Abstract
Human Nude/SCID (severe combined immunodeficiency) is the first severe combined immunodeficiency caused by mutation of the winged–helix–nude (WHN) gene, which is expressed in the thymus but not in the hematopoietic lineage. The disease is characterized by a T-cell defect, congenital alopecia, and nail dystrophy. A Nude/SCID patient who underwent bone marrow transplantation from the human leukocyte antigen–identical heterozygote brother was studied to investigate, in this unique model, the role of the thymus in immunologic reconstitution. Despite an increase in CD3+, CD4+, and CD8+cells, CD4+ CD45 RA naive lymphocytes were not regenerated. Conversely, naive CD8+ cells were normal. After an initial recovery, lymphocyte proliferation to mitogens progressively declined compared with controls and genotypically identical donor cells grown in the WHN+/−environment. Analysis of the T-cell receptor (TCR) repertoire of CD4+ cells revealed that only 3 of 18 Vβ families had an altered CDR3 heterogeneity length profile. Conversely, CD8+lymphocytes showed an abnormal distribution in most Vβ families. These data indicate that the thymus is differentially required in the reconstitution of CD4+ and CD8+ naive subsets and in the maintenance of their TCR repertoire complexity. Taken together, these findings suggest that bone marrow transplantation is ineffective in the long-term cure of this form of SCID.
Introduction
Severe combined immunodeficiency (SCID) encompasses a wide spectrum of disorders of both cell-mediated and humoral immunity.1 Defects in lymphoid differentiation may lead to the complete absence of T and B cells (T−B−phenotype) or, alternatively, may predominantly affect T lymphocyte development (T-B+ phenotype).1 In a few patients with SCID with a predominant T-cell defect, T cells are detectable in peripheral blood but are not functional because of abnormalities in the cell activation process rather than because of differentiation defects.2
In recent years, the identification of several molecular alterations has led to a better understanding of the mechanisms underlying SCID. Alterations in at least 8 genes have been identified and associated with a clinical phenotype of SCID. Abnormalities of signaling molecules, such as the common γ element of several cytokine receptors, the α chain of IL-7 receptor,3 and the JAK-3 tyrosine kinase that associates with the γ chain, are responsible for the X-linked or the autosomal recessive forms of T-B+SCID.4,5 Mutations of RAG-1 andRAG-2 have been described in autosomal recessive forms of the disease.6 Mutations of elements of the T-cell receptor (TCR)/CD3 multimeric complex, such as CD3ε or CD3γ chains,7,8 or of the 70-kd kinase that associates with the ζ chain of the complex, also result in the less severe forms of activation defects.9 It is noteworthy that all the genes so far implicated in the pathogenesis of SCID encode for proteins selectively expressed in hematopoietic tissue. This implies that in most patients, the affected cell line can be replaced by stem cells from healthy donors. However, an appropriate, productive immune response also requires appropriate intercellular connections. A relation between the hematopoietic precursor and the thymic epithelial component plays a crucial role in cell ontogeny and in negative and positive selection.10
We previously reported on 2 sisters affected by SCID because of a T-cell defect associated with congenital alopecia and nail dystrophy.11 This complex phenotype was considered the human homologue of the mouse Nude/SCID phenotype.11Subsequently, the siblings were found to carry a nonsense mutation in the highly conserved winged–helix–nude (WHN) gene.12 Alteration of this gene is responsible for mouse Nude/SCID.13-15WHN is a forkhead–winged helix transcription factor whose expression is restricted to the thymic epithelium, epidermis, and hair follicle.16 17 Human Nude/SCID is the first example of a human SCID caused by a gene not expressed in the hematopoietic cells. Six years ago, before the molecular alteration was identified, the younger of the 2WHN−/− siblings underwent allogeneic human leukocyte antigen (HLA)-related bone marrow transplantation (BMT) because of her critical clinical and immunologic conditions. The current study was undertaken to evaluate, in this unique model, the role of the thymus in immunologic reconstitution and the long-term efficacy of BMT in the human Nude/SCID phenotype.
