Abstract
Murine granulocytes and precursors express low-affinity IgG Fc receptors (FcγR). We investigated the effects of FcγR ligation on the development of eosinophils in cultures of normal murine bone marrow. Eosinophilopoiesis was induced by culture of bone marrow cells in the presence of cytokines (granulocyte-macrophage colony-stimulating factor [GM-CSF], interleukin-3 [IL-3], and IL-5). Addition to the cultures of 2.4G2, a rat monoclonal antibody (mAb) that reacts with FcγRII (CD32) and FcγRIII (CD16), induced granulocyte apoptosis within 24 hours. Granulocytes in cultures that contained 2.4G2 showed chromatin condensation, binding of Annexin-V, and fas induction, and by electron microscopy, apoptosis was most commonly observed in cells of the eosinophil lineage. Since murine granulocytes can express both FcγRII (CD32) and FcγRIII (CD16), we investigated the effect of 2.4G2 on cultures of bone marrow obtained from FcγRIII (CD16) gene–disrupted mice and found that the apoptosis induced with 2.4G2 was CD16-independent. Studies with bone marrow cultures from B6MLR-lpr/lpr and C3H/HEJ-gld/gld mice established that the FcγRII (CD32)-triggered apoptosis was fas-fasL–dependent. When mature eosinophils isolated from hepatic granulomas of Schistosoma mansoni–infected mice were cultured in cytokines in the presence of 2.4G2, the eosinophils underwent apoptosis within 24 hours. These findings identify a previously unknown linkage between FcγR on eosinophils and fas-mediated apoptosis, a connection that could be relevant to mechanisms by which eosinophils mediate tissue injury and antibody-dependent cellular cytotoxicity reactions.
MUCH IS KNOWN about the IgG Fc receptors (FcγR) on mature lymphoid and myeloid cells and about the functions they mediate through interactions with their nominal ligands, IgG and IgG:antigen complexes.1,2 Considerably less is known about the functions of FcγR on immature lymphoid and myeloid cells, even though it is well established that the precursor cells in both lineages coexpress the low-affinity FcγR CD16 and CD32.3-5 Both receptors are encoded by distinct genes and are members of the Ig gene superfamily. The amino acid sequences of the extracellular segments of CD16 and CD32 are 95% identical, but their transmembrane and intracytoplasmic segments are totally different.6 CD16 consists of a single α-chain that associates with the signal-transducing, ITAM-containing γ-chain that is also a component of other receptors such as the high-affinity IgE receptor FcεRI, and the TCR, FcγRI, FcγRIIIA, and FcαRI.7-10 CD32 occurs in two isoforms, β1 and β2, both of which contain a phosphotyrosine motif, designated ITIM, that inhibits ITAM-induced signal transduction.11 12 The near identity of the structures of the extracellular domains of CD16 and CD32 implies that these receptors recognize similar ligands, and the total nonidentity of the transmembrane and cytoplasmic portions of CD16 and CD32 provide the structural basis for their distinct functional properties.
In previous studies,13 we proposed that FcγR expressed on murine T-cell precursors bind alternative, non-Ig ligands expressed on thymic and marrow stromal cells, and that these interactions modulate the rate of lymphoid cell differentiation prior to rearrangement of TCR genes. Consistent with this view is the finding that in T-lineage cells CD16 is downregulated as TCR genes undergo rearrangement. Thus, FcγR on murine lymphoid cells appear to mediate different functions and interact with different ligands depending on the developmental stage of the lymphoid cell.14 Since FcγR are expressed on myeloid cell precursors and on mature myeloid cells, we were interested to know whether FcγR, in addition to their well-known effector functions on mature myeloid cells, might also play a role in the development of myeloid cells. To test this possibility, we investigated the effect of ligation of FcγR by monoclonal anti-FcγR antibody on the development of myeloid cells in an in vitro murine bone marrow culture system in which the presence of interleukin-3 (IL-3), IL-5, and granulocyte macrophage colony-stimulating factor (GM-CSF ) yields cultures in which approximately one third of the cells are eosinophils. These eosinophils are functionally intact, as reflected by their production of eosinophil peroxidase15-18 and of transcripts that encode major basic protein, a mediator of cytotoxicity expressed exclusively by eosinophils.19 Interestingly, eosinophils derived from these conditions, lose their FcγR after 3 days of incubation with the cytokines already cited, suggesting that the expression of FcγR on murine eosinophils is an inducible event.19
We found that ligation of FcγR in this system aborted the development of eosinophils through a fas-dependent, CD32-dependent, CD16-independent induction of apoptosis. Furthermore, mature eosinophils isolated from hepatic granulomas of Schistosoma mansoni–infected mice also underwent apoptosis in response to FcγR ligation by anti-FcγR antibody.
MATERIALS AND METHODS
Mice.Balb/c and CBA mice were purchased from Harlan Sprague-Dawley (Indianapolis, IN). C57BL/6-CD16 gene–disrupted mice20 were constructed by Dr Sjef Verbeek, Department of Immunology, University of Utrecht, The Netherlands. B6MLR-lpr/lpr and C3H/HEJ-gld/gld mice were obtained from Dr Morris Dailey, Department of Pathology, University of Iowa.
