiovascular events in the studied cohort.
Introduction
Anemia is a common problem that disproportionally affects the elderly and has been linked to increased mortality, frailty, and reduced quality of life.1-4 A specific cause cannot be found in approximately 37% to 45% of cases of anemia in the elderly, despite investigations in search of an etiology.5-7 The terms “anemia of unknown etiology” (AUE) and “unexplained anemia in the elderly” are used to describe this population of patients with unexplained or senile anemia.
Erythropoietin (EPO), a hormone that is mainly released by the kidneys in response to hypoxic conditions,8 plays a central role in the regulation of erythropoiesis. Erythropoietin binds to EPO receptors located on erythroid progenitor cells that inhibit apoptosis and promote differentiation into mature red blood cells.9,10 We and others have suggested that that an inadequate EPO response to anemia may be a central factor in the pathophysiology of AUE.11-13 A meta-analysis conducted by our group demonstrated low endogenous EPO levels in AUE compared with iron deficiency,12 and our retrospective cohort study demonstrated that EPO levels are inappropriately low in AUE even after accounting for confounders such as hemoglobin level, renal function, and comorbidities.13 These findings suggest that the pharmacologic replacement of EPO may play a therapeutic role in patients with AUE.
An erythropoiesis-stimulating agent (ESA) is a drug that mimics endogenous EPO by binding to the EPO receptor, thereby promoting erythropoiesis.14 Epoetin alfa was the first recombinant human erythropoietin (rHuEPO) available commercially, and it has the same amino acid sequence as endogenous EPO with only small variations in the sugar profile.10 It is considered to be biologically equivalent to the endogenous form of EPO.15 Darbepoetin alfa is a hyperglycosylated form of rHuEPO that has an increased half-life and potency, allowing for less frequent administration compared with other ESAs.14,16 The approved indications for treatment with an ESA are anemia resulting from chronic kidney disease (CKD), treatment with chemotherapy, treatment of HIV-positive individuals with zidovudine, and for the reduction of allogeneic blood transfusions in elective surgery.17,18 In addition to increasing hematocrit, ESAs have been shown to improve energy and physical functioning in anemic patients with non-dialysis–dependent CKD.19 Because both AUE and CKD are associated with low EPO levels, it follows that patients with AUE may benefit symptomatically from treatment with ESAs.
The main concern with the use of ESAs is the associated risk of cardiovascular events. In CKD, treatment with ESAs has been shown to increase the risk of stroke, hypertension, and vascular access thrombosis.20 Higher doses of epoetin alfa (>10 950 units per week) have also been associated with an increased risk of the composite of myocardial infarction, stroke, hospitalizations for heart failure, and death.21 However, many studies include only patients with CKD and do not account for all confounders that may affect the incidence of cardiovascular events and mortality.
To determine whether ESAs may be a safe and effective treatment of AUE, we examined hemoglobin and cardiovascular outcomes among elderly anemic patients who were tested for EPO levels and treated with ESAs.
Methods
Study design and patient population
We conducted a retrospective cohort study of all consecutive patients referred to the Division of Hematology at the London Health Sciences Centre, a university-affiliated academic center in London, Ontario, Canada, who had EPO levels determined between 1 January 2005 and 31 December 2013. EPO levels were determined by chemiluminescence using an immunoenzymatic method (Access EPO Erythropoietin) with an Access 2 Analyzer (Beckman Coulter, Brea, CA) with a reference range of 2.59 to 18.50 IU/L. We included patients age 60 years or older who met the World Health Organization criteria for anemia (<130 g/L in men; <120 g/L in women),22 and we excluded patients with insufficient electronic medical records. Our group previously published a study of EPO levels in the same patient population,13 and data for both studies was extracted from the patient charts concurrently. To assess cardiovascular outcomes, patients were divided into 2 groups on the basis of whether or not they were treated with an ESA at any time. To assess for treatment response, we included only the patients who received an ESA and excluded patients in whom the response could not be assessed because of missing data, such as pre- or posttreatment hemoglobin levels. The study was approved by the Health Sciences Research Ethics Board at Western University and was performed in accordance with the Declaration of Helsinki.
Determination of anemia etiology
The etiology of each patient’s anemia was adjudicated to 1 of 4 diagnostic groups: CKD, myelodysplastic syndrome (MDS), AUE, or other etiology. Three investigators independently reviewed each patient’s electronic medical records and assigned an etiology on the basis of predefined criteria (supplemental Table 1). The etiology reported by at least 2 of the 3 reviewers was used in the analysis, with differences resolved by consensus after consultation with a fourth reviewer.