Patient, materials, and methods
Patient history
The patient was born of consanguineous Italian parents with a complete absence of hair and dystrophic nails, as seen in her elder sister. Thereafter, like her older sister, she was found to have severe functional T-cell immunodeficiency. No thymic shadow was evident in either child's radiologic examination. The first child died at 12 months of recurrent bronchopneumonia. The clinical and immunologic phenotype has been reported as a new human syndrome (MIM 601705).11 At the age of 5 months, the second child underwent HLA-identical total bone marrow transplantation from her unaffected brother, after only 2 doses of antilymphocyte rabbit globulin at a dose of 2.5 mg/kg and no immunosuppressive therapy or myeloablation by irradiation. The engraftment of donor cells was established through the analysis of highly polymorphic DNA regions using the DIS80 and DQ α markers. Six years after BMT the patient is healthy, but the alopecia has persisted, indicating that it was primitive in nature. Tonsils were not evident, whereas lymph nodes were present both before and after BMT.
Immunophenotype and proliferative responses
The immunophenotype was performed at regular intervals during the post-BMT follow-up using the following monoclonal antibodies in 2-color immunofluorescence: anti-CD3 (Leu-4), anti-CD4 (Leu-3a), anti-CD8 (Leu-2a), anti-CD19 (Leu-12), anti-CD45 RA, anti-CD45 RO, anti-CD11b (Leu-15), and anti-CD28 (Leu-28) (Becton Dickinson, San Jose, CA). A B-D flow cytometer was used. For the proliferation assays, peripheral blood mononuclear cells (PBMC) (2 × 105) were obtained by Ficoll-Hypaque (Biochrom, Berlin, Germany) density gradient separation and cultured at 37°C for 72 hours with medium alone or with 10 μg/mL PHA (Difco Laboratories, Detroit, MI), 8.25 μg/mL concanavalin-A (ConA) (Difco), 10 μg/mL pokeweed (PwM) (Life Technologies, Paisley, United Kingdom), 20 ng/mL phorbol-myristate acetate, and 0.5 mM ionomycin (Sigma Chemical, St. Louis, MO). CD3 cross-linking (CD3 X-L) was performed by precoating tissue culture plates with 1 or 10 ng/mL anti-CD3 monoclonal antibody (Ortho Diagnostics, Raritan, NJ); rIL-2 was used at a concentration of 100 U/mL. Cultures were pulsed with 1 μCi per well of [3H]-thymidine (Amersham International, Buckinghamshire, United Kingdom) 18 hours before harvesting.
RNA extraction and cDNA preparation
Peripheral blood mononuclear cells were isolated by density gradient centrifugation as described above. Magnetic beads coated with anti-CD8+ monoclonal antibody (Dynal Biotech, Oslo, Norway) were used for the positive and negative sorting of T cells. Total RNA was extracted from positively or negatively sorted lymphocytes by using Tryazol (Gibco-BRL, Bethesda, MD). Reverse transcription–polymerase chain reaction (RT-PCR) was performed by using the one-step RT-PCR kit (Gibco-BRL) according to the manufacturer's instructions.
CDR3 heterogeneity length analysis
Conditions for CDR3 size distribution analysis are described elsewhere.18 Briefly, cDNA was amplified through PCR with specific primers to 24 different Vβ families and a fluorescent Cβ primer. One microliter amplified product was mixed with 1.5 μL 100% formamide and 0.5 μL size standard (Genescan-500 ROX, ABI 373; PerkinElmer, Urayasu, Japan), heated at 90°C for 3 minutes, and electrophoresed in a 6.75% denaturing polyacrylamide gel. The distribution of CDR3 size within the amplified product of each Vβ subfamily was evaluated by using an automatic sequencer (ABI 377; PerkinElmer) equipped with a computer program that allows the determination of the fluorescence intensity of each band. Results are depicted as peaks corresponding to the intensity of the fluorescence. CDR3 size patterns that did not have a bell-shaped distribution but had prominent peaks (less than 5) were judged to be abnormal.