Monoclonal antibodies (MoAbs).The MoAbs used in this study were labeled with fluorescein isothiocyanate (FITC), phycoerythin (PE), or cyanine 5.18 (Cy5). They included RB6-8C5 (a rat IgG2b specific for the granulocyte marker, Gr-1) from Pharmingen (San Diego, CA); 6B2 (a rat IgG2a anti–murine CD45R/B220) from Pharmingen; anti–mouse IgM (a hamster IgG) from Jackson ImmunoResearch (Wheat Grove, PA); Fas/Apo-1 (a hamster IgG anti–murine CD95) from Pharmingen; and the isotype match for fas (a polyclonal hamster IgG) from Pharmigen. M1/70 (a rat IgG2a anti–murine Mac-1) and 15.1 (a rat IgG2a anti–murine Ly6C) were obtained from Dr. Thomas Waldschmidt, Department of Pathology, University of Iowa; 2.4G2 (a rat IgG2b anti-FcγRII/RIII) and an irrelevant IgG2b were used in bone marrow (BM) cultures. All MoAbs were dialyzed against phosphate-buffered saline (PBS) and sterile-filtered before addition to the cultures. No endotoxin levels were detected in 2.4G2 MoAbs by the Lumulus amebocyte lysate assay (Pyrochrome, Cape Cod, MA).
Cultures of BM from normal mice.Balb/c male mice (6 to 9 weeks old) were maintained in a horizontal laminar-flow cabinet and provided with sterile food and water ad libitum. Femoral and tibial BMs of 6- to 9-week-old mice were obtained, and a single-cell suspension was prepared in HBSS following a previously described procedure.15 After three washes, 2 × 105 cells/mL were resuspended in RPMI 1640 supplemented with 10% bovine calf serum 0.2 mmol/L-glutamine, 0.1 mmol/L essential and non essential amino acids, 0.1 mmol/L Na-pyruvate, 5 × 10−5 mol/L 2-βME, and an antibiotic-antimycotic (GIBCO-BRL, Grand Island, NY) and incubated in 24-well tissue culture plates (Costar, Cambridge, MA) in the presence of 10 U/mL recombinant GM-CSF, 20 U/mL rIL-5, and 10 U/mL rIL-3 (Genzyme, Cambridge, MA), conditions that have been shown to promote the production of eosinophils.15 Viability of the cultures was determined by Trypan blue dye exclusion.
Flow cytometric analysis.Cells were suspended at a concentration of 107/mL in HBSS buffer containing 10% bovine calf serum, 10 mmol/L HEPES, and 0.02% Na-azide (FACS staining buffer). Cells were stained with conjugated Abs in 50 μL for 40 minutes at 4°C, washed three times, fixed in 2% paraformaldehyde in PBS (pH 7.3), and analyzed on a Coulter (Hialeah, FL) Epics 753 flow cytometer. Listmode data were analyzed using Coulter Elite software (University of Iowa Flow Cytometry Facility). Forward and side scatter and two or three fluorescence parameters were collected on 10,000 cells. For sort purification, cells were stained at a concentration of 1.5 to 2 × 107/mL in FACS staining buffer. A minimum of 3 to 4 × 105 cells were collected.
Apoptotic analysis.Fas and Annexin-V were chosen as apoptotic markers of bone marrow granulocytes. Staining with anti-Fas antibody was performed as described earlier. In parallel, Annexin-V binding was performed following the manufacturer's directions (R & D Systems, Minneapolis, MN). Briefly, BM cells were stained with Gr-1 (PE-labeled) and anti-IgM (Cy5-labeled); after three washes with FACS buffer, Annexin-V (FITC-labeled, 3 μL), propidium iodide (10 μL), and binding buffer (50 μL) were added to a final volume of 500 μL. Early in the process of apoptosis, cells express phosphatidylserine on the outer leaflet of the cell surface membrane and consequently bind Annexin-V. Dead cells do not exclude propidium iodide, which enters and binds to the cellular DNA. In our experiments, cells stained as described were then immediately analyzed by flow cytometry. Cells that were stained with propidium iodide were excluded in all experiments performed.
Electron microscopic studies.Elecron microscopy was performed on cell samples from BM cultures fixed with 2.5% glutaraldehyde in 0.1 mol/L phosphate buffer, pH 7.3, using a Philips (Eindhoven, The Netherlands) CM 10 transmission electron microscope.
Eosinophil purification from liver granulomas of mice infected with S mansoni. Liver granulomas were obtained as previously described.21 Briefly, CBA mice (7 to 8 weeks old) were infected with 35 cercariae of S mansoni, and their livers were harvested 8 to 10 weeks later. Following homogenization of the livers, the intact granulomas were allowed to sediment, treated with collagenase, physically dispersed, and passed through sterile gauze. Inflammatory cells from hepatic granulomas were passed through a nylon wool column to remove macrophages, and nonadherent cells were eluted with RPMI 1640. The nonadherent cell population contained approximately 60% eosinophils, 35% lymphocytes, and 5% macrophages. Murine granuloma eosinophils were purified by centrifugation in a discontinuous Percoll gradient harvesting eosinophils at the interface of 60% and 55% Percoll layers.22 This yields a 95% pure population of eosinophils. Eosinophils were suspended in RPMI 1640 supplemented with 10% bovine calf serum (Hyclone, Logan, UT) and 5 × 10−5 mol/L 2-βME under sterile conditions. Viability was greater than 95% as measured by Trypan blue exclusion.
