Key Points
Fixed-duration epcoritamab plus R2 showed deep, durable responses with favorable long-term outcomes in R/R FL regardless of risk features.
Epcoritamab plus R2 demonstrated a manageable safety profile with mostly low-grade CRS events.
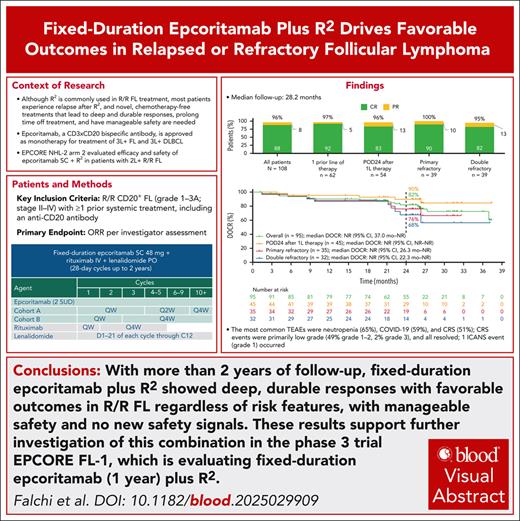
Visual Abstract
Epcoritamab is a subcutaneous CD3×CD20 bispecific antibody approved as monotherapy for relapsed/refractory (R/R) follicular lymphoma (FL). We evaluated fixed-duration epcoritamab with rituximab plus lenalidomide (R2) in R/R FL in arm 2 of EPCORE NHL-2 (phase 1b/2). Patients received epcoritamab (2 step-up doses, then 48-mg full doses) for up to 2 years, and R2 for up to 12 cycles (28 days per cycle). The primary end point was overall response rate (ORR) per investigator assessment (Lugano criteria). As of 21 September 2024, 108 patients received ≥1 epcoritamab dose in expansion (median follow-up, 28.2 months). Median age was 65 years; 57% had 1 previous line of therapy. ORR and complete response (CR) rate were 96% and 88%, respectively; CR rates in patients with high-risk features were 90% (primary refractory), 82% (refractory to anti-CD20 and an alkylating agent), and 83% (disease progression within 24 months of first-line therapy). Two-year estimates for remaining in CR, progression-free survival, overall survival, and not starting next antilymphoma therapy were 82%, 76%, 90%, and 84%, respectively. Minimal residual disease negativity was observed in 86% of evaluable patients (clonoSEQ assay). Common treatment-emergent adverse events (TEAEs) included neutropenia (65%), COVID-19 (59%), and cytokine release syndrome (CRS; 51%). Grade ≥3 TEAEs occurred in 87% of patients; 5 had grade 5 TEAEs (all COVID-19). CRS events were mostly low grade (grade 1, 38%; grade 2, 11%; grade 3, 2%), all resolved, and none led to epcoritamab discontinuation. Fixed-duration epcoritamab plus R2 demonstrated deep, durable responses with manageable safety and favorable outcomes in R/R FL, irrespective of risk features. This trial was registered at www.ClinicalTrials.gov as #NCT04663347.
Introduction
Follicular lymphoma (FL) is the most common subtype of indolent non-Hodgkin lymphoma and is incurable in most patients.1-4 The natural history of this neoplasm is associated with continuous relapses and shorter remission duration with increasing lines of treatment, with the disease eventually becoming refractory to approved treatment.1-3 There is no definitive standard of care for patients with relapsed/refractory (R/R) FL,2,4 and there exists a clear need for improvement of outcomes.
Rituximab plus lenalidomide (R2) is a commonly used treatment approach approved in the United States and Europe for patients with R/R FL.4,5 In preclinical studies, lenalidomide has been shown to repair FL-induced T-cell dysfunction,6 and rituximab has been shown to promote antibody-dependent cellular phagocytosis and natural killer cell–mediated antibody-dependent cellular cytotoxicity against tumor cells.7 In the phase 3 AUGMENT trial, R2 resulted in an overall response rate (ORR) of 78% and complete response (CR) rate of 34% in patients with R/R FL or marginal zone lymphoma not previously refractory to rituximab; estimated 2-year progression-free survival (PFS) rate was 58%.8 Although R2 alone is an active treatment for patients with R/R FL, novel therapies that may provide deeper and more durable responses for a broader population of patients with FL are needed. In particular, patients with high-risk features, including disease progression within 24 months (POD24) of first-line (1L) therapy, disease refractory to 1L antilymphoma therapy (primary refractory disease), and disease refractory to both anti-CD20 and an alkylating agent (double-refractory disease), have a high unmet need.1,9,10
Epcoritamab, a subcutaneously administered CD3×CD20 bispecific antibody, has been approved as monotherapy for R/R FL and different types of R/R large B-cell lymphoma after ≥2 lines of systemic therapy.11-13 The approval for R/R FL is primarily based on results from the FL dose-expansion cohort of the phase 1/2 EPCORE NHL-1 trial (ClinicalTrials.gov identifier: NCT03625037), in which treatment with single-agent epcoritamab until progression or unacceptable toxicity led to a clinically meaningful ORR of 82% and CR rate of 63% with manageable safety in patients with heavily pretreated FL (median previous lines of treatment [pLOT], 3 [interquartile range, 2-4]).11,12,14 Deep responses were observed even in patients with historically difficult-to-treat FL, such as POD24 after 1L therapy (CR rate, 64%) and double-refractory disease (CR rate, 56%).14 Epcoritamab monotherapy was approved for R/R FL with no mandatory hospitalization requirements based on results demonstrating that it can be safely and effectively administered in an outpatient setting with a 3-step step-up dosing (SUD) regimen in cycle 1.11-14
To improve outcomes and convenience in a broader population of patients with FL, and based on each agent’s proven clinical benefit and their mechanisms of action, which appear synergistic preclinically,15 we evaluated fixed-duration epcoritamab, given for up to 2 years, plus R2 in patients with R/R FL after ≥1 line of systemic therapy in arm 2 of the EPCORE NHL-2 trial (NCT04663347).
Methods
Study design and patients
EPCORE NHL-2 arm 2 is a phase 1b/2, open-label, multicenter, global trial that enrolled patients with R/R CD20+ FL (per 2016 World Health Organization classification)16 who had received at least 1 previous systemic antineoplastic agent (GCT3013-02; ClinicalTrials.gov identifier: NCT04663347). Eligible patients had Ann Arbor stage II to IV disease, an Eastern Cooperative Oncology Group performance status of 0 to 2, acceptable organ function, measurable disease, and a need for treatment based on symptoms and/or disease burden per Groupe d’Etude des Lymphomes Folliculaires criteria.17 Patients were excluded from the study if they had FL grade 3B, evidence of transformation to an aggressive lymphoma, contraindication to rituximab, or previous treatment with a bispecific antibody targeting CD3 or CD20. Patients with known active infections, including COVID-19, were not eligible to enroll in EPCORE NHL-2. However, the study protocol did not require treatment discontinuation if patients acquired COVID-19 during the trial.