Results
Lymphocyte reconstitution in the WHN−/−patient after allogeneic BMT
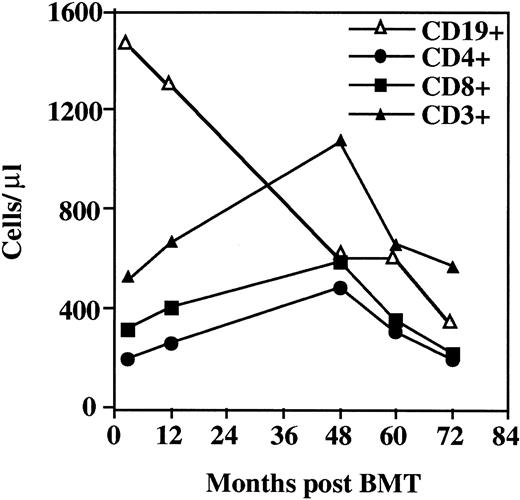
The patient's immunophenotype before BMT is reported elsewhere.11 Briefly, the WHN−/−patient had CD3+ lymphocytes of reduced number and percentage (650 cells/μL and 25%, respectively), a low number of CD4+ cells, and a low-normal number of CD8+cells.11 The T-cell subset and B-lymphocyte profiles during the 6-year post-BMT follow-up are shown in Figure1. Consequent to the administration of antilymphocyte globulin, 12 days after BMT, CD3+, CD4+, and CD8+ cell levels were markedly reduced, whereas the CD19+ cell level was higher than normal (1400 cells/μL). The number of CD3+ cells started to increase progressively 3 months after BMT. Twelve months after BMT, T cells reached a level of 700/μL (27% of PBMCs). CD8+cells increased faster than CD4+ lymphocytes. One year after BMT, chimerism analysis revealed that T cells were from the donor and B cells were from the host (data not shown). Four years after BMT, T cells remained of donor origin, and CD levels were as follows: CD3+cells, 1100/μL; CD8+ cells, 500/μL; and CD4+ cells, 400/μL (18% of PBMCs). Thereafter, CD3+, CD4+, and CD8+ levels progressively declined. The CD4+/CD8+ ratio persisted below 1.
Immunologic reconstitution in the
WHN−/− patient after allogeneic BMT. Immunophenotype analysis of CD19+, total CD3+, CD4+, and CD8+ lymphocyte subsets in the WHN−/− patient after allogeneic BMT. Results are expressed as absolute values.
Immunologic reconstitution in the
WHN−/− patient after allogeneic BMT. Immunophenotype analysis of CD19+, total CD3+, CD4+, and CD8+ lymphocyte subsets in the WHN−/− patient after allogeneic BMT. Results are expressed as absolute values.
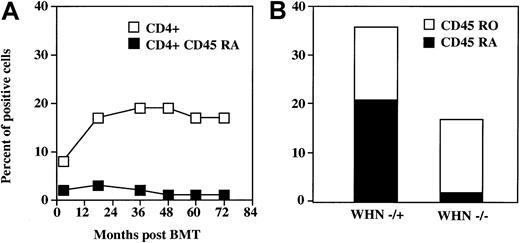
Regeneration of the CD45 RA “naive” phenotype within CD4 and CD8 compartments in the WHN−/−environment after BMT
Despite the increase of CD3+ cells and, albeit to a lower extent, of mature CD3+CD4+ cells, nearly all these cells showed a memory CD45 RO phenotype, whereas only 1% to 3% of lymphocytes expressed the CD4+ CD45 RA “naive” phenotype (Figure 2A). At 6 years of follow-up, no changes were observed in the CD4+ CD45 RA–CD4+ CD45 RO ratio. Because all T cells in theWHN−/− patient were ofWHN+/− donor origin, we compared the memory and naive phenotype of CD4+ cells developed in vivo in the donor with those developed in the recipient environment. Six years after BMT, the CD4+ CD45 RA compartment was significantly reduced only in the CD4+ cells obtained from the recipientWHN−/− environment (Figure 2B). This resulted in a very low RA:RO ratio compared with cells from the donorWHN+/− environment (0.06 and 1.37, respectively). These findings indicate that a functional thymus is necessary to renew the naive CD4+ subset.