Reverse transcription polymerase chain reaction.Oligonucleotides for polymerase chain reaction (PCR) were synthesized by standard procedures. Poly(A) mRNA was prepared using the Micro-Fast Track kit (Invitrogen, San Diego, CA) and was reverse-transcribed according to the manufacturer's protocol. The 40-μL reaction mixture contained enzyme buffer, 40 U/μL RNasin, 0.5 mmol/L dNTP, 0.5 μg oligo-dT/18mer, and 400 U M-MLV reverse transcriptase. The reaction mixture was incubated for 1 hour at 42°C. After incubation, the mixture was diluted to 250 μL with DEPC-water. Then, the reverse-transcribed products were amplified with Taq polymerase (Boehringer Mannheim Corp, Indianapolis, IN). The 50 μL PCR reaction mixture consisted of PCR buffer (10 mmol/L Tris HCl, pH 8.3, 100 mmol/L KCl, and 1.5 mmol/L MgCl2 ), 1.25 mmol/L of each dNTP, 10 μL of the reverse-transcribed product, 1.25 U DNA polymerase, and 150 nmol/L of each primer. Thirty cycles of PCR were performed using a DNA thermal cycler (PTC-100; M.J. Research Inc, Watertown, MA). Each cycle consisted of 1 minute of denaturation at 94°C, 1 minute of annealing at 60°C, and 1 minute of extension at 72°C. After the final cycle, the temperature was held at 72°C for 8 minutes to allow reannealing of the amplified products. The sequence of the primers and the size of the PCR product are presented in Table 1.
List of the Primers
| Receptor . | Sequence (5′-3′ ) . | Reference . | PCR . | Product . |
|---|---|---|---|---|
| FcγRI | ||||
| Forward | CTG CAG GAG TGT CCA TCA CGG TGA AAG A | 350 bp | ||
| Reverse | GGA TGT GAA ACC AGA CAG GAG CTG ATG A | |||
| FcγRII | ||||
| Forward | GCT GGA GGA ACA AAC TAC TGA ACA G | 23 | β1 | 477 bp |
| Reverse | GCA GCT TCT TCC AGA TCA GGA GGA | β2 | 339 bp | |
| FcγRIII | ||||
| Forward | AGT CAC AGT GGG GAC TAC TAC TGC A | 23 | 268 bp | |
| Reverse | CAC TTG TCT TGA GGA GCC TGG TGC T | |||
| γ-Chain | ||||
| Forward | CTC AGC CGT GAT CTT GTT CTT GC | 23 | 238 bp | |
| Reverse | GCT TCA GAG TCT CAT ATG TCT CC | |||
| β-Actin | ||||
| Forward | ATG GAT GAC GAT ATC GCT | 24 | 487 bp | |
| Reverse | ATG AGG TAG TCT GTC AGG |
| Receptor . | Sequence (5′-3′ ) . | Reference . | PCR . | Product . |
|---|---|---|---|---|
| FcγRI | ||||
| Forward | CTG CAG GAG TGT CCA TCA CGG TGA AAG A | 350 bp | ||
| Reverse | GGA TGT GAA ACC AGA CAG GAG CTG ATG A | |||
| FcγRII | ||||
| Forward | GCT GGA GGA ACA AAC TAC TGA ACA G | 23 | β1 | 477 bp |
| Reverse | GCA GCT TCT TCC AGA TCA GGA GGA | β2 | 339 bp | |
| FcγRIII | ||||
| Forward | AGT CAC AGT GGG GAC TAC TAC TGC A | 23 | 268 bp | |
| Reverse | CAC TTG TCT TGA GGA GCC TGG TGC T | |||
| γ-Chain | ||||
| Forward | CTC AGC CGT GAT CTT GTT CTT GC | 23 | 238 bp | |
| Reverse | GCT TCA GAG TCT CAT ATG TCT CC | |||
| β-Actin | ||||
| Forward | ATG GAT GAC GAT ATC GCT | 24 | 487 bp | |
| Reverse | ATG AGG TAG TCT GTC AGG |
RESULTS
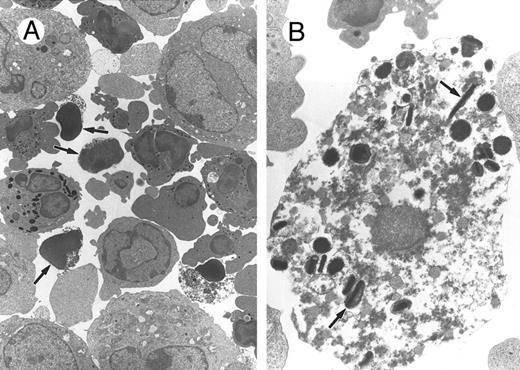
Effect of 2.4G2 on the viability of BM cell cultures.BM cells from normal Balb/c mice were cultured in the presence of IL-3, IL-5, and GM-CSF to induce eosinophil differentiation.15 Addition of 2.4G2, the anti-FcγRII/RIII MoAb, modestly but reproducibly reduced the viability of these cultures. At 48 hours, cell viability of the cultures treated with 25 μg 2.4G2 was 77% ± 9% (P < .0001, n = 20) compared with cultures to which 2.4G2 was not added (94% ± 2%). Different doses of 2.4G2 were analyzed (0.1 to 25 μg/mL), with the highest effect observed at doses of 5 to 25 μg/mL. Addition of irrelevant control rat IgG did not decrease viability of the cultures. The morphology of BM cells cultured with and without 2.4G2 was evaluated by Giemsa-Wright–stained smears and by electron microscopy. Many of the granulocytes in 2.4G2-containing cultures showed morphologic alterations characteristic of apoptosis (cytoplasmic shrinkage, membrane blebbing, chromatin condensation, and nuclear and cellular fragmentation; Fig 1A, arrows). Apoptotic alterations were not seen in other cells, suggesting that the apoptotic effects of 2.4G2 on cultured BM cells were cell lineage–restricted. Figure 1B shows a higher magnification of one apoptotic cell that still contains characteristic eosinophilic granules (arrows).