Eligible patients received subcutaneous epcoritamab in 28-day cycles. Epcoritamab was administered with a 2-step (0.16 mg/0.8 mg) SUD regimen in cycle 1, followed by 48-mg full doses. In cohort 2a, epcoritamab was administered weekly in cycles 1 to 3, every 2 weeks in cycles 4 through 9, and every 4 weeks thereafter for up to 2 years. In cohort 2b, patients received epcoritamab weekly for cycles 1 and 2, then every 4 weeks in cycle 3 and beyond, for up to 2 years. Rituximab 375 mg/m2 was administered IV weekly in cycle 1 and every 4 weeks in cycles 2 through 5. Lenalidomide was administered at a starting dose of 20 mg orally daily for 21 of 28 days for up to 12 cycles.
Cytokine release syndrome (CRS) prophylaxis in cycle 1 consisted of 50 mg IV/oral diphenhydramine and 650 to 1000 mg oral acetaminophen/paracetamol on day 1, 8, 15, and 22, and 15 mg IV dexamethasone 30 to 120 minutes before epcoritamab administration on day 1 through 4, day 8 through 11, day 15 through 18, and day 22 through 25. This regimen was developed before the currently approved optimized cycle 1 3-step SUD regimen.11,14 Hospitalization for at least 24 hours was mandatory for the first full dose of epcoritamab (on cycle 1, day 15 and during repriming cycles as applicable).
Patients received prophylactic antithrombotic therapy according to previous medical history (supplemental Methods, available on the Blood website).
End points and assessments
The protocol-specified primary end point was ORR per Lugano criteria by investigator assessment.18 Key secondary efficacy end points included CR rate, duration of response (DOR), duration of CR (DOCR), time to response, time to next antilymphoma therapy, PFS, overall survival (OS), and rate of minimal residual disease (MRD) negativity. Additional secondary end points were related to safety (incidence and severity of adverse events [AEs]) and pharmacodynamics (biomarkers of efficacy in blood samples and within tumors).
Efficacy was assessed by positron emission tomography–computed tomography at screening, every 6 weeks for 24 weeks, every 12 weeks through 48 weeks, and then every 24 weeks until disease progression. Prespecified groups of interest, including by pLOT and high-risk disease features (POD24, primary refractory, double refractory, and high Follicular Lymphoma International Prognostic Index [3-5]), were analyzed. Definitions of prespecified groups of interest are provided in “Results” and supplemental Methods.
AEs were classified using Medical Dictionary for Regulatory Activities version 27.0, and severity was graded using National Cancer Institute Common Terminology Criteria for Adverse Events, version 5.0. The grouped term “COVID-19,” which included COVID-19, COVID-19 pneumonia, post–acute COVID-19 syndrome, and suspected COVID-19, was applied to capture all COVID-19–related terms. CRS and immune effector cell–associated neurotoxicity syndrome were graded using American Society for Transplantation and Cellular Therapy criteria,19 and clinical tumor lysis syndrome was graded using Cairo-Bishop criteria.20 The relationship between AEs and treatment was determined by the investigator. Serious AEs were defined per International Council for Harmonisation E2A guidelines.21
MRD analysis
MRD analysis was performed on genomic DNA isolated from longitudinal peripheral blood mononuclear cell samples collected from patients at prespecified time points (supplemental Methods) using the clonoSEQ next-generation sequencing assay (Adaptive Biotechnologies, Seattle, WA). Screening tumor biopsies were used to identify trackable tumor clones, with samples quantified as tumor clones detected per 106 nucleated cells.22
Statistical analysis
Data were analyzed using SAS software version 9.4 or higher (SAS Institute, Inc, Cary, NC). Genmab authors analyzed the data, and all authors had access to primary clinical trial data.
Additional methods
Further details are provided in supplemental Methods.
Ethics statement
Site-specific institutional review boards or institutional or central ethics committees approved the protocol before study initiation. The study was conducted in accordance with International Council for Harmonisation E6(R2) guidelines on good clinical practice and the principles of the Declaration of Helsinki. Before enrollment, all patients reviewed and signed informed consent forms.
Results
Patient characteristics and disposition
Between 20 May 2021 and 21 September 2024, 108 patients with R/R FL were treated with epcoritamab plus R2 and included in efficacy and safety analyses. The median age was 65 years, 61 patients (56%) had Follicular Lymphoma International Prognostic Index score of 3 to 5, and 65 (60%) had Ann Arbor stage IV disease (Table 1; dosing cohorts [cohort 2a, n = 24; cohort 2b, n = 84] are reported in supplemental Table 1). The median number of pLOT was 1 (range, 1-7), and 46 patients (43%) had ≥2 pLOT. All patients had previous anti-CD20 antibodies, and 101 (94%) had previous alkylating agents. One patient received previous R2 alone and 1 patient received previous R2 plus bendamustine. Fifty-four patients (50%) had POD24 after 1L therapy, which included 28 patients (26%) who received epcoritamab plus R2 in the second-line (2L) setting. Thirty-nine patients (36%) had primary refractory disease and 39 (36%) had double-refractory disease. Some patients were included in >1 high-risk subgroup.