Reconstitution of CD4+ CD45 RA cells in the
WHN−/− patient after allogeneic BMT. (A) Time-course of the reconstitution of CD4+CD45 RA cells in the WHN−/− patient during the 72 months after allogeneic BMT. (B) Comparison of CD4+ CD45 RA and RO subsets between the WHN−/− recipient and the WHN+/− donor environment 6 years after BMT. Lymphocytes were double stained and analyzed by flow cytometry. Results are expressed as percentage of positive cells.
Reconstitution of CD4+ CD45 RA cells in the
WHN−/− patient after allogeneic BMT. (A) Time-course of the reconstitution of CD4+CD45 RA cells in the WHN−/− patient during the 72 months after allogeneic BMT. (B) Comparison of CD4+ CD45 RA and RO subsets between the WHN−/− recipient and the WHN+/− donor environment 6 years after BMT. Lymphocytes were double stained and analyzed by flow cytometry. Results are expressed as percentage of positive cells.
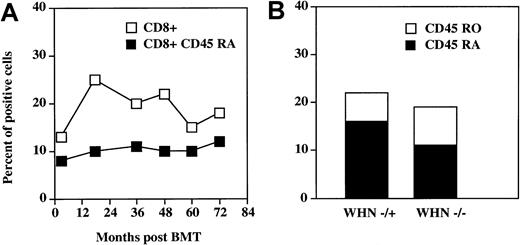
During the post-BMT period there was a marked increase in the percentage and number of CD3+CD8+ cells, which reached 27% and paralleled the increase of the whole CD3+population (Figure 3A). This subset did not contain the CD3−CD8+ natural killer population, which represented 6% of PBMCs. In contrast to the CD4 compartment, the percentage of CD8+ CD45 RA cells was 12% and remained stable during follow-up. Six years after BMT, the CD45 RA–CD45 RO ratio was normal (1.30) (Figure 3B). This suggests that CD8+ CD45 RA lymphocytes are generated even in the absence of a functional thymus. Indeed, the CD8+ CD45 RA–CD8+ CD45 RO ratio was also normal (1.66) also before BMT. CD8+ cells were also stained for the expression of CD28 and CD11b molecules. Fifty percent of CD8+ cells expressed CD28, whereas only 4% of these cells bore CD11b.
Reconstitution of CD8+ CD45 RA T cells in the
WHN−/− patient after the BMT. (A) Time-course of the reconstitution of CD8+ CD45 RA cells in the WHN−/− patient during the 72 months after allogeneic BMT. (B) Comparison of CD8+ CD45 RA and RO subsets between the WHN−/− recipient and theWHN+/−donor environment 6 years after BMT. Lymphocytes were stained and analyzed as described in Figure 2 and in “Materials and methods.”
Reconstitution of CD8+ CD45 RA T cells in the
WHN−/− patient after the BMT. (A) Time-course of the reconstitution of CD8+ CD45 RA cells in the WHN−/− patient during the 72 months after allogeneic BMT. (B) Comparison of CD8+ CD45 RA and RO subsets between the WHN−/− recipient and theWHN+/−donor environment 6 years after BMT. Lymphocytes were stained and analyzed as described in Figure 2 and in “Materials and methods.”
Proliferative responses in the WHN−/−patient after allogeneic BMT
To assess the functional reconstitution of peripheral lymphocytes after BMT, we analyzed at regular intervals during the 6-year follow-up the proliferative response of PBMC from theWHN−/− patient to CD3 cross-linking. From the third month after BMT there was a progressive improvement in the proliferative response to CD3 cross-linking (Figure4A). The response was completely normal 2 years after BMT. Proliferative capability began to decline 48 months after BMT. Again, because all T cells in the recipient were of donor origin, we compared the proliferative response to common mitogens of PBMC obtained from the WHN−/− recipient with the genotypically identical donor cells developed in vivo in theWHN+/− environment. Six years after BMT a remarkable deficiency of cells from theWHN−/− recipient was evident (Figure 4B). Even though exogenous IL-2 induced an approximately 3-fold increase of the response to CD3 cross-linking, the response never reached normal levels.