Morphologic analysis of bone marrow cells treated with the MoAb anti-FcγR, 2.4G2. BM (2 × 105 cells/mL) was cultured for 24 hours with rGM-CSF (10 U/mL), rIL-3 (10 U/mL), and rIL-5 (20 U/mL) with 2.4G2 (25 μg/mL), and the morphology of the cells was analyzed by transmission electron microscopy. (A) Representative example of cells cultured in the presence of 2.4G2. Arrows indicate cells with apoptotic features. (B) Higher magnification of 1 apoptotic cell where the characteristic eosinophilic granules are still intact (arrows).
Morphologic analysis of bone marrow cells treated with the MoAb anti-FcγR, 2.4G2. BM (2 × 105 cells/mL) was cultured for 24 hours with rGM-CSF (10 U/mL), rIL-3 (10 U/mL), and rIL-5 (20 U/mL) with 2.4G2 (25 μg/mL), and the morphology of the cells was analyzed by transmission electron microscopy. (A) Representative example of cells cultured in the presence of 2.4G2. Arrows indicate cells with apoptotic features. (B) Higher magnification of 1 apoptotic cell where the characteristic eosinophilic granules are still intact (arrows).
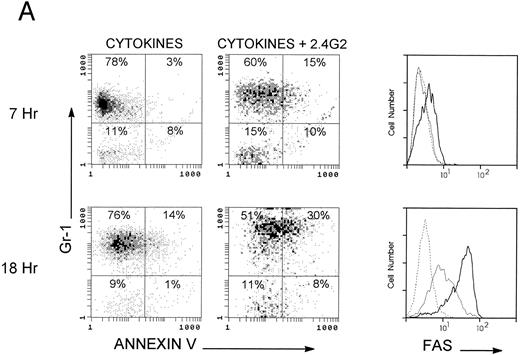
2.4G2 induces apoptosis in granulocytes but not in lymphocytes.To further analyze which populations were undergoing apoptosis, cytofluorimetric studies using Annexin-V and anti-fas antibody both labeled with FITC were performed on different BM populations. Anti–Gr-1 (PE-labeled) and anti-IgM (Cy5-labeled) were used as markers of granulocytes and B lymphocytes, respectively. PI was used to exclude dead cells. Figure 2A shows results obtained with the granulocyte population. Seven hours after incubation with 2.4G2, Annexin-V bound to granulocytes (upper panels), and this reached a maximum level of binding by 18 to 24 hours of incubation (lower panels, n = 11). Figure 2A shows expression of fas in the granulocyte gate on cells cultured with and without 2.4G2. Upon harvesting from normal donors, fas expression was detected on 12% ± 4% (n = 5) of granulocytes. After incubation with cytokines alone for 18 hours, 53% ± 7% (n = 5) of the granulocytes expressed fas, whereas cells incubated with 2.4G2 showed fas expression on 80% ± 6% (P < .0005, n = 5) of the granulocytes. Moreover, the level of fas expression on granulocytes cultured with 2.4G2 was higher (MFI, 20 ± 2.5 v 11 ± 1; Table 2) in the absence of 2.4G2. Figure 2A shows a representative experiment from five with similar results. At the same time, we studied the effect of 2.4G2 on cells in the lymphoid gate. Figure 2B shows a representative experiment with essentially no detectable binding of Annexin-V on cytokine-cultured IgM+ and IgM− lymphocytes at 7 and 18 hours. Likewise, fas expression did not change on IgM+ or IgM− lymphocyte populations cultured with cytokines, and the presence of 2.4G2 had no effect. Annexin-V binding and fas expression in the lymphoid gate did not change at 24 and 48 hours of culture with or without addition of 2.4G2 (n = 9; data not shown). The same results were found using Thy-1 as a T-cell lymphocyte marker (data not shown). All of these experiments demonstrate that after addition of 2.4G2, only cells in the granulocyte population were driven to undergo apoptosis. In separate experiments, purified mature eosinophils (95% pure) from liver granulomas of S mansoni–infected CBA mice were cultured with IL-3, IL-5, and GM-CSF for 20 hours, at which time the basal level of binding to Annexin-V or fas antibody was 17.5% ± 10% and 5.2% ± 4% (n = 3), respectively. However, when 2.4G2 was present in the cultures over the 20-hour incubation, enhanced binding of Annexin-V and anti-fas antibody was observed (42.5% ± 10% and 18% ± 5%, n = 3), demonstrating that the induction of apoptosis by 2.4G2 is not restricted to granulocyte BM precursors, but also occurs in mature eosinophils from a site of active inflammation.