Data collection
All data were obtained from the electronic medical records as previously described.13 Briefly, information on age, comorbidities, and laboratory values were collected at the time the EPO level was first measured. The presence of another cytopenia was defined as an absolute neutrophil count <1.8 × 109/L or a platelet count <100 × 109/L. Comorbidity was assessed by using Charlson’s comorbidity index as modified by Quan et al.23
The CKD-Epidemiology Collaboration24 formula was used to estimate the glomerular filtration rate (GFR) in each patient because it has been shown to more accurately classify future risk of end-stage renal disease and mortality compared with the Modification of Diet in Renal Disease formula.25 A GFR cutoff of <30 mL/min/1.73 m2 was chosen because Mercadal et al26 demonstrated that in anemic patients there is a negative correlation between GFR and hemoglobin at GFRs below 30 mL/min/1.73 m2 but no correlation with GFRs above 30 mL/min/1.73 m2, suggesting that the physiologic response to anemia is somewhat preserved in patients with GFRs >30 mL/min/1.73 m2.
ESA treatment response.
Treatment with an ESA was defined as having received either epoetin alfa or darbepoetin alfa, regardless of the dose or duration of treatment. The decision to administer an ESA was made by the treating physician on the basis of patient preferences and access to funding and was generally reserved for patients with symptomatic anemia that affected quality of life. The mean weekly dose was calculated by dividing the total quantity of ESA that was administered by the total duration of treatment.
We recorded the hemoglobin levels before treatment with an ESA and approximately 1, 3, 6, and 12 months after the initiation of treatment. The number of transfusions in the 8 weeks before and the 8 weeks after the initiation of treatment were estimated on the basis of clinic notes and by the treating physician and transfusion records. Treatment response was defined as an increase of at least 15 g/L in the hemoglobin level from the pretreatment level or a decrease of at least 4 transfusions over 8 weeks compared with pretreatment per the International Working Group response criteria in myelodysplasia.27
Cardiovascular events.
Cardiovascular events were recorded starting at the time of the first EPO measurement and ending at the most recent electronic record available at the time of data collection. The date of the first treatment with an ESA was not used as a starting point to allow for equal treatment of data from both the treated and untreated groups. A cardiovascular event was defined as a documented ischemic stroke, myocardial infarction (ST elevation or non-ST elevation), pulmonary embolism, deep vein thrombosis, or portal vein thrombosis. Death was documented if a death summary or other death documentation was present in the electronic chart.
Statistical analysis
Interobserver agreement was assessed by using the Fleiss’ κ statistic.28 The treatment response in CKD, MDS, and AUE was compared with the reference group (other etiology) by using logistic regression, and odds ratios (ORs) were reported. To adjust for potential confounders, we constructed stepwise logistic regression models to estimate the effect of anemia etiology on treatment response. Models were assessed by residual analysis and adjusted for sex, age, weight, Charlson’s comorbidity index, hemoglobin level, estimated GFR (eGFR), the presence of additional cytopenias, and dichotomized EPO level. The EPO level was transformed into a dichotomous variable by using an optimal cut point that was statistically significant and maximized the OR.
Incidence rates (IRs) per 1000 patient-years were calculated for cardiovascular events and mortality. We generated Kaplan-Meier curves comparing cardiovascular events and cardiovascular event-free survival between the treated and untreated groups. The P values were calculated by the Breslow method. To adjust for potential confounders, we calculated hazard ratios (HRs) by using Cox regression analysis. Models were assessed by residual analysis and adjusted for sex, age, weight, Charlson’s comorbidity index, hemoglobin level, eGFR, the presence of additional cytopenias, and EPO level.
P < .05 was considered statistically significant. Statistical analyses were completed by using Excel 2007 (Microsoft, Redmond, WA) or SPSS Statistics version 22 (IBM, Armonk, NY).