Proliferative responses in the
WHN−/− patient or theWHN+/− healthy donor brother.(A) Proliferative response evaluated through [3H]-thymidine incorporation after stimulation of peripheral blood mononuclear cells by CD3 cross-linking. PBMCs from the WHN−/−patient were analyzed at regular intervals during the 6 years after allogeneic BMT. The results are expressed as cpm. Dotted lines indicate mean ± SD of control values. (B) Comparison 6 years after BMT of the proliferative response to several mitogens between cells grown in the WHN−/− recipient and in theWHN+/− donor environment. Exogenous rIL-2 was used at a concentration of 100 U/mL.
Proliferative responses in the
WHN−/− patient or theWHN+/− healthy donor brother.(A) Proliferative response evaluated through [3H]-thymidine incorporation after stimulation of peripheral blood mononuclear cells by CD3 cross-linking. PBMCs from the WHN−/−patient were analyzed at regular intervals during the 6 years after allogeneic BMT. The results are expressed as cpm. Dotted lines indicate mean ± SD of control values. (B) Comparison 6 years after BMT of the proliferative response to several mitogens between cells grown in the WHN−/− recipient and in theWHN+/− donor environment. Exogenous rIL-2 was used at a concentration of 100 U/mL.
Humoral immunity studies
To evaluate whether immunologic reconstitution, as revealed by cell number and proliferative response to mitogens, was associated with normal capability to generate specific antibody production, an immunization program was scheduled in the post-BMT period. To discriminate between a response due to committed donor cells and that due to newly in vivo–primed lymphocytes, the program included immunizations both to known antigens to the donor at the time of BMT and to neoantigens such as HBsAg. Table 1illustrates the comparison of humoral immunity in the pre- and post-BMT (36 months) periods and, in particular, antibody responses in theWHN−/− (recipient) versus theWHN+/− (donor) environment. A full immunologic reconstitution was achieved 2 years after BMT as revealed by the rise in anti-tetanus toxoid antibodies. Notably, even in theWHN−/− environment, B cells were able to generate a specific antibody response toward antigens unknown at the time of the transplantation. In fact, 2 boosters of HBsAg led to the appearance of specific antibodies, which demonstrates that efficient priming also occurs in the WHN−/−environment.
Humoral immunity studies
| . | WHN−/−recipient . | WHN+/− donor . |
|---|---|---|
| Serum IgG (g/L) | 4.94 | 6.55 |
| Serum IgA (g/L) | 0.49 | 0.77 |
| Serum IgM (g/L) | 0.80 | 0.74 |
| Autoantibodies | abs | abs |
| Specific antibodies | ||
| Allohemoagglutinins | ||
| Pre-BMT | abs | anti-A/B (1:128) |
| Post-BMT | anti-A (1:8) | |
| Tetanus toxoid | ||
| Pre-BMT | abs | pos |
| Post-BMT | pos | pos |
| HbsAg | ||
| Pre-BMT | abs | abs |
| Post-BMT | pos | pos |
| . | WHN−/−recipient . | WHN+/− donor . |
|---|---|---|
| Serum IgG (g/L) | 4.94 | 6.55 |
| Serum IgA (g/L) | 0.49 | 0.77 |
| Serum IgM (g/L) | 0.80 | 0.74 |
| Autoantibodies | abs | abs |
| Specific antibodies | ||
| Allohemoagglutinins | ||
| Pre-BMT | abs | anti-A/B (1:128) |
| Post-BMT | anti-A (1:8) | |
| Tetanus toxoid | ||
| Pre-BMT | abs | pos |
| Post-BMT | pos | pos |
| HbsAg | ||
| Pre-BMT | abs | abs |
| Post-BMT | pos | pos |
abs, absent; pos, positive.