Cytofluorimetric analysis of fas and Annexin-V on different bone marrow populations. Bone marrow cells (2 × 105/mL) were cultured 7 to 18 hours with rGM-CSF, rIL-3, and rIL-5 in the presence of 2.4G2 (25 μg/mL). Cells were stained with Gr-1 (PE) as a marker of the granulocyte population and with IgM (Cy5) as a marker of the lymphocyte population; Annexin-V (FITC) or fas (FITC) were added, and PI-positive cells were excluded in all experiments. A granulocyte gate and a lymphocyte gate were made based on the orthogonal and forward scatter. Fas expression was studied by comparing isotype-matched antibody (broken line), BM cells cultured in the presence of cytokines (thin line), and cells cultured with cytokines plus 2.4G2 (thick line). (A) Apoptotic analysis of the granulocytic cells. (B) Apoptotic analysis on the lymphocyte gate. The figure shows a representative experiment from 5 with similar results.
Cytofluorimetric analysis of fas and Annexin-V on different bone marrow populations. Bone marrow cells (2 × 105/mL) were cultured 7 to 18 hours with rGM-CSF, rIL-3, and rIL-5 in the presence of 2.4G2 (25 μg/mL). Cells were stained with Gr-1 (PE) as a marker of the granulocyte population and with IgM (Cy5) as a marker of the lymphocyte population; Annexin-V (FITC) or fas (FITC) were added, and PI-positive cells were excluded in all experiments. A granulocyte gate and a lymphocyte gate were made based on the orthogonal and forward scatter. Fas expression was studied by comparing isotype-matched antibody (broken line), BM cells cultured in the presence of cytokines (thin line), and cells cultured with cytokines plus 2.4G2 (thick line). (A) Apoptotic analysis of the granulocytic cells. (B) Apoptotic analysis on the lymphocyte gate. The figure shows a representative experiment from 5 with similar results.
Induction of Annexin-V Binding and Fas Surface Expression on Granulocytes From Balb/c KO CD16, MRL-lpr/lpr, and C3H/HEJ-gld/gld Mice at 24 Hours of Culture
| Stimuli . | MFI . | |
|---|---|---|
| . | Annexin-V . | Fas . |
| Balb/c | ||
| Cytokines | 11 ± 1 | 8.2 ± 1 |
| +2.4G2 | 20 ± 2.5 | 18.2 ± 4 |
| KO CD16 | ||
| Cytokines | 11.5 ± 3 | 14 ± 4 |
| +2.4G2 | 20 ± 4 | 23 ± 5 |
| MRL-lpr/lpr | ||
| Cytokines | 10.3 ± 1 | <3 |
| +2.4G2 | 9.7 ± 1 | <3 |
| HEJ-gld/gld | ||
| Cytokines | 5 ± 2 | 9 ± 2 |
| +2.4G2 | 8 ± 1 | 12.5 ± 1 |
| Stimuli . | MFI . | |
|---|---|---|
| . | Annexin-V . | Fas . |
| Balb/c | ||
| Cytokines | 11 ± 1 | 8.2 ± 1 |
| +2.4G2 | 20 ± 2.5 | 18.2 ± 4 |
| KO CD16 | ||
| Cytokines | 11.5 ± 3 | 14 ± 4 |
| +2.4G2 | 20 ± 4 | 23 ± 5 |
| MRL-lpr/lpr | ||
| Cytokines | 10.3 ± 1 | <3 |
| +2.4G2 | 9.7 ± 1 | <3 |
| HEJ-gld/gld | ||
| Cytokines | 5 ± 2 | 9 ± 2 |
| +2.4G2 | 8 ± 1 | 12.5 ± 1 |
Specificity of the apoptotic effect of 2.4G2.To determine the specificity of the effect of 2.4G2, we selected Mac-1(CD11b/CD18), another rat MoAb directed to a cell surface receptor also present on 80% to 90% of granulocytes at the beginning of the culture period. Bone marrow cultures were treated for 24 hours with the combination of the cytokines cited plus addition of 2.4G2, anti–Mac-1, or, as an isotype-matched control, MoAb rat IgG2b. A granulocyte gate was established, and apoptotic analysis was made on Gr-1+ cells. Calculations were made by comparing results from the cells cultured with cytokines alone versus those cultured with cytokines and one of the other MoAbs. Results presented in Table 3 demonstrate that enhanced Annexin-V binding and induction of fas were present when 2.4G2 was added to the cultures, but were not present on granulocytes cultured with anti–Mac-1 or rat IgG2b. The lower part of Table 3 shows the dose dependency of the effect of 2.4G2 on fas induction and on Annexin-V binding by granulocytes. We did not detect any apoptotic effect at doses less than 1 μg/mL.