Results
Patient population
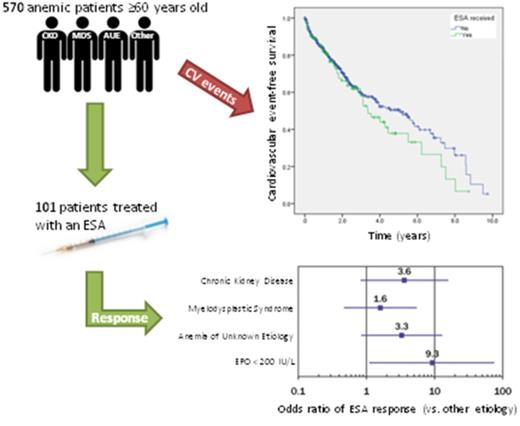
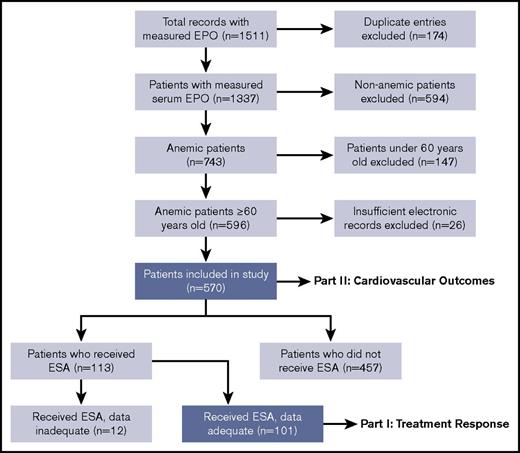
During the study period, there were 1511 requests for the measurement of serum EPO concentration. The most common indications for EPO measurement were anemia and polycythemia. We excluded 941 instances (174 duplicate entries, 594 non-anemic patients, 147 patients younger than age 60 years, and 26 patients with insufficient records), and 570 patients were ultimately included in the assessment of cardiovascular outcomes. Of the 570 patients in the study, 113 patients received an ESA. After excluding 12 patients with inadequate data, 101 patients were included in the assessment of treatment response. A flowchart of cohort construction is provided in Figure 1.
Flowchart of cohort integration. Serum EPO was measured in 1511 patients of whom 101 could be assessed for treatment response and 570 could be assessed for cardiovascular outcomes.
Flowchart of cohort integration. Serum EPO was measured in 1511 patients of whom 101 could be assessed for treatment response and 570 could be assessed for cardiovascular outcomes.
The 3-way interrater agreement between the adjudicated etiologies measured by Fleiss’ κ was 0.62 for all patients included in the study and equal to 0.47 in the subgroup assessed for treatment response.
Treatment response and predictors.
Of the 101 patients studied for response to treatment with an ESA, CKD was the primary etiology in 13 patients, MDS in 46 patients, AUE in 19 patients, and other etiology in 23 patients. The mean age was 75.4 years and 59% were male. The mean Charlson’s comorbidity index was 2.2 and mean hemoglobin was 89.2 g/L. The mean EPO concentration was 31.4 IU/L in CKD, 146.9 in MDS, 86.4 in AUE, and 68.1 in other etiologies. Baseline patient characteristics for each group are listed in Table 1.
Baseline characteristics of patients treated with an ESA
| Characteristic . | CKD . | MDS . | AUE . | Other etiology . | Total . | |||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| No. . | % . | Mean . | SD . | No. . | % . | Mean . | SD . | No. . | % . | Mean . | SD . | No. . | % . | Mean . | SD . | No. . | % . | Mean . | SD . | |
| No. of patients* | 13 | 46 | 19 | 23 | 101 | |||||||||||||||
| Male sex | 5 | 38 | 29 | 63 | 12 | 63 | 13 | 57 | 59 | 59 | ||||||||||
| Age, y | 78.8 | 8.4 | 76.7 | 7.6 | 72.6 | 9.5 | 73.3 | 9.8 | 75.4 | 8.7 | ||||||||||
| Weight, kg | 77.8 | 23.3 | 77.8 | 13.5 | 80.8 | 26.2 | 78.1 | 20.4 | 78.4 | 19.3 | ||||||||||
| Comorbidity index | 2.7 | 1.8 | 1.6 | 1.5 | 2.3 | 1.1 | 3.1 | 2.1 | 2.2 | 1.7 | ||||||||||
| Hemoglobin, g/L | 90.2 | 9.4 | 85.1 | 9.4 | 92.4 | 10.3 | 87.6 | 11.7 | 89.2 | 12.9 | ||||||||||
| EPO, IU/L | 31.4 | 50.4 | 147 | 145 | 86.4 | 178 | 68.1 | 99.2 | 103 | 140 | ||||||||||
| eGFR, mL/min/1.73 m2 | 18.1 | 8.8 | 56.8 | 24.9 | 42.4 | 7.4 | 40.0 | 23.4 | 45.3 | 24.2 | ||||||||||
| Other cytopenia | 4 | 31 | 28 | 61 | 4 | 21 | 3 | 13 | 39 | 39 | ||||||||||
| Characteristic . | CKD . | MDS . | AUE . | Other etiology . | Total . | |||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| No. . | % . | Mean . | SD . | No. . | % . | Mean . | SD . | No. . | % . | Mean . | SD . | No. . | % . | Mean . | SD . | No. . | % . | Mean . | SD . | |
| No. of patients* | 13 | 46 | 19 | 23 | 101 | |||||||||||||||