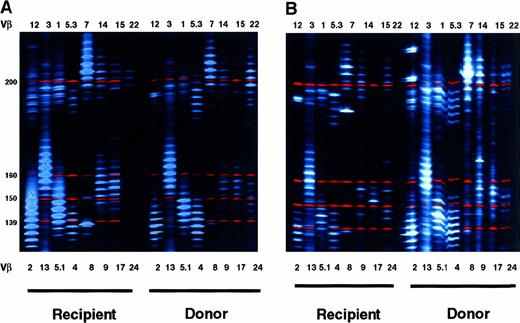
Maintenance of T-cell receptor diversity in theWHN−/−environment
The biologic mechanisms ensuring maintenance of T-cell repertoire diversity in mature lymphocytes are not completely known and involve central and peripheral lymphoid tissue. In this context, the analysis of TCR diversity in our WHN−/− patient after BMT provides a unique opportunity to analyze the role of peripheral factors in maintaining T-lymphocyte diversity. We evaluated the TCR repertoire by analyzing Vβ CDR3 heterogeneity length (spectratyping) 6 years after BMT in genotypically identical T cells obtained from theWHN−/− recipient and from theWHN+/− donor. Figure5A shows the CDR3 length profiles of 18 Vβ families in the CD4+ cells. In cells from theWHN−/− recipient there was a marked alteration in the CDR3 length profile in only 3 of the 18 Vβ families.8,12 15 Notably, in 2 of them (families 8 and 15), the altered profile overlapped in donor and recipient cells. Spectratyping analysis of CD8+ lymphocytes (Figure 5B) revealed that most of the Vβ families displayed an altered profile (11 of the 18 families). Most of them (families 5.3, 8, 9, 12, 14, 17) exhibited oligoclonal expansion. The complexity of the TCR repertoire was reduced in the remaining families. Even though all the families were altered in both WHN+/− andWHN−/− cells, within individual families the distribution pattern differed between recipient and donor, unlike what occurred in CD4+ cells. Spectratyping analysis confirmed the different behavior of CD4+ and CD8+ cells in the absence of a functional thymus.
TCR diversity of recipient or donor T cells.
Comparative analysis of TCR-Vβ CDR3 heterogeneity length in (A) CD8-depleted (CD8−) or (B) CD8-enriched (CD8+) lymphocyte suspensions derived 72 months after BMT from theWHN−/− recipient and theWHN+/− donor environment. RT-PCR was performed on RNA isolated from purified T-cell subsets. PCR products were separated on DNA sequencing polyacrylamide gel by using an automated ABI PRISM 377 apparatus (PerkinElmer). Band intensity was evaluated with the Gene Scan software. Size markers are indicated. TCR-Vβ families showing fewer than 5 major peaks were considered altered.
TCR diversity of recipient or donor T cells.
Comparative analysis of TCR-Vβ CDR3 heterogeneity length in (A) CD8-depleted (CD8−) or (B) CD8-enriched (CD8+) lymphocyte suspensions derived 72 months after BMT from theWHN−/− recipient and theWHN+/− donor environment. RT-PCR was performed on RNA isolated from purified T-cell subsets. PCR products were separated on DNA sequencing polyacrylamide gel by using an automated ABI PRISM 377 apparatus (PerkinElmer). Band intensity was evaluated with the Gene Scan software. Size markers are indicated. TCR-Vβ families showing fewer than 5 major peaks were considered altered.
Discussion
We used a unique model, the human counterpart of the Nude/SCID phenotype recently described by our group,11 to explore the role of the thymus in post-BMT immunologic reconstitution. Nude/SCID is the first form of SCID whose immunodeficiency is substantially and exclusively related to an intrinsic abnormality of the thymus.19,20 The gene involved in this disease is theWHN gene, which encodes a forkhead–winged helix transcription factor and is associated with both the murine and human Nude/SCID phenotypes.12-14 Because WHN is selectively expressed in the thymic epithelia and not in hematopoietic cells, this rare and novel condition in humans is a reliable model with which to investigate the role of intrathymic intercellular connections, and namely the connections between thymic epithelia and hematopoietic progenitors, in the regeneration and maintenance of T-cell responses after allogeneic BMT. The analysis of immunologic reconstitution during the 6-year follow-up after BMT in the patient described herein revealed that the lack of a functional thymus did not prevent the repopulation kinetics of CD3+, CD4+, and CD8+ cells.