Induction of Apoptosis on the Granulocyte Population From BM Cultures by Different Stimuli
| Stimuli . | % Annexin-V . | % Fas . |
|---|---|---|
| Cytokines | 13 ± 2 | 6 ± 2 |
| + Mac-13-151 | 8 ± 1 | 4 ± 2 |
| + Rat IgG2b3-151 | 15 ± 2 | 8 ± 2 |
| + 2.4G2 | ||
| 1 μg/mL | 15 ± 4 | 5 ± 2 |
| 5 μg/mL | 17 ± 5 | 17 ± 33-150 |
| 25 μg/mL | 28 ± 83-150 | 36 ± 103-150 |
| 50 μg/mL | 26 ± 63-150 | 40 ± 83-150 |
| Stimuli . | % Annexin-V . | % Fas . |
|---|---|---|
| Cytokines | 13 ± 2 | 6 ± 2 |
| + Mac-13-151 | 8 ± 1 | 4 ± 2 |
| + Rat IgG2b3-151 | 15 ± 2 | 8 ± 2 |
| + 2.4G2 | ||
| 1 μg/mL | 15 ± 4 | 5 ± 2 |
| 5 μg/mL | 17 ± 5 | 17 ± 33-150 |
| 25 μg/mL | 28 ± 83-150 | 36 ± 103-150 |
| 50 μg/mL | 26 ± 63-150 | 40 ± 83-150 |
P < .05.
Ab was used at a dose of 25 μg/mL.
Effect of 2.4G2 on the growth and differentiation of granulocytes in cultures of BM from CD16/FcγRIII knockout mice.Because 2.4G2 does not distinguish between FcγRII (CD32) and FcγRIII (CD16), a series of experiments were conducted using CD16 knockout (KO) mice to determine the relative contribution of each receptor to the triggering of apoptosis. CD16 KO mice lack the α-subunit of the FcγRIII, but the γ-subunit is expressed normally in these mice.20 Following the same protocol to promote eosinophilic differentiation, BM cells were cultured with IL-3, IL-5, and GM-CSF to determine whether CD16, which is expressed very early in ontogeny, plays any role in the apoptosis of granulocytes caused by addition of 2.4G2. Table 2 shows the results obtained in a series of experiments in which normal Balb/c or C57BL/6-KO CD16 BM cells were cultured in parallel with or without 2.4G2. In the presence of 2.4G2, granulocytes from normal Balb/c and from KO CD16 mice showed the same magnitude of increase in Annexin-V binding and the same magnitude of increased expression of fas. In KO CD16 granulocytes, there is a higher background level of fas expression, but the incremental increase in the cells cultured with 2.4G2 was the same as in the normal Balb/c that express both CD16 and CD32. These findings show that in the absence of CD16, ligation of CD32 by 2.4G2 alone triggers the apoptotic mechanism that occurs in BM granulocytes from normal mice.
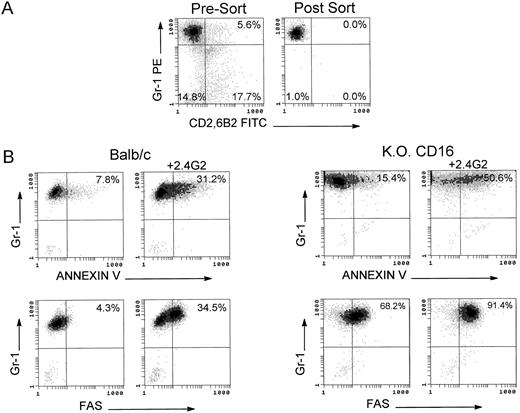
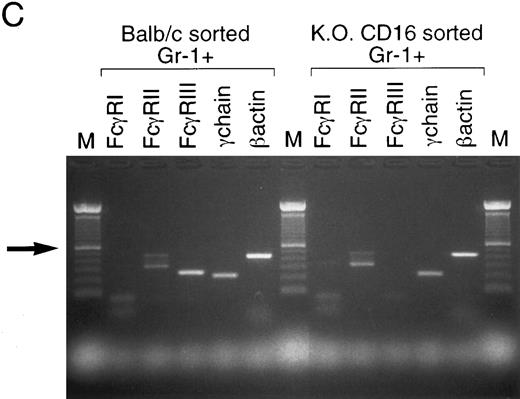
2.4G2 induces apoptosis by direct binding to FcγR on the granulocytic population.To address the question of whether 2.4G2 interacts directly with FcγR in the granulocytic population in the BM cultures, or, alternatively, the apoptotic mechanism is an indirect effect mediated by a cytokine-driven mechanism affecting only the granulocytes, sorting experiments were performed with BM granulocytes from Balb/c and KO CD16 mice. Using a granulocytic gate based on forward and orthogonal scatter, Gr-1 (PE)-positive/CD2 (FITC)-6B2 (FITC)–negative cells were sorted with 99% purity. Sorted Gr-1+ cells were incubated for 24 hours with the combination of cytokines with or without 2.4G2. Figure 3B shows that Gr-1+ cells undergo apoptosis as evidenced by the binding of Annexin-V and the induction of fas. Furthermore, the absence of CD16 on sorted Gr-1+ cells did not decrease the magnitude of the apoptosis detected on these cells compared with Gr-1+ cells from normal Balb/c. RT-PCR analyses of FcγR were performed on sorted Gr-1+ cells (Fig 3C) demonstrating that both Balb/c and C57BL/6-KO CD16 cells expressed FcγRII β1 and β2, thus making each of the FcγRII isoforms candidates for the receptor responsible for initiation of the apoptosis.