| Male sex | 5 | 38 | 29 | 63 | 12 | 63 | 13 | 57 | 59 | 59 | ||||||||||
| Age, y | 78.8 | 8.4 | 76.7 | 7.6 | 72.6 | 9.5 | 73.3 | 9.8 | 75.4 | 8.7 | ||||||||||
| Weight, kg | 77.8 | 23.3 | 77.8 | 13.5 | 80.8 | 26.2 | 78.1 | 20.4 | 78.4 | 19.3 | ||||||||||
| Comorbidity index | 2.7 | 1.8 | 1.6 | 1.5 | 2.3 | 1.1 | 3.1 | 2.1 | 2.2 | 1.7 | ||||||||||
| Hemoglobin, g/L | 90.2 | 9.4 | 85.1 | 9.4 | 92.4 | 10.3 | 87.6 | 11.7 | 89.2 | 12.9 | ||||||||||
| EPO, IU/L | 31.4 | 50.4 | 147 | 145 | 86.4 | 178 | 68.1 | 99.2 | 103 | 140 | ||||||||||
| eGFR, mL/min/1.73 m2 | 18.1 | 8.8 | 56.8 | 24.9 | 42.4 | 7.4 | 40.0 | 23.4 | 45.3 | 24.2 | ||||||||||
| Other cytopenia | 4 | 31 | 28 | 61 | 4 | 21 | 3 | 13 | 39 | 39 | ||||||||||
SD, standard deviation.
A total of 113 patients received ESAs, but 12 patients (2 of whom had AUE) did not have sufficient data to analyze the treatment response and are not included in this table.
Overall, 88 patients were treated with epoetin alfa, with a mean dose of 28.2 × 103 IU per week over a mean duration of 473 days. Nineteen patients were treated with darbepoetin alfa, with a mean dose of 53 μg per week over a mean duration of 787 days. Note that 6 patients were treated with both epoetin alfa and darbepoetin alfa. The average epoetin alfa dose was 12.9 × 103 IU per week in AUE, 10.9 × 103 IU in CKD, and 40.4 × 103 IU in MDS compared with 24.5 × 103 IU in other etiologies.
Overall, 36 of the 101 patients treated with an ESA had a treatment response. Thirty patients met the hemoglobin criteria, and 6 patients met the transfusion criteria (5 with MDS and 1 with CKD). There was a nonsignificant but quantitatively better treatment response in the CKD and AUE groups (54% and 47%, respectively) compared with other etiologies (22%) and MDS (26%); the Pearson χ2P value was equal to .083.
The mean initial and posttreatment hemoglobin levels in the AUE group were 92.4 g/L (SD, 10.3 g/L) and 108.3 g/L (SD, 11.3 g/L), respectively, which was a mean increase of 15.8 g/L (SD, 12.8 g/L). The hemoglobin increased by 15 g/L or more in 9 (47%) of 19 patients. One patient in the AUE group was transfused with 4 or more units in the 8 weeks before treatment and continued to require transfusions in the 8 weeks after treatment. Three patients were transfused with fewer than 4 units before treatment, all of whom required no transfusions within the 8 weeks after treatment. None of the patients in the AUE group (0%) met the transfusion criteria for a treatment response.
The ORs of a response to ESA treatment relative to other etiologies, both adjusted and unadjusted for confounders, were calculated (Table 2). After adjustment for confounders, a superior treatment response was seen in CKD (OR, 3.60; 95% confidence interval [CI], 0.815-15.9; P = .091) and AUE (OR, 3.30; 95% CI, 0.838-13.0; P = .088) compared with other etiologies, although it did not reach statistical significance. Erythropoietin level was the only statistically significant confounder, whereas sex, age, weight, comorbidity index, hemoglobin, eGFR, and presence of another cytopenia were not significant. Erythropoietin cut points of 50, 100, 150, and 200 IU/L were tested. The treatment response was optimal for EPO <200 IU/L with an OR of 9.28 (95% CI, 1.14-75.4).