In the absence of a functional thymus, in theWHN−/− environment no CD4+CD45+ RA naive lymphocytes were detected in the periphery. Moreover, the long-term follow-up revealed that the defect in the generation of the naive CD4 population was not restored by BMT. Conversely, the WHN−/− patient was able to regenerate and maintain a normal amount of CD8+CD45 RA T lymphocytes. Our findings are in keeping with the recent observation in cancer patients who underwent thymectomy, that after BMT the reconstitution of the CD4+ CD45 RA subset relies on a functional host thymus.21 As did our patient, patients who undergo thymectomy retain the capacity to regenerate the naive CD8+ subset, suggesting the presence of a thymus-independent pathway for the generation of this subset.21 Indeed, recovery of the CD4+ CD45 RO memory subset after BMT in thymus-deficient patients presumably results from expansion of the donor's mature lymphocytes. Taken together, these findings also argue against the possibility that in humans CD4+ CD45 RO cells can convert to the naive CD45 RA phenotype.22 The prompt recovery of the CD8+CD45 RA naive cells may indicate the involvement of extrathymic lymphopoiesis. These cells could also originate from conversion of the CD45 RO phenotype.23 However, it should be noted that the CD8+ CD45 RA compartment is heterogeneous in that it contains unprimed and primed cells.24 Unlike unprimed naive cells, primed cells express low levels of CD27 and CD28 and high levels of the CD11b adhesion molecule. Of note, a large fraction of CD3+CD8+ cells in our patient expressed CD28 and only a small percentage expressed CD11b, which suggests that they were unprimed naive CD8+ T cells. In patients with DiGeorge syndrome, BMT can lead to donor T-cell engraftment,25 though it is unclear whether reconstitution is stable over time and whether donor T cells represent an expansion of mature T lymphocytes differentiated before transplantation.26 However, in DiGeorge syndrome, there is residual thymic function as testified by the post-BMT increase in CD4+ CD45 RA+ cells.25
During the 6-year post-BMT period, the proliferative responses of the recipient peripheral blood cells to common mitogens progressively declined to 20% of the genotypically identical donor cells grown in the WHN+/− environment. Feasibly, in the absence of the thymus, T-lymphocyte survival and homeostasis depend principally on continuous stimulation by lymphoid tissue accessory cells.27-29 Because all T cells were of donor origin, this progressive functional impairment could reflect accelerated accumulation of aged lymphocytes that have a low proliferative capability, somewhat similar to what occurs in the elderly.30 31
The thymus plays a major role in T-cell ontogeny.32,33 In this organ, T cell precursors undergo a complex mechanism of positive and negative selection to shape the TCR repertoire of mature lymphocytes. Mature cells become capable of recognizing exogenous peptides in the context of self-major histocompatibility complex molecules. Cortical epithelial cells of the thymus are unique in their ability to mediate positive selection efficiently,10whereas bone marrow–derived dendritic cells are efficient mediators of negative selection; they are not involved at all in the positive selection process.34 Patients with DiGeorge syndrome have a marked reduction in lymphopoiesis and severe alterations in the lymphocyte repertoire.35 In this context, our analysis of T-cell repertoire complexity provides insights into the mechanisms involved in the maintenance and regeneration of T-cell repertoire diversity. The WHN−/− environment did not influence the long-term maintenance of TCR repertoire diversity in the CD4+ population, which consisted mostly of memory cells. Furthermore, the altered Vβ families had a similar profile in host and donor lymphocytes, suggesting that the donor's repertoire is maintained in the host environment. Unlike the CD4 compartment, the complexity of the TCR CDR3 profile was reduced in most CD8+cells, in a few cases indicating oligoclonal expansion. In addition, within individual Vβ families and unlike what was observed in the CD4 compartment, recipient and donor cells had different CDR3 profiles. Whether extrathymic lymphopoiesis or peripheral mechanisms may have contributed to the CD8+ reconstitution is an intriguing question.36 37
It is noteworthy that 6 years after BMT, and despite the progressive immunodeficiency status and the absence of anti-infectious prophylaxis, the patient remains free of infection, probably because of a residual immune response estimated to be 20% of donor cell proliferative potential. In addition, Nude/SCID mice can clear some viral infections. This is presumably because of a T-cell–independent antibody response and mechanisms of innate immunity.38,39 Clearance of the polyoma virus in T-cell–deficient mice results from T-cell–independent antibody-mediated responses.39 In our patient HBsAg immunization led to specific antibody production. The generation of specific antibody responses toward a neoantigen unknown to the donor cells at the time of BMT may indicate that donor-derived CD4+ cells can provide help for B cells. However, it should be noted that we observed specific antibody responses during the phase of optimal immune response; we did not test the response when immunity declined. Alternatively, the generation of a B-cell response against HBsAg may be obtained in a thymus-independent fashion.
In conclusion, our findings on T-cell reconstitution in theWHN−/− environment indicate that a functional thymus is essential to regenerate the CD4+ naive subset. The long-term maintenance of TCR diversity of the memory–effector CD4+ lymphocytes may be ensured by a peripheral mechanism that does not depend mainly on antigen-driven clonal expansion.28 29 Conversely, the regeneration of CD8+ naive lymphocytes after BMT is less thymus dependent and also occurs in the WHN−/− environment. Lastly, despite the favorable clinical course after BMT, our data indicate that this therapeutic procedure is ineffective in the long-term cure of this form of SCID.
Supported by Biomed 2 grant CT983007, MURST-99-PRIN, and Telethon E0934; J.F. received support from Bundesministerium für Bildung und Forschung (BMBF) grants 01 KS 9503/9 and 01 KX 9820F.
The publication costs of this article were defrayed in part by page charge payment. Therefore, and solely to indicate this fact, this article is hereby marked “advertisement” in accordance with 18 U.S.C. section 1734.
References
Author notes
Claudio Pignata, Department of Pediatrics, Unit of Immunology, “Federico II” University, Via S. Pansini 5-80131, Naples, 80127 Italy; e-mail: pignata@unina.it.

![Fig. 4. Proliferative responses in the. / WHN−/− patient or theWHN+/− healthy donor brother.(A) Proliferative response evaluated through [3H]-thymidine incorporation after stimulation of peripheral blood mononuclear cells by CD3 cross-linking. PBMCs from the WHN−/−patient were analyzed at regular intervals during the 6 years after allogeneic BMT. The results are expressed as cpm. Dotted lines indicate mean ± SD of control values. (B) Comparison 6 years after BMT of the proliferative response to several mitogens between cells grown in the WHN−/− recipient and in theWHN+/− donor environment. Exogenous rIL-2 was used at a concentration of 100 U/mL.](https://ash.silverchair-cdn.com/ash/content_public/journal/blood/97/4/10.1182_blood.v97.4.880/5/m_h80410717004.jpeg?Expires=1765897872&Signature=uLhv03y7qkGxRYyTec6lVZk0ODWpBDg68KpR3ecFLSzQhc4rrakHWqSTqMuwAQ5nc2UYFsSxKRhXWiTnKEbyw8VG7Ql6UsN3BH9s7Q5sHFs~vMy1cnrpIQG2j2LPx5eF8VN4Yw0QvsaM9zBQJabtOtty0nm9HFFCmByMNntP56KG2yUTISB3ODZmHpYd7FmxjVN8ThEd0XbzIFxOBKpeQd8xwIgaD3q7aJf5GimVMSqI6SSseAPGz7BjGybWrgzQMjuRTOTgoHLnK8JjE5P4Sn2-u8UT6FzKYgmpd-ye1sp58gbonY~82-jlLjOjPbNFGdIdg8PrfBLkuW~9MeFC9w__&Key-Pair-Id=APKAIE5G5CRDK6RD3PGA)
This feature is available to Subscribers Only
Sign In or Create an Account Close Modal