Apoptotic mechanism on sorted Gr-1+ cells from bone marrow cultures of Balb/c and C57BL/6-KO CD16 mice. (A) Fresh BM cells were stained with Gr-1(PE), CD2(FITC), and 6B2(FITC). A granulocyte gate was used based on the orthogonal and forward scatter to perform the sorting. Gr-1+ CD2−, 6B2− cells were collected postsorting. (B) Apoptotic analysis of sorted Gr-1+ cells on Balb/c and C57BL/6-KO CD16 mice. Sorted cells (2 × 105/mL) were cultured for 24 hours with rGM-CSF, rIL-3, and rIL-5 in the presence of 2.4G2 (25 μg/mL). Annexin-V and fas expression were analyzed. The cursor was placed to compare the cultures with cytokines alone or in combination with 2.4G2. (C) RT-PCR analyses of FcγR were performed on sorted Gr-1+ population (1 × 106 cells). Arrow shows 600-bp standard in the 100-bp ladder. FcγRIIβ1 and β2 transcripts correspond to the bands at 477 and 339 bp, respectively.
Apoptotic mechanism on sorted Gr-1+ cells from bone marrow cultures of Balb/c and C57BL/6-KO CD16 mice. (A) Fresh BM cells were stained with Gr-1(PE), CD2(FITC), and 6B2(FITC). A granulocyte gate was used based on the orthogonal and forward scatter to perform the sorting. Gr-1+ CD2−, 6B2− cells were collected postsorting. (B) Apoptotic analysis of sorted Gr-1+ cells on Balb/c and C57BL/6-KO CD16 mice. Sorted cells (2 × 105/mL) were cultured for 24 hours with rGM-CSF, rIL-3, and rIL-5 in the presence of 2.4G2 (25 μg/mL). Annexin-V and fas expression were analyzed. The cursor was placed to compare the cultures with cytokines alone or in combination with 2.4G2. (C) RT-PCR analyses of FcγR were performed on sorted Gr-1+ population (1 × 106 cells). Arrow shows 600-bp standard in the 100-bp ladder. FcγRIIβ1 and β2 transcripts correspond to the bands at 477 and 339 bp, respectively.
Granulocyte apoptosis triggered by ligation of FcγRII (CD32) is dependent on fas and fas ligand.Granulocytes are known to express fas on their surface without undergoing apoptosis.25 26 To test the hypothesis that the induction of fas surface expression on granulocytes caused by addition of 2.4G2 resulted in fas-mediated apoptosis and consequent granulocyte death, fas-defective mice (MRL-lpr/lpr) were used in some experiments. Table 2 summarizes the findings obtained in these experiments. First, the viability of cultured BM cells treated with cytokines alone or with cytokines plus 2.4G2 remained unchanged from 24 hours to 6 days (91% ± 2%, n = 3). Furthermore, no significant Annexin-V binding was detected in the granulocyte population, an observation consistent with the sustained viability of these cells in the presence of 2.4G2. Similar experiments were made with fas ligand–defective mice, C3H/HEJ-gld/gld. The data in Table 2 show that in the absence of the fas counterreceptor, fas ligand, the granulocyte population does not bind Annexin-V, even though an induction of fas expression is detected when 2.4G2 is added to the cultures. Although the intensity of fas expression on cells is not significantly greater in the presence of 2.4G2 (9 ± 2 v 12.5 ± 1), in the presence of 2.4G2 there was almost a threefold increase in the percentage of cells that express fas (7% ± 4% v 23% ± 4% n = 3). Furthermore, the viability of these cultures remained unchanged (91% ± 3%, n = 3). All of these data lead to the conclusion that the strong induction of fas on the granulocyte population caused by addition of 2.4G2 requires interaction with fas ligand for apoptosis to occur.