ORs of ESA treatment response compared with other etiology, unadjusted and adjusted for confounders
| Etiology . | OR* . | 95% CI . | P . |
|---|---|---|---|
| Unadjusted model | |||
| CKD | 4.20 | 0.962-18.3 | .056 |
| MDS | 1.27 | 0.387-4.17 | .693 |
| AUE | 3.24 | 0.849-12.4 | .085 |
| Adjusted model, EPO linear† | |||
| CKD | 3.60 | 0.815-15.9 | .091 |
| MDS | 1.61 | 0.472-5.47 | .448 |
| AUE | 3.30 | 0.838-13.0 | .088 |
| EPO <200 IU/L | 9.28 | 1.14-75.4 | .037 |
| Etiology . | OR* . | 95% CI . | P . |
|---|---|---|---|
| Unadjusted model | |||
| CKD | 4.20 | 0.962-18.3 | .056 |
| MDS | 1.27 | 0.387-4.17 | .693 |
| AUE | 3.24 | 0.849-12.4 | .085 |
| Adjusted model, EPO linear† | |||
| CKD | 3.60 | 0.815-15.9 | .091 |
| MDS | 1.61 | 0.472-5.47 | .448 |
| AUE | 3.30 | 0.838-13.0 | .088 |
| EPO <200 IU/L | 9.28 | 1.14-75.4 | .037 |
OR of treatment response is relative to other etiology.
Includes only EPO level as a confounder. Sex, age, weight, comorbidity index, hemoglobin, eGFR, and presence of another cytopenia were not found to be statistically significant predictors of response.
Cardiovascular events.
Of the 570 patients, 113 patients were treated with an ESA and 457 patients were not treated. The mean pretreatment hemoglobin levels were 88.5 g/L in the treated group and 97.7 g/L in the untreated group. The mean EPO levels were 135.2 IU/L in the treated group and 187.2 IU/L in the untreated group. The median time between the measurement of the EPO level and starting an ESA was 79 days. Baseline patient characteristics for both groups are shown in Table 3. There were a total of 21 cardiovascular events and 44 confirmed deaths in the ESA group (IRs were 68 and 143 per 1000 patient-years, respectively). Among the 457 untreated patients, there were 43 cardiovascular events and 140 deaths (IRs were 46 and 151 per 1000 patient-years, respectively). No cardiovascular events were documented between the measurement of the EPO level and starting an ESA. The absolute numbers and rates of cardiovascular events and deaths are shown in supplemental Table 2.
Baseline characteristics of all patients in the study
| Characteristic . | Received ESA . | Did not receive ESA . | ||||||
|---|---|---|---|---|---|---|---|---|
| No. . | % . | Mean . | SD . | No. . | % . | Mean . | SD . | |
| Total No. of patients | 113 | 457 | ||||||
| CKD | 13 | 12 | 12 | 3 | ||||
| MDS | 52 | 46 | 147 | 32 | ||||
| AUE | 21 | 19 | 96 | 21 | ||||
| Other etiology | 27 | 24 | 202 | 44 | ||||
| Male sex | 68 | 60 | 274 | 60 | ||||
| Age, y | 75.1 | 8.7 | 75.9 | 8.1 | ||||
| Weight, kg | 78.5 | 19.7 | 74.6 | 18.0 | ||||
| Comorbidity index | 2.2 | 1.7 | 1.4 | 1.4 | ||||
| Hemoglobin, g/L | 88.5 | 12.8 | 97.7 | 15.9 | ||||
| EPO, IU/L | 135.2 | 420.0 | 187.2 | 539.3 | ||||
| eGFR, mL/min/1.73 m2 | 46.6 | 24.3 | 61.0 | 22.4 | ||||
| Other cytopenia | 45 | 40 | 175 | 38 | ||||
| Characteristic . | Received ESA . | Did not receive ESA . | ||||||
|---|---|---|---|---|---|---|---|---|
| No. . | % . | Mean . | SD . | No. . | % . | Mean . | SD . | |
| Total No. of patients | 113 | 457 | ||||||
| CKD | 13 | 12 | 12 | 3 | ||||
| MDS | 52 | 46 | 147 | 32 | ||||
| AUE | 21 | 19 | 96 | 21 | ||||
| Other etiology | 27 | 24 | 202 | 44 | ||||
| Male sex | 68 | 60 | 274 | 60 | ||||
| Age, y | 75.1 | 8.7 | 75.9 | 8.1 | ||||
| Weight, kg | 78.5 | 19.7 | 74.6 | 18.0 | ||||
| Comorbidity index | 2.2 | 1.7 | 1.4 | 1.4 | ||||
| Hemoglobin, g/L | 88.5 | 12.8 | 97.7 | 15.9 | ||||
| EPO, IU/L | 135.2 | 420.0 | 187.2 | 539.3 | ||||
| eGFR, mL/min/1.73 m2 | 46.6 | 24.3 | 61.0 | 22.4 | ||||
| Other cytopenia | 45 | 40 | 175 | 38 | ||||
Of the 117 patients with AUE, 21 were treated with an ESA and 96 were not treated. In total, there were 7 cardiovascular events and 6 confirmed deaths in the ESA group (IRs were 110 and 94 per 1000 patient-years, respectively). Among the untreated patients with AUE, there were 6 cardiovascular events and 15 deaths (IRs were 27 and 68 per 1000 patient-years, respectively).