Demographic and baseline clinical characteristics
| Characteristic . | Overall N = 108 . |
|---|---|
| Age, median (range), y | 65 (30-80) |
| Age group (y), n (%) | |
| <65 | 52 (48.1) |
| 65 to <75 | 44 (40.7) |
| ≥75 | 12 (11.1) |
| Sex at birth, n (%) | |
| Male | 55 (50.9) |
| Female | 53 (49.1) |
| Race,∗n (%) | |
| White | 78 (72.2) |
| Black or African American | 2 (1.9) |
| Asian | 2 (1.9) |
| Other | 2 (1.9) |
| Ethnicity,†n (%) | |
| Hispanic or Latino | 3 (2.8) |
| Not Hispanic or Latino | 23 (21.3) |
| Enrollment, n (%) | |
| Europe | 79 (73.1) |
| North America | 26 (24.1) |
| Australia | 3 (2.8) |
| ECOG performance status, n (%) | |
| 0 | 69 (63.9) |
| 1 | 36 (33.3) |
| 2 | 3 (2.8) |
| FLIPI, n (%) | |
| 0-1 | 14 (13.0) |
| 2 | 33 (30.6) |
| 3-5 | 61 (56.5) |
| GELF criteria‡ | 104 (96.3) |
| Ann Arbor stage, n (%) | |
| I or II | 19 (17.6) |
| III | 24 (22.2) |
| IV | 65 (60.2) |
| Bulky disease per investigator, n (%) | |
| ≥7 cm | 29 (26.9) |
| <7 cm | 79 (73.1) |
| LDH | |
| High | 34 (31.5) |
| Low | 1 (0.9) |
| Normal | 73 (67.6) |
| pLOT, n (%) | |
| 1 | 62 (57.4) |
| 2 | 28 (25.9) |
| 3 | 9 (8.3) |
| ≥4 | 9 (8.3) |
| Previous systemic treatment, n (%) | |
| Anti-CD20 | 108 (100) |
| Alkylating agents | 101 (93.5) |
| Anthracyclines | 70 (64.8) |
| Bendamustine | 42 (38.9) |
| Immunomodulatory drugs | 4 (3.7) |
| CAR T-cell therapy | 3 (2.8) |
| R2§ | 2 (1.9) |
| POD24 after 1L therapy, n (%) | 54 (50.0) |
| POD24 2L|| | 28 (25.9) |
| Primary refractory,¶,# n (%) | 39 (36.1) |
| Double refractory,¶,∗∗ n (%) | 39 (36.1) |
| Characteristic . | Overall N = 108 . |
|---|---|
| Age, median (range), y | 65 (30-80) |
| Age group (y), n (%) | |
| <65 | 52 (48.1) |
| 65 to <75 | 44 (40.7) |
| ≥75 | 12 (11.1) |
| Sex at birth, n (%) | |
| Male | 55 (50.9) |
| Female | 53 (49.1) |
| Race,∗n (%) | |
| White | 78 (72.2) |
| Black or African American | 2 (1.9) |
| Asian | 2 (1.9) |
| Other | 2 (1.9) |
| Ethnicity,†n (%) | |
| Hispanic or Latino | 3 (2.8) |
| Not Hispanic or Latino | 23 (21.3) |
| Enrollment, n (%) | |
| Europe | 79 (73.1) |
| North America | 26 (24.1) |
| Australia | 3 (2.8) |
| ECOG performance status, n (%) | |
| 0 | 69 (63.9) |
| 1 | 36 (33.3) |
| 2 | 3 (2.8) |
| FLIPI, n (%) | |
| 0-1 | 14 (13.0) |
| 2 | 33 (30.6) |
| 3-5 | 61 (56.5) |
| GELF criteria‡ | 104 (96.3) |
| Ann Arbor stage, n (%) | |
| I or II | 19 (17.6) |
| III | 24 (22.2) |
| IV | 65 (60.2) |
| Bulky disease per investigator, n (%) | |
| ≥7 cm | 29 (26.9) |
| <7 cm | 79 (73.1) |
| LDH | |
| High | 34 (31.5) |
| Low | 1 (0.9) |
| Normal | 73 (67.6) |
| pLOT, n (%) | |
| 1 | 62 (57.4) |
| 2 | 28 (25.9) |
| 3 | 9 (8.3) |
| ≥4 | 9 (8.3) |
| Previous systemic treatment, n (%) | |
| Anti-CD20 | 108 (100) |
| Alkylating agents | 101 (93.5) |
| Anthracyclines | 70 (64.8) |
| Bendamustine | 42 (38.9) |
| Immunomodulatory drugs | 4 (3.7) |
| CAR T-cell therapy | 3 (2.8) |
| R2§ | 2 (1.9) |
| POD24 after 1L therapy, n (%) | 54 (50.0) |
| POD24 2L|| | 28 (25.9) |
| Primary refractory,¶,# n (%) | 39 (36.1) |
| Double refractory,¶,∗∗ n (%) | 39 (36.1) |
Baseline characteristics were similar between patients who received different dosing regimens (supplemental Table 1).
ECOG, Eastern Cooperative Oncology Group; FLIPI, Follicular Lymphoma International Prognostic Index; GELF, Groupe d’Etude des Lymphomes Folliculaires; LDH, lactate dehydrogenase.
Race category was not reported for 24 patients (22.2%).
Ethnicity was missing for 82 patients (75.9%).
GELF criteria include: (1) 3 nodes in 3 distinct areas, with each node ≥3 cm in diameter; (2) a tumor ≥7 cm in diameter; (3) presence of systemic symptoms; (4) symptomatic spleen enlargement; (5) serous effusion; (6) cytopenias; (7) leukemia; and (8) local risk of compression (epidural, ureteral, etc). Patients who did not require treatment per GELF criteria but had symptomatic follicular lymphoma could be enrolled.
Includes R2 plus bendamustine.
Patients had POD24 after first-line therapy and received epcoritamab plus R2 in the 2L.
Refractory: disease progression or stable disease as best response to therapy or disease progression within 6 months after completion of therapy.
Primary refractory: refractory to first-line antilymphoma therapy.
Double refractory: refractory to both anti-CD20 and an alkylating agent.
At a median study follow-up of 28.2 months (range, 2.4-39.0; 95% confidence interval [CI], 27.9-30.0), 53 patients (49%) completed treatment per protocol and 1 (1%) was still on treatment (supplemental Table 2). A total of 54 patients (50%) discontinued treatment. Primary reasons for treatment discontinuation were AEs (n = 26 [24%]), progressive disease (PD; n = 17 [16%]), withdrawal by patient (n = 8 [7%]), and other (n = 3 [3%]; recurrent infections, treatment-related immunosuppression and related infections, and reached maximum time on study [n = 1 each]). Median duration of epcoritamab treatment was 22 months (range, 0-26), and most patients completed R2 treatment per protocol; supplemental Table 3 shows treatment exposure. Median relative dose intensity for lenalidomide was ≥81% throughout the 12 treatment cycles.
Efficacy
Responses
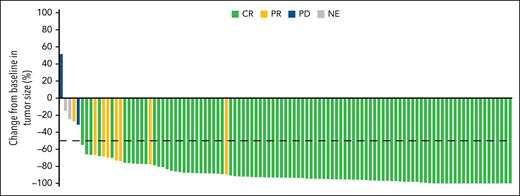
Among the 108 patients who received treatment, ORR was 96% (95% CI, 91-99) and CR rate was 88% (95% CI, 80-93). Two patients experienced PD with no response per Lugano criteria, and 2 patients were not evaluable for response (1 patient died and 1 patient discontinued treatment due to an AE before undergoing response assessment). Median time to response was 1.4 months (range, 1.0-3.1) and median time to CR was 1.4 months (range, 1.0-8.4), coinciding with the first response assessment. Almost all patients evaluable for change from baseline in tumor size (95%) had a meaningful (≥50%) reduction in target lesions (Figure 1).
Waterfall plot. Best percentage change from baseline in the sum of products of perpendicular diameters of target lesions for patients with R/R FL by best overall response per Lugano criteria by investigator assessment is shown. Patients evaluable for change from baseline in tumor size (n = 107) are shown; 1 patient was excluded due to the absence of baseline tumor measurements. Dashed line indicates a 50% reduction in tumor size compared with baseline. NE, not evaluable.
Waterfall plot. Best percentage change from baseline in the sum of products of perpendicular diameters of target lesions for patients with R/R FL by best overall response per Lugano criteria by investigator assessment is shown. Patients evaluable for change from baseline in tumor size (n = 107) are shown; 1 patient was excluded due to the absence of baseline tumor measurements. Dashed line indicates a 50% reduction in tumor size compared with baseline. NE, not evaluable.
