Abstract
Altered expression of proteins of the fibrinolytic and coagulation cascades in obesity may contribute to the cardiovascular risk associated with this condition. In spite of this, the zymogenic nature of some of the molecules and the presence of variable amounts of activators, inhibitors, and cofactors that alter their activity have made it difficult to accurately monitor changes in the activities of these proteins in tissues where they are synthesized. Thus, as a first approach to determine whether tissue factor (TF) expression is altered in obesity, this study examined changes in TF mRNA in various tissues from lean and obese (ob/ob and db/db) mice. TF gene expression was elevated in the brain, lung, kidney, heart, liver, and adipose tissues of both ob/ob and db/db mice compared with their lean counterparts. In situ hybridization analysis indicated that TF mRNA was elevated in bronchial epithelial cells in the lung, in myocytes in the heart, and in adventitial cells lining the arteries including the aortic wall. Obesity is associated with insulin resistance and hyperinsulinemia, and administration of insulin to lean mice induced TF mRNA in the kidney, brain, lung, and adipose tissue. These observations suggest that the hyperinsulinemia associated with insulin-resistant states, such as obesity and noninsulin-dependent diabetes mellitus, may induce local TF gene expression in multiple tissues. The elevated TF may contribute to the increased risk of atherothrombotic disease that accompanies these conditions.
Introduction
Obesity and related noninsulin-dependent diabetes mellitus (NIDDM) are among the most common health problems in industrialized societies and are associated with an increased incidence of thrombosis and accelerated atherosclerosis.1,2Interestingly, a number of clinical studies have demonstrated dysregulation of both the coagulation and fibrinolytic systems in obesity/NIDDM,3-8 which suggests that these changes may contribute to the cardiovascular complications in these disorders. In this regard, several studies have shown an increase in tissue factor (TF)–mediated coagulation and/or in factor VII activity or antigen in obese patients and those with NIDDM.4,9-15 TF is the major cellular initiator of the coagulation cascade and also serves as a cell-surface receptor for the activation of factor VII.16-19 Activation of the coagulation cascade by aberrant expression of TF may promote thrombosis in patients with a variety of clinical disorders. These disorders include Gram-negative sepsis17,20 and atherosclerosis,21-23 as well as adult respiratory distress syndrome, systemic lupus erythematosus, Crohn disease, rheumatoid arthritis, and various forms of cancer.17 TF is expressed in human atherosclerotic plaques and may play a significant role in the thrombotic complications associated with plaque rupture.17,22,23 These observations suggest that the increase in TF in obesity and NIDDM could promote the development of a hypercoagulable state and thereby contribute to the cardiovascular complications associated with these conditions. Interestingly, a number of recent reports have demonstrated TF activity in plasma.24-27 The origin of this activity and its biologic significance remain to be established.
We previously demonstrated that levels of plasminogen activator inhibitor-1 (PAI-1)28 and TF29 gene expression were elevated in adipose tissues of genetically obese (ob/ob) mice. These mice cannot produce leptin,30 and, as a consequence, they experience early-onset obesity, insulin resistance, and hyperinsulinemia.30,31 In this report, we used ob/ob mice together with obese db/db mice (which lack the leptin receptor) to determine whether TF also was elevated in other tissues of the obese mice. Because of the strong correlation between obesity and hyperinsulinemia, we also asked whether these effects were mediated by insulin. Our results demonstrate that TF mRNA is significantly elevated in the brain, lung, kidney, liver, and heart of both ob/ob and db/db mice. Moreover, we show that insulin can contribute to the increase in TF gene expression in some of these tissues. The coordinated increase in TF and PAI-1 in obesity3 28 would thus be expected to increase coagulation and impair fibrinolysis, thereby promoting a state that favors thrombosis.