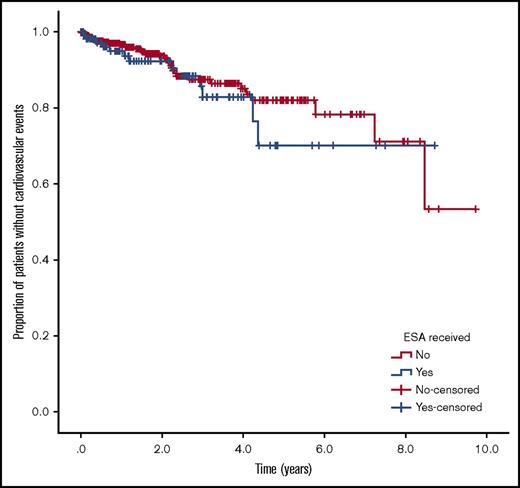
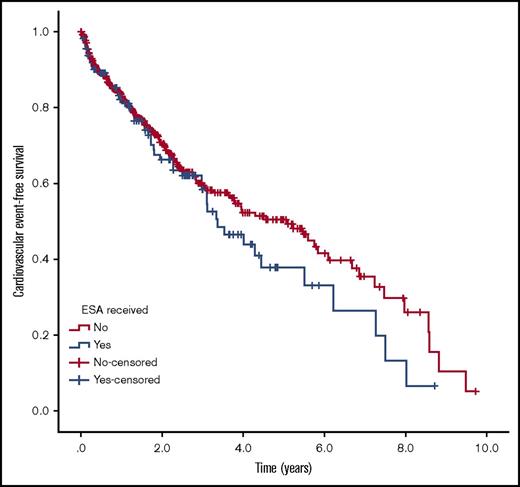
Figures 2 and 3 show the Kaplan-Meier curves of cardiovascular events and cardiovascular event-free survival in the ESA-treated and untreated groups. We found no statistically significant difference between groups for either analysis.
Kaplan-Meier curve of cardiovascular events. Proportion of patients without a cardiovascular event for ESA-treated and untreated patients. No difference was observed between groups (P = .425)
Kaplan-Meier curve of cardiovascular events. Proportion of patients without a cardiovascular event for ESA-treated and untreated patients. No difference was observed between groups (P = .425)
Kaplan-Meier curve of cardiovascular event-free survival. Proportion of surviving patients without a cardiovascular event for ESA-treated and untreated patients. No difference was observed between groups (P = .599)
Kaplan-Meier curve of cardiovascular event-free survival. Proportion of surviving patients without a cardiovascular event for ESA-treated and untreated patients. No difference was observed between groups (P = .599)
HRs for cardiovascular events and cardiovascular events or death were calculated (Table 4). There was no statistically significant difference in the rate of cardiovascular events in the group treated with ESAs compared with the untreated group (HR, 1.28; 95% CI, 0.68-2.42; P = .445), even after adjusting for the presence of another cytopenia (HR, 1.24; 95% CI, 0.66-2.34; P = .507). There was no statistically significant difference in the rate of the composite outcome of cardiovascular event and death in the group treated with ESAs compared with the untreated group (HR, 1.21; 95% CI, 0.88-1.66; P = .234), even after adjusting for relevant confounders (HR, 0.94; 95% CI, 0.67-1.31; P = .693). Sex, age, Charlson’s comorbidity index, hemoglobin level, and the presence of another cytopenia were statistically significant confounders.
Cox regression of cardiovascular events and cardiovascular event-free survival for ESA-treated vs untreated patients, adjusted for confounders
| . | HR . | 95% CI . | P . |
|---|---|---|---|
| Cardiovascular event | |||
| Unadjusted | |||
| ESA treatment | 1.28 | 0.68-2.42 | .445 |
| Adjusted* | |||
| ESA treatment | 1.24 | 0.66-2.34 | .507 |
| Presence of another cytopenia | 1.92 | 1.07-3.42 | .028 |
| Cardiovascular event or death | |||
| Unadjusted | |||
| ESA treatment | 1.21 | 0.88-1.66 | .234 |
| Adjusted† | |||
| ESA treatment | 0.94 | 0.67-1.31 | .693 |
| Presence of another cytopenia | 2.06 | 1.54-2.75 | < .001 |
| Hemoglobin, g/L | 0.98 | 0.97-0.99 | < .001 |
| Age, years | 1.03 | 1.01-1.06 | .001 |
| Male sex | 1.55 | 1.15-2.08 | .004 |
| Comorbidity index | 1.13 | 1.03-1.23 | .006 |
| . | HR . | 95% CI . | P . |
|---|---|---|---|
| Cardiovascular event | |||
| Unadjusted | |||
| ESA treatment | 1.28 | 0.68-2.42 | .445 |
| Adjusted* | |||
| ESA treatment | 1.24 | 0.66-2.34 | .507 |
| Presence of another cytopenia | 1.92 | 1.07-3.42 | .028 |
| Cardiovascular event or death | |||
| Unadjusted | |||
| ESA treatment | 1.21 | 0.88-1.66 | .234 |
| Adjusted† | |||
| ESA treatment | 0.94 | 0.67-1.31 | .693 |
| Presence of another cytopenia | 2.06 | 1.54-2.75 | < .001 |
| Hemoglobin, g/L | 0.98 | 0.97-0.99 | < .001 |
| Age, years | 1.03 | 1.01-1.06 | .001 |
| Male sex | 1.55 | 1.15-2.08 | .004 |
| Comorbidity index | 1.13 | 1.03-1.23 | .006 |
Adjusted for presence of a cytopenia.
Adjusted for presence of a cytopenia, hemoglobin, age, sex, and comorbidity index.
Discussion
The use of ESAs specifically in patients with AUE has garnered very little research. Previous studies have suggested that EPO levels are inappropriately low in AUE,12 even after accounting for potential confounders.13 This suggests that the exogenous administration of EPO may have a therapeutic role in elderly patients with AUE; however, treatment responses in this population have not been adequately studied. In a population of predominantly older African American women with anemia, 1 randomized controlled trial demonstrated hemoglobin response and improvements in fatigue and quality of life.29 In this study, 47% of patients with AUE and 54% of patients with CKD met the criteria for a response to ESA treatment. This suggests that ESAs may be effective at increasing hemoglobin levels in AUE. Logistic regression analysis did not show statistically significant differences between groups, but the ORs of response were 3.3 in AUE and 3.6 in CKD compared with other etiologies.
As expected, patients with CKD responded well to treatment with ESAs, with treatment responses higher in CKD and the required ESA doses lower. This agrees with the current understanding of anemia in CKD as a deficiency in EPO secondary to kidney fibrosis.30 The patients with AUE also demonstrated good response rates when treated with ESAs. Treatment responses and ESA doses were similar to those seen in the group with CKD. Although the response to ESAs was not statistically significantly superior in AUE compared with other etiologies, the high absolute response rates provide further evidence that AUE is secondary to inadequate levels of endogenous EPO, rather than being a result of insensitivity to EPO or a decreased marrow reserve.
Logistic regression analysis revealed that patients with an EPO level <200 IU/L have significantly increased odds of response (OR, 9.3) compared with patients above this cutoff, independent of the etiology of the anemia. This suggests that patients with elevated serum EPO levels have insensitivity to EPO or saturated EPO receptors and therefore respond less reliably to treatment with ESAs. Previous studies in MDS and chronic myelomonocytic leukemia have established that a lower EPO level is associated with superior responses to ESAs.31,32 The findings in this study imply that the EPO level, specifically a cutoff of 200 IU/L, may have merit in predicting response to ESAs in AUE in addition to in MDS.