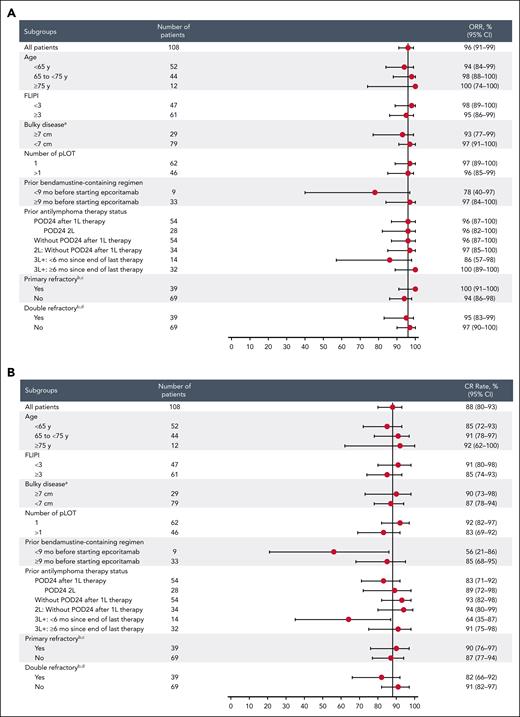
ORR and CR rates were consistently high across risk groups (Figure 2) defined based on pLOT (1 pLOT and >1 pLOT, CR rate of 92% and 83%, respectively), POD24 status in the 2L setting (with/without POD24, CR rate of 89%/94%), and disease refractoriness (nonprimary refractory/primary refractory, CR rate of 87%/90%; nondouble refractory/double refractory, CR rate of 91%/82%).
Response per Lugano criteria by investigator assessment in prespecified groups. (A) ORRs and (B) CR rates. aBulky disease per investigator. bRefractory: disease progression or stable disease as best response to therapy or disease progression within 6 months after completion of therapy. cPrimary refractory: refractory to 1L antilymphoma therapy. dDouble refractory: refractory to both anti-CD20 and an alkylating agent. 3L+, received epcoritamab plus R2 in the third line or later; FLIPI, Follicular Lymphoma International Prognostic Index; POD24 2L, progression of disease within 24 months after 1L therapy and received epcoritamab plus R2 in the 2L.
Response per Lugano criteria by investigator assessment in prespecified groups. (A) ORRs and (B) CR rates. aBulky disease per investigator. bRefractory: disease progression or stable disease as best response to therapy or disease progression within 6 months after completion of therapy. cPrimary refractory: refractory to 1L antilymphoma therapy. dDouble refractory: refractory to both anti-CD20 and an alkylating agent. 3L+, received epcoritamab plus R2 in the third line or later; FLIPI, Follicular Lymphoma International Prognostic Index; POD24 2L, progression of disease within 24 months after 1L therapy and received epcoritamab plus R2 in the 2L.
Time-to-event outcomes
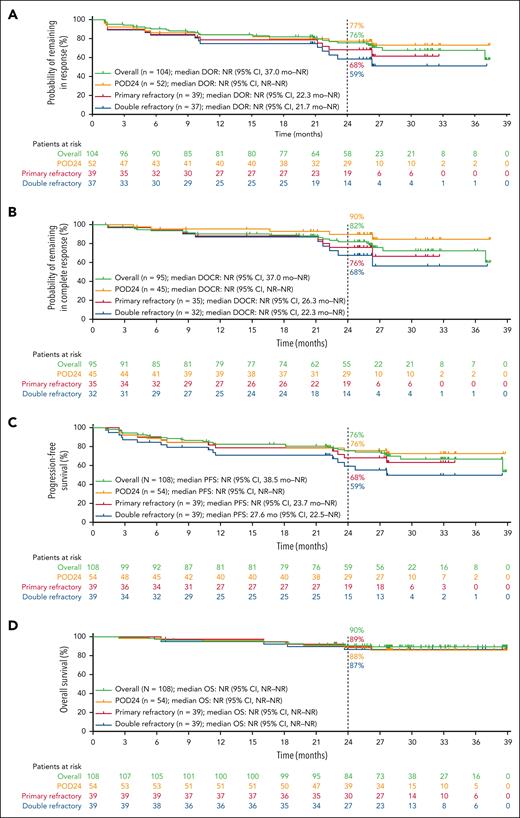
Median follow-up was 26.2 months for DOR and DOCR, 27.6 months for PFS, 28.2 months for OS, and 28.0 months for time to next antilymphoma therapy. Median DOR, DOCR, PFS, OS, and time to next antilymphoma therapy were not reached. Two-year Kaplan-Meier estimates for patients with an objective response (CR or partial response [PR]) remaining in response and patients with CR remaining in CR were 76% (95% CI, 65-83; Figure 3A) and 82% (95% CI, 72-89; Figure 3B), respectively; 2-year estimates for PFS, OS, and patients not starting next antilymphoma therapy were 76% (95% CI, 66-83; Figure 3C), 90% (95% CI, 83-95; Figure 3D), and 84% (95% CI, 75-90; supplemental Figure 1), respectively. Efficacy outcomes for the 2 dosing cohorts (supplemental Table 4) were consistent with those of the overall population. Time-to-event outcomes in groups of patients with difficult-to-treat disease (Figure 3) and by pLOT (supplemental Figure 2) were consistent with those of the overall population.
Patient outcomes. Kaplan-Meier plots of DOR in patients with CR or PR per Lugano criteria by investigator assessment (A); DOCR in patients with CR per Lugano criteria by investigator assessment (B); PFS by investigator assessment (C); and OS overall and in patients with POD24, primary refractory disease (disease refractory to 1L antilymphoma therapy), and double-refractory disease (disease refractory to both anti-CD20 and an alkylating agent) (D). Kaplan-Meier estimates are shown. NR, not reached.
Patient outcomes. Kaplan-Meier plots of DOR in patients with CR or PR per Lugano criteria by investigator assessment (A); DOCR in patients with CR per Lugano criteria by investigator assessment (B); PFS by investigator assessment (C); and OS overall and in patients with POD24, primary refractory disease (disease refractory to 1L antilymphoma therapy), and double-refractory disease (disease refractory to both anti-CD20 and an alkylating agent) (D). Kaplan-Meier estimates are shown. NR, not reached.
Fifty-two patients with CR and 1 patient with PR as their best overall response completed epcoritamab treatment per protocol (2 years of 28-day cycles; supplemental Figure 3). Three patients with CR experienced disease progression after completing epcoritamab treatment, and all other patients who completed treatment remained in response at data cutoff.