DISCUSSION
The major new finding in the present report is that ligation of FcγRII/CD32 on developing and mature murine eosinophils induces their destruction through apoptosis. This conclusion is based on several types of observations: (1) Murine BM cultured with GM-CSF, IL-3, and IL-5 produces large numbers of eosinophils, but the presence of anti-FcγR MoAb in the cultures totally blocks production of eosinophils; (2) Electron microscopy of bone marrow cultures showed the characteristic morphologic changes of apoptosis in eosinophilic granulocytes; (3) Cytofluorimetric analysis of bone marrow cells demonstrated that only the granulocytic population bound Annexin-V; (4) Cultures containing anti-FcγR Ab showed a significant induction of fas on Gr-1+ cells; and (5) Experiments with mature purified eosinophils from S mansoni hepatic granulomas demonstrated that FcγRII/RIII-mediated apoptosis also occurs with these mature inflammatory eosinophils. These findings might have implications for the mechanisms by which eosinophils are cleared from sites of inflammation in vivo, an issue about which there is currently no information. Previous in vitro studies27-31 showed that ligation of FcγR on eosinophils or neutrophils resulted in the release of inflammatory mediators. Furthermore, purified eosinophils from S mansoni hepatic granulomas can release O−2 by ligation of FcγR using opsonized zymosan and 2.4G2.19 Conceivably, immune complexes in vivo might cross-link CD32 on eosinophils, resulting in mediator release and elimination of the mature eosinophil by apoptosis even in the presence of GM-CSF, IL-3, and IL-5, factors that would continue to promote development and recruitment of new eosinophils to the inflammatory site. GM-CSF, IL-3, and IL-5 can prolong survival and activate mature eosinophils.15-18,32-36 It has been shown that mature eosinophils produce and release a number of cytokines, including GM-CSF and IL-5,37,38 and this could provide an autocrine circuit at the inflammatory site. Conceivably, CD32 could modulate this process, since it has been shown in lymphocytes and mast cells that CD32 contains an inhibitory motif that can block certain phosphotyrosine-mediated activation signals.12 The present finding that CD32 can induce apoptosis identifies a mechanism that could override the autocrine circuit in the eosinophil. Recent data from our laboratory confirm the absence of FcεRII/CD23 on eosinophils derived from BM cultures19 even in the presence of IL-5 and GM-CSF, which can upregulate CD23 in eosinophilic lineage cells,39 ruling out any possible contribution of CD23 in the apoptotic effect in murine eosinophils. While it is clear in our studies that FcγR-mediated granulocyte apoptosis occurred even in the absence of CD16, it is interesting that Azzoni et al40 showed that in human NK cells direct activation of CD16 induces a c-myc–dependent apoptotic mechanism.
Interaction between fas and fas ligand on the cell surface initiates a signal transduction pathway leading to apoptosis in T cells.41,42 Expression of fas and fas ligand on granulocytes30 31 strongly suggests that this interaction may be critical in the life/death decision in granulocytes. The induction of apoptosis in our study was dependent on fas and fas ligand expression, as shown by the failure to induce granulocyte apoptosis in the fas mutant murine strain (MRL-lpr/lpr). The requirement for participation of the fas ligand was shown by the absence of apoptosis even in the presence of induction of fas expression on Gr-1+ cells from the fas ligand mutant mouse strain C3H/HEJ-gld/gld.
Since the MoAb 2.4G2 does not distinguish between CD16 and CD32, the studies in CD16 KO mice were particularly informative because they clearly showed that FcγR-triggered apoptosis could be induced in the absence of CD16. Because both β1 and β2 isoforms of CD32 are expressed on the Gr-1+ population of normal and CD16 KO BM cells, it is not yet determined whether the induction of apoptosis in granulocytes by ligation of CD32 is dependent on the β1, β2, or both forms of the receptor. By sorting Gr-1+ cells, we demonstrated that the apoptosis caused by 2.4G2 was due to direct binding to FcγR expressed on Gr-1+ cells, ruling out an indirect effect mediated by Gr-1− cells in the BM cultures. Our findings identify a previously unknown function of FcγR on eosinophils. In this regard, it is interesting that FcγR on T-lineage cells also have functions not previously appreciated. Early in lymphocyte development, CD16 and CD32 are coexpressed on T-lineage precursors, but are downregulated in thymocytes with the onset of productive rearrangement of TCR genes.13 Before the rearrangement of antigen receptor genes in T-lymphoid cells, FcγR appear to modulate lymphoid cell maturation, possibly through interaction with alternative non-Ig ligands expressed on stromal cells. Thus, in addition to their classic effector functions, FcγR on lymphoid and myeloid cells participate in mechanisms that influence the growth, development, and death of these cell populations.
At the present time, it is not possible to extrapolate precisely to humans these findings in the mouse. Murine CD32 is encoded by a single gene and two isoforms are generated, β1 and β2, both containing the ITIM sequence. In humans, CD32 is encoded by three genes and two (FcγRIIA and FcγRIIC) contain ITAM sequences, while FcγRIIB contains an ITIM sequence. Additional information about the amounts of the isoforms and the functions of both CD32 and CD16 in human eosinophils is needed before it will be feasible to relate our findings in mouse eosinophils to the situation in humans.
In summary, these studies identify a new role for FcγR on granulocytes. In addition to their participation in granulocyte effector functions such as phagocytosis and degranulation reactions, it is now clear that FcγR are linked to apoptotic pathways in granulocytes. We propose that after eosinophils are degranulated, ligation of CD32 by IgG-Ag complexes triggers an apoptotic mechanism via fas-fas ligand that results in elimination of these cells, perhaps by the macrophages present in high numbers at the inflammatory site.
ACKNOWLEDGMENT
We are grateful to J. Fishbaugh and G. Hess for expert technical assistance in the flow cytometric studies, and to C. Bray for assistance with electron microscopy.
Address reprint requests to Richard G. Lynch, MD, Department of Pathology, College of Medicine, MRC375, University of Iowa, Iowa City, IA 52246.


This feature is available to Subscribers Only
Sign In or Create an Account Close Modal