To the best of our knowledge, this is the first study of ESAs that investigates cardiovascular outcomes in a heterogeneous group of patients that includes patients with AUE. Previous studies in patients with CKD have identified an increase in cardiovascular events with ESA treatment, which is associated with the use of higher target hemoglobin levels,20,21 but a meta-analysis by Palmer et al33 did not show a statistically significant effect on mortality or major cardiovascular events. ESAs may be associated with decreased mortality in low-risk MDS, although clear supporting evidence is lacking.34 We are not aware of any existing safety data for ESAs in AUE. Our Cox regression analysis showed no statistically significant increase in the risk for cardiovascular events or for the composite end point of cardiovascular events or death in our heterogeneous population of elderly anemic patients after adjustment for confounders. We cannot exclude the possibility of a type II error, and we acknowledge that the study may not have the statistical power to detect meaningful differences between the treated and untreated groups. The risk of the composite outcome of cardiovascular event or death was increased by the presence of another cytopenia, lower hemoglobin level, advanced age, male sex, and the presence of comorbidities. It has previously been shown that mortality increases proportionally with anemia severity.35 As expected, the presence of comorbidities is associated with increased mortality, and Charlson’s comorbidity index is already a validated tool for predicting mortality in a wide variety of clinical settings.23
A potential limitation of this study is the use of other etiology as a reference group to compare treatment responses. This group is highly heterogeneous and even includes cases of anemia that have multifactorial etiologies. Other etiology was chosen as the reference group because combining the patients who did not have CKD, MDS, or AUE into a single group allowed for a larger comparison population for statistical analysis. However, this strategy was consistent with the purpose of the study, which was to test the hypothesis that ESAs are more effective in AUE than in many other etiologies. In addition, we cannot rule out the possibility of some degree of misclassification in the anemia etiologies. However, we maximized the objectiveness of the groups by assigning etiologies on the basis of defined clinical criteria, and we have demonstrated reasonable agreement between the adjudicators. We were also limited by the heterogeneity within each etiologic group. Although we classified AUE as a single etiology, it is possible that the pathogenesis of anemia in this group is highly heterogeneous. Many of the patients assigned to the AUE group had renal impairment that was not severe enough to meet the eGFR cutoff of less than 30 mL/min/1.73 m2. In fact, two-thirds of the AUE patients in the study, and all 19 AUE patients included in the analysis of treatment response had eGFRs between 30 and 60 mL/min/1.73 m2. This study placed only those patients with eGFRs <30 mL/min/1.73 m2 in the CKD etiologic group, because patients with eGFRs greater than the cutoff are unlikely to have CKD as the sole cause of the anemia.26
We recognize that the inclusion of only the patients with measured EPO levels may introduce bias into the analysis of treatment responses and cardiovascular outcomes. However, this bias could not be avoided, given our a priori interest in the role of EPO levels in predicting the response to treatment with ESAs. Because it is common practice for EPO to be measured in patients with MDS to predict response to ESAs, the studied population included a disproportionate number of patients with MDS (32% of patients with EPO measurements). MDS was also the most common indication that a hematologist would prescribe an ESA for (46% of patients treated with an ESA), so the high proportion of patients with MDS reflects the distribution of etiologies of patients under consideration for ESA treatment.
Finally, although we conducted extensive analyses to try to control for the study limitations, potential explanations for the lack of association between ESA treatment and cardiovascular events include the possibility of residual confounding in previous studies, the risk of selection bias in our retrospective study, the heterogeneity of the population compared with previous studies that included mainly CKD patients, and the small sample size potentially leading to a failure to detect real differences between groups (type II error). We recognize that because of its retrospective nature, our study cannot definitively prove response to ESA treatment in AUE. Because there are no prospective or retrospective studies, our retrospective study provides new evidence that should serve as a basis for planning and funding a prospective study. Despite its limitations, this study is the first to assess the response to ESAs in AUE and the first to explicitly include patients with AUE in the investigation of cardiovascular outcomes and mortality.
In conclusion, our results suggest that ESAs may be effective in treating elderly patients with AUE. Treatment responses may be similar to those in seen in CKD, although superiority in AUE and CKD versus other etiologies was not shown with statistical significance. This supports the notion that a relative EPO deficiency is probably related to the pathogenesis of AUE. Although treatment may be associated with increased cardiovascular events and mortality, this was not found to be significant in our cohort, although caution is needed when considering this therapeutic modality. Because this is one of the first studies of ESAs to date that includes patients with AUE, it provides new evidence that should serve as the basis for prospective studies that explore the safety and effectiveness of ESAs specifically in the treatment of AUE.
Presented in part at the 58th annual meeting of the American Society of Hematology, San Diego, CA, 3-6 December 2016.
The full-text version of this article contains a data supplement.
Authorship
Contribution: Z.G. collected and analyzed data and drafted and reviewed the manuscript; S.S., A.M., and A.X. collected data and drafted and analyzed the manuscript; and A.L.-L. designed the study, collected and analyzed data, and drafted and analyzed the manuscript.
Conflict-of-interest disclosure: The authors declare no competing financial interests.
Correspondence: Alejandro Lazo-Langner, 800 Commissioners Rd E, Room E5-216, London, ON N6A 5W9, Canada; e-mail: alejandro.lazolangner@lhsc.on.ca.