The 34 patients with CR who discontinued epcoritamab treatment early due to AEs (n = 23) or other reasons (n = 11, including withdrawal of consent [n = 3] and investigator decision [n = 5]) had a median epcoritamab treatment exposure of 10.0 months. Of these 34 patients, 29 (85%) remained in CR and 1 (3%) had initiated their next line of antilymphoma therapy as of data cutoff.
MRD analyses
Of 72 MRD-evaluable patients (67%), 62 (86%) had MRD negativity with an assay cutoff of 10−6 at any time point (see supplemental Table 5 for definitions used in MRD assessments). Most MRD-evaluable patients with a best response of CR were MRD negative (n/n = 60/65 [92%]). Only 1 patient with CR and MRD negativity became MRD positive at a subsequent assessment (cycle 5, day 1), and this patient later experienced radiographic PD. Among MRD-evaluable patients with a best response of PR, 33% (n/n = 2/6) were MRD negative; 1 of the 2 patients had an indeterminate MRD assessment at cycle 3, day 1, and negative MRD assessments at cycle 5, day 1 and cycle 7, day 1, whereas the other patient had 1 MRD-negative assessment at cycle 3, day 1, with no subsequent MRD assessments. MRD-negativity rates among MRD-evaluable patients with POD24 (n = 38), primary refractory disease (n = 28), and double-refractory disease (n = 27) were 87%, 82%, and 78%, respectively.
Safety
The most common treatment-emergent AEs (TEAEs) were neutropenia (65%; 3 patients [3%] had febrile neutropenia), COVID-19 (including COVID-19 pneumonia, post–acute COVID-19 syndrome, and suspected COVID-19; 59%), and CRS (51%; Table 2; dosing cohorts are reported in supplemental Table 6; a summary of TEAEs stratified by age group is shown in supplemental Table 7). Ninety-four patients (87%) had grade ≥3 TEAEs. TEAEs led to treatment discontinuation in 33 patients (31%); the most common was COVID-19 (n = 13 [12%]; see supplemental Figure 4 for a swimmer plot including these patients). After the initial 12 cycles of combination therapy, in the first 6 months of epcoritamab monotherapy, 6 patients (6% overall) discontinued treatment due to AEs, the most common of which were COVID-19 (n = 2) and pneumonia (n = 2). Between 6 and 12 months of epcoritamab monotherapy, 5 patients (5% overall) discontinued treatment due to AEs, most commonly COVID-19 (n = 3). TEAEs leading to R2 dose modification (including dose delay, reduction, increase, interruption, or omission) were reported in 94 patients (87%), including 89 patients (82%) with lenalidomide dose modification and 58 (54%) with rituximab dose modification. Of 53 patients who completed treatment per protocol, 51 had TEAEs leading to dose modification of any trial drug, including 43 with TEAEs leading to epcoritamab dose delay and 48 with TEAEs leading to R2 dose modification. Median treatment duration for these 51 patients was 23.4 months (95% CI, 23.3-23.5).
Summary of TEAEs
| . | Any grade, n (%) . | Grade ≥3, n (%) . |
|---|---|---|
| Any AE | 108 (100) | 94 (87.0) |
| Serious AE | 77 (71.3) | 54 (50.0) |
| AE leading to treatment discontinuation∗ | 33 (30.6) | NA |
| Fatal AE | 5 (4.6) | 5 (4.6) |
| AEs in ≥15% of patients | ||
| Hematologic | ||
| Neutropenia† | 70 (64.8) | 59 (54.6) |
| Anemia | 23 (21.3) | 4 (3.7) |
| Thrombocytopenia‡ | 23 (21.3) | 6 (5.6) |
| Nonhematologic | ||
| COVID-19§ | 64 (59.3) | 26 (24.1) |
| CRS|| | 55 (50.9) | 2 (1.9) |
| Injection-site reaction¶ | 52 (48.1) | 0 |
| Diarrhea | 50 (46.3) | 3 (2.8) |
| Fatigue | 43 (39.8) | 3 (2.8) |
| Constipation | 38 (35.2) | 1 (0.9) |
| Pyrexia | 33 (30.6) | 1 (0.9) |
| Muscle spasms | 27 (25.0) | 1 (0.9) |
| Cough | 26 (24.1) | 0 |
| Rash | 26 (24.1) | 2 (1.9) |
| Nausea | 24 (22.2) | 0 |
| Headache | 23 (21.3) | 1 (0.9) |
| Pneumonia# | 23 (21.3) | 13 (12.0) |
| Upper respiratory tract infection | 23 (21.3) | 1 (0.9) |
| Infusion-related reaction | 21 (19.4) | 0 |
| Arthralgia | 20 (18.5) | 0 |
| Hypogammaglobulinemia | 20 (18.5) | 1 (0.9) |
| Urinary tract infection | 19 (17.6) | 3 (2.8) |
| Asthenia | 17 (15.7) | 0 |
| Peripheral edema | 17 (15.7) | 0 |
| . | Any grade, n (%) . | Grade ≥3, n (%) . |
|---|---|---|
| Any AE | 108 (100) | 94 (87.0) |
| Serious AE | 77 (71.3) | 54 (50.0) |
| AE leading to treatment discontinuation∗ | 33 (30.6) | NA |
| Fatal AE | 5 (4.6) | 5 (4.6) |
| AEs in ≥15% of patients | ||
| Hematologic | ||
| Neutropenia† | 70 (64.8) | 59 (54.6) |
| Anemia | 23 (21.3) | 4 (3.7) |
| Thrombocytopenia‡ | 23 (21.3) | 6 (5.6) |
| Nonhematologic | ||
| COVID-19§ | 64 (59.3) | 26 (24.1) |
| CRS|| | 55 (50.9) | 2 (1.9) |
| Injection-site reaction¶ | 52 (48.1) | 0 |
| Diarrhea | 50 (46.3) | 3 (2.8) |
| Fatigue | 43 (39.8) | 3 (2.8) |
| Constipation | 38 (35.2) | 1 (0.9) |
| Pyrexia | 33 (30.6) | 1 (0.9) |
| Muscle spasms | 27 (25.0) | 1 (0.9) |
| Cough | 26 (24.1) | 0 |
| Rash | 26 (24.1) | 2 (1.9) |
| Nausea | 24 (22.2) | 0 |
| Headache | 23 (21.3) | 1 (0.9) |
| Pneumonia# | 23 (21.3) | 13 (12.0) |
| Upper respiratory tract infection | 23 (21.3) | 1 (0.9) |
| Infusion-related reaction | 21 (19.4) | 0 |
| Arthralgia | 20 (18.5) | 0 |
| Hypogammaglobulinemia | 20 (18.5) | 1 (0.9) |
| Urinary tract infection | 19 (17.6) | 3 (2.8) |
| Asthenia | 17 (15.7) | 0 |
| Peripheral edema | 17 (15.7) | 0 |
NA, not available.