Materials and methods
Animals and tissue preparation
Adult male obese mice (C57BL/6J ob/ob, weight 49 ± 2.3 g; C57BL/KsJ db/db, weight 46 ± 1.9 g) at 3 months of age and their lean littermates (C57BL/6J+/+ and C57BL/KsJ+/+, weight 18.1 ± 1.1 g) were obtained from Jackson Labs (Bar Harbor, ME). For insulin experiments, lean mice (C57BL/6J+/+) were injected intraperitoneally with 10 U regular human insulin (Himulin R; Eli Lilly, Indianapolis, IN), and the controls were injected with an equivalent volume of saline alone. At the conclusion of each experiment, mice were anesthetized by metofane (Pitman-Moore, Mundelein, IL), and various tissues were removed and processed either for in situ hybridization analysis or for the isolation of total RNA as described previously.32
Quantitative reverse transcriptase–polymerase chain reaction
The concentration of TF mRNA in tissues was determined by quantitative reverse transcriptase–polymerase chain reaction (RT-PCR) using a competitor cRNA containing upstream and downstream primers for TF and β-actin (internal control), as described previously.33-35 After reverse transcription (using 105 molecules of cRNA for TF and 107 molecules for β-actin, as determined in preliminary experiments) and PCR using32P-end–labeled 5′ primers, 20 μL of the PCR products was subjected to electrophoresis on 2.5% agarose gels. The appropriate bands corresponding to the internal standard cRNA product and the target mRNA product were excised from the gel, and the incorporated radioactivity was quantified using a scintillation counter. A standard curve for the internal control cRNA was constructed and used to determine the specific activity of the target mRNA, as described previously.32 34 Variations in sample loading were assessed by comparison with β-actin mRNA.
Riboprobe preparation and in situ hybridization
A subclone containing 821 bp of the mouse TF cDNA (nucleotides 229-1049) cloned into the vector pGEM-3Z36 was used to prepare a riboprobe for in situ hybridization.37 This vector was linearized and used as a template for in vitro transcription of radiolabeled antisense or sense riboprobes with the use of SP6 or T7 RNA polymerase, respectively, in the presence of [35S]UTP (greater than 1200 Ci/mmol [37 TBq/mm]; Amersham, Arlington Heights, IL). Both sense and antisense probes were routinely labeled to specific activities between 0.5 and 2 × 108 cpm/mg RNA. In situ hybridization was performed as described previously.37 Slides were exposed in the dark at 4°C for 4 to 12 weeks and then developed and stained with hematoxylin and eosin.
Statistical analysis
Statistical comparison of results was performed using the unpaired Student t test.
Results
TF mRNA levels in tissues from lean and obese mice
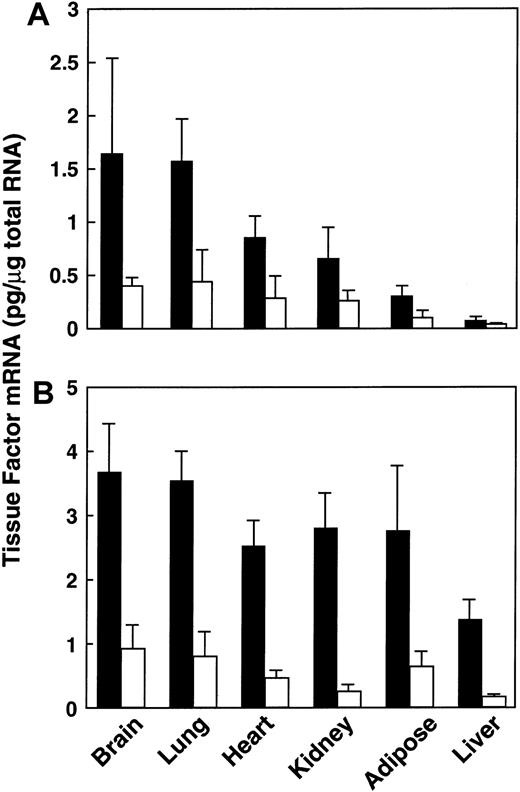
We previously demonstrated that TF gene expression was elevated in adipose tissues of obese (ob/ob) mice.29 To determine whether it was also elevated in other tissues, we compared the levels of TF mRNA in a variety of tissues from lean and ob/ob mice (Figure1). Tissues were removed from 3-month-old mice, and total RNA was prepared and analyzed for TF mRNA by quantitative RT-PCR. TF mRNA levels were elevated in all of the tissues examined from the ob/ob mice when compared with their lean counterparts (Figure 1A). For example, in the 3-month-old ob/ob mice, TF mRNA was increased by approximately 4-fold in the brain (P < .04, n = 6) and lung (P < .0001, n = 6), by 3-fold in the heart (P < .0001, n = 6), and by 2-fold in the kidney (P < .02, n = 6) and liver (P < .001, n = 6). We next determined whether TF mRNA was elevated in tissues of the db/db mouse, a different model of genetic obesity. Again, TF mRNA levels were elevated in all of the tissues examined (Figure 1B), including the brain (4.7-fold; P < .003, n = 6), the lung (5-fold; P < .002, n = 6), the heart (5-fold;P < .0007, n = 6), the kidney (6-fold;P < .001, n = 6), the adipose tissue (3.5-fold;P < .03, n = 6), and the liver (3.7-fold;P < .01, n = 6). These results indicate that elevated TF mRNA is not unique to the ob/ob mouse.