AEs leading to treatment discontinuation were COVID-19 (n = 13; includes COVID-19 pneumonia); neutropenia (includes neutropenia and decreased neutrophil count; n = 6); fatigue (n = 3); malaise, pneumonia, and thrombocytopenia (thrombocytopenia includes thrombocytopenia and decreased platelet count; n = 2); and cerebral hemorrhage, colon cancer, CRS, cytomegalovirus colitis, dementia, depression, diarrhea, edema, fall, Haemophilus infection, hypogammaglobulinemia, insomnia, mania, muscle spasms, organizing pneumonia, parainfluenza virus infection, peripheral sensory neuropathy, Pneumocystis jirovecii pneumonia, pneumonitis, pulmonary embolism, rash, rhinitis, rhinovirus infection, and squamous cell carcinoma of the tongue (all n = 1). Some patients had >1 AE leading to treatment discontinuation.
Neutropenia includes neutropenia and decreased neutrophil count.
Thrombocytopenia includes thrombocytopenia and decreased platelet count.
COVID-19 includes COVID-19, COVID-19 pneumonia, post–acute COVID-19 syndrome, and suspected COVID-19.
Grade 1, n = 41 (38%); grade 2, n = 12 (11%); grade 3, n = 2 (2%).
Injection-site reaction includes injection-site reaction, erythema, pain, pruritus, rash, and swelling.
Excludes COVID-19 pneumonia, which was included in the grouped term “COVID-19.” Among the 23 patients with non–COVID-19 pneumonia, microorganisms were identified for 3 patients (Rhodococcus equi and respiratory syncytial virus, n = 1; Haemophilus influenzae, n = 1; and Moraxella catarrhalis, n = 1). For the remaining 20 patients, the microorganisms causing pneumonia were not identified, not confirmed, or not reported. Among these 20 patients, 1 patient had a concurrent COVID-19 infection.
Grade ≥3 COVID-19 occurred in 26 patients (24%), and the 5 patient deaths due to TEAEs were all due to COVID-19. Seventy-nine patients (73%) had gastrointestinal TEAEs, the most common of which were diarrhea (n = 50), constipation (n = 38), nausea (n = 24), abdominal pain (n = 14), and dyspepsia (n = 12). Twenty patients (19%) had hypogammaglobulinemia, and 37 (34%) received immunoglobulin supplementation. Neutropenia was primarily grade 3 to 4 (grade 3, n = 23 [21%]; grade 4, n = 36 [33%]), and led to treatment discontinuation in 6 patients. Fifty-six patients (52%) received granulocyte colony-stimulating factor, including 28 patients (26%) who were treated prophylactically.
CRS events were mostly low grade (38% grade 1, 11% grade 2, 2% grade 3; see supplemental Results for descriptions of patients with grade 3 cytokine release syndrome) for additional information on patients with grade 3 CRS). As previously observed with epcoritamab monotherapy,14,23 most CRS events occurred after the first full epcoritamab dose on cycle 1, day 15 (supplemental Figure 5). Median time to CRS onset after the first full dose was 2 days (range, 1-9). Thirty-six patients (33%) experienced serious CRS events; these events were considered serious due to hospitalization. All CRS events resolved (median time to resolution, 2 days [range, 1-23]), and none led to epcoritamab discontinuation. Fourteen patients (13%) received tocilizumab, 11 of whom (10%) received 1 dose. A grade 1 immune effector cell–associated neurotoxicity syndrome event occurred at the time of the first full dose in a patient with no baseline cognitive impairment (a description of a patient with immune effector cell–associated neurotoxicity syndrome can be found in the supplemental Results). The event resolved without dose modification or treatment. No clinical tumor lysis syndrome events were reported. Thromboembolic events occurred in 12 patients (11%), including 1 patient with grade 4 pulmonary embolism that led to treatment discontinuation.
Translational analyses
We explored the pharmacodynamic profile of epcoritamab plus R2 longitudinally in the peripheral blood from patients with available samples. Time points at which samples were collected to measure circulating immune cell counts and cytokine levels are provided in supplemental Table 8. Median B-cell levels reached undetectable levels (≤10 B cells per μL blood) by cycle 1, day 8, and remained undetectable in subsequent cycles (supplemental Figure 6A). Conversely, circulating total T cells showed substantial expansion compared with baseline between cycle 2 and cycle 7 (supplemental Figure 6B), whereas circulating natural killer cells showed substantial expansion between cycle 2, day 8 and cycle 7 (supplemental Figure 6C). In addition, we conducted an analysis of T-cell memory subsets (supplemental Figure 7). Within the CD4+ T-cell population, naive, T central memory, and T effector memory subsets showed substantial expansion between approximately cycle 1, day 22 and cycle 7, day 1. In contrast, CD4+ T effector memory cells reexpressing CD45RA did not show any meaningful changes on treatment. Within the CD8+ T-cell population, naive and all memory subsets evaluated showed substantial expansion between approximately cycle 1, day 22, and cycle 7, day 1.
We also assessed the levels of cytokines associated with the occurrence of CRS. Circulating interleukin 6, interleukin 10, tumor necrosis factor α, and interferon gamma levels showed peak elevation on cycle 1, day 16, ∼24 hours after the first full dose (supplemental Figure 8).
Discussion
In this phase 1b/2 study, we evaluated the combination of fixed-duration epcoritamab and R2 in patients with R/R FL because both therapies have been shown to be safe and effective alone for R/R FL.8,14 With the largely nonoverlapping safety profiles of epcoritamab and R2 and the potential to improve outcomes with this combination, it was compelling to clinically evaluate this novel regimen as a safe and potent chemotherapy-free treatment option. With a median follow-up of >2 years, the combination of epcoritamab and R2 demonstrated clinically meaningful antitumor activity, with CR rates that, to our knowledge, are higher than any previously published for other bispecific antibody–based regimens in R/R FL, although crosstrial comparisons must be interpreted with caution. The clinical benefit of epcoritamab plus R2 was observed across groups of patients with both standard-risk and high-risk disease features. Analyses based on pLOT showed that the CR rate was higher in the 2L group compared with the group receiving epcoritamab plus R2 in the third line or later, suggesting an advantage to using epcoritamab regimens earlier. Most patients with a poor prognosis, such as those with primary refractory or double-refractory disease, whose FL is typically resistant to common therapies including rituximab, had a CR with epcoritamab plus R2.
Durable responses were observed in nearly all patients who completed treatment per protocol, underscoring the ability of epcoritamab plus R2 to provide sustained clinical benefit as a fixed-duration treatment regimen for patients with R/R FL. The median follow-up is still relatively short for FL, which limits the interpretation of long-term outcomes, and it will be important to continue to assess patients treated with epcoritamab plus R2 in this trial and others to evaluate long-term efficacy.
Some patients with CR did not complete treatment per protocol, instead receiving a median of almost 1 year of epcoritamab treatment, and most had durable responses, with 29 of 34 (85%) remaining in CR at data cutoff. In cohort 2a, 9 of 24 (38%) patients completed treatment per protocol and were followed-up for a median of 38.6 months, with long-term outcomes consistent with those of the overall population, with medians for time-to-event outcomes having not been reached with >3 years of follow-up. The observation that most patients in CR who discontinued treatment due to AEs remained in CR at data cutoff is encouraging and supports the use of a fixed treatment duration in this setting. Furthermore, the rate of treatment discontinuation excluding discontinuations due to an AE of COVID-19 was 38%, which is similar to a previously published treatment discontinuation rate for R2 (30%).8
Although maintenance therapy for patients with R/R FL is a US Food and Drug Administration–approved standard and is supported by results from phase 3 trials including GADOLIN24 and EORTC 2098125 (providing rationale for the epcoritamab maintenance used in EPCORE NHL-2 arm 2), the limitations of B-cell–depleting maintenance therapy for patients with hematologic malignancies are now better understood in the postpandemic era. Our study was designed before the COVID-19 pandemic; therefore, the duration of epcoritamab maintenance in this trial may no longer be optimal by current standards and shortening it may prevent infectious complications. These learnings, along with the findings observed with the dosing regimens in cohorts 2a and 2b, led to epcoritamab plus R2 being investigated as a 1-year fixed-duration therapy for patients with R/R FL in the ongoing phase 3 EPCORE FL-1 trial (ClinicalTrials.gov identifier: NCT05409066).
The depth of responses was emphasized by high rates of MRD negativity among MRD-evaluable patients overall (86%), and in MRD-evaluable patients with POD24 (87%), primary refractory disease (82%), and double-refractory disease (78%). Along with published MRD reports from the epcoritamab monotherapy trial (EPCORE NHL-1),14 the MRD analyses from our study contribute to the growing body of evidence supporting the use of this biomarker in patients with R/R FL.
The AEs observed with epcoritamab plus R2 were consistent with the known safety profiles of the individual components8,14 and were manageable overall. The median relative dose intensity for lenalidomide remained high (≥81%) throughout the 12 treatment cycles, indicating the tolerability of this combination regimen.
COVID-19 occurred in 59% of patients in the study, with grade ≥3 COVID-19 occurring in 24% of patients. With patients enrolled and treated during the global COVID-19 pandemic, including the peak of the Omicron variant, some patients discontinued treatment due to COVID-19 or died due to COVID-19 infection while on study. There is an increased risk of morbidity and mortality due to infections for patients treated with B-cell–depleting therapies, including bispecific antibodies, and COVID-19 also had an impact on other studies of B-cell–depleting therapies during the same time period.26-30 Furthermore, immune responses to COVID-19 vaccines have been reported to be reduced in individuals who are immunocompromised, including patients with hematologic malignancies.31 The findings in EPCORE NHL-2 arm 2 and other trials evaluating epcoritamab suggest that, although trial participants had an elevated risk of severe outcomes from COVID-19, observed rates of infection and mortality did not significantly exceed expected rates based on epidemiological projections. Because the protocol was developed before the start of the COVID-19 pandemic, COVID-19 vaccination status was not required for enrollment in this trial. Future studies aimed at assessing the impact of vaccination on outcomes with epcoritamab are warranted.
With a 2-SUD regimen in cycle 1, CRS events with epcoritamab plus R2 were mostly low grade; however, 2 grade 3 CRS events were reported (details of patients with grade 3 cytokine release syndrome can be found in supplemental Results). An optimized 3-step SUD cycle 1 regimen that includes adequate hydration and prophylactic dexamethasone has been shown to substantially reduce the incidence and severity of CRS with epcoritamab monotherapy in patients with R/R FL with ≥2 pLOT.14 This 3-step SUD regimen was approved after the design of our trial11; however, it is being investigated with epcoritamab plus R2 in the EPCORE FL-1 trial.
Translational analyses showed that the mechanism of action of epcoritamab in combination with R2 was similar to that observed with epcoritamab monotherapy. Although the limitations of crosstrial comparisons must be considered, median interleukin 6 levels with epcoritamab plus R2 were lower than those observed with epcoritamab monotherapy with the 2-SUD regimen,14 in line with the lower CRS rate observed with epcoritamab plus R2. Although the reason for this phenomenon was not evaluated in this study, it is possible that the administration of rituximab weekly during cycle 1 contributed to eliminating target B cells from the peripheral blood and lymphoid tissue and dampening the production of cytokines associated with CRS.
The AUGMENT trial has provided a benchmark for R2 as a treatment approach.8 Although AUGMENT and EPCORE NHL-2 arm 2 were conducted in different treatment landscapes and enrolled slightly different patient populations (patients with previous lenalidomide or with rituximab-refractory disease were excluded from AUGMENT), deep responses appear more frequent with epcoritamab plus R2 than with R2 alone (CR rate was 88% in EPCORE NHL-2 arm 2 vs 34% in AUGMENT).8 In a trial investigating a CD20-targeted monoclonal antibody (obinutuzumab) in combination with lenalidomide in patients with R/R FL (GALEN; phase 2), the 2-year PFS estimate was 65% (vs 76% in EPCORE NHL-2 arm 2).32 In a phase 3 randomized trial of IV tafasitamab, an anti-CD19 monoclonal antibody, plus R2 vs R2 alone, CR rates were 49% vs 40%, and median PFS was 22.4 months vs 13.9 months (hazard ratio, 0.43), respectively.33 Although the median follow-up of the study was limited (14 months), the initial data suggest that combining a novel monoclonal antibody with R2 may improve PFS compared with R2 alone.33
These are, to our knowledge, the first published results for an off-the-shelf, subcutaneous CD3×CD20 bispecific antibody in combination with an anti-CD20 monoclonal antibody–containing, chemotherapy-free regimen. The CR rate with epcoritamab plus R2 was 88%. Glofitamab plus obinutuzumab in patients with R/R FL led to a CR rate of 74%,34 whereas mosunetuzumab plus lenalidomide led to a CR rate of 66%.35 ORR and CR rates with the chimeric antigen receptor (CAR) T-cell therapies tisagenlecleucel, axicabtagene ciloleucel, and lisocabtagene maraleucel were 86% and 69% (efficacy analysis set), 94% and 79% (per-protocol population), and 93% and 91% (intention-to-treat population), respectively, and remissions with CAR T-cell therapy were durable.36-38 Although the limitations of crosstrial comparisons in unmatched populations must be considered when comparing results, in contrast to CAR T-cell therapies, bispecific antibodies do not require apheresis, engineering before administration, or referral to a treatment center specializing in cell therapy. Moreover, unlike the other approved CD3×CD20 bispecific antibodies, epcoritamab is approved for subcutaneous administration,11 which requires less time and resource use and may be safer than IV administration.39
Strengths of this study include the large sample size and its international nature. The limitations are those of an open-label, single-arm design with no comparator, now being addressed with the randomized phase 3 trial. Although this trial demonstrated deep and durable responses with an epcoritamab combination regimen in a population comprised largely of patients with 1 pLOT, there are no comparable data in the 2L setting for epcoritamab monotherapy, highlighting a data gap that should be addressed with future studies.