Expression of TF mRNA in tissues from lean and obese mice.
Total RNA was extracted from various tissues of male lean (■) and obese (▪) animals at 3 months of age. TF mRNA was determined using quantitative RT-PCR analysis, as described in “Materials and methods.” For each condition, n = 6, mean ± SD. (A) Lean versus ob/ob mice. (B) Lean versus db/db mice.
Expression of TF mRNA in tissues from lean and obese mice.
Total RNA was extracted from various tissues of male lean (■) and obese (▪) animals at 3 months of age. TF mRNA was determined using quantitative RT-PCR analysis, as described in “Materials and methods.” For each condition, n = 6, mean ± SD. (A) Lean versus ob/ob mice. (B) Lean versus db/db mice.
Cellular localization of TF mRNA in tissues from obese and lean mice
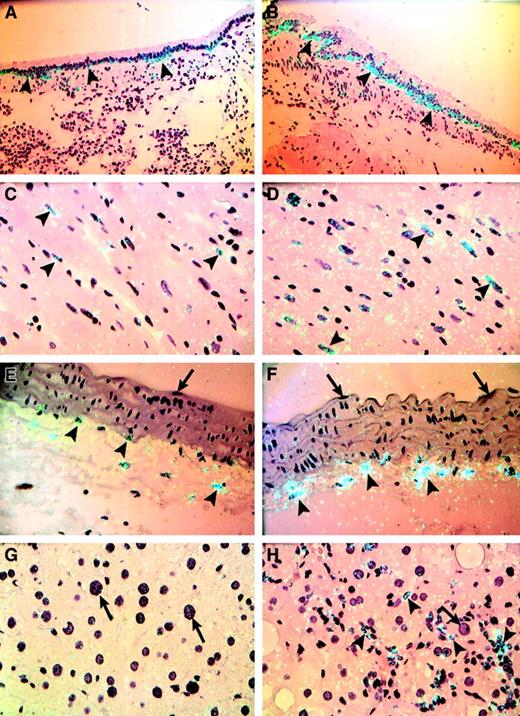
In situ hybridization experiments were performed to determine the cellular distribution of TF mRNA within various tissues from lean and ob/ob mice (Figures 2,3). TF mRNA was detected in bronchiolar epithelial cells in the lungs of lean mice (Figure 2A), and this signal was markedly elevated in the same cells in lungs from obese mice (Figure 2B). A weak but cell-specific signal for TF mRNA was observed in cardiomyocytes in the heart of lean mice (Figure 2C). In heart tissue from ob/ob mice, a larger proportion of the myocytes expressed TF mRNA, and the intensity of this signal was increased as well (Figure2D). TF expression was observed in adventitial cells lining the aorta (Figure 2E) and other arteries (Figure 3A) of lean mice, and this signal for TF was also elevated in vessels from obese mice (Figure 2F, Figure 3B). It is well established that TF is expressed in adventitial fibroblasts surrounding blood vessels under normal and pathologic conditions. For example, in 1989, Wilcox et al22 reported that TF mRNA was expressed in fibroblastlike cells in the adventitia surrounding normal vessels. Since then, several other investigators have confirmed the expression of TF mRNA and antigen in fibroblastic cells in vascular adventitia.18,19 38-43 The TF-positive adventitial cells observed in this study (Figure 2F, Figure 3B) are thus, in all likelihood, stromal fibroblasts of the vascular adventitia. The fact that these cells did not stain with the smooth muscle cell–specific marker α-actin (Dako, Carpinteria, CA; data not shown) and the macrophage marker F4/80 (Bachem, Philadelphia, PA; data not shown), 2 other cell types likely to be found in this location, supports this hypothesis. It should be noted that we did not observe TF expression in large-vessel endothelial cells in any of the tissues examined from either lean or obese mice (Figure 2E,F, arrows; Figure 3). In the liver, hepatocytes did not express TF in the lean or obese mice (Figure 2G,H, arrows). However, patches of inflammatory/Kupffer cells in the obese liver appeared to express TF mRNA (Figure 2H, arrowheads).
Cellular distribution of TF mRNA in tissues from lean and obese (ob/ob) mice.
In situ hybridization was performed on sections of paraffin-embedded tissues from lean mice (A,C,E,G) and obese mice (B,D,F,H) using35S-labeled TF riboprobes, as described in “Materials and methods.” Representative sections are shown. (A,B) Lung; (C,D) heart; (E,F) aorta; (G,H) liver. Arrowheads indicate examples of positive hybridization signals, and arrows indicate the absence of a hybridization signal. Slides were exposed for 4 weeks at 4°C and stained with hematoxylin and eosin. Original magnification is × 400 for all sections.
Cellular distribution of TF mRNA in tissues from lean and obese (ob/ob) mice.
In situ hybridization was performed on sections of paraffin-embedded tissues from lean mice (A,C,E,G) and obese mice (B,D,F,H) using35S-labeled TF riboprobes, as described in “Materials and methods.” Representative sections are shown. (A,B) Lung; (C,D) heart; (E,F) aorta; (G,H) liver. Arrowheads indicate examples of positive hybridization signals, and arrows indicate the absence of a hybridization signal. Slides were exposed for 4 weeks at 4°C and stained with hematoxylin and eosin. Original magnification is × 400 for all sections.
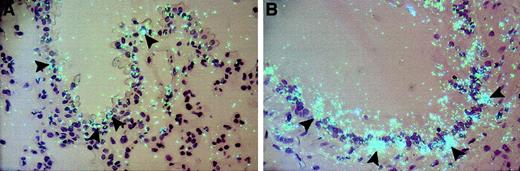
Cellular distribution of TF mRNA in an artery in the perinephric fat from lean and obese (ob/ob) mice.
In situ hybridization was performed on paraffin-embedded perinephric fat from lean (A) and obese (B) mice using 35S-labeled TF riboprobes, as described in “Materials and methods.” Arrowheads indicate examples of positive hybridization signals, and arrows show the absence of hybridization signal in endothelial cells. Slides were exposed for 4 weeks at 4°C and stained with hematoxylin and eosin. Original magnification is × 400 for both sections.
Cellular distribution of TF mRNA in an artery in the perinephric fat from lean and obese (ob/ob) mice.
In situ hybridization was performed on paraffin-embedded perinephric fat from lean (A) and obese (B) mice using 35S-labeled TF riboprobes, as described in “Materials and methods.” Arrowheads indicate examples of positive hybridization signals, and arrows show the absence of hybridization signal in endothelial cells. Slides were exposed for 4 weeks at 4°C and stained with hematoxylin and eosin. Original magnification is × 400 for both sections.
Regulation of TF mRNA by insulin in vivo
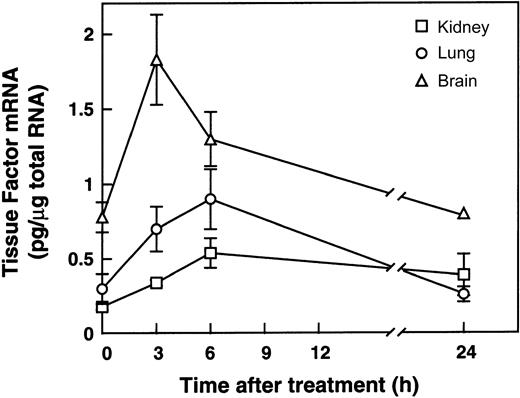
Insulin is increased in the plasma of obese insulin-resistant ob/ob and db/db mice because of the compensatory hyperinsulinemia that usually accompanies insulin resistance in these models.44In previous studies, we demonstrated that intraperitoneal administration of insulin to lean or ob/ob mice increased PAI-1 expression in the plasma and adipose tissues.45 We therefore hypothesized that the elevated insulin might also induce TF gene expression in these mice. To begin to test this hypothesis, we determined the effect of exogenously administered insulin on TF gene expression in lean mice. A variety of tissues were removed 3, 6, and 24 hours after intraperitoneal administration of 10 U insulin, and total RNA was prepared and analyzed for TF mRNA by quantitative RT-PCR (Figure 4). Insulin induced TF mRNA in the kidney (2.5-fold; P < .004, n = 3), lung (3-fold;P < .02, n = 3), brain (2.5-fold;P < .05, n = 3), and adipose tissues (2-fold;P < .04, n = 3; data not shown). Insulin did not induce TF mRNA in the heart, and TF expression in the liver decreased 3-fold after insulin treatment (data not shown). In situ hybridization analysis failed to detect specific cellular signals for TF mRNA in the kidney, lung, or brain of insulin-treated lean mice (data not shown). In the lung, however, insulin increased TF mRNA in the bronchiolar epithelial cells (Figure 5B). This pattern of TF expression in the lungs from insulin-treated lean mice was similar to the pattern of expression observed in the lungs from obese mice (Figure 2B). Taken together, these results are consistent with the hypothesis that the hyperinsulinemia associated with obesity may, in part, be responsible for the local elevation of TF mRNA observed in some tissues of the obese mice.
Induction of TF mRNA expression in lean mice by insulin.
Male lean mice (C57BL/6J+/+) 6 to 8 weeks old were injected intraperitoneally with 10 U human insulin (Himulin) or saline, and various tissues were removed 3, 6, and 24 hours later. Total RNA was prepared and analyzed for TF gene expression by quantitative RT-PCR. n = 3, mean ± SD.
Induction of TF mRNA expression in lean mice by insulin.
Male lean mice (C57BL/6J+/+) 6 to 8 weeks old were injected intraperitoneally with 10 U human insulin (Himulin) or saline, and various tissues were removed 3, 6, and 24 hours later. Total RNA was prepared and analyzed for TF gene expression by quantitative RT-PCR. n = 3, mean ± SD.
Effect of insulin on the cellular distribution of TF mRNA in the lungs of lean mice.
In situ hybridization was performed on paraffin sections from the lungs of untreated (A) and insulin-treated (Himulin, 10 U; 6 hours) (B) lean (C57BL/6J) mice. Slides were exposed for 4 weeks at 4°C and stained with hematoxylin and eosin. Arrowheads indicate positive cells. Original magnification is × 400 for both sections.
Effect of insulin on the cellular distribution of TF mRNA in the lungs of lean mice.
In situ hybridization was performed on paraffin sections from the lungs of untreated (A) and insulin-treated (Himulin, 10 U; 6 hours) (B) lean (C57BL/6J) mice. Slides were exposed for 4 weeks at 4°C and stained with hematoxylin and eosin. Arrowheads indicate positive cells. Original magnification is × 400 for both sections.
Discussion
Thrombotic episodes associated with various diseases, including atherosclerosis, septic shock, and cancer, are often correlated with increased expression of TF.17,21-23 Obese/NIDDM patients are at a higher risk for developing atherothrombotic disease,1,2 and several studies have documented abnormalities in the coagulation system in these patients, including increases in the plasma concentrations of factor VII.8Although factor VII increases in the plasma of obese individuals, little information is available about whether TF, the cellular receptor for factor VII and the primary initiator of the coagulation cascade,16-19 is also elevated. In previous studies, we demonstrated that the ob/ob mouse is a potentially useful model of human obesity because it provided novel insights into the elevation and abnormal regulation of PAI-1 gene expression in this condition.28 Moreover, when compared with lean mice, genetically obese mice have higher levels of TF gene expression in their adipose tissues.29 In the experiments described in the present study, we used the same model system (ie, ob/ob mice) as well as an additional model of murine obesity (db/db mice) to investigate whether TF gene expression in obese mice was altered in other tissues besides the fat. Because hyperinsulinemia is associated with obesity and appears to be an independent risk factor for cardiovascular disease,3 46-48 we also investigated the effects of insulin on TF activity in plasma and on TF gene expression in tissues.
Our results demonstrate that TF mRNA is elevated in several tissues of obese mice compared with their lean counterparts, including the brain, lung, kidney, heart, adipose tissue, and liver (Figure 1A,B). In situ hybridization demonstrated elevated TF expression in extravascular cells in most of these tissues (Figure 2). For example, elevated TF mRNA was observed in the bronchiolar epithelial cells of the lung, in myocytes of the heart, in adventitial cells (probably stromal fibroblasts) of blood vessels, in tubular epithelial cells of the kidney (data not shown), and in astrocytes of the brain (data not shown). The increased expression of TF mRNA in tissues from obese mice appears to result from increased synthesis by the same cells that constitutively produce it in lean mice.18,19,36,39,41,42Many studies have demonstrated the extravascular activation of the TF-dependent coagulation pathway.49-54 Thus, the increase in TF expression by extravascular cells in many tissues of the obese mice could conceivably promote a local hypercoagulable state in these tissues and thereby promote local fibrin deposition. Recent studies have demonstrated the presence of circulating and potentially active TF in the blood of healthy subjects, and this plasma TF may be involved in thrombus propagation at the site of vascular injury.24Whether elevated TF mRNA observed in tissues of obese mice in this study actually leads to elevated TF activity in the blood remains to be determined. An increase in plasma TF antigen and activity has been observed in a number of disease states, such as myocardial infarction,25 unstable angina,26 and sickle cell disease.27 Plasma TF activity also was observed in patients with diabetes mellitus, with the concentrations being significantly higher in patients with retinopathy or nephropathy than in patients with no complications.55 Finally, hypercoagulable states as a result of shedding of TF-rich microvesicles from cell surfaces have been demonstrated in cancer56 and disseminated intravascular coagulation,57,58 as well as in collagen disease, diabetic microangiopathy, and chronic renal failure.59
Experiments were performed to identify mechanisms that may contribute to the elevated levels of TF mRNA in tissues of the obese mice. The ob/ob and db/db mice are insulin resistant and hyperinsulinemic,44 and both of these features appear to represent important risk factors for cardiovascular disease.3,46-48 This hyperinsulinemia may promote atherosclerosis by a number of mechanisms. For example, high insulin levels may stimulate mitogenic signaling pathways leading to the proliferation of vascular endothelial and smooth muscle cells.60-62 Moreover, insulin regulates lipoprotein metabolism60,63 and stimulates the synthesis of endothelin and PAI-1,28,60,64 both of which are atherogenic molecules. In this study, we therefore asked whether insulin could also induce TF expression in various tissues from lean mice. Intraperitoneal injection of 10 U regular insulin into lean mice increased plasma insulin to levels observed in obese mice.28 TF mRNA expression was increased in several tissues, including kidney, lung, brain, and adipose tissue (Figure 4). However, except in the lung, we were unable to detect insulin-mediated increases in TF mRNA in these tissues by using in situ hybridization. A possible explanation for this failure may be that in these tissues, TF mRNA is widely distributed and thus diluted below the detection threshold of the in situ technique. According to this idea, TF mRNA would still be detectable by the more sensitive PCR assay. In the lung, we observed an increase in TF in patches of bronchiolar epithelial cells (Figure 5). Induction of TF by insulin in tissues such as the kidney may create a prothrombotic milieu, thus contributing to the diabetic nephropathy and glomerulosclerosis often associated with obesity and NIDDM. In this regard, recent human studies have demonstrated that hyperinsulinemic patients have a reduced capacity to release tissue factor pathway inhibitor (TFPI), the inhibitor of factor VIIa/tissue factor complex,65 from endothelial cells in response to heparin. Thus, hyperinsulinemia appears to promote a prothrombotic state not only by increasing TF expression, but also by inhibiting the release of TFPI.
The observed changes in TF may be an epistatic effect caused by the absence of leptin (ob/ob) or leptin signaling (db/db) rather than obesity per se. However, we have observed that the amount of TF mRNA in adipose tissues of ob/ob mice,29 and in other tissues such as the brain, lung, kidney, heart, and liver (data not shown), increases as the animals become more obese with age. These observations support the hypothesis that it is obesity per se that leads to elevated TF expression in this model. Analyzing TF gene expression in other models of obesity (eg, fat/fat, tub/tub, or diet induced), in which obesity does not depend on functional leptin, will be the ultimate proof that increased TF expression is a general feature of the obese phenotype. In summary, the mechanisms that promote hemostatic imbalance in obese and diabetic conditions are obviously complex and may involve the dysregulation of several genes of the coagulation and fibrinolytic cascades. Our data clearly demonstrate that TF mRNA expression is elevated in several tissues of obese mice when compared with those from lean mice and that this expression may be regulated by insulin in some tissues. These changes in TF expression together with elevated PAI-1 levels in obesity28 may simultaneously compromise normal fibrin clearance mechanisms and lead to a procoagulant state. These observations thus raise the possibility that increased coagulation and impaired fibrinolysis may contribute to the increased cardiovascular risk associated with conditions such as obesity and NIDDM.
We thank T. Thinnes for her excellent technical assistance and Alicia Palestini for her expert secretarial skills. This is the Scripps Research Institute manuscript number 13140-VB.
Supported by grants from the National Institutes of Health (HL 47819) and Novartis Pharmaceuticals.
The publication costs of this article were defrayed in part by page charge payment. Therefore, and solely to indicate this fact, this article is hereby marked “advertisement” in accordance with 18 U.S.C. section 1734.
References
Author notes
David J. Loskutoff, Department of Vascular Biology, The Scripps Research Institute, 10550 North Torrey Pines Rd, VB-3, La Jolla, CA 92037; e-mail: loskutof@scripps.edu.


This feature is available to Subscribers Only
Sign In or Create an Account Close Modal