With >2 years of follow-up, fixed-duration epcoritamab plus R2 demonstrated highly encouraging results, with deep and durable responses observed across all patient populations treated, including patients with high-risk disease features. The high MRD-negativity rates further substantiate the depth of responses achieved with this potent regimen. The safety profile of this chemotherapy-free combination was manageable and consistent with previous reports, and there were no new safety signals.
Acknowledgments
The authors thank the patients and their families for their participation in this study. The authors also thank the participating study sites, investigators, data monitoring committee, and other research personnel for their support of this trial, and Işıl Altıntaş of Genmab for valuable contributions. Medical writing assistance was provided by Rachel O’Keefe and Braden Roth of Peloton Advantage, LLC, an OPEN Health company, and funded by Genmab A/S and AbbVie.
K.M.L. is supported by the National Institute for Health and Care Research Manchester Biomedical Research Centre (NIHR203308). This research was funded in part by a grant from Blood Cancer United (formerly the Leukemia & Lymphoma Society) and through the National Cancer Institute, National Institutes of Health Cancer Center Support Grant P30 CA008748 (institution of L.F.).
Authorship
Contribution: L.F., D.H., A.R., A.A., and J.M. were responsible for conception, design, and planning of the study; D.H., A.R., A.A., and J.M. were responsible for data acquisition; L.F. had final responsibility for the decision to submit for publication; and all authors contributed to data interpretation, writing of the manuscript, critical review and revision of the manuscript, and had full access to all of the data in the study.
Conflict-of-interest disclosure: L.F. reports consultancy for AbbVie, Genentech, AstraZeneca, Genmab, and Roche; honoraria from AbbVie, Genentech, AstraZeneca, Genmab, Roche, Sanofi, and EvolveImmune; research funding from AbbVie, Genentech, AstraZeneca, Genmab, Roche, BeiGene, and Innate; and membership on an entity’s board of directors or advisory committees for AbbVie, Genentech, ADC Therapeutics, Ipsen, Johnson & Johnson, and Seagen. A.S. reports consultancy role with Takeda, Bristol Myers Squibb (BMS), Novartis, Janssen, Merck Sharp & Dohme (MSD), Amgen, GlaxoSmithKline (GSK), Sanofi, Kite, and Mundipharma; honoraria from Takeda, BMS, Novartis, Janssen, MSD, Amgen, GSK, Sanofi, and Kite; membership on an entity’s board of directors or advisory committees for Takeda, BMS, Novartis, Janssen, Amgen, Bluebird, Sanofi, and Kite; travel expenses from Takeda, BMS, and Roche; research funding from Takeda; and a speaker’s bureau role for Takeda, BMS, Novartis, Janssen, MSD, Amgen, GSK, Sanofi, and Kite. S.L. reports membership on an entity’s board of directors or advisory committees for AbbVie, BeiGene, Genmab, Gilead, Incyte, Novartis, Orion, Roche, and Sobi; research funding (paid to institution) from Bayer, BMS, Genmab, Hutchmed, Novartis, Nordic Nanovector, and Roche; and honoraria from Gilead, Incyte, and Novartis. J.S.P.V. reports consultancy role with Secura Bio. M.N. reports membership on an advisory board for AbbVie; and research funding from Nordic Nanovector and Takeda. H.H. reports membership on an advisory board for Incyte, Novartis, Roche, and Takeda; membership on a safety committee for Genmab and Nordic Nanovector; consultancy for Gilead and Roche; and research funding from Incyte. R.W.M. reports consultancy for AbbVie, Adaptive Biotechnologies, BMS, Epizyme, Genmab, and Intellia; and research funding (paid to institution) from BMS, Genentech/Roche, Genmab, and Merck. P.J.L. reports research grants from Takeda and Servier; advisory honoraria from BMS, Roche, Takeda, Genmab, AbbVie, Incyte, Regeneron, and Sandoz; and consultancy honoraria from Y-mAbs Therapeutics. P.A. reports consultancy and speaker’s bureau roles for AbbVie, AstraZeneca, BMS, and Janssen. K.M.L. reports membership on the Epcoritamab Global Council for Genmab; consulting or advisory roles for AbbVie, BeiGene, BMS, Genmab, Kite/Gilead, and Roche; a speakers bureau role for AbbVie and BMS; research funding (paid to institution) from AbbVie, ADC Therapeutics, AstraZeneca, BeiGene, BMS, CellCentric, Genmab, Janssen, Kite/Gilead, MorphoSys, MSD, Nurix, Regeneron, Roche, Step Pharma, and Viracta; and travel expenses from BMS. G.S. reports current employment at AbbVie. D.H., A.R., and A.A. report employment at Genmab at the time the analysis was conducted. J.M., Y.H., A.J.S., and C.M. report current employment at Genmab. M.H. reports membership on scientific advisory boards for AbbVie, BMS, Genmab, Janssen, Roche, and Takeda; and research support (paid to institution) from BMS, Genentech, Genmab, Incyte, Janssen, Novartis, Roche, and Takeda. D.B. reports consultancy for AbbVie, BMS, Genmab, Gilead, MorphoSys, Roche, Sobi, and Takeda; honoraria from AbbVie, Gilead, Janssen, MorphoSys, Roche, and Takeda; and travel expenses/accommodations from AbbVie, Recordati, and Roche. The remaining authors declare no competing financial interests.
The current affiliation for D.H. is Hematology/Oncology, Clinical Development, AstraZeneca, New York, NY.
The current affiliation for A.R. is Late Stage Oncology Global Clinical Development, Merck, Rahway, NJ.
The current affiliation for A.A is Clinical Science, Daiichi Sankyo, Basking Ridge, NJ.
Correspondence: Lorenzo Falchi, Lymphoma Service, Memorial Sloan Kettering Cancer Center, 530 E 74th St, New York, NY 10021; email: falchil@mskcc.org; and David Belada, 4th Department of Internal Medicine, Haematology, Charles University, Hospital and Faculty of Medicine, Sokolská 581, 500 05 Hradec Králové, Czech Republic; email: beladad@lfhk.cuni.cz.
References
Author notes
On behalf of the Lunenburg Lymphoma Phase I/II Consortium-HOVON/LLPC.
Deidentified individual participant data collected during the trial will not be available upon request for further analyses by external independent researchers. Aggregated clinical trial data from the trial are provided via publicly accessible study registries/databases as required by law. For more information, please contact clinicaltrials@genmab.com.
The online version of this article contains a data supplement.
There is a Blood Commentary on this article in this issue.
The publication costs of this article were defrayed in part by page charge payment. Therefore, and solely to indicate this fact, this article is hereby marked “advertisement” in accordance with 18 USC section 1734.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal